Influence of Acetylcholinesterase Inhibitors Used in Alzheimer’s Disease Treatment on the Activity of Antioxidant Enzymes and the Concentration of Glutathione in THP-1 Macrophages under Fluoride-Induced Oxidative Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Cultivation and Treatment of Cells
2.3. Enzyme Activity
2.4. Glutathione (GSH) Concentration
2.5. Fluorescent Studies
Visualization and Quantitative Estimation of ROS Formation Within the Cells
2.6. Protein Assay
2.7. Statistic Evaluation
3. Results
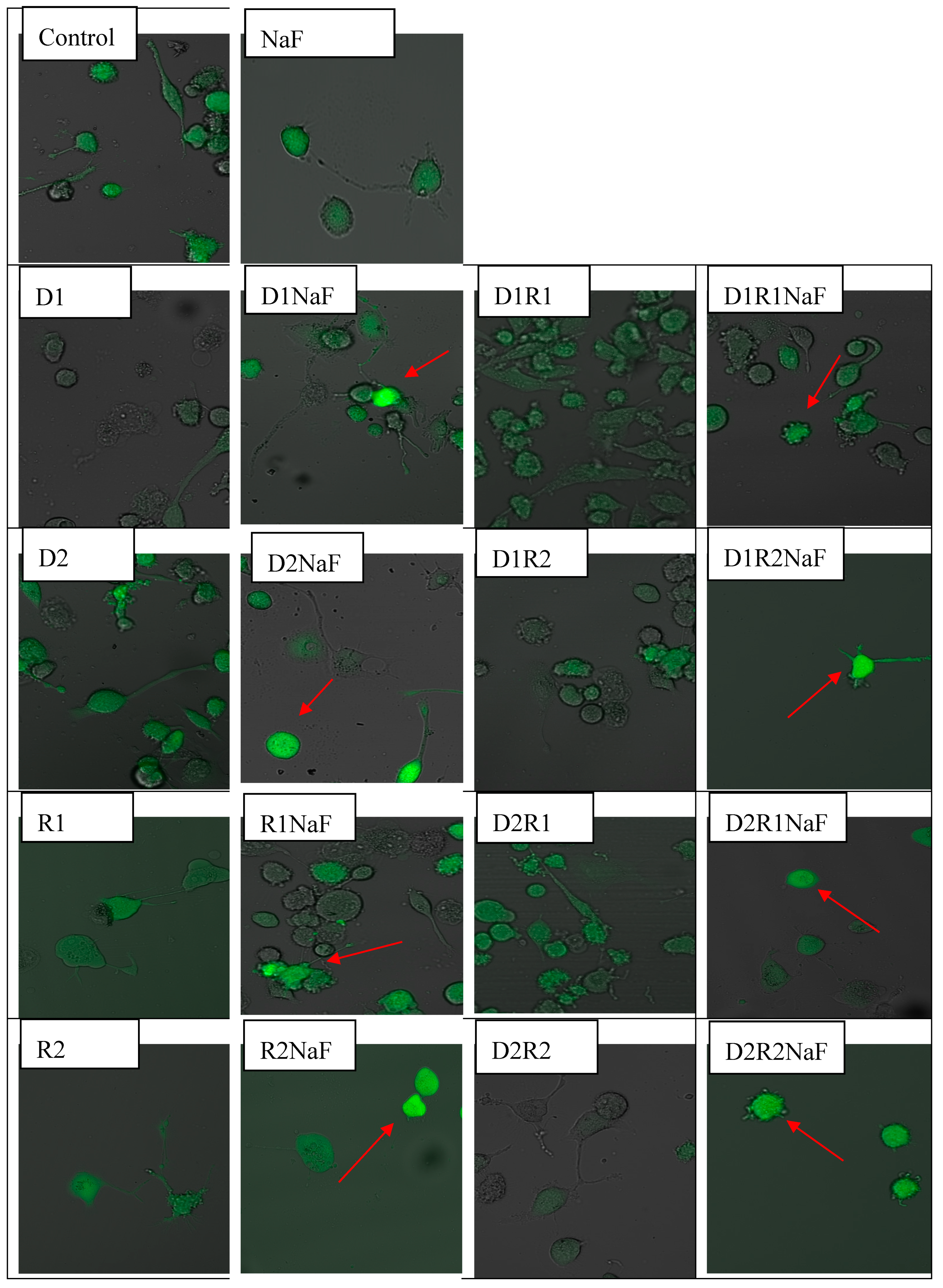
3.1. Effect of Donepezil and Rivastigmine on Intracellular ROS Generation in Macrophages
Rivastigmine and Donepezil Inhibited ROS Generation in Macrophages
3.2. The Effect of Donepezil and Rivastigmine on Intracellular ROS Generation in Fluoride-Exposed Macrophages
Prooxidative Fluoride Condition Increased ROS Quantity in Macrophages
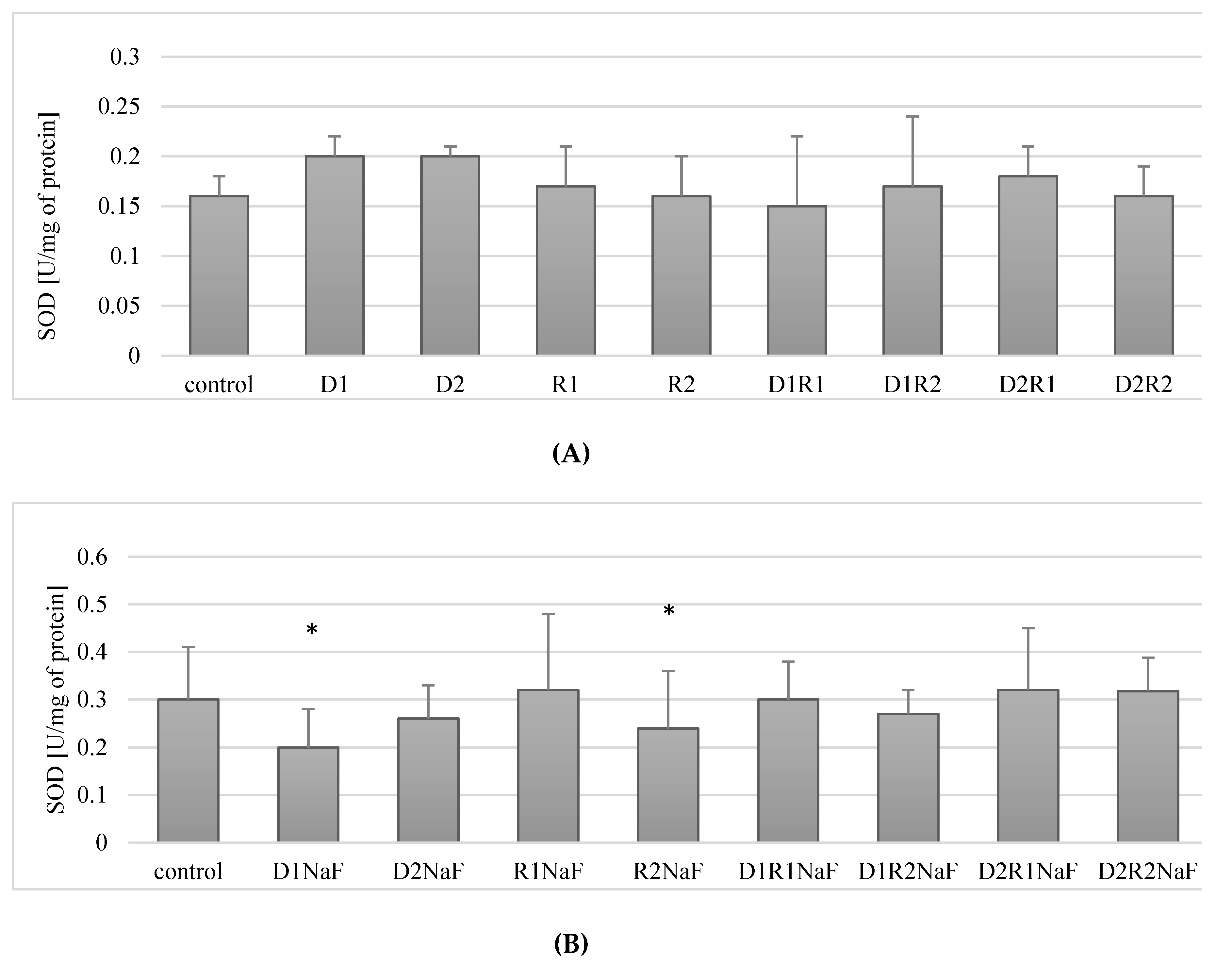
3.3. Donepezil and Rivastigmine Exerted Influence on Superoxide Dismutase Activity (SOD) in Macrophages
3.3.1. Rivastigmine and Donepezil did not Affect SOD Activity in Macrophages
3.3.2. Rivastigmine and Donepezil Used Separately Inhibited SOD Activity in Fluoride-Exposed Macrophages
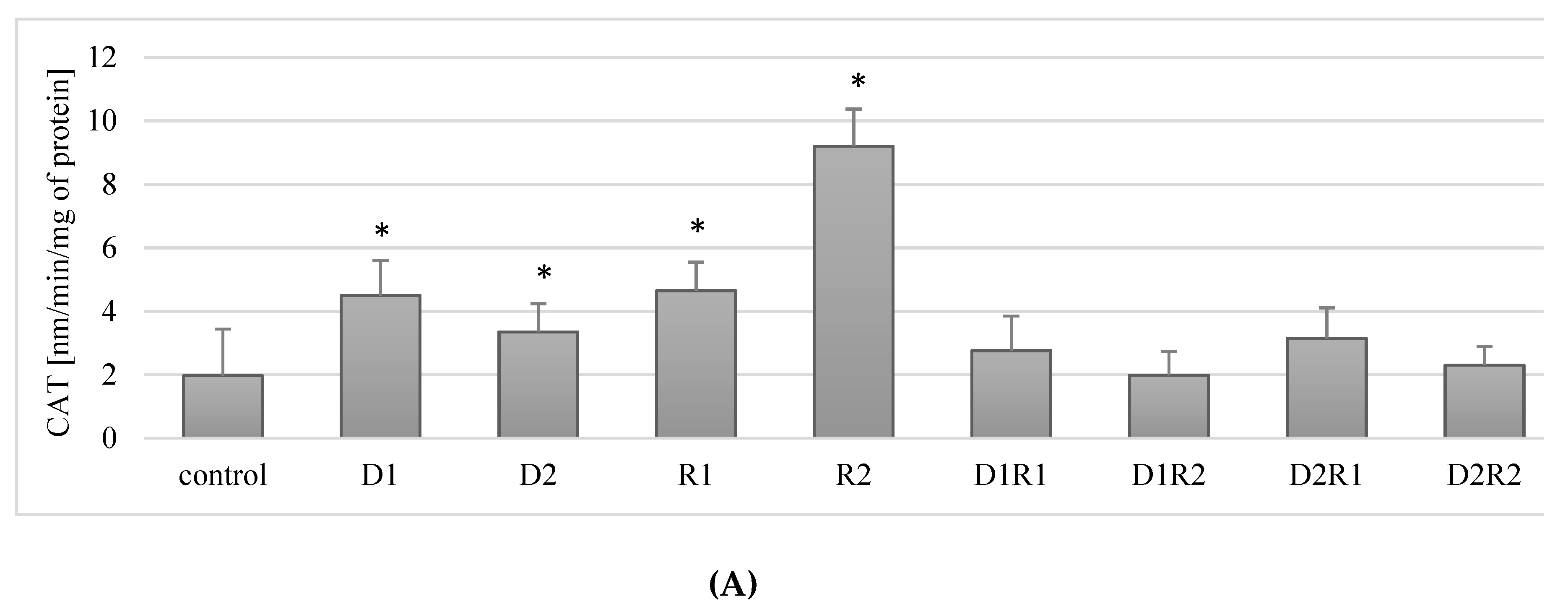
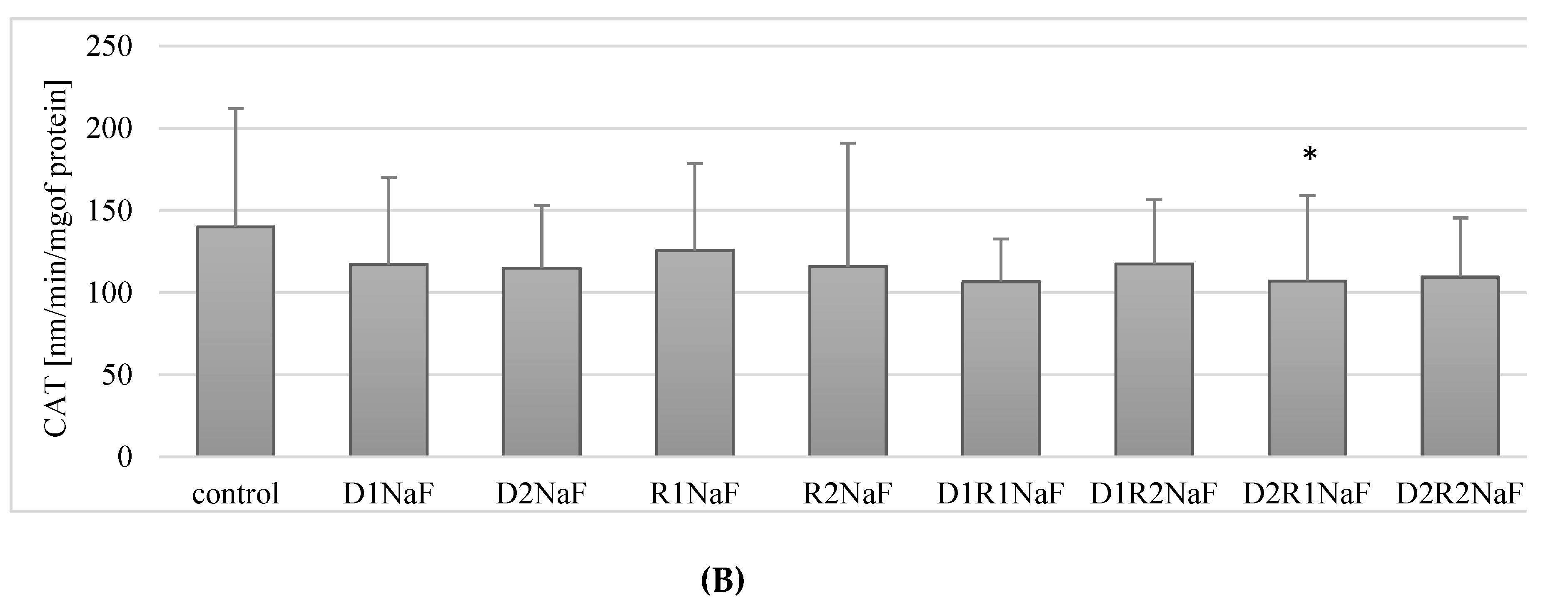
3.4. Donepezil and Rivastigmine Modulation of Catalase Activity (CAT) in Macrophages
3.4.1. Rivastigmine as well as Donepezil Used Separately Increased the Activity of CAT in Macrophages
3.4.2. Rivastigmine and Donepezil Inhibited CAT Activity in Fluoride-Exposed Macrophages
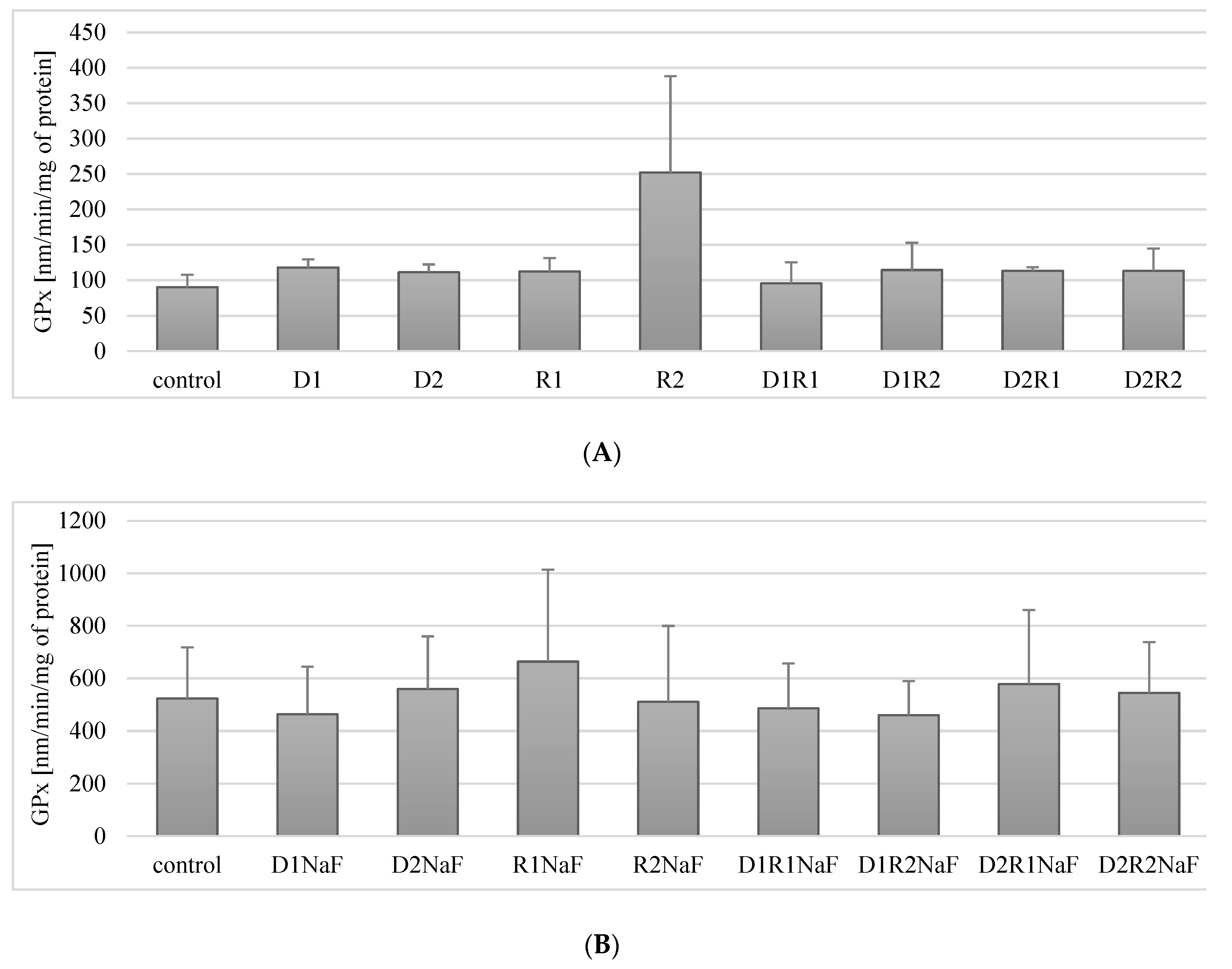
3.5. Effect of Donepezil and Rivastigmine on Glutathione Peroxidase (GPx) Activity in Macrophages
3.5.1. Rivastigmine and Donepezil Did Not Affect GPx Activity in Macrophages
3.5.2. Rivastigmine and Donepezil Did Not Affect GPx Activity in Fluoride-Exposed Macrophages
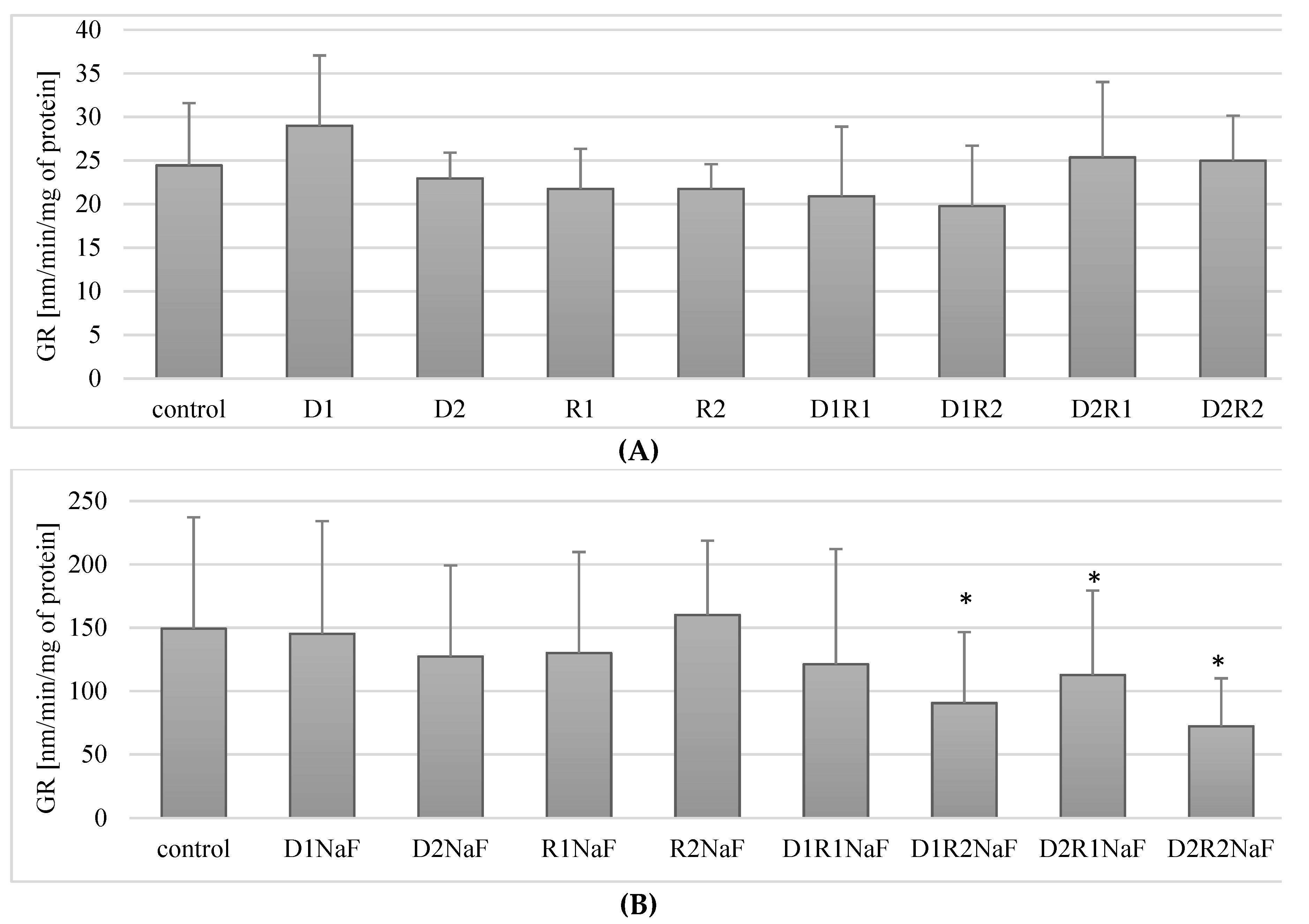
3.6. Influence of Donepezil and Rivastigmine on Glutathione Reductase (GR) Activity in Macrophages
3.6.1. Rivastigmine and Donepezil Did Not Affect GR Activity in Macrophages
3.6.2. Rivastigmine and Donepezil Decreased GR Activity in Macrophages in Fluoride-Exposed Macrophages
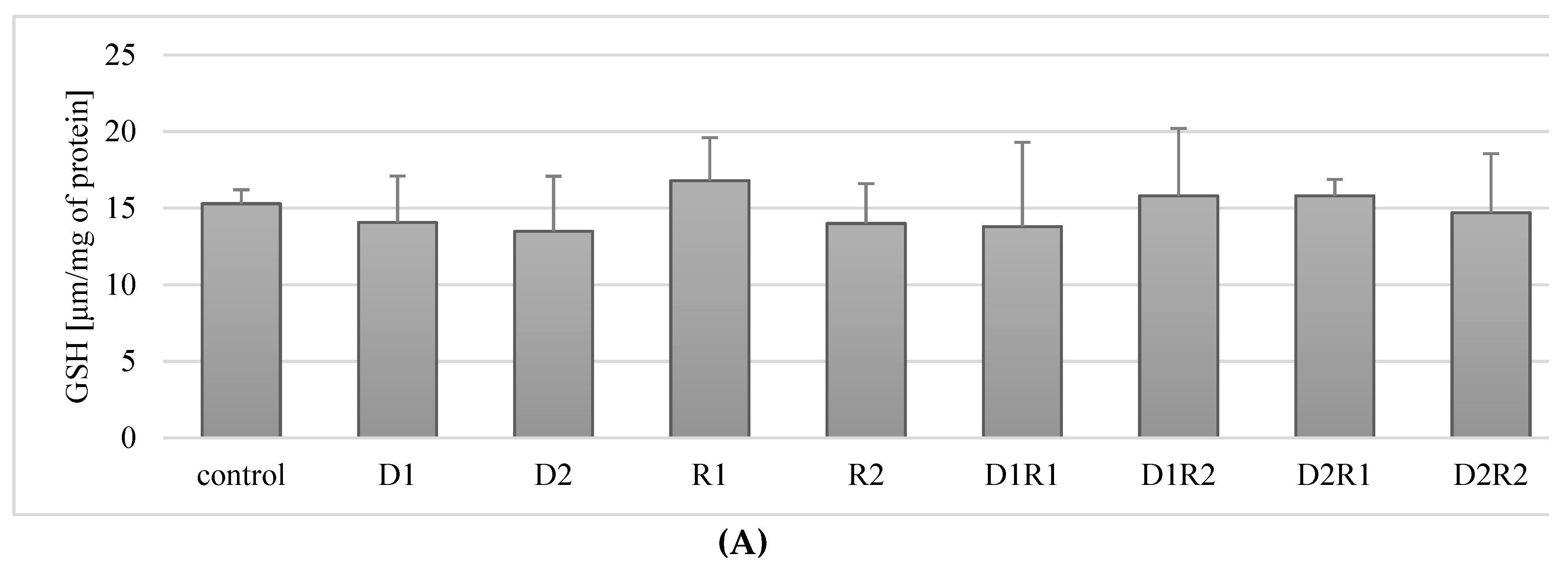
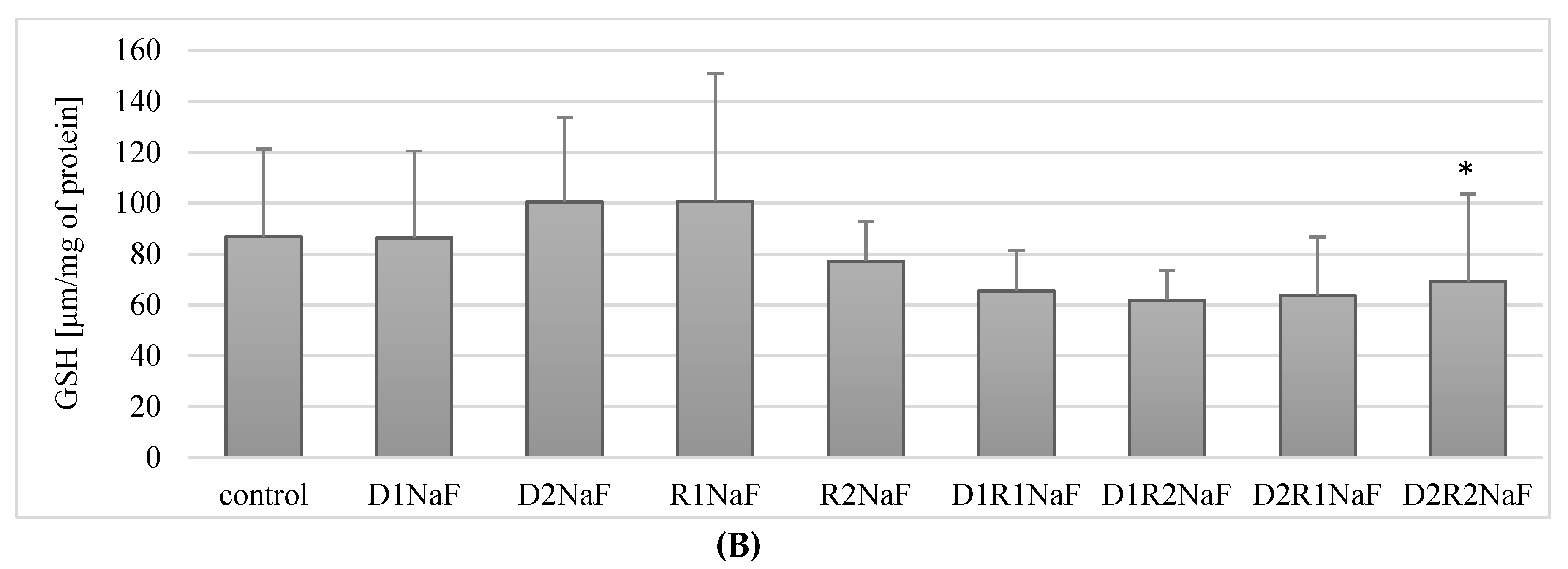
3.7. Effect of Donepezil and Rivastigmine on the Concentration of the Reduced Form of GSH Glutathione in Macrophages
3.7.1. Rivastigmine and Donepezil Had No Effect on GSH Concentration in Macrophages
3.7.2. The Combination of Rivastigmine and Donepezil at the Highest Concentrations Reduced the Concentration of the Reduced Form of Glutathione (GSH) in Fluoride-Exposed Macrophages
4. Discussion
4.1. Acetylcholinesterase Inhibitors-Induced Changes in SOD Activity
4.2. Catalase (CAT) Activity Alterations in Response to Inhibitors of Acetylcholinesterase
4.3. The Effect of Acetylcholinesterase Inhibitors on the Activity of Glutathione Peroxidase (GPx), Glutathione Reductase (GR) and the Concentration of the Reduced Form of Glutathione (GSH)
4.4. Potential Mechanism of Inhibitory Effects of Fluoride on Acetylcholinesterase Inhibitors
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Andersen, J.K. Oxidative stress in neurodegeneration: Cause or consequence? Neurodegeneration 2004, 10, S18–S25. [Google Scholar] [CrossRef] [PubMed]
- Emerit, J.; Edeas, M.; Bricaire, F. Neurodegenerative diseases and oxidative stress. Biomed. Pharmacother. 2004, 58, 39–46. [Google Scholar] [CrossRef]
- Butterfield, D.A.; Castegna, A.; Lauderback, C.M.; Drake, J. Evidence that amyloid β-peptide-induced lipid peroxidation and its sequelae in Alzheimer’s Disease brain contribute to neuronal death. Neurobiol. Aging 2002, 23, 655–664. [Google Scholar] [CrossRef]
