Mumps in the Vaccination Age: Global Epidemiology and the Situation in Germany
Abstract
1. Introduction
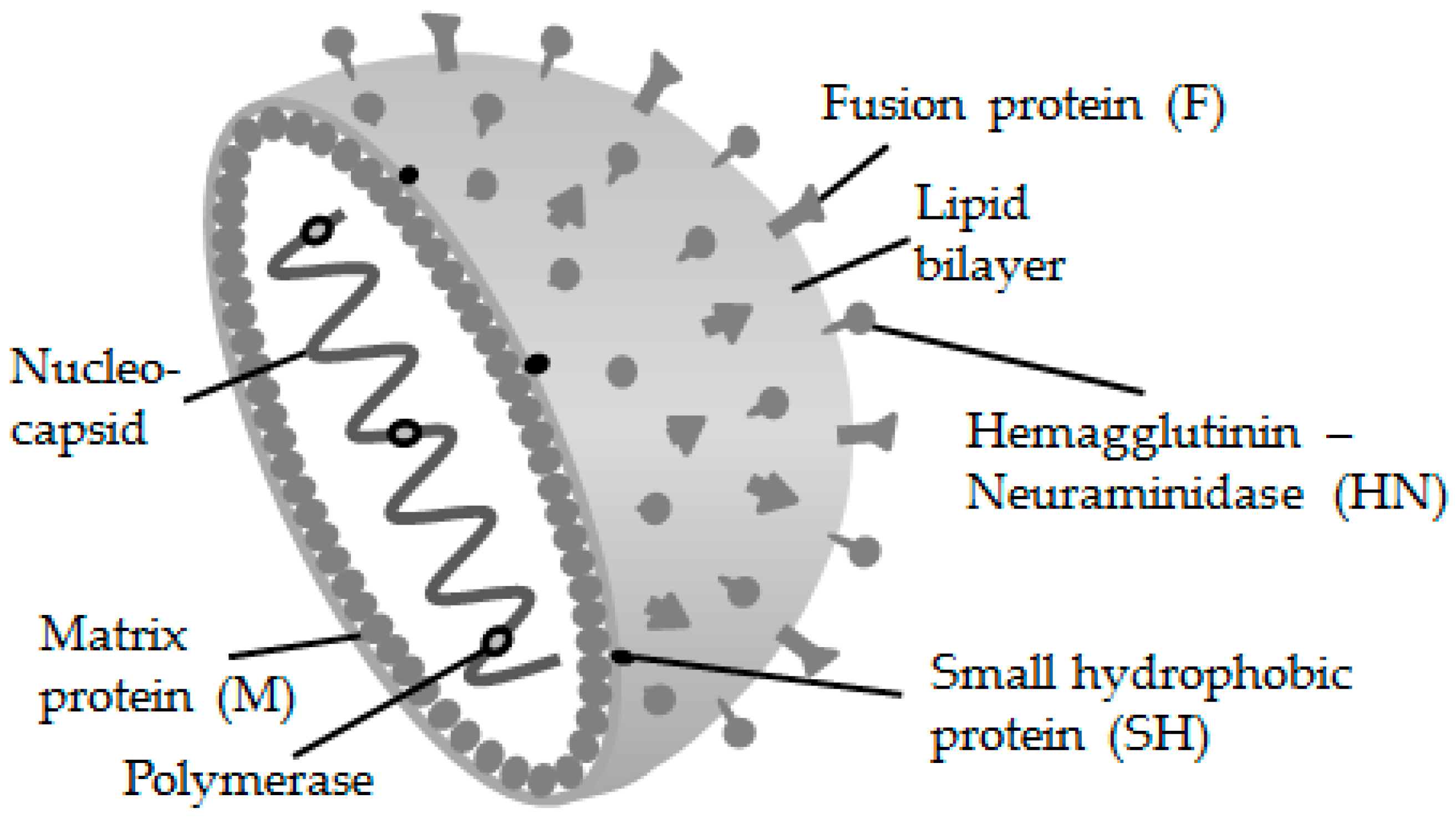
2. The Mumps Virus and Its Genotypes
3. Vaccination against the MuV
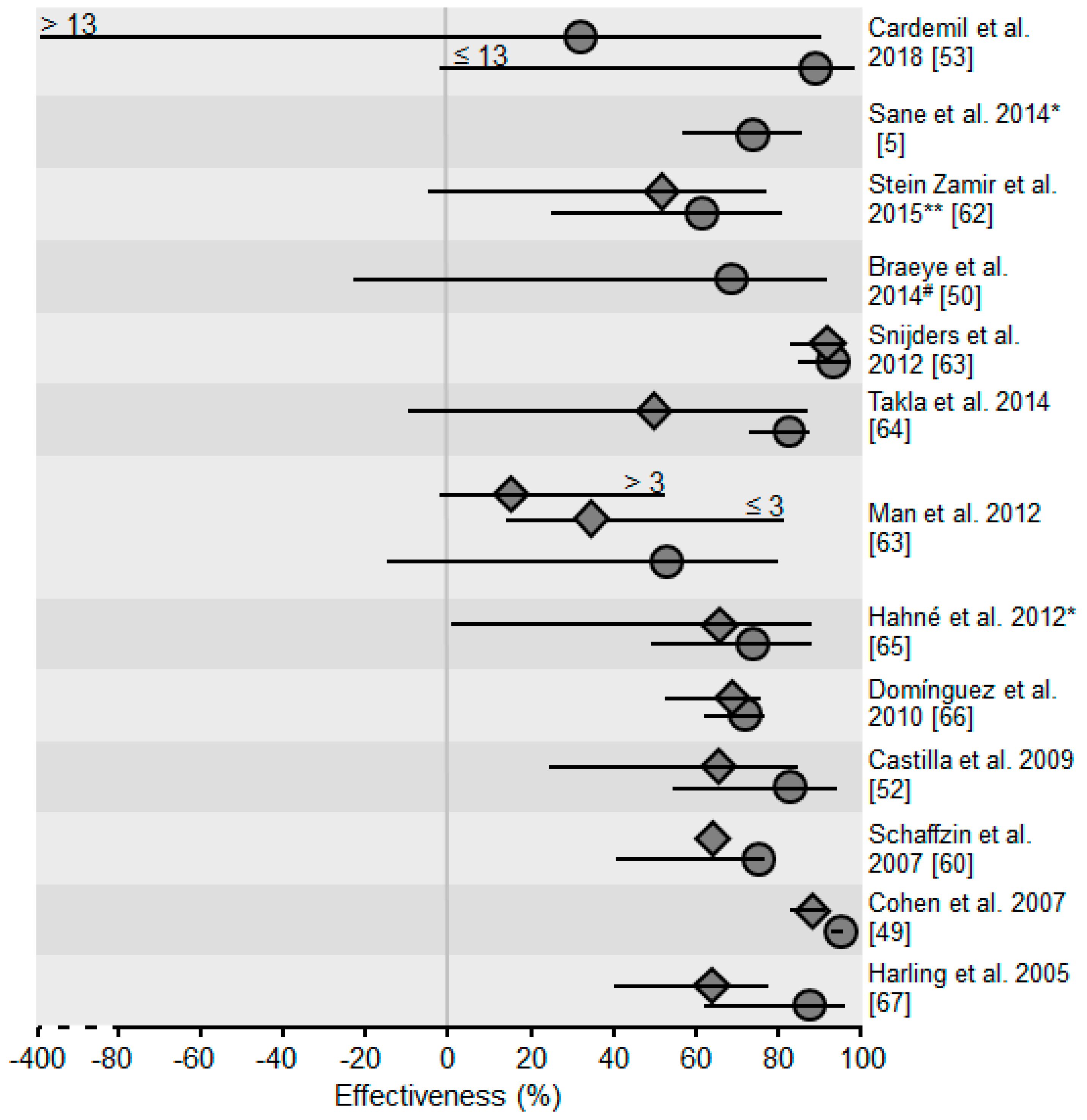
4. Outbreaks in the Vaccination Age
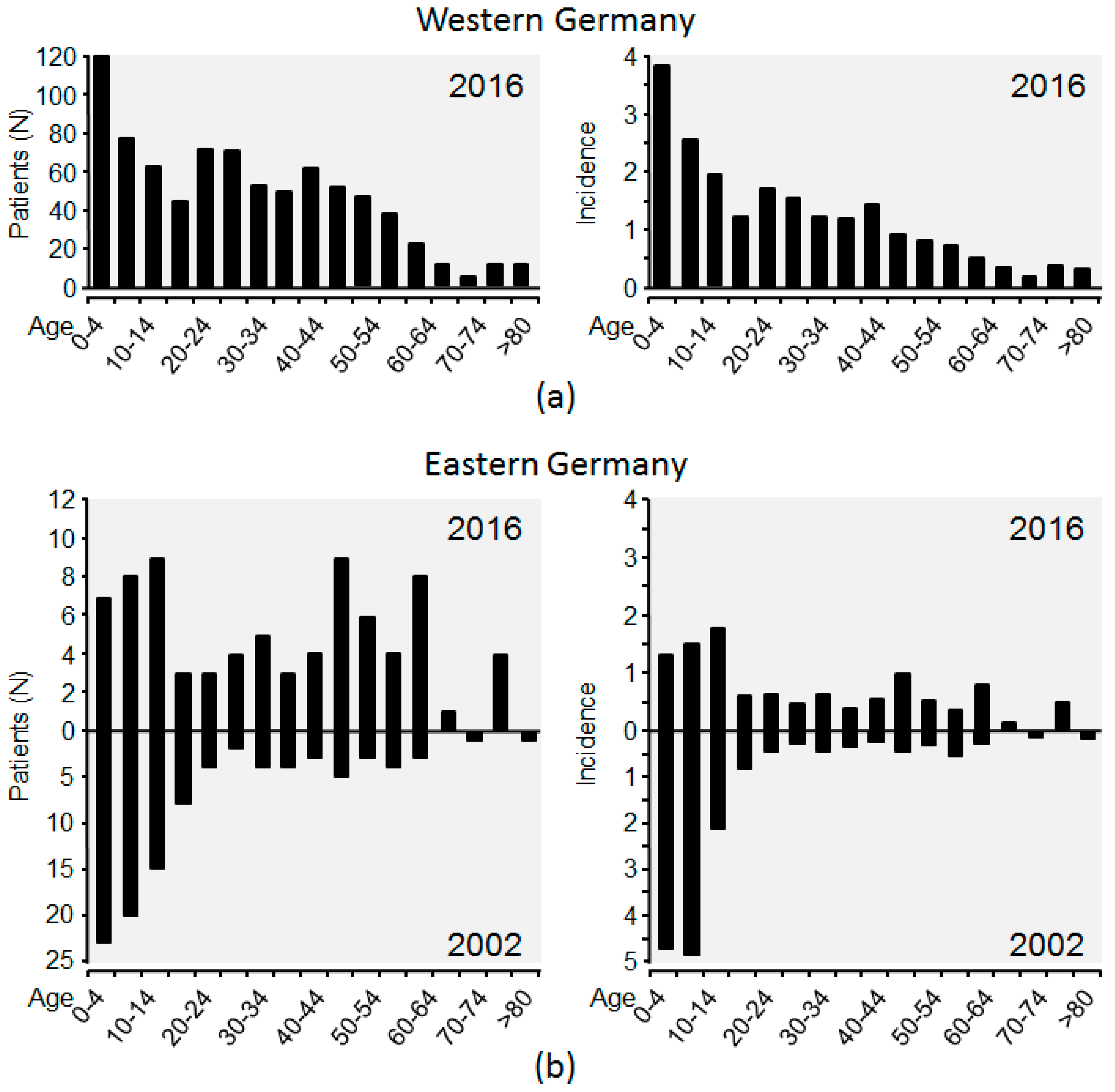
5. The Epidemiologic Situation in Germany
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dittrich, S.; Hahné, S.; van Lier, A.; Kohl, R.; Boot, H.; Koopmans, M.; van Binnendijk, R. Assessment of serological evidence for mumps virus infection in vaccinated children. Vaccine 2011, 29, 9271–9275. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Updated recommendations for isolation of persons with mumps. MMWR Morb. Mortal. Wkly. Rep. 2008, 57, 1103–1105. [Google Scholar]
