The Effectiveness and Cost-Effectiveness of Screening for and Vaccination Against Hepatitis B Virus among Migrants in the EU/EEA: A Systematic Review
Abstract
1. Introduction
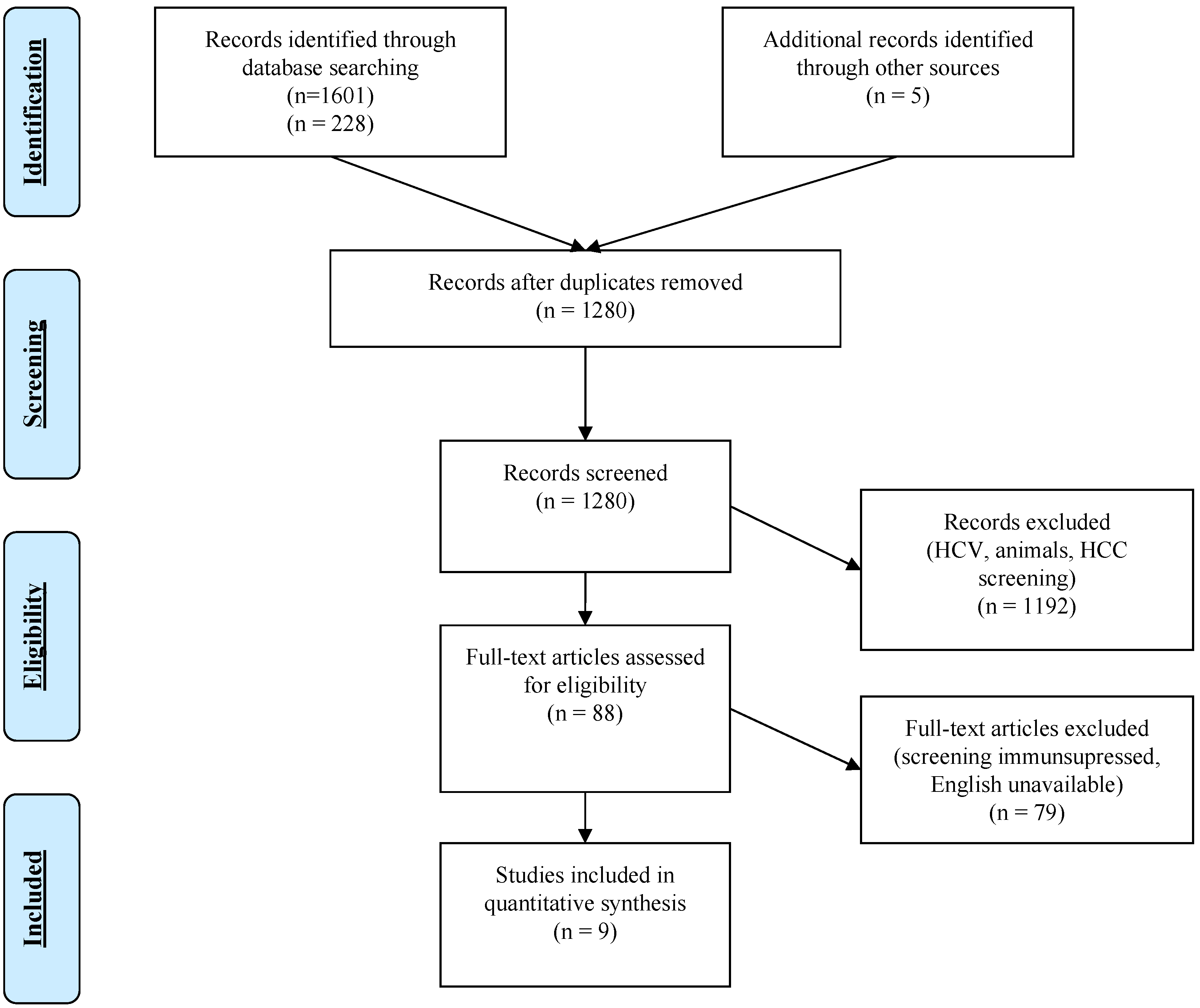
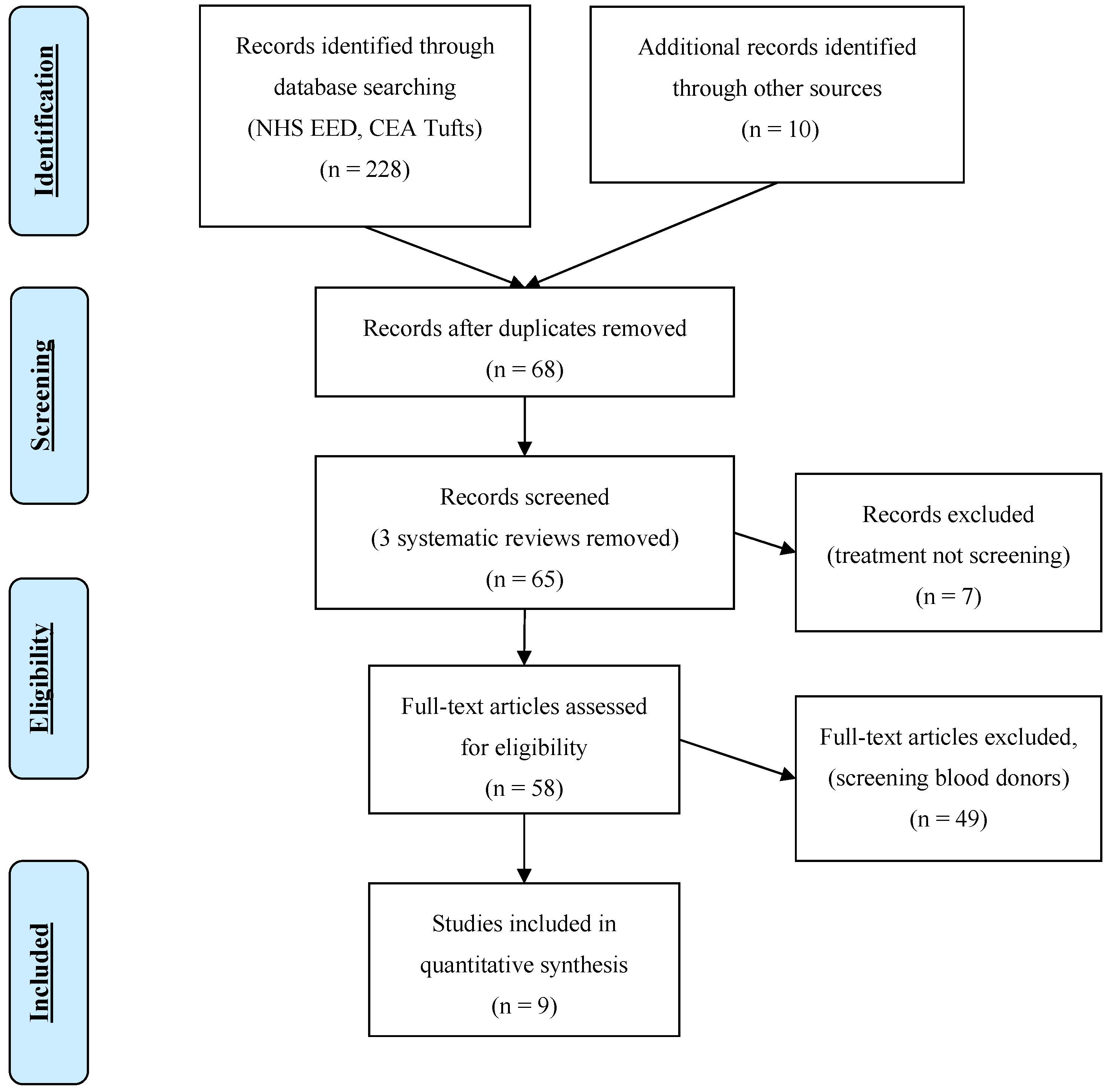
2. Materials and Methods
- Is screening for HBV infection (and subsequent management) associated with decreased morbidity and mortality in migrant populations?
- What is the effectiveness of HBV vaccination programs in migrant populations?
- What is the cost-effectiveness of screening and vaccination programs for HBV?
3. Results
3.1. Effectiveness of Screening for CHB
3.2. Vaccination Against HBV
3.3. Cost-Effectiveness of Screening and Subsequent Management for Hepatitis B
3.4. Cost-Effectiveness of Vaccination
4. Discussion
4.1. Implementation Considerations
4.2. Strengths and Weaknesses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix 1. Search Terms
| 1 exp Hepatitis B/(49954) |
| 2 (CHB or HBV or HepB).mp. (32246) |
| 3 ((hep or hepatitis) adj3 B).mp. (82681) |
| 4 hbsag.tw. (16191) |
| 5 (hbs adj2 ag).tw. (720) |
| 6 hb-s-ag.tw. (24) |
| 7 ((serum or type b) adj2 hepatitis).tw. (3299) |
| 8 or/1–7 (86774) |
| 9 exp Mass Screening/(108496) |
| 10 (screened or screening? or tested or testing or tests).tw. (1733475) |
| 11 Early Diagnosis/(19328) |
| 12 ((case? or early) adj2 (detected or detection? or diagnos$ or discover$)).tw. (153478) |
| 13 exp Population Surveillance/(56663) |
| 14 (disease? adj2 surveillance).tw. (4191) |
| 15 Contact Tracing/ (3561) |
| 16 contact tracing.tw. (1176) |
| 17 or/9–16 (1940435) |
| 18 meta analysis.mp,pt. (96656) |
| 19 review.pt. (2060002) |
| 20 search$.tw. (266555) |
| 21 guideline.pt. (15761) |
| 22 guideline/(15761) |
| 23 guidelines as topic/(34049) |
| 24 practice guideline.pt. (21200) |
| 25 practice guideline/(21200) |
| 26 practice guidelines as topic/(91709) |
| 27 (CPG or CPGs or guidance or guideline? or recommend$ or standard?).ti. (147079) |
| 28 exp clinical pathway/(5268) |
| 29 exp clinical protocol/(139279) |
| 30 ((care or clinical) adj2 pathway?).tw. (5122) |
| 31 or/18-30 (2570832) |
| 32 8 and 17 and 31 (2192) |
| 33 animals/ not (humans/ and animals/) (4214239) |
| 34 32 not 33 (2176) |
| 35 34 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$).ed. (675) |
| 36 remove duplicates from 35 (reviews and guidelines) (652) |
| 37 exp “costs and cost analysis”/ (197842) |
| 38 cost$.mp. (467557) |
| 39 cost effective$.tw. (83015) |
| 40 cost benefit analys$.mp. (67281) |
| 41 health care costs.mp. (37134) |
| 42 or/37–41 (476890) |
| 43 8 and 17 and 42 (888) |
| 44 animals/not (humans/and animals/)(4214239) |
