Sepiolite-Based Adsorbents for the Removal of Potentially Toxic Elements from Water: A Strategic Review for the Case of Environmental Contamination in Hunan, China
Abstract
1. Introduction
2. Metal Pollution in Hunan, China
3. Natural Sepiolite
4. Modification of Sepiolite
4.1. Acid Treatment
4.2. Thermal Treatment
4.3. Magnetic Modification
4.4. Organic Modification
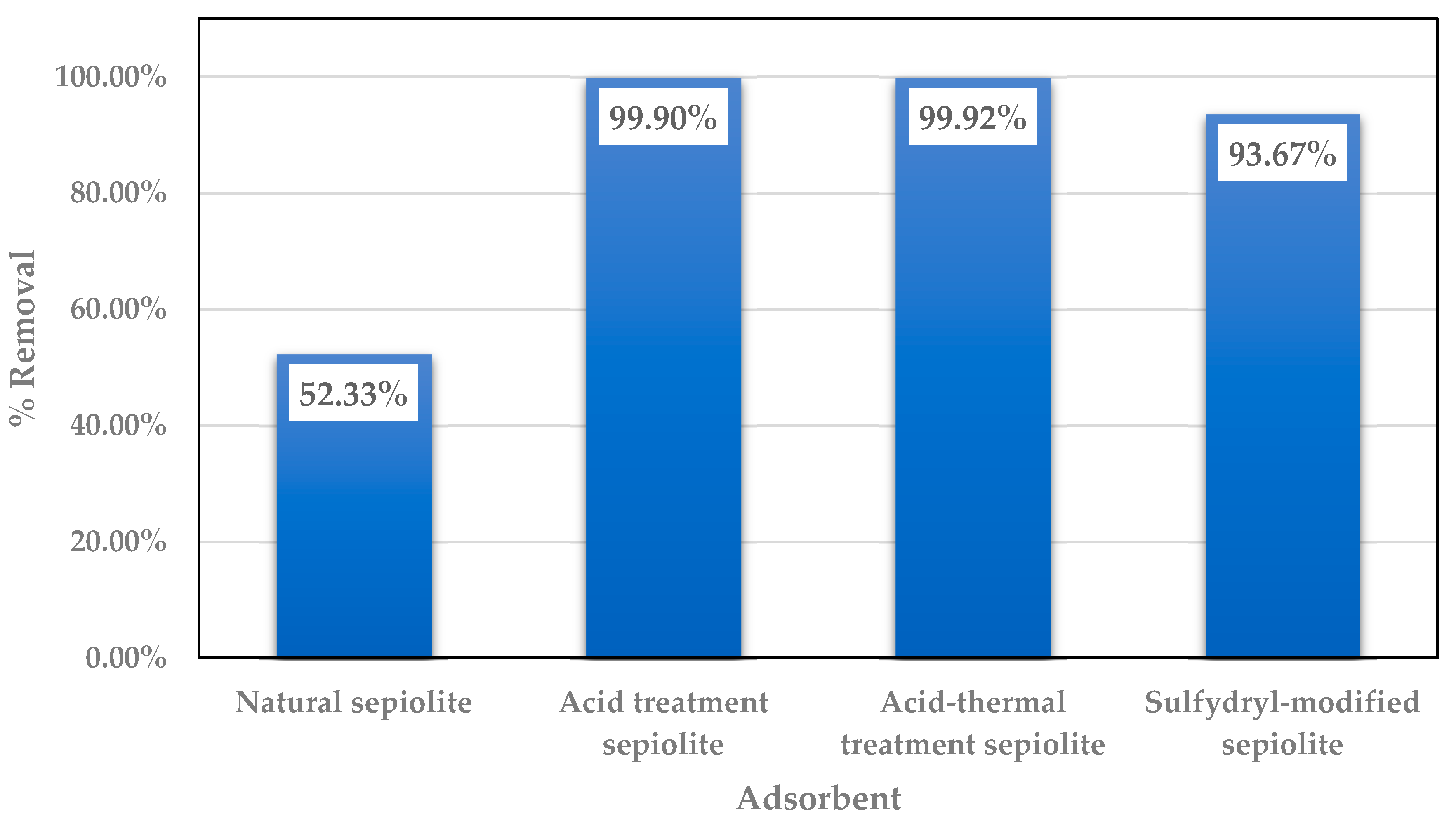
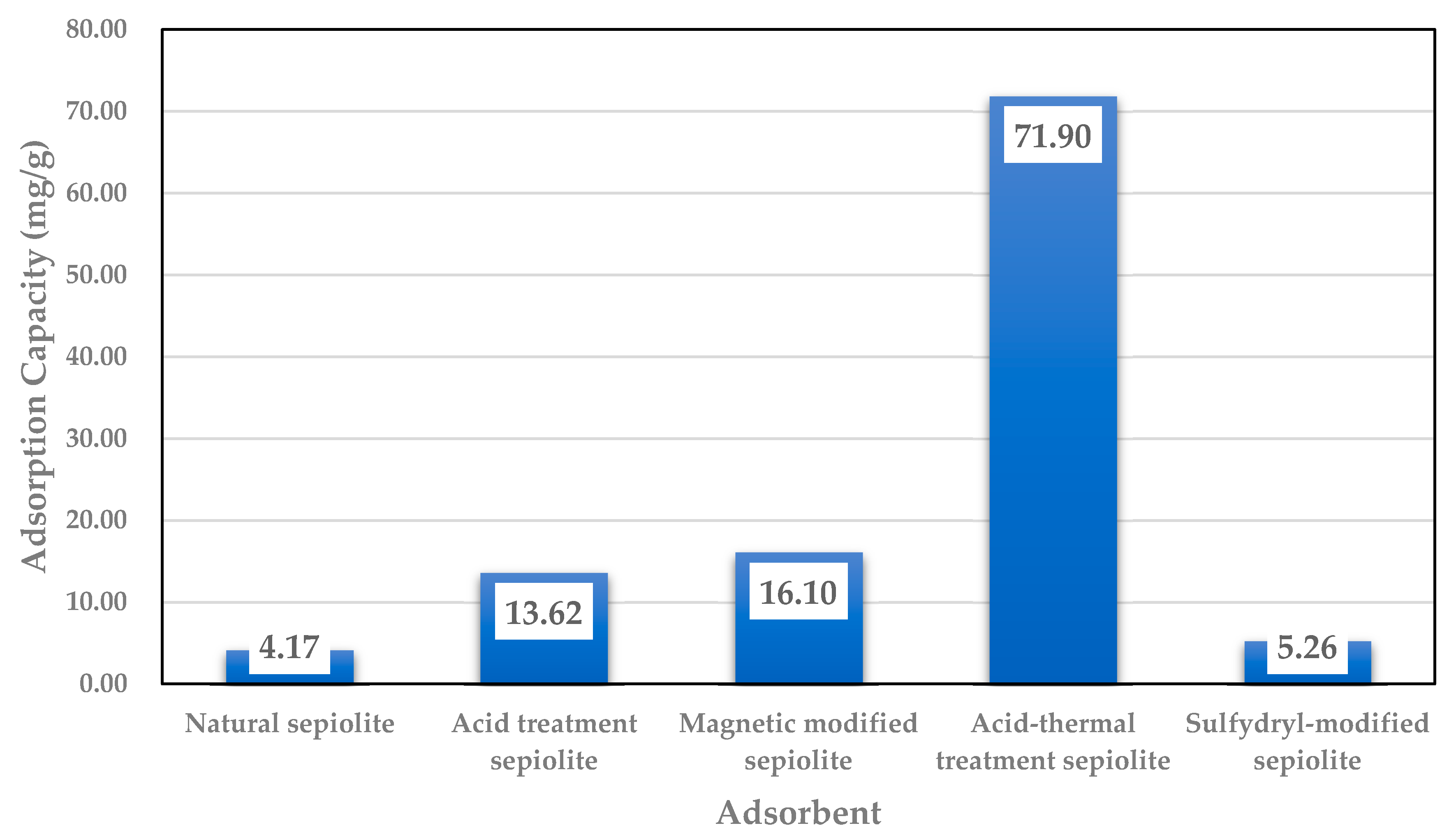
4.5. Acid Thermal Treatment
5. Examples of the Application of Sepiolite to Potentially Toxic Element Removals from Aqueous Environmental Systems
5.1. The Removal Hg2+ from Wastewater
5.2. The Removal of Cd from Wastewater
5.3. The Removal of Pb from Wastewater
5.4. The Application of Remove Cr in the Wastewater by Sepiolite
6. Regeneration of Sepiolite
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paul, D. Research on heavy metal pollution of river Ganga: A review. Ann. Agrar. Sci. 2017, 15, 278–286. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Zhang, W.; Li, Z.; Li, J.; Ge, C.; Liu, J.; Bai, X.; Feng, H.; Yu, L. Assessment of heavy metal pollution in Red River surface sediments, Vietnam. Mar. Pollut. Bull. 2016, 113, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Xiao, D. Status and control technology of heavy metal pollution. Energy Energy Conserv. 2012, 2, 49–50. (In Chinese) [Google Scholar]
- Wang, H.; Fang, F.; Xie, H. Research situation and outlook on heavy metal pollution in water environment of China. Guangdong Trace Elements Sci. 2010, 1, 14–18. (In Chinese) [Google Scholar]
- Yue, X.; Liu, K.; Lin, X.; Zhou, Q.; Mao, G.; Zhou, B.; Zhao, J. Current status of heavy metal pollution in seven major water systems in China. Prev. Med. Trib. 2014, 3, 209–213. (In Chinese) [Google Scholar]
- Duan, Q.; Lee, J.; Liu, Y.; Chen, H.; Hu, H. Distribution of heavy metal pollution in surface soil samples in China: A graphical review. Bull. Environ. Contam. Toxicol. 2016, 97, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Morton-Bermea, O.; Hernández-Álvarez, E.; González-Hernández, G.; Romero, F.; Lozano, R.; Beramendi-Orosco, L.E. Assessment of heavy metal pollution in urban topsoils from the metropolitan area of Mexico City. J. Geochem. Explor. 2009, 101, 218–224. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, L.; Zhao, Z.; Cai, Y. Heavy metal contamination in surface sediments of representative reservoirs in the hilly area of southern China. Environ. Sci. Pollut. Res. 2017, 24, 26574–26585. [Google Scholar] [CrossRef] [PubMed]
- Paul, D.; Sinha, S.N. Isolation and characterization of a phosphate solubilizing heavy metal tolerant bacterium from River Ganga, West Bengal, India. Songklanakarin J. Sci. Technol. 2015, 37, 651–657. [Google Scholar]
- Rajbanshi, A. Study on heavy metal resistant bacteria in Guheswori Sewage Treatment Plant. Our Nat. 2008, 6, 52–57. [Google Scholar] [CrossRef]