- Anderson, R.M.; Crombie, J.A.; Grenfell, B.T. The epidemiology of mumps in the UK: A preliminary study of virus transmission, herd immunity and the potential impact of immunization. Epidemiol. Infect. 1987, 99, 65–84. [Google Scholar] [CrossRef] [PubMed]
- Beard, C.M.; Benson, R.C.; Kelalis, P.P.; Elveback, L.R.; Kurland, L.T. The incidence and outcome of mumps orchitis in Rochester, Minnesota, 1935 to 1974. Mayo Clin. Proc. 1977, 52, 3–7. [Google Scholar] [PubMed]
- Sane, J.; Gouma, S.; Koopmans, M.; de Melker, H.; Swaan, C.; van Binnendijk, R.; Hahné, S. Epidemic of mumps among vaccinated persons, the Netherlands, 2009–2012. Emerg. Infect. Dis. 2014, 20, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Örvell, C.; Myers, R.; Rota, P.A.; Nakayama, T.; Forcic, D.; Hiebert, J.; Brown, K.E. Genomic diversity of mumps virus and global distribution of the 12 genotypes. Rev. Med. Virol. 2015, 25, 85–101. [Google Scholar] [CrossRef] [PubMed]
- Drexler, J.F.; Corman, V.M.; Müller, M.A.; Maganga, G.D.; Vallo, P.; Binger, T.; Gloza-Rausch, F.; Rasche, A.; Yordanov, S.; Seebens, A.; et al. Bats host major mammalian paramyxoviruses. Nat. Commun. 2012, 3, 796. [Google Scholar] [CrossRef] [PubMed]
- Katoh, H.; Kubota, T.; Ihara, T.; Maeda, K.; Takeda, M.; Kidokoro, M. Cross-neutralization between human and African bat mumps viruses. Emerg. Infect. Dis. 2016, 22, 703–706. [Google Scholar] [CrossRef] [PubMed]
- Rubin, S.A.; Qi, L.; Audet, S.A.; Sullivan, B.; Carbone, K.M.; Bellini, W.J.; Rota, P.A.; Sirota, L.; Beeler, J. Antibody induced by immunization with the jeryl lynn mumps vaccine strain effectively neutralizes a heterologous wild-type mumps virus associated with a large outbreak. J. Infect. Dis. 2008, 198, 508–515. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information (NCBI). Taxonomy browser. Available online: https://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?id=1979165 (accessed on 7 April 2018).