| 45 43 not 44 (883) |
| 46 45 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$).ed. (266) |
| 47 remove duplicates from 46 (costing) (254) |
| 1 exp hepatitis B/(79653) |
| 2 (CHB or HBV or HepB).mp. (53611) |
| 3 ((hep or hepatitis) adj3 B).mp. (132717) |
| 4 hbsag.tw. (24116) |
| 5 (hbs adj2 ag). tw. (1126) |
| 6 hb-s-ag.tw (626) |
| 7 ((serum or type b) adj2 hepatitis).tw. (4201) |
| 8 or/1–7 (140818) |
| 9 exp mass screening/(182894) |
| 10 (screened or screening? or tested or testing or tests).tw. (2429779) |
| 11 anonymous testing/(223) |
| 12 early diagnosis/(83109) |
| 13 ((case? or early) adj2 (detected or detection? or diagnos$ or discover$)).tw. (235744) |
| 14 exp health survey/(184232) |
| 15 (disease? adj2 surveillance).tw. (5252) |
| 16 contact examination/(2867) |
| 17 contact tracing.tw. (1512) |
| 18 or/9-17 (2853474) |
| 19 meta analysis.mp,pt. (163362) |
| 20 review.pt. (2163167) |
| 21 search$.tw. (371891) |
| 22 guideline.pt. (0) |
| 23 guideline/(144) |
| 24 guidelines as topic/(229891) |
| 25 practice guideline.pt. (0) |
| 26 practice guideline/(275498) |
| 27 practice guidelines as topic/(171087) |
| 28 (CPG or CPGs or guidance or guideline? or recommend$ or standard?).ti. (203281) |
| 29 exp clinical pathway/(6983) |
| 30 exp clinical protocol/(75932) |
| 31 ((care or clinical) adj2 pathway?).tw. (9455) |
| 32 or/19-31 (2897811) |
| 33 8 and 18 and 32 (3886) |
| 34 (exp animal/ or animal.hw. or nonhuman/) not (exp human/ or human cell/ or (human or humans).ti.) (5865316) |
| 35 33 not 34 (3822) |
| 36 (immigra$ or migrant$ or migration$ or refugee$).mp. (337937) |
| 37 35 and 36 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$).dd. (68) |
| 38 remove duplicates from 37 (reviews and guidelines) (67) |
| 39 cost effectiveness analysis/(114261) |
| 40 cost.tw. (387424) |
| 41 costs.tw. (208729) |
| 42 or/39-41 (544762) |
| 43 8 and 18 and 42 (1580) |
| 44 (exp animal/ or animal.hw. or nonhuman/) not (exp human/ or human cell/ or (human or humans).ti.) (5865316) |
| 45 43 not 44 (1556) |
| 46 (immigra$ or migrant$ or migration$ or refugee$).mp. (337937) |
| 47 45 and 46 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$).dd. (59) |
References
- World Health Organization (WHO). Guidelines for the Screening, Care and Treatment of Persons with Chronic Hepatitis C Infection; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- World Health Organization. WHO Guidelines on Hepatitis B and C Testing; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- European Centre for Disease Prevention and Control (ECDC). Epidemiological Assessment of Hepatitis B and C among Migrants in the EU/EEA; ECDC: Stockholm, Sweden, 2016.