- Wu, W.; Wu, P.; Yang, F.; Sun, D.L.; Zhang, D.X.; Zhou, Y.K. Assessment of heavy metal pollution and human health risks in urban soils around an electronics manufacturing facility. Sci. Total Environ. 2018, 630, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Li, L.; Qu, Z.; Xu, H.; Xu, J.; Yan, N. A novel method for the sequential removal and separation of multiple heavy metals from wastewater. J. Hazard. Mater. 2018, 342, 617–642. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Gao, G.; Pan, B.; Zhang, W.; Lv, L. A new combined process for efficient removal of Cu (II) organic complexes from wastewater: Fe (III) displacement/UV degradation/alkaline precipitation. Water Res. 2015, 87, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Carro, L.; Barriada, J.L.; Herrero, R.; Vicente, M.E.S.D. Interaction of heavy metals with Ca-pretreated Sargassum muticum algal biomass: Characterization as a cation exchange process. Chem. Eng. J. 2015, 264, 181–187. [Google Scholar] [CrossRef]
- Zhang, W.; Li, J.; Tang, J.; Wang, B.; Huang, F. Advance of the treatment of heavy metal wastewater by adsorption. Chem. Ind. Eng. Prog. 2013, 11, 2749–2756. [Google Scholar]
- Naushad, M.; Ahamad, T.; Al-Maswari, B.; Alqadami, A.A.; Alsheri, S.M. Nickel ferrite bearing nitrogen-doped mesoporous carbon as efficient adsorbent for the removal of highly toxic metal ion from aqueous medium. Chem. Eng. J. 2017, 330, 1351–1360. [Google Scholar] [CrossRef]
- Alqadami, A.A.; Naushad, M.; Alothman, Z.A.; Ghfar, A.A. Novel metal-organic framework (MOF) based composite material for the sequestration of U(VI) and Th (IV) metal ions from aqueous environment. Appl. Mater. Interfaces 2017, 9, 36026–36037. [Google Scholar] [CrossRef] [PubMed]
- Vaizogullar, A.I.; Ugurlu, M.; Kula, I. Comparing adsorption activity of raw sepiolite and CTAB modified sepiolite: Kinetic and adsorption study for removal of Hg2+. Int. J. Environ. 2015, 4, 19–31. [Google Scholar] [CrossRef][Green Version]
- Renu; Agarwal, M.; Singh, K. Heavy metal removal from wastewater using various adsorbents: A review. J. Water Reuse Desalin. 2017, 7, 387–419. [Google Scholar] [CrossRef]
- Alaba, P.A.; Oladoja, N.A.; Sani, Y.M.; Ayodele, O.B.; Mohammed, I.Y.; Olupinla, S.F.; Daud, W.M.W. Insight into wastewater decontamination using polymeric adsorbents. J. Environ. Chem. Eng. 2018, 6, 1651–1672. [Google Scholar] [CrossRef]
- Malik, D.S.; Jain, C.K.; Yadav, A.K. Removal of heavy metals from emerging cellulosic low-cost adsorbents: A review. Appl. Water Sci. 2017, 7, 2113–2136. [Google Scholar] [CrossRef]
- Ko, D.; Mines, P.D.; Jakobsen, M.H.; Yavuz, C.T.; Hansen, H.C.B.; Andersen, H.R. Disulfide polymer grafted porous carbon composites for heavy metal removal from stormwater runoff. Chem. Eng. J. 2018, 348, 685–692. [Google Scholar] [CrossRef]
- Jlassi, K.; Abidi, R.; Benna, M.; Chehimi, M.M.; Kasak, P.; Krupa, I. Bentonite-decorated calix 4 arene: A new, promising hybrid material for heavy-metal removal. Appl. Clay Sci. 2018, 161, 15–22. [Google Scholar] [CrossRef]
- Zhou, Y.Y.; Wang, Z.H.; Hursthouse, A.; Ren, B.Z. Gemini surfactant-modified activated carbon for remediation of hexavalent chromium from water. Water 2018, 10. [Google Scholar] [CrossRef]