- Galazka, A.M.; Robertson, S.E.; Kraigher, A. Mumps and mumps vaccine: A global review. Bull. WHO 1999, 77, 3–14. [Google Scholar] [PubMed]
- Cheng, W.Y.; Liu, M.T. Molecular characteristics of mumps viruses isolated in Taiwan from 2006 to 2016. Heliyon 2018, 4, e00518. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Immunization coverage. Available online: http://www.who.int/mediacentre/factsheets/fs378/en/ (accessed on 10 April 2018).
- Lewnard, J.A.; Grad, Y.H. Vaccine waning and mumps re-emergence in the United States. Sci. Transl. Med. 2018, 10, eaao5945. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control (ECDC). Vaccine Scheduler | ECDC. Available online: https://vaccine-schedule.ecdc.europa.eu (accessed on 12 April 2018).
- Koskiniemi, M.; Vaheri, A. Effect of measles, mumps, rubella vaccination on pattern of encephalitis in children. Lancet 1989, 1, 31–34. [Google Scholar] [CrossRef]
- Dayan, G.H.; Rubin, S. Mumps outbreaks in vaccinated populations: Are available mumps vaccines effective enough to prevent outbreaks? Clin. Infect. Dis. 2008, 47, 1458–1467. [Google Scholar] [CrossRef] [PubMed]
- Peltola, H.; Kulkarni, P.S.; Kapre, S.V.; Paunio, M.; Jadhav, S.S.; Dhere, R.M. Mumps outbreaks in Canada and the United States: Time for new thinking on mumps vaccines. Clin. Infect. Dis. 2007, 45, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Perez-Vilar, S.; Weibel, D.; Sturkenboom, M.; Black, S.; Maure, C.; Castro, J.L.; Bravo-Alcántara, P.; Dodd, C.N.; Romio, S.A.; de Ridder, M.; et al. Enhancing global vaccine pharmacovigilance: Proof-of-Concept study on aseptic meningitis and immune thrombocytopenic purpura following measles-mumps containing vaccination. Vaccine 2018, 36, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Cui, A.; Brown, D.W.G.; Xu, W.; Jin, L. Genetic variation in the HN and SH genes of mumps viruses: A comparison of strains from mumps cases with and without neurological symptoms. PLoS ONE 2013, 8, e61791. [Google Scholar] [CrossRef] [PubMed]
- Da Cunha, S.S.; Rodrigues, L.C.; Barreto, M.L.; Dourado, I. Outbreak of aseptic meningitis and mumps after mass vaccination with MMR vaccine using the leningrad-zagreb mumps strain. Vaccine 2002, 20, 1106–1112. [Google Scholar] [CrossRef]
- Da Silveira, C.M.; Kmetzsch, C.I.; Mohrdieck, R.; Sperb, A.F.; Prevots, D.R. The risk of aseptic meningitis associated with the leningrad-zagreb mumps vaccine strain following mass vaccination with measles-mumps-rubella vaccine, Rio Grande Do Sul, Brazil, 1997. Int. J. Epidemiol. 2002, 31, 978–982. [Google Scholar] [CrossRef] [PubMed]
- Sharma, H.J.; Aly Oun, S.; Abou Bakr, S.S.; Kapre, S.V.; Jadhav, S.S.; Dhere, R.M.; Bhardwaj, S. No demonstrable association between the leningrad-zagreb mumps vaccine strain and aseptic meningitis in a large clinical trial in Egypt. Clin. Microbiol. Infect. 2010, 16, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Cizman, M.; Mozetic, M.; Radescek-Rakar, R.; Pleterski-Rigler, D.; Susec-Michieli, M. Aseptic meningitis after vaccination against measles and mumps. Pediatr. Infect. Dis. J. 1989, 8, 302–308. [Google Scholar] [PubMed]
- Medindia. Drug—Tresivac—0.5ml (Measles, Mumps and Rubella (mmr) Vaccine) Price List or Costs of Medication|Medindia. Available online: https://www.medindia.net/Drug-Price/Measles-Mumps-and-Rubella-Mmr-Vaccine/Tresivac.html (accessed on 16 April 2018).