- Hatzakis, A.; Wait, S.; Bruix, J.; Buti, M.; Carballo, M.; Cavaleri, M.; Colombo, M.; Delarocque-Astagneau, E.; Dusheiko, G.; Esmat, G.; et al. The State of Hepatitis B and C in Europe: Report from the Hepatitis B and C Summit Conference. J. Viral Hepat. 2011, 18 (Suppl. 1), 1–16. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Hepatitis B and C in the EU Neighbourhood: Prevalence, Burden of Disease and Screening Policies; ECDC: Stockhlom, Sweden, 2010.
- Blachier, M.; Leleu, H.; Peck-Radosavljevic, M.; Valla, D.C.; Roudot-Thoraval, F. The Burden of Liver Disease in Europe: A Review of Available Epidemiological Data. J. Hepatol. 2013, 58, 593–608. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Antenatal Screening for HIV, Hepatitis B, Syphilis and Rubella Susceptibility in the EU/EEA; ECDC: Stockholm, Sweden, 2016.
- Rossi, C.; Shrier, I.; Marshall, L.; Cnossen, S.; Schwartzman, K.; Klein, M.B.; Schwarzer, G.; Greenaway, C. Seroprevalence of Chronic Hepatitis B Virus Infection and Prior Immunity in Immigrants and Refugees: A Systematic Review and Meta-Analysis. PLoS ONE 2012, 7, e44611. [Google Scholar] [CrossRef] [PubMed]
- Duffell, E.F.; Hedrich, D.; Mardh, O.; Mozalevskis, A. Towards Elimination of Hepatitis B and C in European Union and European Economic Area Countries: Monitoring the World Health Organization’s Global Health Sector Strategy Core Indicators and Scaling up Key Interventions. Eurosurveillance 2017, 22, 30476. [Google Scholar] [CrossRef] [PubMed]
- LeFevre, M.L. Screening for Hepatitis B Virus Infection in Nonpregnant Adolescents and Adults: U.S. Preventive Services Task Force Recommendation Statement. Ann. Intern. Med. 2014, 161, 58–66. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Vaccine Schedule. Available online: https://vaccine-schedule.ecdc.europa.eu/ (accessed on 17 August 2018).
- Hutton, D.W.; Tan, D.; So, S.K.; Brandeau, M.L. Cost-Effectiveness of Screening and Vaccinating Asian and Pacific Islander Adults for Hepatitis B. Ann. Intern. Med. 2007, 147, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Edmunds, W.J.; Medley, G.F.; Nokes, D.J.; Hall, A.J.; Whittle, H.C. The Influence of Age on the Development of the Hepatitis B Carrier State. Proc. R. Soc. B Biol. Sci. 1993, 253, 197–201. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Global Health Sector Strategy on Viral Hepatitis 2016–2021. Towards Ending Viral Hepatitis; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- European Centre for Disease Prevention and Control. Hepatitis B and C Testing Activities, Needs, and Priorities in the EU/EEA; ECDC: Stockholm, Sweden, 2017.