- Pedersen, W.A.; Fu, W.; Keller, J.N.; Markesbery, W.R.; Appel, S.; Smith, R.G.; Kasarskis, E.; Mattson, M.P. Protein modification by the lipid peroxidation product 4-hydroxynonenal in the spinal cords of amyotrophic lateral sclerosis patients. Ann. Neurol. 1998, 45, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Dexter, D.T.; Carter, C.J.; Wells, F.R.; Javoy-Agid, F.; Agid, Y.; Lees, A.; Jenner, P.; Marsden, C.D. Basal lipid peroxidation in substantia nigra is increased in Parkinson’s Disease. J. Neurochem. 1989, 52, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Cho, S. Microglia and monocyte-derived macrophages in stroke. Neurotherapeutics 2016, 13, 702–718. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.C.; Chien, C.; Kuo, Y.H.; Jeyashoke, N.; Chen, J.; Dong, W.C.; Chao, L.K.; Hua, K.F. Osthole regulates inflammatory mediator expression through modulating NF-κB, mitogen-activated protein kinases, protein kinase C, and reactive oxygen species. J. Agric. Food Chem. 2010, 58, 10445–10451. [Google Scholar] [CrossRef]
- Lushchak, V.I. Free radicals, reactive oxygen species, oxidative stress and its classification. Chem. Biol. Interact. 2014, 224, 164–175. [Google Scholar] [CrossRef]
- Pietarinen-Runtti, P.; Lakari, E.; Raivio, K.O.; Kinnula, V.L. Expression of antioxidant enzymes in human inflammatory cells. Am. J. Physiol. Cell Physiol. 2000, 278, C118–C125. [Google Scholar] [CrossRef]
- Moosman, B.; Behl, C. Antioxidants as treatment for neurodegenerative diseases. Expert Opin. Investig. Drugs 2002, 11, 1407–1435. [Google Scholar] [CrossRef]
- Warner, D.S.; Sheng, H.; Batinic-Heberle, I. Oxidants antioxidants and the ischemic brain. J. Exp. Biol. 2004, 207, 3221–3231. [Google Scholar] [CrossRef] [PubMed]
- Gałecka, E.; Jacewicz, R.; Mrowicka, M.; Florkowski, A.; Gałecki, P. Enzymy antyoksydacyjne—Budowa, właściwości, funkcje. Pol. Merk. Lek. 2008, XXV, 266–268. [Google Scholar]
- Yoshioka, Y.; Kitao, T.; Kisino, T.; Yamamuro, A.; Maeda, S. Nitric oxide protects macrophages from hydrogen peroxide—induced apoptosis by inducing the formation of catalase. J. Immunol. 2006, 176, 4675–4681. [Google Scholar] [CrossRef] [PubMed]
- Novo, E.; Parola, M. Redox mechanisms in hepatic chronic wound healing and fibrogenesis. Fibrogenes. Tissue Repair. 2008, 1, 5. [Google Scholar] [CrossRef] [PubMed]
- Grossberg, G.T. Cholinesterase inhibitors for the treatment of Alzheimer’s Disease: Getting on and staying on. Curr. Ther. Res. 2003, 64, 216–235. [Google Scholar] [CrossRef]
- Pohanka, M. Inhibitors of Acetylcholinesterase and Butyrylcholinesterase Meet Immunity. Int. J. Mol. Sci. 2014, 15, 9809–9825. [Google Scholar] [CrossRef] [PubMed]
- Kurz, A.; Farlow, M.; Lefevre, G. Pharmakokinetics of a novel transdermal rivastigmine patch for the treatment of Alzheimer’s Disease. Int. J. Clin. Pract. 2009, 63, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Birks, J. Cholinesterase inhibitors for Alzheimer Disease. Cochrane Database Syst. Rev. 2006, 1, CD005593. [Google Scholar] [CrossRef]
- Farlow, M.R. Use of antidementia agents in vascular dementia: Beyond Alzheimer Disease. Mayo Clin. Proc. 2006, 81, 1350–1358. [Google Scholar] [CrossRef]
- Bai, D.L.; Tang, X.C.; He, X.C. Huperzine A, a potenitial therapeutic agent for treatment of Alzheimer’s Disease. Curr. Med. Chem. 2000, 7, 355–374. [Google Scholar] [CrossRef]
- Cheewakriengkrai, L.; Gauthier, S. A 10-year perspective on donepezil. Expert Opin. Pharmacother. 2013, 14, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Hwang, H.; Lee, H.W.; Suk, K. Microglia signaling as a target of donepezil. Neuropharmacology 2010, 58, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Lefevre, G.; Bȕche, M.; Sędek, G.; Maton, S.; Enz, A.; Lorch, U.; Sagan, C.; Appel-Dingemanse, S. Similar rivastigmine pharmacodynamics in Japanese and white healthy participants following the application of novel rivastigmine patch. J. Clin. Pharmacol. 2009, 49, 430–443. [Google Scholar] [CrossRef] [PubMed]
- Hansen, R.A.; Gartlehner, G.; Webb, A.P.; Morgan, L.C.; Moore, C.G.; Jonas, D.E. Efficiacy and safety of donepezil, galantamine and rivastigmine for the treatment of Alzheimer’s disease: A systemic review and meta-analysis. Clin. Interv. Aging 2008, 3, 211–225. [Google Scholar] [PubMed]