- Unicef.org. Measles Mumps & Rubella: Technical and Supply Information note. Available online: https://www.Unicef.Org/Supply/Files/MMR_Technical note.pdf (accessed on 28 April 2018).
- Rowhani-Rahbar, A.; Fireman, B.; Lewis, E.; Nordin, J.; Naleway, A.; Jacobsen, S.J.; Jackson, L.A.; Tse, A.; Belongia, E.A.; Hambidge, S.J.; et al. Effect of age on the risk of fever and seizures following immunization with measles-containing vaccines in children. JAMA Pediatr. 2013, 167, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, S.E.; Dover, D.C.; Simmonds, K.A.; Svenson, L.W. Risk of febrile seizures after first dose of measles-mumps-rubella-varicella vaccine: A population-based cohort study. CMAJ 2014, 186, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Streng, A.; Liese, J.G. Decline of varicella vaccination in German surveillance regions after recommendation of separate first-dose vaccination for varicella and measles-mumps-rubella. Vaccine 2014, 186, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Bauchau, V.; Van Holle, L.; Cohen, C. Modelling hospitalisation ratios for febrile convulsions and severe varicella under combined measles, mumps, rubella, and varicella (MMRV—Priorix-TetraTM) Compared to Separate MMR + V Vaccination. Drug Saf. 2015, 38, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Macartney, K.; Gidding, H.F.; Trinh, L.; Wang, H.; Dey, A.; Hull, B.; Orr, K.; McRae, J.; Richmond, P.; Gold, M.; et al. Evaluation of combination measles-mumps-rubella-varicella vaccine introduction in Australia. JAMA Pediatr. 2017, 171, 992–998. [Google Scholar] [CrossRef] [PubMed]
- Demicheli, V.; Rivetti, A.; Debalini, M.G.; Di Pietrantonj, C. Vaccines for measles, mumps and rubella in children. Evid. Based Child Health 2013, 8, 2076–2238. [Google Scholar] [CrossRef]
- France, E.K.; Glanz, J.; Xu, S.; Hambidge, S.; Yamasaki, K.; Black, S.B.; Marcy, M.; Mullooly, J.P.; Jackson, L.A.; Nordin, J.; et al. Risk of immune thrombocytopenic purpura after measles-mumps-rubella immunization in children. Pediatrics 2008, 121, e687–692. [Google Scholar] [CrossRef] [PubMed]
- Madsen, K.M.; Hviid, A.; Vestergaard, M.; Schendel, D.; Wohlfahrt, J.; Thorsen, P.; Olsen, J.; Melbye, M.A. Population-based study of measles, mumps, and rubella vaccination and autism. N. Engl. J. Med. 2002, 347, 1477–1482. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.L.; Kramarz, P.; Bohlke, K.; Benson, P.; Thompson, R.S.; Mullooly, J.; Black, S.; Shinefield, H.; Lewis, E.; Ward, J.; et al. Measles-mumps-rubella and other measles-containing vaccines do not increase the risk for inflammatory bowel disease: A case-control study from the vaccine safety datalink project. Arch. Pediatr. Adolesc. Med. 2001, 155, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Hooker, B.S.; Geier, D.; Kern, J.; Geier, M.; Boyle, C.; Boulet, S.; Schieve, L.; Cohen, R.; Blumberg, S.; Yeargin-Allsopp, M.; et al. Measles-mumps-rubella vaccination timing and autism among young African American Boys: A reanalysis of CDC data. Transl. Neurodegener. 2014, 3, 16. [Google Scholar] [CrossRef] [PubMed]
- Hooker, B.S. Retraction: Measles-mumps-rubella vaccination timing and autism among young African American boys: a reanalysis of CDC data. Transl Neurodegener. 2014, 3, 22. [Google Scholar] [CrossRef]
- Koch, J.; Takla, A. Effects of the MMR vaccination on the epidemiology of mumps in Germany. Bundesgesundheitsblatt Gesundheitsforsch. Gesundheitssch. 2013, 56, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Davidkin, I.; Jokinen, S.; Broman, M.; Leinikki, P.; Peltola, H. Persistence of measles, mumps, and rubella antibodies in an MMR-Vaccinated Cohort: A 20-year follow-up. J. Infect. Dis. 2008, 197, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Cortese, M.M.; Barskey, A.E.; Tegtmeier, G.E.; Zhang, C.; Ngo, L.; Kyaw, M.H.; Baughman, A.L.; Menitove, J.E.; Hickman, C.J.; Bellini, W.J.; et al. Mumps antibody levels among students before a mumps outbreak: In search of a correlate of immunity. J. Infect. Dis. 2011, 204, 1413–1422. [Google Scholar] [CrossRef] [PubMed]
- De Wit, J.; Emmelot, M.E.; Poelen, M.C.M.; van Binnendijk, R.S.; van der Lee, S.; van Baarle, D.; Han, W.G.H.; van Els, C.A.; Kaaijk, P. Mumps infection but not childhood vaccination induces persistent polyfunctional CD8 + T-cell memory. J. Allergy Clin. Immunol. 2018, 141, 1908–1911. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information (NCBI). Home—PubMed—NCBI. Available online: http://www.ncbi.nlm.nih.gov/pubmed (accessed on 24 March 2018).
