Compliance with Tuberculosis Screening in Irregular Immigrants
Abstract
:1. Introduction
2. Materials and Methods
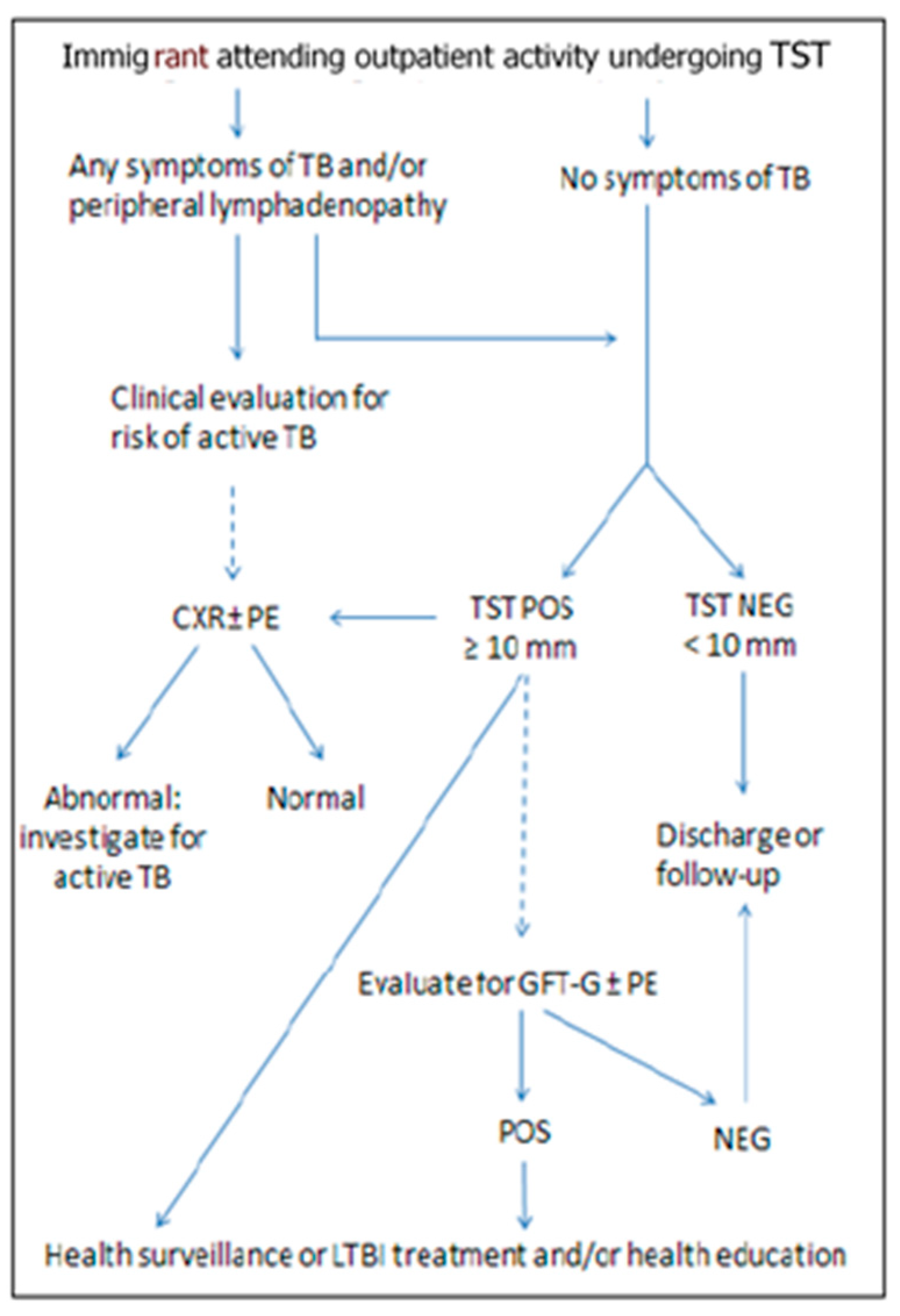
2.1. Setting and Clinical Procedures
2.2. Study Design and Definitions
2.3. Data Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Floyd, K.; Glaziou, P.; Zumla, A.; Raviglione, M. The global tuberculosis epidemic and progress in care, prevention, and research: An overview in year 3 of the End TB era. Lancet Respir. Med. 2018, 6, 299–314. [Google Scholar] [CrossRef]
- WHO. Global Tuberculosis Report 2016; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Matteelli, A.; Rendon, A.; Tiberi, S.; Al-Abri, S.; Voniatis, C.; Carvalho, A.C.C.; Centis, R.; D’Ambrosio, L.; Visca, D.; Spanevello, A.; et al. Tuberculosis elimination: Where are we now? Eur. Respir. Rev. 2018, 27. [Google Scholar] [CrossRef] [PubMed]
- Lonnroth, K.; Migliori, G.B.; Abubakar, I.; D’Ambrosio, L.; de Vries, G.; Diel, R.; Douglas, P.; Falzon, D.; Gaudreau, M.A.; Goletti, D.; et al. Towards tuberculosis elimination: An action framework for low-incidence countries. Eur. Respir. J. 2015, 45, 928–952. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Montalva, A.; Salvador, F.; Molina-Morant, D.; Molina, I. Tuberculosis and immigration. Enfermedades Infecciosas y Microbiologia Clinica 2018, 36, 446–455. [Google Scholar] [CrossRef] [PubMed]
- WHO. The End TB Strategies; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Kaushik, N.; Lowbridge, C.; Scandurra, G.; Dobler, C.C. Post-migration follow-up programme for migrants at increased risk of developing tuberculosis: A cohort study. ERJ Open Res. 2018, 4. [Google Scholar] [CrossRef] [PubMed]
- IsdseAsesdRE-R. La Tubercolosi in Italia, Rapporto 2008; Ministero della Salute: Roma, Italy, 2010.
- Salute, M.D. Aggiornamento Delle Raccomandazioni per le Attività di Controllo Della Tubercolosi. Gestione dei Contatti e Della Tubercolosi in Ambito Assistenziale; Istituto Nazionale Malattie Infettive Lazzaro Spallanzani IRCCS: Roma, Italy, 2010. [Google Scholar]
- Salute, M.D. Aggiornamento Delle Raccomandazioni per le Attività di Controllo Della Tubercolosi: Politiche Efficaci a Contrastare la Tubercolosi Nella Popolazione Immigrata; Ministero della Salute: Roma, Italy, 2010.
- CDC. Targeted Tubercolin Testing and Treatment of Latent Tubercolosis Inferction; CDC: Atlanta, GA, USA, 2000.