- Yang, Z.Z.; Zhang, Y.; Wu, K.; Wang, Z.; Qi, X. Tissue distribution and pharmacodynamics of rivastigmine after Intranasal and intravenous administration in rats. Curr. Alzheimer Res. 2012, 9, 315–325. [Google Scholar] [CrossRef]
- Darreh-Shori, T.; Hellstrom-Lindahl, E.; Flores-Flores, C.; Guan, Z.Z.; Soreq, H.; Nordberg, A. Long-lasting acetylocholinesterase splice variations in anticholinesterse-treated Alzheimer’s Disease patients. J. Neurochem. 2004, 88, 1102–1113. [Google Scholar] [CrossRef] [PubMed]
- Nizri, E.; Irony-Tur-Sinai, M.; Franesh, N.; Lavon, I.; Lavi, E.; Weinstock, M.; Brenner, T. Suppression of neuroinflammation and immunomodulation by acetylcholinesterase inhibitor rivastigmine. J. Neuroimmunol. 2008, 203, 12–22. [Google Scholar] [CrossRef]
- Gutowska, I.; Baranowska-Bosiacka, I.; Goschorska, M.; Kolasa, A.; Łukomska, A.; Jakubczyk, K.; Dec, K.; Chlubek, D. Fluoride as a factor initiating and potentiating inflammation in THP-1 differentiated monocytes/macrophages. Toxicol. In Vitro 2015, 29, 1661–1668. [Google Scholar] [CrossRef]
- Cabral Pinto, M.M.S.; Marinho-Reis, A.P.; Almeida, A.; Ordens, C.M.; Silva, M.M.V.G.; Freitas, S.; Simoes, M.R.; Moreira, P.I.; Dinis, P.A.; Diniz, L.; et al. Human predisposition to cognitive impairment and its relation with environmental exposure to potentially toxic elements. Environ. Geochem. Health 2018, 40, 1767–1784. [Google Scholar] [CrossRef]
- Dec, K.; Łukomska, A.; Maciejewska, D.; Jakubczyk, K.; Baranowska-Bosiacka, I.; Chlubek, D.; Wąsik, A.; Gutowska, I. The influence of fluorine on the disturbances of homeostasis in the central nervous system. Biol. Trace Elem. Res. 2017, 177, 224–234. [Google Scholar] [CrossRef]
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R.T. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream Antioxidant therapeutic options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.M.; Raina, R.; Dubey, N.; Verma, P.K. Effect of deltamethrin and fluoride co-exposure on the brain antioxidant status and cholinesterase activity in Wistar rats. Drug Chem. Toxicol. 2018, 41, 123–127. [Google Scholar] [CrossRef]
- Baselt, R.; Shaw, R.; McEvilly, R. Effect of Sodium Fluoride on Cholinesterase Activity in Postmortem Blood. J. Forensic Sci. 1985, 30, 1206–1209. [Google Scholar] [CrossRef] [PubMed]
- Moss, D.E.; Perez, R.G.; Kobayashi, H. Cholinesterase Inhibitor Therapy in Alzheimer’s: The limits and tolerability of Irreversible CNS-selective Acetylcholinesterase inhibition in Primates. J. Alzhiemers Dis. 2017, 55, 1285–1294. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Nakano, T.; Moss, D.E.; Tadahiko, S. Effects of a central acetylcholinesterase, methanesulfonyl fluoride on the cerebral cholinergic system and behavior in mice: Comparison with an organophosphate DDVP. J. Heath Sci. 1999, 45, 191–202. [Google Scholar] [CrossRef]
- Borlongan, C.V.; Sumaya, I.C.; Moss, D.E. Methanesulfonyl fluoride, an acetylcholinesterase inhibitor attenuates simple learning and memory deficits in ischemic rats. Brain Res. 2005, 1038, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Mildner, A.; Schmidt, H.; Nitsche, M.; Merkler, D.; Hanisch, U.K.; Mack, M.; Heikenwalder, M.; Brück, W.; Priller, J.; Prinz, M. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nat. Neurosci. 2007, 10, 1544–1553. [Google Scholar] [CrossRef]
- Greter, M.; Merad, M. Regulation of microglia development and homeostasis. Glia 2013, 61, 121–127. [Google Scholar] [CrossRef]
- Streit, W.J.; Miller, K.R.; Lopes, K.O.; Njie, E. Microglial degeneration in the aging brain—Bad news for neurons? Front. Biosci. 2008, 13, 3423–3438. [Google Scholar] [CrossRef]
- Qin, Z. The use of THP-1 cells as a model for mimicking the function and regulation of monocytes and macrophages in the vasculature. Atherosclerosis 2012, 221, 2–11. [Google Scholar] [CrossRef]