- Pottie, K.; Mayhew, A.; Morton, R.; Greenaway, C.; Akl, E.; Rahman, P. Prevention and Assessment of Infectious Diseases among Children and Adult Migrants Arriving to the European Union/European Economic Association: A Protocol for a Suite of Systematic Reviews for Public Health and Health Systems. BMJ Open 2017, 7, e014608. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaf, J.; Altman, D.G.; Group, T.P. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Shea, B.J.; Grimshaw, J.M.; Well, G.A.; Boers, M.; Andersson, N.; Hamel, C.; Porter, A.C.; Tugwell, P.; Moher, D.; Bouter, L.M. Development of AMSTAR: A Measurement Tool to Assess the Methodological Quality of Systematic Reviews. BMC Med. Res. Methodol. 2007, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Schünemann, H.J.; Wiercioch, W.; Brozek, J.; Etxeandia-Ikobaltzeta, I.; Mustafa, R.A.; Manja, V.; Brignardello-Petersen, R.; Neumann, I.; Falavigna, M.; Alhazzani, W.; et al. GRADE Evidence to Decision (EtD) Frameworks for Adoption, Adaptation, and de Novo Development of Trustworthy Recommendations: GRADE-ADOLOPMENT. J. Clin. Epidemiol. 2017, 81, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Larsson, L.; Hendricksen, C. Health Economics Information Resources: A Self-Study Course: Module 4; U.S. National Library of Medicine: Bethesda, MD, USA, 2014. [Google Scholar]
- Chou, R.; Dana, T.; Bougatsos, C.; Blazina, I.; Khangura, J.; Zakher, B. Screening for Hepatitis B Virus Infection in Adolescents and Adults: A Systematic Review to Update the U.S. Preventive Services Task Force Recommendation. Ann. Intern. Med. 2014, 161, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.L.H.; Yiu, K.K.L.; Wong, V.W.S.; Tsoi, K.K.F.; Chan, H.L.Y. Meta-Analysis: Reduction in Hepatic Events Following Interferon-Alfa Therapy of Chronic Hepatitis B. Aliment. Pharmacol. Ther. 2010, 32, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- EASL. EASL 2017 Clinical Practice Guidelines on the Management of Hepatitis B Virus Infection. J. Hepatol. 2017, 67, 370–398. [Google Scholar] [CrossRef] [PubMed]
- Graham, S.; Guy, R.J.; Cowie, B.; Wand, H.C.; Donovan, B.; Akre, S.P.; Ward, J.S. Chronic Hepatitis B Prevalence among Aboriginal and Torres Strait Islander Australians since Universal Vaccination: A Systematic Review and Meta-Analysis. BMC Infect. Dis. 2013, 13, 403. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.H.; You, S.L.; Chen, C.J.; Liu, C.J.; Lee, C.M.; Lin, S.M.; Chu, H.C.; Wu, T.C.; Yang, S.S.; Kuo, H.S.; et al. Decreased Incidence of Hepatocellular Carcinoma in Hepatitis B Vaccinees: A 20-Year Follow-up Study. J. Natl. Cancer Inst. 2009, 101, 1348–1355. [Google Scholar] [CrossRef] [PubMed]
- Rossi, C.; Schwartzman, K.; Oxlade, O.; Klein, M.B.; Greenaway, C. Hepatitis B Screening and Vaccination Strategies for Newly Arrived Adult Canadian Immigrants and Refugees: A Cost-Effectiveness Analysis. PLoS ONE 2013, 8, e78548. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.W.L.; Woo, G.; Heathcote, E.J.; Krahn, M. Cost Effectiveness of Screening Immigrants for Hepatitis B. Liver Int. 2011, 31, 1179–1190. [Google Scholar] [CrossRef] [PubMed]
- Veldhuijzen, I.K.; Toy, M.; Hahné, S.J.M.; De Wit, G.A.; Schalm, S.W.; de Man, R.A.; Richardus, J.H. Screening and Early Treatment of Migrants for Chronic Hepatitis B Virus Infection Is Cost-Effective. Gastroenterology 2010, 138, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Rein, D.B.; Lesesne, S.B.; Smith, B.D.; Weinbaum, C.M. Models of Community-Based Hepatitis B Surface Antigen Screening Programs in the U.S. and Their Estimated Outcomes and Costs. Public Health Rep. 2011, 126, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Jazwa, A.; Coleman, M.S.; Gazmararian, J.; Wingate, L.T.; Maskery, B.; Mitchell, T.; Weinberg, M. Cost-Benefit Comparison of Two Proposed Overseas Programs for Reducing Chronic Hepatitis B Infection among Refugees: Is Screening Essential? Vaccine 2015, 33, 1393–1399. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, M.; Cicchetti, A.; Gasbarrini, A. The Cost-Effectiveness of Alternative Strategies against HBV in Italy. Health Policy 2011, 102, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Eckman, M.H.; Kaiser, T.E.; Sherman, K.E. The Cost-Effectiveness of Screening for Chronic Hepatitis B Infection in the United States. Clin. Infect. Dis. 2011, 52, 1294–1306. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Xie, Q.; Liu, Y.; Toy, M.; Onder, F.O. Cost-Effectiveness of Early Detection of Inactive and Treatment of Active Cases in a High Endemic Chronic Hepatitis B Region. J. Antivir. Antiretrovir. 2013, 5, 154–159. [Google Scholar] [CrossRef]
- Seedat, F.; Hargreaves, S.; Nellums, L.B.; Ouyang, J.; Brown, M.; Friedland, J.S. How Effective Are Approaches to Migrant Screening for Infectious Diseases in Europe? A Systematic Review. Lancet Infect. Dis. 2018, 18, e259–e271. [Google Scholar] [CrossRef]
- Chang, M.-H.; Chen, C.-J.; Lai, M.-S.; Hsu, H.-M.; Wu, T.-C.; Kong, M.-S.; Liang, D.-C.; Shau, W.-Y.; Chen, D.-S. Universal Hepatitis B Vaccination in Taiwan and the Incidence of Hepatocellular Carcinoma in Children. N. Engl. J. Med. 1997, 336, 1855–1859. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.H.; Chang, M.H.; Huang, L.M.; Chen, H.L.; Hsu, H.Y.; Chiu, T.Y.; Tsai, K.S.; Chen, D.S. Hepatitis B Virus Infection in Children and Adolescents in a Hyperendemic Area: 15 Years after Mass Hepatitis B Vaccination. Ann. Intern. Med. 2001, 135, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.S.; Mcmahon, B.J.; Lok, A.S.F.; Wong, J.B.; Ahmed, A.T.; Mouchli, M.A.; Wang, Z.; Prokop, L.J.; Murad, M.H.; Mohammed, K. Antiviral Therapy in Chronic Hepatitis B Viral Infection during Pregnancy: A Systematic Review and Meta-Analysis. Hepatology 2016, 63, 319–333. [Google Scholar] [CrossRef] [PubMed]