- Xie, X.; Wang, F.; Wang, G.; Mei, R.; Wang, C. Study on heavy metal pollution in surface water in China. Environ. Sci. Manag. 2017, 2, 31–34. (In Chinese) [Google Scholar]
- Cao, C.; Li, Y. Heavy metal pollution in Hunan is shocking. Environ. Educ. 2014, 12, 27–30. (In Chinese) [Google Scholar]
- Tian, B. Interpretation of “Hunan experiment” for heavy metal removal. People’s Friend 2012, 8, 15–16. (In Chinese) [Google Scholar]
- Liu, J.; Chen, M. Construction of heavy metal monitoring technical system during twelfth five-year plan in China. Environ. Sci. Manag. 2014, 39, 125–128. (In Chinese) [Google Scholar]
- Qiao, R. Study on the Legal Problems of Water Treatment in Xiangjiang River Basin. Master’s Thesis, University of Geosciences, Beijing, China, 2017. (In Chinese). [Google Scholar]
- Lei, M.; Qin, P.; Tie, B. The Present Situation and Analysis of Heavy Metal Pollution in Hunan Xiangjiang River Basin. In Proceedings of the Third National Symposium on Agricultural Environmental Science, Tianjing, China, 23–26 October 2009. (In Chinese). [Google Scholar]
- Wang, Q.; Wang, S.; Liu, M. Study evaluation on pollution of Xiang River Valley in Hunan Province. China Water Wastewater 2004, 8, 104–106. (In Chinese) [Google Scholar]
- Lei, D. Analysison heavy metals pollution status in Hunan Province and its remediation strategy. Hunan Nonferrous Met. 2012, 1, 57–60. (In Chinese) [Google Scholar]
- Liu, Y.; Gao, L.; Li, Z.; Liu, S.; Huang, K.; Li, J. Analysis on heavy metals pollution status and reasons in Xiangjiang River and discussion on its countermeasures. Environ. Prot. Sci. 2010, 36, 26–29. (In Chinese) [Google Scholar] [CrossRef]
- Wang, L.; Guo, Z.; Xiao, X.; Chen, T.; Liao, X.; Song, J.; Wu, B. Heavy metal pollution of soils and vegetables in the midstream and downstream of the Xiangjiang River, Hunan Province. J. Geogr. Sci. 2008, 18, 353–362. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, Y.; Hu, J.; Peng, G.; Xie, H.; Li, Z.; Peng, G. Characteristics and potential ecological risk assessment of heavy metal pollution in the sediments of middle reaches of Xiangjiang River. J. Ecol. Rural Environ. 2017, 33, 135–141. (In Chinese) [Google Scholar]
- Zhang, K.; Yang, X.; Wu, Y.; Wu, B.; Kuang, X. Pollution characteristics and ecological risk assessment of heavy metals in surface sediments in Changsha-Zhuzhou-Xiangtan Reach, Xiang Jiang River, China. J. Agric. Resour. Environ. 2015, 32, 60–65. (In Chinese) [Google Scholar]
- Fang, X.; Tian, D.; Xie, R. Soil physical and chemical properties of the wasteland in Xiangtan manganese mine. Xiangtan Manganese Min. 2006, 5, 1494–1501. (In Chinese) [Google Scholar]
- Tian, Q.; Zhang, G.; Xie, Y.; Mo, Y. Distribution and ecological risk assessment of heavy metals in surface sediments from main tributary entrances of Dongting Lake. Asian J. Ecotoxicol. 2017, 12, 191–200. (In Chinese) [Google Scholar]
- Zhou, Z. Overview of the resources of sepiolite clay in China and abroad. Land Resour. Herald 1985, 1, 148–156. (In Chinese) [Google Scholar]