- Olszowski, T.; Baranowska-Bosiacka, I.; Gutowska, I.; Piotrowska, K.; Korbecki, J.; Kurzawski, M.; Chlubek, D. The effect of cadmium on COX-1 and COX-2 gene, protein expression and enzymatic activity in THP-1 macrophages. Biol. Trace Elem. Res. 2015, 165, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Ross, A.C. Retinoic acid regulates cell cycle progression and cell differentiation in human monocytic THP-1 cells. Exp. Cell Res. 2004, 297, 68–81. [Google Scholar] [CrossRef] [PubMed]
- Auwerx, J. The human leukemia cell line, THP1: A multifaceted model for study monocyte-macrophage differentiation. Experimentia 1991, 47, 22–31. [Google Scholar] [CrossRef]
- Dooley, M.; Lamb, H.M. Donepezil. A review of its use in Alzheimer’s Disease. Drugs Aging 2000, 16, 199–226. [Google Scholar] [CrossRef] [PubMed]
- Sadowsky, C.H.; Micca, J.L.; Grossberg, G.T.; Velting, D.M. Rivastigmine from capsules to patch: Therapeutic advances in the management of Alzheimer Disease and Parkinson Disease dementia. Prim. Care Companion CNS Disord. 2014, 16, 10. [Google Scholar] [CrossRef] [PubMed]
- Ota, T.; Shinotoh, H.; Fukushi, K.; Kikuchi, T.; Sato, K.; Tanaka, N.; Shimada, H.; Hirano, S.; Miyoshi, M.; Arai, H.; et al. Estimation of plasma IC50 of donepezil for cerebral acetylocholinesteraze inhibition in patients with Alzheimer Disease using positron emission tomography. Clin. Neuropharmacol. 2010, 33, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Chou, M.C.; Chen, C.H.; Liu, C.K.; Chen, S.H.; Wu, S.J.; Yang, Y.H. Concentrations of rivastygmine and NAP 226-90 and the cognitive response in Taiwanese Alzheimer’s Disease Patients. J. Alzheimers Dis. 2012, 31, 857–864. [Google Scholar] [CrossRef]
- Tiseo, P.J.; Rogers, S.L.; Friedhoff, L.T. Pharmakokinetic and pharmacodynamic profile of donepezil HCl following evening administration. Br. J. Clin. Pharmacol. 1998, 46, 13–18. [Google Scholar] [CrossRef]
- Schulz, M.; Schimoldt, A. Therapeutic and toxic blood concentrations of more than 800 drugs and other xenobiotics. Pharmacie 2003, 58, 447–474. [Google Scholar]
- Kalyanalakshmi, P.; Vijayabhaskar, M.; Dhananjaya Naidu, M. Lipid peroxidation and antioxidant enzyme status of adult males with skeletal fluorosis in Andhra Pradesh, India. Fluoride 2007, 40, 42–45. [Google Scholar]
- De la Fuente, B.; Vázquez, M.; Rocha, R.A.; Devesa, V.; Vélez, D. Effect of sodium fluoride on immune response in murine macrophages. Toxicol. In Vitro 2016, 34, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Gutowska, I.; Baranowska-Bosiacka, I.; Siennicka, A.; Baśkiewicz, M.; Machaliński, B.; Stachowska, E.; Chlubek, D. Fluoride and generation of proinflammatory factors in human macrophages. Fluoride 2011, 44, 125–134. [Google Scholar]
- Gutowska, I.; Baranowska-Bosiacka, I.; Siennicka, A.; Telesiński, A.; Stańczyk-Dunaj, M.; Wesołowska, T.; Gąssowska, M.; Kłos, P.; Zakrzewska, H.; Machaliński, B.; et al. Activation of phospholipase A2 by low levels of fluoride in THP1 macrophages via altered Ca2+ and cAMP concentration. PLEFA 2012, 86, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Aam, B.B.; Fonnum, F. ROS scavenging effects of organic extract of diesel exhaust particles on human neutrophil granulocytes and rat alveolar macrophages. Toxicology 2007, 230, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, S.S.; Eligini, S.; Barmbilla, M.; Tremoli, E.; Colli, S. Reactive oxygen species mediate cyclooxygenase-2 induction during monocyte to macrophage differentiation: Critical role of NADPH oxidase. Cardiovasc. Res. 2003, 60, 187–197. [Google Scholar] [CrossRef]
- Bartosz, G. Use of spectroscopic probes for detection of reactive oxygen species. Clin. Chim. Acta 2006, 368, 53–76. [Google Scholar] [CrossRef] [PubMed]
- Eligini, S.; Barbieri, S.S.; Cavalca, V.; Camera, M.; Brambilla, M.; De Franceschi, M.; Tremoli, E.; Colli, S. Diversity and similarity in signaling events leading to rapid Cox-2 induction by tumor necrosis factor-alpha and phorbol ester in human endothelial cells. Cardiovasc. Res. 2005, 65, 683–693. [Google Scholar] [CrossRef]