- WHO. Definitions and Reporting Framework for Tuberculosis—2013 Revision; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Morano, J.P.; Walton, M.R.; Zelenev, A.; Bruce, R.D.; Altice, F.L. Latent tuberculosis infection: Screening and treatment in an urban setting. J. Community Health 2013, 38, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Bennett, R.J.; Brodine, S.; Waalen, J.; Moser, K.; Rodwell, T.C. Prevalence and treatment of latent tuberculosis infection among newly arrived refugees in San Diego County, January 2010-October 2012. Am. J. Public Health 2014, 104, e95–e102. [Google Scholar] [CrossRef] [PubMed]
- Bodenmann, P.; Vaucher, P.; Wolff, H.; Favrat, B.; de Tribolet, F.; Masserey, E.; Zellweger, J.P. Screening for latent tuberculosis infection among undocumented immigrants in Swiss healthcare centres; a descriptive exploratory study. BMC Infect. Dis. 2009, 9, 34. [Google Scholar] [CrossRef] [PubMed]
- El-Hamad, I.; Casalini, C.; Matteelli, A.; Casari, S.; Bugiani, M.; Caputo, M.; Bombana, E.; Scolari, C.; Moioli, R.; Scarcella, C.; et al. Screening for tuberculosis and latent tuberculosis infection among undocumented immigrants at an unspecialised health service unit. Int. J. Tuberc. Lung Dis. 2001, 5, 712–716. [Google Scholar] [PubMed]
- Schepisi, M.S.; Gualano, G.; Piselli, P.; Mazza, M.; D’Angelo, D.; Fasciani, F.; Barbieri, A.; Rocca, G.; Gnolfo, F.; Olivani, P.; et al. Active Tuberculosis Case Finding Interventions Among Immigrants, Refugees and Asylum Seekers in Italy. Infect. Dis. Rep. 2016, 8, 6594. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines on the Management of Latent Tuberculosis Infection; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Suphanchaimat, R.; Kantamaturapoj, K.; Putthasri, W.; Prakongsai, P. Challenges in the provision of healthcare services for migrants: A systematic review through providers’ lens. BMC Health Serv. Res. 2015, 15, 390. [Google Scholar] [CrossRef] [PubMed]
- Cheng, I.H.; Drillich, A.; Schattner, P. Refugee experiences of general practice in countries of resettlement: A literature review. Br. J. Gen. Pract. 2015, 65, e171–e176. [Google Scholar] [CrossRef] [PubMed]
- Saracino, A.; Scotto, G.; Fornabaio, C.; Martinelli, D.; Faleo, G.; Cibelli, D.; Tartaglia, A.; Di Tullio, R.; Fazio, V.; Prato, R.; et al. QuantiFERON-TB Gold In-Tube test (QFT-GIT) for the screening of latent tuberculosis in recent immigrants to Italy. New Microbiol. 2009, 32, 369–376. [Google Scholar] [PubMed]
| Steps of the Algorithm | N Performed/Prescribed | % |
|---|---|---|
| TST read | 349/368 | 94.8 |
| CXR performed | 134/151 | 88.7 |
| QFT performed | 89/110 | 80.9 |
| PE attended | 78/84 | 92.9 |
| Screening TB (TST, CXR, PE) completed | 332/368 | 90.2 |
| negative TST with CXR indication | 12/12 | 100 |