- Wang, C.; Wang, G.; Tao, T.; Zhu, M.; Qu, F. Research progress and application status of sepiolite functional green building materials. Bull. Chin. Ceram. Soc. 2017, 10, 3285–3291. (In Chinese) [Google Scholar]
- Zhuang, G.; Gao, J.; Chen, H.; Zhang, Z. A new one-step method for physical purification and organic modification of sepiolite. Appl. Clay Sci. 2018, 153, 1–8. [Google Scholar] [CrossRef]
- Carmona, J.A.; Ramírez, P.; Trujillo-Cayado, L.A.; Caro, A.; Muñoz, J. Rheological and microstructural properties of sepiolite gels. Influence of the addition of ionic surfactants. J. Ind. Eng. Chem. 2018, 59, 1–7. [Google Scholar] [CrossRef]
- Wang, C. Research on Low Density Cement Slurry System for Complex Well Cementing. Master’s Thesis, Southwest Petroleum University, Chengdu, China, 2004. (In Chinese). [Google Scholar]
- Liang, K. The Mineralogy Research of the Sepiolite and Its Application in Environment Conservation. Ph.D. Thesis, Central South University, Changsha, China, 2008. (In Chinese). [Google Scholar]
- Duan, E.; Han, J.; Song, Y.; Guan, Y.; Zhao, W.; Yang, B.; Guo, B. Adsorption of styrene on the hydrothermal-modified sepiolite. Mater. Lett. 2013, 111, 150–153. [Google Scholar] [CrossRef]
- Jia, M.; Dai, Y.; Du, T.; Liu, C. Preparation of magnetically modified sepiolite and adsorption of hexavalent chromium. Environ. Chem. 2011, 30, 1546–1552. (In Chinese) [Google Scholar]
- Liu, C.; Huang, Y.; Yu, F.; Sun, X.; Xu, F.; Wu, W.; Zhong, M. Sepiolite modification on Pb adsorption characteristics. Environ. Chem. 2013, 32, 2024–2029. (In Chinese) [Google Scholar]
- Li, C.; Yu, J. Modification and application of sepiolite in chromium-containing wastewater treatment. Plat. Finish. 2013, 35, 17–22. (In Chinese) [Google Scholar]
- Feng, Y. Study on the Adsorption of Lead and Cadmium by Sepiolite. Master’s Thesis, University of South, Hengyang, China, 2007. (In Chinese). [Google Scholar]
- Feng, Y.; He, S.; Gao, W.; Li, H. Adsorption of organic matter and heavy metals in wastewater on sepiolite. Water Purif. Technol. 2006, 25, 63–66. (In Chinese) [Google Scholar]
- Shi, T.; Jia, S.; Chen, Y.; Wen, Y.; Chen, J.; Huang, R.; Wang, Z.; Liu, Y. Removal of heavy metal ions from wastewater by modified natural ores as adsorbents: A review. Technol. Water Treat. 2009, 35, 18–23. (In Chinese) [Google Scholar]
- Zhang, Y.; Wang, L.; Wang, F.; Liang, J.; Ran, S.; Sun, J. Phase transformation and morphology evolution of sepiolite fibers during thermal treatment. Appl. Clay Sci. 2017, 143, 205–211. [Google Scholar] [CrossRef]
- Li, H. Study on kinetic and thermodynamics for heating modified sepiolite adsorbing acid blue 62 from aqueous solution. Pop. Sci. Technol. 2017, 19, 36–38. (In Chinese) [Google Scholar]
- Miura, A.; Nakazawa, K.; Takei, T.; Kumada, N.; Kinomura, N.; Ohki, R.; Koshiyama, H. Acid-, base-, and heat-induced degradation behavior of Chinese sepiolite. Ceram. Int. 2012, 38, 4677–4684. (In Chinese) [Google Scholar] [CrossRef]
- Valentin, J.L.; Lopez-Manchado, M.A.; Rodriguez, A.; Posadas, P.; Ibarra, L. Novel anhydrous unfolded structure by heating of acid pre-treated sepiolite. Appl. Clay Sci. 2007, 36, 245–255. [Google Scholar] [CrossRef]