- McMahon, B.J.; Helminiak, C.; Wainwright, R.B.; Bulkow, L.; Trimble, B.A.; Wainwright, K. Frequency of Adverse Reactions to Hepatitis B Vaccine in 43,618 Persons. Am. J. Med. 1992, 92, 254–256. [Google Scholar] [CrossRef]
- Hahné, S.J.; Veldhuijzen, I.K.; Wiessing, L.; Lim, T.-A.; Salminen, M.; van de Laar, M. Infection with Hepatitis B and C Virus in Europe: A Systematic Review of Prevalence and Cost-Effectiveness of Screening. BMC Infect. Dis. 2013, 13, 181. [Google Scholar]
- Hacker, K.; Anies, M.; Folb, B.L.; Zallman, L. Barriers to Health Care for Undocumented Immigrants: A Literature Review. Risk Manag. Healthc. Policy 2015, 8, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Mostert, M.C.; Richardus, J.H.; De Man, R.A. Referral of Chronic Hepatitis B Patients from Primary to Specialist Care: Making a Simple Guideline Work. J. Hepatol. 2004, 41, 1026–1030. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.; Bates, G.; McCoy, E.; Beynon, C.; McVeigh, J.; Bellis, M. A Systematic Review of the Effectiveness and Cost-Effectiveness of Interventions Aimed at Raising Awareness and Engaging with Groups Who Are at an Increased Risk of Hepatitis B and C Infection—Final Report; Liverpool John Moores University: Liverpool, UK, 2012. [Google Scholar]
- Seedat, F.; Hargreaves, S.; Friedland, J.S. Engaging New Migrants in Infectious Disease Screening: A Qualitative Semi-Structured Interview Study of UK Migrant Community Health-Care Leads. PLoS ONE 2014, 9, e108261. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, S.; Seedat, F.; Car, J.; Escombe, R.; Hasan, S.; Eliahoo, J.; Friedland, J.S. Screening for Latent TB, HIV, and Hepatitis B/C in New Migrants in a High Prevalence Area of London, UK: A Cross-Sectional Study. BMC Infect. Dis. 2014, 14, 657. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, S.; Lillis, D.; Cotter, A.; O’Dea, S.; Tuite, H.; Fleming, C.; Crowley, B.; Fitzgerald, I.; Dalby, L.; Barry, H.; et al. Opt-out Panel Testing for HIV, Hepatitis B and Hepatitis C in an Urban Emergency Department: A Pilot Study. PLoS ONE 2016, 11, e0150546. [Google Scholar] [CrossRef] [PubMed]
- Robotin, M.C.; George, J. Community-Based Hepatitis B Screening: What Works? Hepatol. Int. 2014, 8, 478–492. [Google Scholar] [CrossRef] [PubMed]
- Coppola, N.; Alessio, L.; Gualdieri, L.; Pisaturo, M.; Sagnelli, C.; Caprio, N.; Maffei, R.; Starace, M.; Angelillo, I.F.; Pasquale, G.; et al. Hepatitis B Virus, Hepatitis C Virus and Human Immunodeficiency Virus Infection in Undocumented Migrants and Refugees in Southern Italy, January 2012 to June 2013. Eurosurveillance 2015, 20, 30009. [Google Scholar] [CrossRef] [PubMed]
- El-Hamad, I.; Pezzoli, M.C.; Chiari, E.; Scarcella, C.; Vassallo, F.; Puoti, M.; Ciccaglione, A.; Ciccozzi, M.; Scalzini, A.; Castelli, F. Point-of-Care Screening, Prevalence, and Risk Factors for Hepatitis B Infection among 3728 Mainly Undocumented Migrants from Non-EU Countries in Northern Italy. J. Travel Med. 2015, 22, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Veldhuijzen, I.K.; Wolter, R.; Rijckborst, V.; Mostert, M.; Voeten, H.A.; Cheung, Y.; Boucher, C.A.; Reijnders, J.G.P.; De Zwart, O.; Janssen, H.L.A. Identification and Treatment of Chronic Hepatitis B in Chinese Migrants: Results of a Project Offering on-Site Testing in Rotterdam, the Netherlands. J. Hepatol. 2012, 57, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Lewis, H.; Burke, K.; Begum, S.; Ushiro-Limb, I.; Foster, G. What is the best method of case finding for chronic viral hepatitis in at-risk migrant communities? J. Hepatol. 2012, 56, S351. [Google Scholar] [CrossRef]
- Gish, R.G.; Cooper, S. Hepatitis B in the Greater San Francisco Bay Area: An Integrated Programme to Respond to a Diverse Local Epidemic. J. Viral Hepat. 2011, 18, e40–e51. [Google Scholar] [CrossRef] [PubMed]
| Study | Quality | Type of Study | Population | Intervention | Results/Outcomes |
|---|---|---|---|---|---|
| Should Hepatitis B Virus (HBV) screening be offered to recently arrived migrants to the EU/EEA? | |||||
| Chou et al. 2014 [21] | AMSTAR * 9/11 | Systematic Review | 12 RCTs | Treatment with Nucelos(t)ide Analogues (NAs) compared to placebo | Reduced rate of intermediate outcomes in NA group (HBV DNA loss, HBeAg loss, Histologic improvement, HBsAg loss). No significant decrease in HCC incidence. No increase in significant adverse events in NA group but higher rates of study withdrawal |
| Wong et al. 2010 [22] | AMSTAR 7/11 | Systematic Review | 11 RCTs | Treatment with pegylated interferon alpha compared to placebo | Decreased rate of Hepatic Events (RR 0.55 95% CI 0.43 to 0.70), cirrhotic complications (RR 0.46 95% CI 0.32 to 0.67) and liver related mortality (RR 0.63 95% CI 0.42 to 0.96) for treatment group |
| EASL 2017 [23] | NA | Guideline | Current Treatment Guidelines for acute and chronic infection with hepatitis B for the EU/EEA | ||
| ECDC 2016 [3] | NA | Technical Document | EU/EEA Migrants/General Population | No intervention | 5.5% of migrants born in intermediate/high prevalence countries infected with CHB infection compared to 1.12% in the general population of EU/EEA Migrants from HBV endemic countries 5.1% of population of the EU/EEA but 25% (range 14–47%) of the total number of CHB cases |
| WHO 2017 [2] | NA | Guideline | Guidelines on hepatitis B testing including information on implementation of screening programs. | ||