| positive TST | 122/139 | 87.8 |
| Screening LTBI (TST, CXR, QFT, PE) completed | 317/363 | 87.3 |
| negative TST with CXR indication | 12/12 | 100 |
| positive TST | 107/134 | 79.9 |
| LTBI treatment | ||
| refusal or voluntary interruption | 8/28 | 28.6 |
| medical contraindication or interruption | 6/28 | 21.4 |
| treatment completed | 14/28 | 50.0 |
| LTBI health surveillance never started | 17/41 | 41.5 |
| voluntary interruption | 18/41 | 43.9 |
| completed | 6/41 | 14.6 |
| TB treatment completed | 5/5 | 100 |
| Whole algorithm completed | 281/368 | 76.4 |
| negative TST with CXR indication | 12/12 | 100 |
| positive TST | 71/139 | 51.1 |
| Sample Characteristics | TB Screening Started/Completed | Bivariate OR (95% CI) | p-Value | Multivariate OR (95% CI) | p-Value | |
|---|---|---|---|---|---|---|
| N | % | |||||
| Sex: | ||||||
| Male | 222/243 | 91.36 | 1.00 (reference) | 1.00 (reference) | ||
| Female | 110/125 | 88.00 | 0.69 (0.34–1.40) | 0.306 | 0.47 (0.22–1.03) | 0.061 |
| Age at TST in Years: | ||||||
| 15–24 | 90/97 | 92.78 | 1.00 (reference) | 1.00 (reference) | ||
| 25–34 | 113/130 | 86.92 | 0.52 (0.21–1.30) | 0.161 | 0.44 (0.16–1.18) | 0.103 |
| 35–44 | 87/95 | 91.58 | 0.85 (0.29–2.43) | 0.756 | 0.57 (0.18–1.84) | 0.344 |
| ≥45 | 42/46 | 91.30 | 0.82 (0.23–2.94) | 0.757 | 0.90 (0.20–4.03) | 0.891 |
| Continuous OR | 0.99 (0.70–1.41) | 0.972 | 0.96 (0.64–1.43) | 0.827 | ||
| Region of Origin: | ||||||
| Africa | 103/111 | 92.79 | 1.00 (reference) | 1.00 (reference) | ||
| Eastern Mediterranean | 75/83 | 90.36 | 0.73 (0.26–2.03) | 0.544 | 0.75 (0.25–2.27) | 0.607 |
| Europe | 60/72 | 83.33 | 0.39 (0.15–1.00) | 0.051 | 0.44 (0.15–1.30) | 0.138 |
| SE Asia/West Pacific | 85/90 | 94.44 | 1.32 (0.42–4.19) | 0.637 | 2.49 (.57–10.87) | 0.226 |
| Americas | 9/12 | 75.00 | 0.23 (0.05–1.04) | 0.056 | 0.27 (0.05–1.44) | 0.125 |
| TB Incidence °: | 1.26 (0.89–1.79) | 0.187 | 1.15 (0.79–1.69) | 0.458 | ||
| Any Symptoms *: | ||||||
| No | 211/236 | 89.41 | 1.00 (reference) | |||
| Yes | 121/132 | 91.67 | 1.30 (0.62–2.74) | 0.485 | ||
| Cough: | ||||||
| No | 249/280 | 88.93 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 83/88 | 94.32 | 2.07 (0.78–5.49) | 0.145 | 3.23 (1.04–10.05) | 0.043 |
| Education **: | 1.36 (0.88–2.12) | 0.168 | 1.49 (0.93–2.40) | 0.101 | ||
| Italian Language **°: | 0.89 (0.52–1.50) | 0.652 | 0.90 (0.51–1.58) | 0.721 | ||
| Employment Status: | ||||||
| No | 234/258 | 90.70 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 90/102 | 88.24 | 0.77 (0.37–1.60) | 0.484 | 0.79 (0.32–1.96) | 0.612 |