- Chen, W.; Ma, L.; Liu, H. Adsorption capacity of magnetic modified sepiolite humic acid in source water. J. Hohai Univ. (Nat. Sci.) 2017, 45, 109–115. (In Chinese) [Google Scholar]
- Li, C.; Xia, Q.; Dai, B. Research on and modification and application of sepiolite in Lead and Zinc contained wastewater treatment. Plat. Finish. 2015, 37, 19–26. (In Chinese) [Google Scholar]
- Li, C.; Xia, Q.; Cao, Y.; Dai, B.; Song, F.; Liu, Z.; Ge, H. Treatment of nickel-containing wastewater with magnetic modified sepiolite. Electroplat. Finish. 2015, 1, 47–52. (In Chinese) [Google Scholar]
- Yu, T.; Dai, Y.; Wang, W.; Jia, M.; Li, F.; Gong, M. Adsorption characteristic and mechanism of heavy metals onto magnetically modified sepiolite. Environ. Chem. 2013, 32, 1566–1570. (In Chinese) [Google Scholar]
- Zhang, Q. Study on Adsorption Characteristic of Sepiolite. Master’s Thesis, Hebei University of Technology, Tianjin, China, 2002. [Google Scholar]
- Zhou, Q. Synthesis of Organo-functionalized Clay Minerals and Their Adsorption Performance for Heavy Metal Ions. Ph.D. Thesis, China University of Geosciences, Beijing, China, 2016. [Google Scholar]
- Liang, X.; Xu, Y.; Sun, G.; Wang, L.; Sun, Y.; Yang, S.; Xu, Q. Preparation and characterization of mercapto functionalized sepiolite and their application for sorption of lead and cadmium. Chem. Eng. J. 2011, 174, 436–444. [Google Scholar] [CrossRef]
- Liang, X.; Xu, Y.; Wang, L.; Sun, Y.; Lin, D.; Sun, Y.; Qin, X.; Wan, Q. Sorption of Pb2+ on mercapto functionalized sepiolite. Chemosphere 2013, 90, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Chen, B.; Zhang, J.; Liu, J. Adsorption of Hg (II) in water by sulfydryl-modified sepiolite. Environ. Sci. 2016, 37, 2187–2194. (In Chinese) [Google Scholar]
- Jin, S.; Yang, W.; Tang, M. Study on surface modification and application of sepiolite. Non-Met. Mines 2001, 4, 23–24. (In Chinese) [Google Scholar]
- Luo, D.; Yi, P.; Chen, A.; Shi, H. Adsorption of modified unchanged meerschaum on Pb2+, Hg2+ and Cd2+ in wastewater. Technol. Water Treat. 2003, 29, 89–91. (In Chinese) [Google Scholar]
- Huang, J.; Wu, Z.; Chen, L.; Sun, Y. The sorption of Cd (II) and U (VI) on sepiolite: a combined experimental and modeling studies. J. Mol. Liquids 2015, 209, 706–712. [Google Scholar] [CrossRef]
- Xu, Y.; Liang, X.; Sun, G.; Sun, Y.; Qin, X.; Wang, L. Surface chemical characteristics of sepiolites and their adsorption mechanisms of Pb2+, Cd2+ and Cu2+. J. Agro-Environ. Sci. 2009, 28, 2057–2063. [Google Scholar]
- Xu, Y.; Liang, X.; Sun, G.; Sun, Y.; Qin, X.; Wang, L.; Dai, X. Effects of acid and heating treatments on the structure of sepiolite and its adsorption of lead and cadmium. Environ. Sci. 2010, 31, 1560–1567. (In Chinese) [Google Scholar]
- Wang, W.; Dai, Y.; Jia, M.; Li, X.; Du, T. Surface point of zero charge of magnetic sepiolite and adsorption characteristics of cadmium. Environ. Chem. 2012, 31, 1691–1696. (In Chinese) [Google Scholar]