- Seagle, E.E.; Bednarczyk, R.A.; Hill, T.; Fiebelkorn, A.P.; Hickman, C.J.; Icenogle, J.P.; Belongia, E.A.; McLean, H.Q. Measles, mumps, and rubella antibody patterns of persistence and rate of decline following the second dose of the MMR vaccine. Vaccine 2018, 36, 818–826. [Google Scholar] [CrossRef] [PubMed]
- LeBaron, C.W.; Forghani, B.; Beck, C.; Brown, C.; Bi, D.; Cossen, C.; Sullivan, B.J. Persistence of mumps antibodies after 2 doses of measles-mumps-rubella vaccine. J. Infect. Dis. 2009, 200, 888–899. [Google Scholar] [CrossRef] [PubMed]
- Briss, P.A.; Fehrs, L.J.; Parker, R.A.; Wright, P.F.; Sannella, E.C.; Hutcheson, R.H.; Schaffner, W. Sustained transmission of mumps in a highly vaccinated population: Assessment of primary vaccine failure and waning vaccine-induced immunity. J. Infect. Dis. 1994, 169, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Hersh, B.S.; Fine, P.E.M.; Kent, W.K.; Cochi, S.L.; Kahn, L.H.; Zell, E.R.; Hays, P.L.; Wood, C.L. Mumps outbreak in a highly vaccinated population. J. Pediatr. 1991, 119, 187–193. [Google Scholar] [CrossRef]
- Schwarz, N.G.; Bernard, H.; Melnic, A.; Bucov, V.; Caterinciuc, N.; An Der Heiden, M.; Andrews, N.; Pebody, R.; Aidyralieva, C.; Hahné, S. Mumps Outbreak in the Republic of Moldova, 2007–2008. Pediatr. Infect. Dis. J. 2010, 29, 703–706. [Google Scholar] [CrossRef] [PubMed]
- Vygen, S.; Fischer, A.; Meurice, L.; Mounchetrou Njoya, I.; Gregoris, M.; Ndiaye, B.; Ghenassia, A.; Poujol, I.; Stahl, J.P.; Antona, D.; et al. Waning immunity against mumps in vaccinated young adults, France 2013. Eurosurveillance 2016, 21, 30156. [Google Scholar] [CrossRef] [PubMed]
- Cohen, C.; White, J.M.; Savage, E.J.; Glynn, J.R.; Choi, Y.; Andrews, N.; Brown, D.; Ramsay, M.E. Vaccine effectiveness estimates, 2004–2005 mumps outbreak, England. Emerg. Infect. Dis. 2007, 13, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Braeye, T.; Linina, I.; De Roy, R.; Hutse, V.; Wauters, M.; Cox, P.; Mak, R. Mumps increase in Flanders, Belgium, 2012–2013: Results from temporary mandatory notification and a cohort study among university students. Vaccine 2014, 32, 4393–4398. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhao, J.; Wang, T.; Hao, L.; Ma, C.; Su, Q.; Luo, H. Mumps-containing vaccine effectiveness during outbreaks in two schools in Guangdong, China, 2012. West. Pac. Surveill. Resp. J. 2012, 3, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Castilla, J.; García Cenoz, M.; Arriazu, M.; Fernández-Alonso, M.; Martínez-Artola, V.; Etxeberria, J.; Irisarri, F.; Barricarte, A. Effectiveness of jeryl lynn-containing vaccine in Spanish children. Vaccine 2009, 27, 2089–2093. [Google Scholar] [CrossRef] [PubMed]
- Cardemil, C.V.; Dahl, R.M.; James, L.; Wannemuehler, K.; Gary, H.E.; Shah, M.; Marin, M.; Riley, J.; Feikin, D.R.; Patel, M.; et al. Effectiveness of a third dose of MMR vaccine for mumps outbreak control. N. Engl. J. Med. 2017, 377, 947–956. [Google Scholar] [CrossRef] [PubMed]
- May, M.; Rieder, C.A.; Rowe, R.J. Emergent lineages of mumps virus suggest the need for a polyvalent vaccine. Int. J. Infect. Dis. 2018, 66, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Yung, C.F.; Andrews, N.; Bukasa, A.; Brown, K.E.; Ramsay, M. Mumps complications and effects of mumps vaccination, England and Wales, 2002–2006. Emerg. Infect. Dis. 2011, 17, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Baugh, V.; Figueroa, J.; Bosanquet, J.; Kemsley, P.; Addiman, S.; Turbitt, D. Ongoing Measles outbreak in orthodox Jewish community, London, UK. Emerg. Infect. Dis. 2013, 19, 1707–1709. [Google Scholar] [CrossRef] [PubMed]