| Years in Italy: | ||||||
| <5 | 223/252 | 88.49 | 1.00 (reference) | 1.00 (reference) | ||
| ≥5 | 102/109 | 93.58 | 1.89 (0.80–4.47) | 0.144 | 3.29 (1.18–9.22) | 0.023 |
| Continuous OR | 1.06 (0.96–1.18) | 0.248 | 1.18 (0.72–1.92) | 0.513 | ||
| Homelessness: | ||||||
| No | 313/345 | 90.72 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 19/23 | 82.61 | 0.49 (0.16–1.52) | 0.213 | 0.35 (0.10–1.25) | 0.107 |
| Refugees: | ||||||
| No | 304/340 | 89.41 | - | |||
| Yes | 28/28 | 100.00 | - | 0.070 *** | ||
| Prostitution: | ||||||
| No | 323/357 | 90.48 | 1.00 (reference) | |||
| Yes | 9/11 | 81.82 | 0.47 (0.10–2.28) | 0.352 | ||
| Pregnancy: | ||||||
| No | 323/359 | 89.97 | - | |||
| Yes | 9/9 | 100.00 | - | 0.319 *** | ||
| TST Result: | ||||||
| Negative | 210/210 | 100.00 | - | |||
| Positive | 122/139 | 87.77 | - | 0.000 *** | ||
| Sample Characteristics | LTBI Screening Started/Completed | Bivariate OR (95% CI) | p-Value | Multivariate OR (95% CI) | p-Value | |
|---|---|---|---|---|---|---|
| N | % | |||||
| Sex: | ||||||
| Male | 211/241 | 87.55 | 1.00 (reference) | 1.00 (reference) | ||
| Female | 106/122 | 86.89 | 0.94 (0.49–1.80) | 0.857 | 0.76 (0.38–1.54) | 0.451 |
| Age at TST in Years: | ||||||
| 15–24 | 88/97 | 90.72 | 1.00 (reference) | 1.00 (reference) | ||
| 25–34 | 107/127 | 84.25 | 0.55 (0.24–1.26) | 0.157 | 0.54 (0.22–1.31) | 0.173 |
| 35–44 | 80/93 | 86.02 | 0.63 (0.26–1.55) | 0.314 | 0.57 (0.21–1.56) | 0.273 |
| ≥45 | 42/46 | 91.30 | 1.07 (0.31–3.69) | 0.910 | 1.39 (0.33–5.86) | 0.658 |
| Continuous OR | 0.98 (0.72–1.34) | 0.892 | 1.01 (0.70 –1.45) | 0.956 | ||
| Region of Origin: | ||||||
| Africa | 97/109 | 88.99 | 1.00 (reference) | 1.00 (reference) | ||
| Eastern Mediter. | 73/83 | 87.95 | 0.90 (0.37–2.20) | 0.823 | 0.93 (0.35–2.47) | 0.892 |
| Europe | 60/72 | 83.33 | 0.62 (0.26–1.47) | 0.275 | 0.72 (0.27–1.91) | 0.509 |
| SE Asia/West Pacific | 78/87 | 89.66 | 1.07 (0.43–2.67) | 0.881 | 2.39 (0.72–7.93) | 0.155 |
| Americas | 9/12 | 75.00 | 0.37 (0.09–1.56) | 0.177 | 0.32 (0.06–1.64) | 0.174 |
| TB Incidence °: | 1.11 (0.82–1.52) | 0.489 | 0.99 (0.70–1.40) | 0.975 | ||
| Any Symptoms *: | ||||||
| No | 203/235 | 86.38 | 1.00 (reference) | |||
| Yes | 114/128 | 89.06 | 1.28 (0.66–2.51) | 0.464 | ||
| Cough: | ||||||
| No | 240/279 | 86.02 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 77/84 | 91.67 | 1.79 (0.77–4.16) | 0.178 | 2.38 (0.92–6.17) | 0.073 |
| Education **: | 1.09 (0.73–1.63) | 0.660 | 1.09 (0.71–1.68) | 0.688 | ||
| Italian Language **°: | 1.13 (0.68–1.87) | 0.636 | 1.15 (0.67–1.95) | 0.616 | ||