- Xie, J. Study on the Adsorption Characteristics of Sulfydryl-Modified Sepiolite to Hg (II) and Cd (II). Master’s Thesis, Southwest University, Chongqing, China, 2016. (In Chinese). [Google Scholar]
- Eren, E.; Gumus, H. Characterization of the structural properties and Pb (II) adsorption behavior of iron oxide coated sepiolite. Desalination 2011, 273, 276–284. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Suzuki, N.; Sato, K.; Fukata, N.; Murakami, M.; Shimizu, T. Active mercury (II) ion removal: Stoichiometrically controlled thiol-functionalized mesoporous silica by a mass production spray dry system. Bull. Chem. Soc. Jpn. 2009, 82, 1039–1043. [Google Scholar] [CrossRef]
- Chen, B. Adsorption of Pb (II) by Sodium Dodecyl Benzene Sulfonate Modified Sepiolite. Master’s Thesis, Hunan University, Changsha, China, 2013. (In Chinese). [Google Scholar]
- Liu, Y.; Jiang, M. Treatment of mult-metals wastewater using sepiolite modified with acid and heating. Guangzhou Chem. Ind. 2011, 21, 137–139. (In Chinese) [Google Scholar]
- Marjanović, V.; Lazarević, S.; Janković-Častvan, I.; Potkonjak, B.; Janaćković, Đ.; Petrović, R. Chromium (VI) removal from aqueous solutions using mercaptosilane functionalized sepiolites. Chem. Eng. J. 2010, 166, 198–206. [Google Scholar] [CrossRef]
- Fu, R.; Yang, Y.; Xu, Z.; Zhang, X.; Guo, X.; Bi, D. The removal of chromium (VI) and lead (II) from groundwater using sepiolite-supported nanoscale zero-valent iron (S-NZVI). Chemosphere 2015, 138, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Marjanović, V.; Lazarević, S.; Janković-Častvan, I.; Jokić, B.; Janaćković, D.; Petrović, R. Adsorption of chromium (VI) from aqueous solutions onto amine-functionalized natural and acid-activated sepiolites. Appl. Clay Sci. 2013, s80–81, 202–210. [Google Scholar] [CrossRef]
- Kocaoba, S. Adsorption of Cd (II), Cr (III) and Mn (II) on natural sepiolite. Desalination 2009, 244, 24–30. [Google Scholar] [CrossRef]
- Fang, J. Preparation of Co-Fe-Sepiolite Sorbent and Its Regenerated by High Temperature Gas Catalytic Oxidation Method. Master’s Thesis, Guangxi Normal University, Guilin, China, 2010. [Google Scholar]
- Feng, J. Study on the Modification of Sepiolite and Its Regeneration by Catalytic Oxidation Process Using High Temperature Gas Flow. Master’s Thesis, Guangxi Normal University, Guilin, China, 2014. [Google Scholar]
- Jia, N.; Wang, H.; Huo, J. The study on adsorption of Zn2+ by modified meerschaum. Chinamining Mag. 2006, 4, 70–72. (In Chinese) [Google Scholar]
- Liang, K.; Wang, D.; Long, L.; Xi, C. Study on the recovery of gallium using from acid lixivium of zinc modified meerschaum residues. Acta Mineral. Sin. 2006, 26, 277–280. (In Chinese) [Google Scholar]
- Zheng, Y.; Xie, Y.; Wang, A. Adsorption of Pb2+ onto chitosan-grafted-poly (acrylic acid)/sepiolite composite. Environ. Sci. 2009, 30, 2575–2579. (In Chinese) [Google Scholar]
- Gaber, S.; Haija, M.A.; Priyabrata, P.; Selvaraj, M.; Banat, F. Removal of iron from industrial lean methyldiethanolamine solvent by adsorption on sepiolite. Sep. Sci. Technol. 2018, 53, 404–416. [Google Scholar] [CrossRef]