- Baranowska-Bosiacka, I.; Gutowska, I.; Marchetti, C.; Rutkowska, M.; Marchlewicz, M.; Kolasa, A.; Prokopowicz, A.; Wiernicki, I.; Piotrowska, K.; Baśkiewicz, M.; et al. Altered energy status of primary cerebellar granule neuronal cultures from rats exposed to lead in the pre- and neonatal period. Toxicology 2011, 280, 24–32. [Google Scholar] [CrossRef]
- Bradford, H.F.; Richards, C.D. Specific release of endogenous glutamate from piriform cortex stimulated in vitro. Brain Res. 1976, 105, 168–172. [Google Scholar] [CrossRef]
- Niedzielska, E.; Smaga, I.; Gawlik, M.; Moniczewski, A.; Stankowicz, P.; Pera, J.; Filip, M. Oxidative stress in neurodegenerative diseases. Mol. Neurobiol. 2016, 53, 4094–4125. [Google Scholar] [CrossRef]
- Richardson, J.S. Free radicals in the Genesis of Alzheimer’s Disease. Ann. N. Y. Acad. Sci. 1993, 695, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Marklund, S.L.; Adolfsson, R.; Gottfries, C.G.; Winblad, B. Superoxide dismutase isoenzymes in normal brains and in brains from patients with dementia of Alzheimer type. J. Neurol. Sci. 1985, 67, 319–325. [Google Scholar] [CrossRef]
- Gsell, W.; Conrad, R.; Hickethier, M.; Sofic, E.; Frölich, L.; Wichart, I.; Jellinger, K.; Moll, G.; Ransmayr, G.; Beckmann, H.; et al. Decreased catalase activity but unchanged superoxide dismutase activity in brains of patients with dementia of Alzheimer type. J. Neurochem. 1995, 64, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Massaad, C.A. Neuronal and vascular oxidative stress in Alzheimer’s Disease. Curr. Neuropharmacol. 2011, 9, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Marcus, D.L.; Strafaci, J.A.; Freedman, M.L. Differential neuronal expression of manganese superoxide dismutase in Alzheimer’s disease. Med. Sci. Monit. 2006, 12, BR8-14. [Google Scholar]
- Murakami, K.; Murata, N.; Noda, Y.; Tahara, S.; Kaneko, T.; Kinoshita, N.; Hatsuta, H.; Murayama, S.; Barnham, K.J.; Irie, K.; et al. SOD1 (Copper/Zinc Superoxide Dismutase) Deficiency Drives Amyloid β Protein Oligomerization and Memory Loss in Mouse Model of Alzheimer Disease. J. Biol. Chem. 2011, 30, 44557–44568. [Google Scholar] [CrossRef]
- Li, X.; Zhao, X.; Mao, X.; Liu, Z.; Guo, L.; Bi, K.; Jia, Y. Schisantherin A recovers Aβ-induced neurodegeneration with cognitive decline in mice. Physiol. Behav. 2014, 132, 10–16. [Google Scholar] [CrossRef]
- Meunier, J.; Ieni, J.; Maurice, T. The anti-amnesic and neuroprotective effects of donepezil against amyloid β25-35 peptide induced toxicity in mice involve an interaction with σ1 receptor. Br. J. Pharmacol. 2006, 149, 998–1012. [Google Scholar] [CrossRef]
- Vani, M.L.; Reddy, K.P. Effects of fluoride accumulation on some enzymes of brain and gastrocnemius muscle in mice. Fluoride 2000, 33, 17–26. [Google Scholar]
- Patel, D.; Chinoy, N.J. Influence of fluoride on biological free radical reactions in ovary of mice and its reversal. Fluoride 1998, 31, S27. [Google Scholar]
- Sun, G.; Qiu, L.; Ding, G.; Qian, C.; Zheng, Q. Effects of β-carotene and SOD on lipid peroxidation induced by fluoride: An experimental study. Fluoride 1998, 31, S29. [Google Scholar]
- Zhang, M.; Wang, A.; He, W.; He, P.; Xu, B.; Xia, T.; Chen, X.; Yang, K. Effects of fluoride on the expression of NCAM, oxidative stress, and apoptosis in primary cultured hippocampal neurons. Toxicology 2007, 17, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Klugman, A.; Naughton, D.P.; Isaac, M.; Shah, I.; Petroczi, A.; Tabet, N. Antioxidant enzymatic activities in Alzheimer’s disease: The relationship to acetylcholinesterase inhibitors. J. Alzheimers Dis. 2012, 30, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.Y.; Tang, X.C. Huperzine B, a novel acetylcholinesterase inhibitor, attenuates hydrogen peroxide induced injury in PC12 cells. Neurosci. Lett. 2000, 29, 41–44. [Google Scholar] [CrossRef]