| Employment Status: | ||||||
| No | 228/257 | 88.72 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 81/98 | 82.65 | 0.61 (0.32–1.16) | 0.131 | 0.56 (0.25–1.26) | 0.162 |
| Years in Italy: | ||||||
| <5 | 215/249 | 86.35 | 1.00 (reference) | 1.00 (reference) | ||
| ≥5 | 95/107 | 88.79 | 1.25 (0.62–2.52) | 0.530 | 1.88 (0.80–4.40) | 0.145 |
| Continuous OR | 1.04 (0.95–1.13) | 0.376 | 0.99 (0.64–1.54) | 0.972 | ||
| Homelessness: | ||||||
| No | 299/340 | 87.94 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 18/23 | 78.26 | 0.49 (0.17–1.40) | 0.185 | 0.35 (0.11–1.12) | 0.076 |
| Refugees: | ||||||
| No | 290/335 | 86.57 | 1.00 (reference) | |||
| Yes | 27/28 | 96.43 | 4.19 (0.56–31.60) | 0.165 | ||
| Prostitution: | ||||||
| No | 309/352 | 87.78 | 1.00 (reference) | |||
| Yes | 8/11 | 72.73 | 0.37 (0.09–1.45) | 0.155 | ||
| Pregnancy: | ||||||
| No | 309/355 | 87.04 | - | |||
| Yes | 8/8 | 100.00 | - | 0.277 *** | ||
| TST result: | ||||||
| Negative | 210/210 | 100.00 | - | |||
| Positive | 107/134 | 79.85 | - | 0.000 *** | ||
| Protocol Started/Completed | Bivariate OR (95% CI) | p-Value | Multivariate | p-Value | ||
|---|---|---|---|---|---|---|
| Sample characteristics | N | % | OR (95% CI) | |||
| Sex: | ||||||
| Male | 183/243 | 75.31 | 1.00 (reference) | 1.00 (reference) | ||
| Female | 98/125 | 78.40 | 1.19 (0.71–1.99) | 0.509 | 1.18 (0.65–2.12) | 0.588 |
| Age at TST in Years: | ||||||
| 15–24 | 83/97 | 85.57 | 1.00 (reference) | 1.00 (reference) | ||
| 25–34 | 97/130 | 74.62 | 0.50 (0.25–0.99) | 0.046 | 0.44 (0.21–0.93) | 0.031 |
| 35–44 | 70/95 | 73.68 | 0.47 (0.23–0.98) | 0.043 | 0.39 (0.17–0.88) | 0.024 |
| ≥45 | 31/46 | 67.39 | 0.35 (0.15–0.81) | 0.014 | 0.26 (0.10–0.68) | 0.006 |
| Continuous OR | 0.74 (0.58–0.94) | 0.014 | 0.67 (0.50–0.89) | 0.007 | ||
| Region of Origin: | ||||||
| Africa | 85/111 | 76.58 | 1.00 (reference) | 1.00 (reference) | ||
| Eastern Mediterranean | 62/83 | 74.70 | 0.90 (0.47–1.75) | 0.763 | 0.87 (0.41–1.85) | 0.723 |
| Europe | 49/72 | 68.06 | 0.65 (0.34–1.26) | 0.205 | 0.90 (0.42–1.97) | 0.801 |
| SE Asia/West Pacific | 76/90 | 84.44 | 1.66 (0.81–3.41) | 0.167 | 4.48 (1.57–12.73) | 0.005 |
| Americas | 9/12 | 75.00 | 0.92 (0.23–3.64) | 0.903 | 0.67 (0.14–3.16) | 0.617 |
| TB Incidence °: | 0.91 (0.72–1.16) | 0.461 | 0.79 (0.59–1.05) | 0.107 | ||
| Any Symptoms *: | ||||||
| No | 179/236 | 75.85 | 1.00 (reference) | |||
| Yes | 102/132 | 77.27 | 1.08 (0.65–1.79) | 0.758 | ||
| Cough: | ||||||