- Yu, S.; Zhai, L.; Zhong, S.; Qiu, Y.; Cheng, L.; Ren, X. Synthesis and structural characterization of magnetite/sepiolite composite and its sorptive properties for Co (II) and Cd (II). J. Taiwan Inst. Chem. Eng. 2016, 59, 221–228. [Google Scholar] [CrossRef]
- Li, S.; Dai, Y.; Li, N.; Shi, L.; Tang, W. Preparation of iron modified sepiolite and its adsorption characteristics of antimony. Technol. Water Treat. 2009, 35, 49–52. (In Chinese) [Google Scholar]
- Li, C.; Yu, J. Modification and application of sepiolite inlead and zinc smelter wastewater treatment. Sci. Technol. Eng. 2013, 13, 9153–9157. (In Chinese) [Google Scholar]
- Yan, H. The Modification of Sepiolite and Its Application in Heavy Metal Wastewater Treatment. Master’s Thesis, Northwest University, Chongqing, China, 2013. (In Chinese). [Google Scholar]
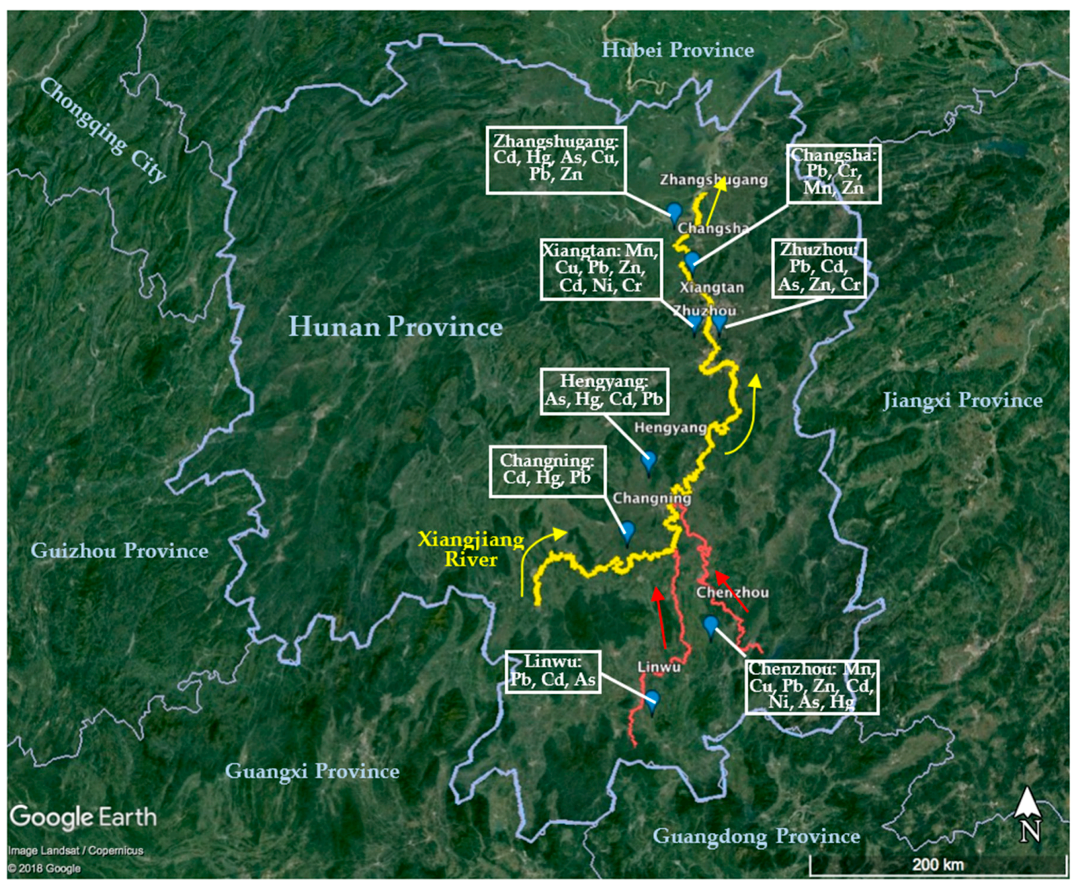
| Monitoring Locations in Xiangjiang River | PTE (s) | Pollution Condition | Reference |
|---|---|---|---|
| Upstream, Chenzhou Reach | Mn, Cu, Pb, Zn, Cd, Ni, As, Hg | Most of the elements exceed the standard e.g., Cd exceeds the discharge standard by about 1.2–9 times. | [31] |
| Gan river, Linwu (A tributary of Xiangjiang River) | Pb, Cd, As | Compared with the limit value of Environmental quality standards for surface water, Pb exceeds 109 times, Cd exceeds 242 times, As exceeds 75.8 times. | [32] |
| Changning Reach | Cd, Hg, Pb | Cd, Hg, and Pb were above the limit of the emission standard 265, 104.2, and 13.8 times, respectively. | [31] |
| Hengyang Reach | As, Hg, Pb, Cd | Compared with the limit value of Environmental quality standards for surface water, As, Hg, Pb, and Cd exceed the standard; 13.58%, 8.94%, 2.32%, and 27.16%, respectively. | [33] |
| Hengyang–Changsha Reach | As, Cd, Pb, Zn, Cu | Compared with the China Environmental Quality Standard for Soil Metals (GB15618-1995, Grade II), in the sample sites, the ratio of exceedance for As, Cd, and Pb was 13.2%, 68.5%, and 8.7% of soil samples, respectively. Cd, Pb, Zn, and Cu were much higher than their respective background values in the soil of Hunan Province, being 83.1–1178.7, 4.46–15.9, 2.88–16.1, and 3.35–6.22 times as high, respectively. | [34,35] |
| Changsha–Zhuzhou–Xiangtan Reach | Mn, Zn, Pb, Cu, Cr, Ni | Serious pollution of Pb and Zn, mild or moderate pollution of Mn and Cu in Xiawan, Zhuzhou Reach; moderate pollution of Pb and Zn, mild or moderate pollution of Cu, Mn, Ni, and Cr in Xiangtan and Changsha Reach. | [36] |
| Zhuzhou Reach | Cd, As, Pb, Zn, Cr | Cd in tobacco leaves is 6.98–37 mg/kg, Cd in cabbage is 15.4–18.3 mg/kg, Cd in rice is 1.03–1.78 mg/kg, Cd in amaranth is 6.03 mg/kg. As levels in vegetables are five times higher than normal. | [31,36] |
| Xiangtan Reach (manganese mine) | Mn, Cu, Pb, Zn, Cd, Ni | The average contents of Mn, Cu, Pb, Zn, Cd, and Ni are as follows: 7990.21 mg/kg, 66.38 mg/kg, 401.15 mg/kg, 640.32 mg/kg, 13.15 mg/kg, and 91.33 mg/kg. Their content is more than the national average worth several times or even dozens of times. | [37] |