- Goverdhan, P.; Sravanthi, A.; Mamatha, T. Neuroprotective effects of meloxicam and selegiline in scopolamine-induced cognitive impairment and oxidative stress. Int. J. Alzheimers Dis. 2012, 2012, 974013. [Google Scholar] [CrossRef] [PubMed]
- Goschorska, M.; Gutowska, I.; Olszewska, M.; Baranowska-Bosiacka, I.; Olszowski, T.; Rać, M.; Chlubek, D. Effect of sodium fluoride on catalase activity in THP-1 macrophages. Fluoride 2015, 48, 274–282. [Google Scholar]
- Barbier, O.; Arreola-Mendoza, L.; DelRazo, L.M. Molecular mechanisms of fluoride toxicity. Chem. Biol. Int. 2010, 188, 319–333. [Google Scholar] [CrossRef]
- Morales-González, J.A.; Gutiérrez-Salinas, J.; García-Ortiz, L.; Del Carmen Chima-Galán, M.; Madrigal-Santillán, E.; Esquivel-Soto, J.; Esquivel-Chirino, C.; González-Rubio, M.G. Effect of sodium fluoride ingestion on malondialdehyde concentration and the activity of antioxidant enzymes in rat erythrocytes. Int. J. Mol. Sci. 2010, 11, 2443–2452. [Google Scholar] [CrossRef]
- Yamaguti, P.M.; Simões, A.; Ganzerla, E.; Souza, D.N.; Nogueira, F.N.; Nicolau, J. Effects of single exposure of sodium fluoride on lipid peroxidation and antioxidant enzymes in salivary glands of rats. Oxid. Med. Cell. Longev. 2013, 674593. [Google Scholar] [CrossRef]
- Ravula, S.; Harinarayan, C.V.; Prasad, U.V.; Ramalakshmi, T.; Rupungudi, A.; Madrol, V. Effect of fluoride on reactive oxygen species and bone metabolism in postmenopausal women. Fluoride 2012, 45, 108–115. [Google Scholar]
- Gubandru, M.; Margina, D.; Tsitsimpikou, C.; Goutzourelas, N.; Tsarouhas, K.; Ilie, M.; Tsatsakis, A.M.; Kouretas, D. Alzheimer’s disease treated patients showed different patterns for oxidative stress and inflammation markers. Food. Chem. Toxicol. 2013, 61, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Liu, L.; Zhu, X.; Wu, W.; Wang, Y. Hesperidin alleviates cognitive impairment, mitochondrial dysfunction and oxidative stress in a mouse model of Alzheimer’s disease. Cell. Mol. Neurobiol. 2014, 34, 1209–1221. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Prakash, A.; Pahwa, D. Galantamine potentiates the protective effect of rofecoxib and caffeic acid against intrahippocampal kainic acid-induced cognitive dysfunction in rat. Brain Res. Bull. 2011, 85, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Khurana, S.; Jain, S.; Mediratta, P.K.; Banerjee, B.D.; Sharma, K.K. Protective role of curcumin on colchicine-induced cognitive dysfunction and oxidative stress in rats. Hum. Exp. Toxicol. 2012, 31, 686–697. [Google Scholar] [CrossRef] [PubMed]
- Shivarajashankara, Y.M.; Shivashankara, A.R.; Bhat, P.G.; Rao, S.H. Effect of fluoride intoxication on lipid peroxidation and antioxidant systems in rats. Fluoride 2001, 34, 108–113. [Google Scholar]
- Inkielewicz, I.; Krechniak, J. Fluoride effects on glutathione peroxidase and lipid peroxidation in rats. Fluoride 2004, 37, 7–12. [Google Scholar]
- Hensley, K.; Carney, J.M.; Mattson, M.P.; Aksenova, M.; Harris, M.; Wu, J.F.; Floyd, R.A.; Butterfield, D.A. A model for beta-amyloid aggregation and neurotoxicity based on free radical generation by the peptide: Relevance to Alzheimer disease. PNAS 1994, 91, 3270–3274. [Google Scholar] [CrossRef]
- Chen, J.; Shan, K.R.; Long, Y.G.; Wang, Y.N.; Nordberg, A.; Guan, Z.Z. Selective decreases of nicotinic acetylcholine receptors in PC12 cells exposed to fluoride. Toxicology 2003, 183, 235–242. [Google Scholar] [CrossRef]
- Jones, S.; Sudweeks, S.; Yakel, J.L. Nicotinic receptors in the brain: Correlating physiology with function. Trends. Neurosci. 1999, 22, 555–561. [Google Scholar] [CrossRef]
- Nordberg, A. Human nicotinic receptors*/their role in aging and dementia. Neurochem. Int. 1994, 25, 93–97. [Google Scholar] [CrossRef]
- Paterson, D.; Nordberg, A. Neuronal nicotinic receptors in the human brain. Prog. Neurobiol. 2000, 61, 75–111. [Google Scholar] [CrossRef]
- Svensson, A.L.; Nordberg, A. Tacrine and donepezil attenuate the neurotoxic effect of Aβ (25-35) in rat PC12 cells. Neuroreport 1998, 9, 1519–1522. [Google Scholar] [CrossRef] [PubMed]