| WHO 2015 [1] | NA | Guideline | Guidelines for the prevention, care and treatment of persons with chronic hepatitis B infection. | ||
| What is the effectiveness of vaccination programs against HBV? | |||||
| Graham et al. 2013 [24] | AMSTAR 4/11 | Systematic Review | Australia: Aboriginal population/general population | Vaccination with HBV vaccine compared to no vaccine | Reduced rate of positive HBsAg in post vaccination cohort 3.96% (95% CI: 3.15–4.77) compared to pre-vaccination cohort 16.72 (95% CI: 7.38–26.06). |
| Chang et al. 2009 [25] | NA | Individual Study | Endemic Country Taiwan (Republic of China) | Vaccination with HBV vaccine compared to no vaccine | Reduced rate of positive HBsAg (RR 0.07 95% CI 0.04 to 0.13) and chronic liver disease mortality (RR 0.34 95% CI 0.25 to 0.45) in post vaccination cohort |
| Rossi et al. 2012 [8] | AMSTAR 6/11 | Systematic Review | Global Migrant Population | No intervention | Prevalence of CHB higher in refugees and asylum seekers compared to immigrants (9.6% vs. 5.1%). 39.7% of migrants demonstrated prior immunity to HBV either through prior infection or vaccination (95% CI: 35.7–43.9%). |
| Study | Certainty of Economic Evidence (Quality) | Design | Population | Intervention | Cost-Effectiveness | Resource Requirements |
|---|---|---|---|---|---|---|
| What is the cost-effectiveness of screening (and subsequent management) migrants for chronic hepatitis B (CHB)? | ||||||
| Rossi et al. 2013 [26] | Allowance was made for uncertainty in the estimates of costs and consequences across plausible ranges. Appropriate statistical analyses (probabilistic sensitivity analyses (PSA) were performed for costs and consequences. Justification was provided for key study parameters and some upper and lower range estimates. Some ranges cited the data sources; other ranges provided an assumption for upper and lower limits but no further justification. The cost-effectiveness results were not very sensitive to changes in the values. Most of time, the intervention was still cost-effective. At a standard willingness to pay threshold, the probability of being cost-effective was 78% (high). | Decision-analytic Markov model, results presented in Canadian dollars. | Vaccination strategies for newly arrived adult Canadian immigrants and refugees | (i) universal vaccination, (ii) screening + vaccination (iii) screening+ treatment (iv) screening +treatment + vaccination | (i) and (ii) were dominated by no intervention (iii) screening and treatment: CAN$40,880 per QALY gained vs no intervention (iv) screen, treatment, vaccination: CAN$437,335 per QALY gained vs no intervention | The intervention has moderate costs. Categories and volumes of resource use were not reported separately. |
| Wong et al. 2011 [27] | All the ranges were provided. Both one way, and probabilistic sensitivity analyses were performed. All data sources for model inputs were provided, with the exception of reference years for all costs. The results were sensitive to the progression rate and discount rate used. The intervention had a 55% probability of being cost-effective compared to a no screening strategy. Certainty in the results was deemed to be moderate overall. | Decision-analytic Markov model; reported in Canadian dollars | Immigrants to Canada | (i) ‘No screening’; (ii) ‘Screen and Treat’ (iii) ‘Screen, Treat and Vaccinate’ | ICER for Screen and treat ranged from CAN$45,221 (Tenofovir, 3% discount) per QALY gained to CAN$101,513 (Entecavir, 5% discount) ICER for Screen, treat and vaccinate ranged from CAN$96,523 (Entecavir, 3% discount) to CAN$3,648,123 per QALY gained (Tenofovir, 5% discount) Favours intervention (ii): screen and treat, CAN$69,209/QALY gained (cost-effective) A vaccination program following the screening program was not cost-effective compared with the screen and treat strategy. | Large costs for strategy ii: screen and treat if using either Entecavir or Tenofovir. Large costs for strategy iii: screen, treat and vaccinate using Entecavir. Other interventions had moderate costs. Resource use was not quantified separately. |
| Hutton et al. 2007 [12] | Allowance for uncertainty was accommodated. Both one way and PSA were conducted. All the data sources were provided for ranges used in sensitivity analyses. The probability of being cost-effective was 82–85% when the values of key variables were changed. Therefore the certainty of the intervention being cost-effective was deemed high. | Decision-analytic Markov model; results in US dollars | Asian and Pacific Islander adult immigrants to the US | (i) No Screening: universal vaccination strategy for all individuals (ii) Screen, Treat and No Vaccination: a screen-and-treat strategy: screen individuals and treat infected persons; (iii) Screen, Treat and Vaccinate: vaccine for non-infected persons (iv) Screen, Treat and Ring Vaccinate: screen for close contacts and vaccinate non-infected persons | The screen-and-treat strategy, intervention (ii) has an incremental cost-effectiveness ratio of US$36,088 per QALY gained compared with the status quo. Screen and treat and ring vaccinate strategy, intervention (iv) has a cost-effectiveness ratio of US$39,903 per QALY gained compared with the screen-and-treat strategy. Universal vaccination, intervention (i) and screen and treat and vaccine were dominated. | Costs were moderate, ranging from US$85,000 per person per lifetime with universal vaccination to US$87,000 per person per lifetime to screen, treat and ring vaccinate. |