| No | 212/280 | 75.71 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 69/88 | 78.41 | 1.16 (0.65–2.07) | 0.604 | 1.75 (0.88–3.46) | 0.110 |
| Education **: | 1.18 (0.86–1.62) | 0.306 | 1.24 (0.88–1.74) | 0.220 | ||
| Italian Language **°: | 1.26 (0.85–1.89) | 0.254 | 1.30 (0.82–2.04) | 0.265 | ||
| Employment Status: | ||||||
| No | 201/258 | 77.91 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 75/102 | 73.53 | 0.79 (0.46–1.34) | 0.377 | 0.59 (0.30–1.17) | 0.133 |
| Years in Italy: | ||||||
| <5 | 188/252 | 74.60 | 1.00 (reference) | 1.00 (reference) | ||
| ≥5 | 88/109 | 80.73 | 1.43 (0.82–2.48) | 0.209 | 2.94 (1.44–6.01) | 0.003 |
| Continuous OR | 1.06 (0.99–1.13) | 0.119 | 1.30 (0.90–1.86) | 0.160 | ||
| Homelessness: | ||||||
| No | 267/345 | 77.39 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 14/23 | 60.87 | 0.45 (0.19–1.09) | 0.077 | 0.35 (0.13–0.96) | 0.042 |
| Refugees: | ||||||
| No | 258/340 | 75.88 | 1.00 (reference) | |||
| Yes | 23/28 | 82.14 | 1.46 (0.54–3.97) | 0.456 | ||
| Prostitution: | ||||||
| No | 273/357 | 76.47 | 1.00 (reference) | |||
| Yes | 8/11 | 72.73 | 0.82 (0.21–3.16) | 0.774 | ||
| Pregnancy: | ||||||
| No | 272/359 | 75.84 | - | |||
| Yes | 9/9 | 91.67 | - | 0.091 *** | ||
| TST Result: | ||||||
| Negative | 210/210 | 100.00 | - | |||
| Positive | 71/139 | 51.08 | - | 0.000 *** | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonvicini, F.; Cilloni, S.; Fornaciari, R.; Casoni, C.; Marchesi, C.; Greci, M.; Monici, L.; Nicolini, F.; Vinceti, M. Compliance with Tuberculosis Screening in Irregular Immigrants. Int. J. Environ. Res. Public Health 2019, 16, 28. https://doi.org/10.3390/ijerph16010028
Bonvicini F, Cilloni S, Fornaciari R, Casoni C, Marchesi C, Greci M, Monici L, Nicolini F, Vinceti M. Compliance with Tuberculosis Screening in Irregular Immigrants. International Journal of Environmental Research and Public Health. 2019; 16(1):28. https://doi.org/10.3390/ijerph16010028
Chicago/Turabian StyleBonvicini, Francesca, Silvia Cilloni, Rossano Fornaciari, Carmen Casoni, Cristina Marchesi, Marina Greci, Lucia Monici, Fausto Nicolini, and Marco Vinceti. 2019. "Compliance with Tuberculosis Screening in Irregular Immigrants" International Journal of Environmental Research and Public Health 16, no. 1: 28. https://doi.org/10.3390/ijerph16010028
APA StyleBonvicini, F., Cilloni, S., Fornaciari, R., Casoni, C., Marchesi, C., Greci, M., Monici, L., Nicolini, F., & Vinceti, M. (2019). Compliance with Tuberculosis Screening in Irregular Immigrants. International Journal of Environmental Research and Public Health, 16(1), 28. https://doi.org/10.3390/ijerph16010028