- Guan, Z.Z.; Zhang, X.; Ravid, R.; Nordberg, A. Decreased protein levels of nicotinic receptor subunits in the hippocampus and temporal cortex of patients with Alzheimer’s disease. J. Neurochem. 2000, 74, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Goschorska, M.; Baranowska-Bosiacka, I.; Gutowska, I.; Piotrowska, K.; Metryka, E.; Safranow, K.; Chlubek, D. Effect of acetylcholinesterase inhibitors donepezil and rivastigmine on the activity and expression of cyclooxygenases in a model of the inflammatory action of fluoride on macrophages obtained from the THP-1 monocytes. Toxicology 2018, 406–407, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Anderson, N.G.; Kilgour, E.; Sturgill, T.W. Activation of mitogen-activated protein kinase in BC3H1 myocytes by fluoroaluminate. J. Biol. Chem. 1991, 266, 10131–10135. [Google Scholar] [PubMed]
- Chabre, M. Aluminofluoride and beryllofluoride complexes: New phosphate analogs in enzymology. Trends Biochem. Sci. 1990, 15, 6–10. [Google Scholar] [CrossRef]
- Refsnes, M.; Skuland, T.; Schwarze, P.E.; Øvrevik, J.; Låg, M. Fluoride-induced IL-8 release in human epithelial lung cells: Relationship to EGF-receptor-, SRC- and MAP-kinase activation. Toxicol. Appl. Pharmacol. 2008, 15, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Thrane, E.V.; Refsnes, M.; Thoresen, G.H.; Lag, M.; Schwarze, P.E. Fluoride-induced apoptosis in epithelial lung cells involves activation of MAP kinases p38 and possibly JNK. Toxicol. Sci. 2001, 61, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, M.; Danon, A.; Rimon, G. Prostaglandin H synthase: Protein synthesis-independent regulation in bovine aortic endothelial cells. Am. J. Physiol. Cell Physiol. 1997, 273, C1749–C1755. [Google Scholar] [CrossRef]
- Griendling, K.K.; Sorescu, D.; Lassegue, B.; Ushio-Fukai, M. Modulation of protein kinase activity and gene expression by reactive oxygen species and their role in physiology and pathology. Arterioscler. Thromb. Vasc. Biol. 2018, 2175–2183. [Google Scholar]
- Blaylock, R.L. Excitotoxicity: A possible central mechanism in fluoride toxicity. Fluoride 2004, 37, 301–314. [Google Scholar]
- Lee, J.; Han, Y.E.; Favorov, O.; Tommerdahl, M.; Whitsel, B.; Lee, C.J. Fluoride induces a volume reduction in CA1 hippocampal slices via MAP kinase pathway through volume regulation anion channels. Exp. Neurobiol. 2016, 25, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Deora, A.A.; Win, T.; Vanhaesebroeck, B.; Lander, H.M. A redox-triggered ras-effector interaction: Recruitment of phosphatidylinositol 39-kinase to Ras by redox stress. J. Biol. Chem. 1998, 273, 29923–29928. [Google Scholar] [CrossRef] [PubMed]
- Takada-Takatori, Y.; Kume, T.; Ohgi, Y.; Fujii, T.; Niidome, T.; Sugimoto, H.; Akaike, A. Mechanisms of alpha7-nicotinic receptor up-regulation and sensitization to donepezil induced by chronic donepezil treatment. Eur. J. Pharmacol. 2008, 590, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Takada-Takatori, Y.; Kume, T.; Ohgi, Y.; Izumi, Y.; Niidome, T.; Fujii, T.; Sugimoto, H.; Akaike, A. Mechanism of neuroprotection by donepezil pretreatment in rat cortical neurons chronically treated with donepezil. J. Neurosci. Res. 2008, 86, 3575–3583. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, H.Y.; Tang, X.C. Cholinergic deficiency involved in vascular dementia: Possible mechanism and strategy for treatment. Acta Pharmacol. Sin. 2009, 30, 879–888. [Google Scholar] [CrossRef]
| Symbol | Drug | Concentration (ng/mL) |
|---|---|---|
| D1 | donepezil | 20 |
| D2 | donepezil | 100 |
| R1 | rivastigmine | 5 |
| R2 | rivastigmine | 25 |
| Group | Procedure | Donepezil Concentration | Rivastigmine Concentration |
|---|---|---|---|
| Control | macrophages cultured with DMSO | - | - |
| D1 | macrophages cultured with donepezil | 20 ng/mL | - |
| D2 | macrophages cultured with donepezil | 100 ng/mL | - |