| Veldhuijzen et al. 2010 [28] | Uncertainty was tested and all ranges were provided Univariate, multivariate, PSA were conducted. Data sources for all the ranges were provided Cost-effectiveness results were robust to changes in model parameters. The probability of being cost-effective was 72%; the certainty was deemed moderate. | Decision-analytic Markov model; results reported in Euros | Migrants to the Netherlands from intermediate and high HBV endemic areas | One-off systematic screening and subsequent treatment of eligible patients, compared with the status quo: i.e., existing pregnancy screening, testing due to medical complaints, contact tracing, and checkup for STIs. | Incremental cost-effectiveness ratio (ICER) of €8966 per QALY gained. Discounted costs at 4% and effects at 1.5%, resulted in a slightly lower ICER of €8823 per QALY gained. | Status quo had low test costs at €458 while the screen and treat strategy had a test cost of €15,954. Referral and follow up costs for the status quo strategy was €838; while the screen and treat strategy had a follow up cost of €3074. i.e., a large difference. Screening and treatment costs per person were ~€130 per person. |
| Rein et al. 2011 [29] | Standard deviations were provided for costs. | Costing study (exploratory, pilot study) | Overseas-born community living in the US | Screening models: (i) Community Clinic (ii) Community Outreach (iii) Outreach partnership (iv) Partnership contract | Cost-effectiveness was not reported | Cost per complete screen ranged from US$40 for the Community Clinic to US$280 for the Partnership Contract model. Low costs for Community based but higher costs for Partnership model. The costs per positive person identified varied from US$609 in the Community model to US$4657 in the Partnership model. Cost per complete screen/cost per newly identified positive case (adj. for prevalence): 1) US$40/$854 ($895) 2) US$102/$2641 ($2698) 3) US$280/$6300 ($6013) 4) US$176/$5709 ($5063) |
| Jazwa et al. 2015 [30] | Allowance was made for uncertainty, all the ranges were provided. Not all statistical tests were reported. Univariate sensitivity analysis was conducted, which is consistent with the study design (cost benefit study, not cost-effectiveness). All the data sources and assumptions were provided. Not applicable. | Cost-benefit analysis | Refugees to the US; costs reported in US dollars | (i) Vaccinate only without HBV screening (ii) Screen, then vaccinate or initiate management | The net benefits of the screen and vaccinate strategy ranged from US$24 million to US$130 million after 5 years from program initiation | The cost per refugee for the vaccination only strategy was low if the screen rate <70%, however after 10 years, if the screening rate was more than 70%, the cost of the vaccination only strategy was moderate: US$706–$968 per person. |
| Ruggeri et al. 2011 [31] | Allowance was made for uncertainty, all the ranges were provided. Both one way and PSA were conducted. All the data sources and assumptions were provided. The results were not sensitive to changes in the model values. The probability of being cost-effective was 70–98%; the certainty of the results was deemed high. | Decision-analytic Markov model; results reported in Euros | Residents of Italy | (i) Screening of Italian patients at risk (assumed prevalence of 7%) and treatment of cases according to protocol; (ii) compared with no screening and treatment of patients with cirrhosis or HCC | ICER of €18,256 per QALY gained (±€387) for screening compared to no screening | High costs for the screening strategy: €67,008 (±€515) per person per year; Low cost for no screening strategy, but moderate costs for the screen and treat strategy. Moderate cost for no screening strategy: €7939 (±€1679) per person per year |
| Eckman et al. 2011 [32] | Allowance was made for uncertainty, all the ranges were provided. PSA was conducted All the data sources and assumptions were provided. The cost-effectiveness results were sensitive to model parameters including cost of treatment, drug resistance, and disease prevalence. The probability of the intervention being CE was 49%; certainty was deemed moderate. | Decision-analytic Markov model; results reported in US dollars | Asymptomatic outpatients in the US | Screening for Hepatitis B surface antigen followed by treatment of appropriate patients with (i) pegylated interferon-a2a for 48 weeks, (ii) a low-cost nucleoside or nucleotide agent with a high rate of developing viral resistance for 48 weeks, (iii) prolonged treatment with low-cost, high-resistance nucleoside or nucleotide, (iv) prolonged treatment with a high-cost nucleoside or nucleotide with a low rate of developing viral resistance; compared with no screening | Intervention (iii) was dominated by the no screening intervention; Intervention (ii) and intervention (v) were dominated by intervention (iv). Intervention (iv) was cost-effective with an ICER of US$29,232 per QALY gained. | Low cost for no screening strategy US$915 per person per year. Moderate cost for screen and treat, ranging from US$1170 (treat with low cost, high resistance nucleoside) to US$1286 (treat with high cost, low resistance nucleoside). Resource use was not reported separately. |