- Muhsen, K.; Shohat, T.; Aboudy, Y.; Mendelson, E.; Algor, N.; Anis, E.; Cohen, D. Sero-Prevalence of mumps antibodies in subpopulations subsequently affected: By a large scale mumps epidemic in Israel. Vaccine 2011, 29, 3878–3882. [Google Scholar] [CrossRef] [PubMed]
- Wielders, C.C.; van Binnendijk, R.S.; Snijders, B.E.; Tipples, G.A.; Cremer, J.; Fanoy, E.; Dolman, S.; Ruijs, W.L.; Boot, H.J.; de Melker, H.E.; et al. Mumps epidemic in orthodox religious low-vaccination communities in the Netherlands and Canada, 2007 to 2009. Eurosurveillance 2011, 16, 19989. [Google Scholar] [PubMed]
- Fanoy, E.B.; Cremer, J.; Ferreira, J.A.; Dittrich, S.; van Lier, A.; Hahné, S.J.H.; Boot, H.J.; Van Binnendijk, R.S. Transmission of mumps virus from mumps-vaccinated individuals to close contacts. Vaccine 2011, 29, 9551–9556. [Google Scholar] [CrossRef] [PubMed]
- Schaffzin, J.K.; Pollock, L.; Schulte, C.; Henry, K.; Dayan, G.; Blog, D.; Smith, P. Effectiveness of previous mumps vaccination during a summer camp outbreak. Pediatrics 2007, 120, e862–e868. [Google Scholar] [CrossRef] [PubMed]
- Barskey, A.E.; Schulte, C.; Rosen, J.B.; Handschur, E.F.; Rausch-Phung, E.; Doll, M.K.; Cummings, K.P.; Alleyne, E.O.; High, P.; Lawler, J.; et al. Mumps outbreak in orthodox Jewish communities in the United States. N. Engl. J. Med. 2012, 367, 1704–1713. [Google Scholar] [CrossRef] [PubMed]
- Stein Zamir, C.; Schroeder, H.; Shoob, H.; Abramson, N.; Zentner, G. Characteristics of a large mumps outbreak: Clinical severity, complications and association with vaccination status of mumps outbreak cases. Hum. Vaccines Immunother. 2015, 11, 1413–1417. [Google Scholar] [CrossRef] [PubMed]
- Snijders, B.E.P.; van Lier, A.; van de Kassteele, J.; Fanoy, E.B.; Ruijs, W.L.M.; Hulshof, F.; Blauwhof, A.; Schipper, M.; van Binnendijk, R.; Boot, H.J.; et al. Mumps vaccine effectiveness in primary schools and households, The Netherlands, 2008. Vaccine 2012, 30, 2999–3002. [Google Scholar] [CrossRef] [PubMed]
- Takla, A.; Böhmer, M.M.; Klinc, C.; Kurz, N.; Schaffer, A.; Stich, H.; Stöcker, P.; Wichmann, O.; Koch, J. Outbreak-related mumps vaccine effectiveness among a cohort of children and of young adults in Germany 2011. Hum. Vaccines Immunother. 2014, 10, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Hahné, S.; Whelan, J.; van Binnendijk, R.; Swaan, C.; Fanoy, E.; Boot, H.; de Melker, H. Mumps vaccine effectiveness against orchitis. Emerg. Infect. Dis. 2012, 18, 191–193. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, A.; Torner, N.; Castilla, J.; Batalla, J.; Godoy, P.; Guevara, M.; Carnicer, D.; Caylà, J.; Rius, C.; Jansà, J.M. Mumps vaccine effectiveness in highly immunized populations. Vaccine 2010, 28, 3567–3570. [Google Scholar] [CrossRef] [PubMed]
- Harling, R.; White, J.M.; Ramsay, M.E.; Macsween, K.F.; van den Bosch, C. The Effectiveness of the mumps component of the MMR vaccine: A case control study. Vaccine 2005, 23, 4070–4074. [Google Scholar] [CrossRef] [PubMed]
- Fiebelkorn, A.P.; Coleman, L.A.; Belongia, E.A.; Freeman, S.K.; York, D.; Bi, D.; Zhang, C.; Ngo, L.; Rubin, S. Mumps antibody response in young adults after a third dose of measles-mumps-rubella vaccine. Open Forum Infect. Dis. 2014, 1, ofu094. [Google Scholar] [CrossRef] [PubMed]