| Entrance of Dongting Lake | Cd, Hg, As, Cu, Pb, Zn | Cd, Hg, As, Cu, Pb, and Zn in the sediments were 3.27, 0.190, 27.10, 39.8, 38.0, and 157.8 mg /kg, respectively. | [38] |
| Regeneration Methods | Metal (s) | Before Regeneration | After Regeneration | Reference |
|---|---|---|---|---|
| HCl | Ga3+ | The removal is 98.8% | The removal is 94.4% after the fourth cycle of adsorption–desorption | [83] |
| Pb2+ | equilibrium absorption capacity is 638.9 mg/g | The equilibrium absorption capacity is 489.2 mg/g after fifth cycle of adsorption–desorption | [84] | |
| HNO3 | Pb2+ | The saturated adsorption capacity is 114.2 mg/g | The saturated adsorption capacity is 97.6 mg/g | [66] |
| Hg2+ | The saturated adsorption capacity is 84.6 mg/g | The saturated adsorption capacity is 64.1 mg/g | [66] | |
| Cd2+ | The saturated adsorption capacity is 71.9 mg/g | The saturated adsorption capacity is 52.5 mg/g | [66] | |
| Fe3+ | / | Removal of iron ions was decreased less than 5% after the fourth cycle of adsorption-desorption | [85] | |
| NaOH | Co2+ | The sorption capacities of Co2+ is 16.02 mg/g, | The sorption capacities of Co2+ is 14.50 mg/g after the sixth cycle of adsorption-desorption | [86] |
| Cd2+ | The sorption capacities of Cd2+ is 12.38 mg/g | The sorption capacities of Cd2+ is 10.99 mg/g after the sixth cycle of adsorption–desorption | [86] | |
| Sb | / | Removal efficiency was decreased less than 7% after the fifth cycle of adsorption–desorption | [87] | |
| NaCl | Pb, Zn | / | Removal efficiency was decreased by 21.27% after the fifth cycle of adsorption–desorption | [88] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; Liao, L.; Hursthouse, A.; Song, N.; Ren, B. Sepiolite-Based Adsorbents for the Removal of Potentially Toxic Elements from Water: A Strategic Review for the Case of Environmental Contamination in Hunan, China. Int. J. Environ. Res. Public Health 2018, 15, 1653. https://doi.org/10.3390/ijerph15081653
Wang Z, Liao L, Hursthouse A, Song N, Ren B. Sepiolite-Based Adsorbents for the Removal of Potentially Toxic Elements from Water: A Strategic Review for the Case of Environmental Contamination in Hunan, China. International Journal of Environmental Research and Public Health. 2018; 15(8):1653. https://doi.org/10.3390/ijerph15081653
Chicago/Turabian StyleWang, Zhenghua, Lina Liao, Andrew Hursthouse, Na Song, and Bozhi Ren. 2018. "Sepiolite-Based Adsorbents for the Removal of Potentially Toxic Elements from Water: A Strategic Review for the Case of Environmental Contamination in Hunan, China" International Journal of Environmental Research and Public Health 15, no. 8: 1653. https://doi.org/10.3390/ijerph15081653
APA StyleWang, Z., Liao, L., Hursthouse, A., Song, N., & Ren, B. (2018). Sepiolite-Based Adsorbents for the Removal of Potentially Toxic Elements from Water: A Strategic Review for the Case of Environmental Contamination in Hunan, China. International Journal of Environmental Research and Public Health, 15(8), 1653. https://doi.org/10.3390/ijerph15081653