| Li et al. 2013 [33] | Allowance was made for uncertainty. All upper bound and lower bound limits were provided. Only univariate sensitivity analysis was conducted. No, justification was provided for the ranges tested for price of treatment, or probability of disease progression. The cost-effectiveness results were not sensitive (i.e., remained robust) to changes in the values of variables. Moderate certainty. | Decision-analytic Markov model; results reported in US dollars. | Residents of Zhoushan Island in mainland China. | Monitor and treat scenarios in 3 patient groups according to treatment eligibility, (1) ineligible (2) borderline (3) eligible compared with natural history (no screening and no antiviral treatment of patients with cirrhosis or HCC) | ICER of the monitor and treat strategy compared to the natural history was US$97 per QALY gained for the ineligible group, US$500/QALY for the borderline group, US$1131/QALY for the eligible group. With a 5% reduction in Entecavir price: the monitor and treat strategy becomes cost saving (ICER < 0) in the ineligible group; the ICER was US$254 for the eligible group, and US$860 for the eligible group. With a 50% reduction in Entecavir price: the monitor and treat strategy was cost saving for all sub groups: (ICER < 0). | For the ineligible group: Difference in costs was small. For example, total costs per patient per lifetime was US$21,229 for natural history strategy, and US$21,550 for Monitor and Treat. For the borderline group: The difference in costs was larger. For natural history strategy, the total costs = per patient lifetime was US$33,280 while the total cost per patient lifetime for the monitor and treat strategy was US$37,043. For the eligible group: The difference in costs was largest. With natural history strategy, total cost per patient lifetime was US$32,430 while total cost per patient lifetime for monitor and treat strategy was US$42,711. |
| Certainty Assessment | Summary of Findings | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| № of participants (studies) Follow-up | Risk of bias | Inconsistency | Indirectness | Imprecision | Publication bias | Overall certainty of evidence | Study event rates (%) | Relative effect (95% CI) | Anticipated absolute effects | ||
| With no vaccine | With HBV vaccine | Risk with no vaccine | Risk difference with HBV vaccine | ||||||||
| HCC mortality | |||||||||||
| 54289638 (1 observational study) | serious a | not serious b | not serious | serious c | none | Very Low | 135/27144819 (0.0%) | 20/27144819 (0.0%) | RR 0.90 (0.75 to 1.09) | 0 per 100,000 | 0 fewer per 100,000 (0 fewer to 0 fewer) |
| Liver cancers (except non-hepatocellular carcinoma) | |||||||||||
| 6898803 (1 observational study) | serious a | serious b | not serious | serious c | none | Very Low | 24/3381519 (0.0%) | 20/3517284 (0.0%) | RR 0.80 (0.42 to 1.48) | 1 per 100,000 | 0 fewer per 100,000 (0 fewer to 0 fewer) |
| HBsAg carriage | |||||||||||
| 1916 (1 observational study) | serious d | not serious | not serious | not serious | strong association e | Low | 39/559 (7.0%) | 9/1357 (0.7%) | RR 0.07 (0.04 to 0.13) | 6977 per 100,000 | 6488 fewer per 100,000 (6698 fewer to 6070 fewer) |
| Anti-HBc | |||||||||||
| 1916 (1 observational study) | serious d | not serious | not serious | not serious | strong association e | Low | 115/559 (20.6%) | 39/1357 (2.9%) | RR 0.11 (0.08 to 0.16) | 20,572 per 100,000 | 18,309 fewer per 100,000 (18,927 fewer to 17,281 fewer) |
| Chronic Liver Disease | |||||||||||
| 54289638 (1 observational study) | serious a | not serious | not serious | not serious | strong association | Low | 407/38702888 (0.0%) | 55/15586750 (0.0%) | RR 0.34 (0.25 to 0.45) | 1 per 100,000 | 1 fewer per 100,000 (1 fewer to 1 fewer) |
| HCC Incidence | |||||||||||
| 54289638 (1 observational study) | serious a | not serious | not serious | not serious | none | Very Low | 712/38702888 (0.0%) | 191/15586750 (0.0%) | RR 0.89 (0.75 to 1.04) | 2 per 100,000 | 0 fewer per 100,000 (0 fewer to 0 fewer) |
| HBsAg (continuous) | |||||||||||
| 8545 (8 observational studies) | serious a | serious f | not serious | not serious | none | Very Low | 5516 (Number of Events) | 3029 (Number of Events) | N/A | The mean HBsAg (continuous) ranged from 5.19–25.99 % | 3.96 % lower (3.15 lower to 4.17 lower) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Myran, D.T.; Morton, R.; Biggs, B.-A.; Veldhuijzen, I.; Castelli, F.; Tran, A.; Staub, L.P.; Agbata, E.; Rahman, P.; Pareek, M.; et al. The Effectiveness and Cost-Effectiveness of Screening for and Vaccination Against Hepatitis B Virus among Migrants in the EU/EEA: A Systematic Review. Int. J. Environ. Res. Public Health 2018, 15, 1898. https://doi.org/10.3390/ijerph15091898
Myran DT, Morton R, Biggs B-A, Veldhuijzen I, Castelli F, Tran A, Staub LP, Agbata E, Rahman P, Pareek M, et al. The Effectiveness and Cost-Effectiveness of Screening for and Vaccination Against Hepatitis B Virus among Migrants in the EU/EEA: A Systematic Review. International Journal of Environmental Research and Public Health. 2018; 15(9):1898. https://doi.org/10.3390/ijerph15091898
Chicago/Turabian StyleMyran, Daniel T, Rachael Morton, Beverly-Ann Biggs, Irene Veldhuijzen, Francesco Castelli, Anh Tran, Lukas P Staub, Eric Agbata, Prinon Rahman, Manish Pareek, and et al. 2018. "The Effectiveness and Cost-Effectiveness of Screening for and Vaccination Against Hepatitis B Virus among Migrants in the EU/EEA: A Systematic Review" International Journal of Environmental Research and Public Health 15, no. 9: 1898. https://doi.org/10.3390/ijerph15091898
APA StyleMyran, D. T., Morton, R., Biggs, B.-A., Veldhuijzen, I., Castelli, F., Tran, A., Staub, L. P., Agbata, E., Rahman, P., Pareek, M., Noori, T., & Pottie, K. (2018). The Effectiveness and Cost-Effectiveness of Screening for and Vaccination Against Hepatitis B Virus among Migrants in the EU/EEA: A Systematic Review. International Journal of Environmental Research and Public Health, 15(9), 1898. https://doi.org/10.3390/ijerph15091898