| R1 | macrophages cultured with rivastigmine | - | 5 ng/mL |
| R2 | macrophages cultured with rivastigmine | - | 25 ng/mL |
| D1R1 | macrophages cultured with both medicaments | 20 ng/mL | 5 ng/mL |
| D1R2 | macrophages cultured with both medicaments | 20 ng/mL | 25 ng/mL |
| D2R1 | macrophages cultured with both medicaments | 100 ng/mL | 5 ng/mL |
| D2R2 | macrophages cultured with both medicaments | 100 ng/mL | 25 ng/mL |
| Group | Procedure | NaF Concentration | Donepezil Concentration | Rivastigmine Concentration |
|---|---|---|---|---|
| Control | macrophages cultured with NaF and DMSO | 3 µM | - | - |
| D1NaF | macrophages cultured with donepezil and NaF | 3 µM | 20 ng/mL | - |
| D2NaF | macrophages cultured with donepezil and NaF | 3 µM | 100 ng/mL | - |
| R1NaF | macrophages cultured with rivastigmine and NaF | 3 µM | - | 5 ng/mL |
| R2NaF | macrophages cultured with rivastigmine and NaF | 3 µM | - | 25 ng/mL |
| D1R1NaF | macrophages cultured with donepezil, rivastigmine and NaF | 3 µM | 20 ng/mL | 5 ng/mL |
| D1R2NaF | macrophages cultured with donepezil, rivastigmine and NaF | 3 µM | 20 ng/mL | 25 ng/mL |
| D2R1NaF | macrophages cultured with donepezil, rivastigmine and NaF | 3 µM | 100 ng/mL | 5 ng/mL |
| D2R2NaF | macrophages cultured with donepezil, rivastigmine and NaF | 3 µM | 100 ng/mL | 25 ng/mL |
| Experimental Conditions | (D) and/or (R) | (D) and/or (R) + NaF | ||
|---|---|---|---|---|
| DCF Fluorescence Intensity # | % Decrease/Increase vs. Control | DCF Fluorescence Intensity # | % Decrease/Increase vs. Control | |
| C (n = 6) | 39.87 ± 1.51 | 46.58 ± 1.78 | ||
| D1 (n = 6) | 38.25 ± 1.17 | −4.06 | 52.26 ± 1.96 | 12.19 * |
| D2 (n = 6) | 38.76 ± 2.76 | −2.78 | 52.45 ± 2.02 | 12.60 * |
| R1(n = 6) | 39.95 ± 3.43 | −0.20 | 54.67 ± 3.67 | 17.36 * |
| R2 (n = 6) | 41.13 ± 1.15 | 3.16 | 55.32 ± 1.43 | 18.76 * |
| D1R1(n = 6) | 40.01 ± 1.24 | 0.35 | 51.43 ± 2.11 | 10.41 * |
| D1R2 (n = 6) | 40.02 ± 1.14 | 0.38 | 50.55 ± 1.32 | 9.93 * |
| D2R1(n = 6) | 39.55 ± 1.22 | −0.80 | 52.21 ± 2.67 | 12.08 * |
| D2R2 (n = 6) | 39.85 ± 2.54 | −0.05 | 50.02 ± 1.54 | 7.39 * |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goschorska, M.; Gutowska, I.; Baranowska-Bosiacka, I.; Piotrowska, K.; Metryka, E.; Safranow, K.; Chlubek, D. Influence of Acetylcholinesterase Inhibitors Used in Alzheimer’s Disease Treatment on the Activity of Antioxidant Enzymes and the Concentration of Glutathione in THP-1 Macrophages under Fluoride-Induced Oxidative Stress. Int. J. Environ. Res. Public Health 2019, 16, 10. https://doi.org/10.3390/ijerph16010010
Goschorska M, Gutowska I, Baranowska-Bosiacka I, Piotrowska K, Metryka E, Safranow K, Chlubek D. Influence of Acetylcholinesterase Inhibitors Used in Alzheimer’s Disease Treatment on the Activity of Antioxidant Enzymes and the Concentration of Glutathione in THP-1 Macrophages under Fluoride-Induced Oxidative Stress. International Journal of Environmental Research and Public Health. 2019; 16(1):10. https://doi.org/10.3390/ijerph16010010
Chicago/Turabian StyleGoschorska, Marta, Izabela Gutowska, Irena Baranowska-Bosiacka, Katarzyna Piotrowska, Emilia Metryka, Krzysztof Safranow, and Dariusz Chlubek. 2019. "Influence of Acetylcholinesterase Inhibitors Used in Alzheimer’s Disease Treatment on the Activity of Antioxidant Enzymes and the Concentration of Glutathione in THP-1 Macrophages under Fluoride-Induced Oxidative Stress" International Journal of Environmental Research and Public Health 16, no. 1: 10. https://doi.org/10.3390/ijerph16010010
APA StyleGoschorska, M., Gutowska, I., Baranowska-Bosiacka, I., Piotrowska, K., Metryka, E., Safranow, K., & Chlubek, D. (2019). Influence of Acetylcholinesterase Inhibitors Used in Alzheimer’s Disease Treatment on the Activity of Antioxidant Enzymes and the Concentration of Glutathione in THP-1 Macrophages under Fluoride-Induced Oxidative Stress. International Journal of Environmental Research and Public Health, 16(1), 10. https://doi.org/10.3390/ijerph16010010