- Latner, D.R.; Parker Fiebelkorn, A.; McGrew, M.; Williams, N.J.; Coleman, L.A.; McLean, H.Q.; Rubin, S.; Hickman, C.J. Mumps virus nucleoprotein and hemagglutinin-specific antibody response following a third dose of measles mumps rubella vaccine. Open Forum Infect. Dis. 2017, 4, ofx263. [Google Scholar] [CrossRef] [PubMed]
- Ogbuanu, I.U.; Kutty, P.K.; Hudson, J.M.; Blog, D.; Abedi, G.R.; Goodell, S.; Lawler, J.; McLean, H.Q.; Pollock, L.; Rausch-Phung, E.; et al. Impact of a third dose of measles-mumps-rubella vaccine on a mumps outbreak. Pediatrics 2012, 130, e1567–e1574. [Google Scholar] [CrossRef] [PubMed]
- Marin, M.; Broder, K.R.; Temte, J.L.; Snider, D.E.; Seward, J.F. Use of combination measles, mumps, rubella, and varicella vaccine: Recommendations of the advisory committee on immunization practices (ACIP). MMWR Recomm. Rep. 2010, 59, 1–12. [Google Scholar] [PubMed]
- Nelson, G.E.; Aguon, A.; Valencia, E.; Oliva, R.; Guerrero, M.L.; Reyes, R.; Lizama, A.; Diras, D.; Mathew, A.; Monforte, M.N.; et al. Epidemiology of a mumps outbreak in a highly vaccinated island population and use of a third dose of measles-mumps-rubella vaccine for outbreak control—Guam 2009 to 2010. Pediatr. Infect. Dis. J. 2013, 32, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.; Quinlisk, P.; Weigel, A.; Riley, J.; James, L.; Patterson, J.; Hickman, C.; Rota, P.A.; Stewart, R.; Clemmons, N.; et al. Mumps outbreak in a highly vaccinated university-affiliated setting before and after a measles-mumps-rubella vaccination campaign-iowa, July 2015–May 2016. Clin. Infect. Dis. 2018, 66, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Robert Koch Institute (RKI). Survstat@RKI 2.0. Available online: https://survstat.rki.de/content/query/create.aspx (accessed on 12 April 2018).
- Mankertz, A.; Beutel, U.; Schmidt, F.J.; Borgmann, S.; Wenzel, J.J.; Ziegler, P.; Weißbrich, B.; Santibanez, S. Laboratory-based investigation of suspected mumps cases submitted to the german national reference centre for measles, mumps, and rubella, 2008 to 2013. Int. J. Med. Microbiol. 2015, 305, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Otto, W.; Mankertz, A.; Santibanez, S.; Saygili, H.; Wenzel, J.; Jilg, W.; Wieland, W.F.; Borgmann, S. Ongoing outbreak of mumps affecting adolescents and young adults in Bavaria, Germany, August to October 2010. Eurosurveillance 2010, 15, 19748. [Google Scholar] [PubMed]
- Borgmann, S.; Schwab, F.; Santibanez, S.; Mankertz, A. Mumps virus infection in vaccinated patients can be detected by an increase in specific IgG antibodies to high titres: A retrospective study. Epidemiol. Infect. 2014, 142, 2388–2396. [Google Scholar] [CrossRef] [PubMed]

| Name | Manufacturer | Viruses | Mumps Virus Strain | Dosage (TCID50) | Costs (Germany) |
|---|---|---|---|---|---|
| MMR VAXPRO | MSD * | MMR | Jeryl Lynn | >20,000 | 48.79 Euro |
| Proquad | MSD * | MMRV | Jeryl Lynn | >20,000 | 103.28 Euro |
| Priorix | GSK ** | MMR | RIT 4385 | >25,000 | 51.30 Euro |
| Priorix Tetra | GSK ** | MMRV | RIT 4385 | >25,000 | 103.47 Euro |
| MMR II | MSD * | MMR | Jeryl Lynn | >12,500 | |
| M-M-RvaxPro | MSD * | MMR | Jeryl Lynn | >12,500 | |
| Tresivac | Serum Institute of India Ltd. | MMR | L-Zagreb | >5000 | 96.25 INR *** (≈1.19 Euro) |
| Study | Observation | ||||
|---|---|---|---|---|---|
| Rubin et al., 2008 [9] | Sera of 88 children who had received their first MMR vaccine between 12 and 24 months and their second vaccine between 4 and 6 years of age had been examined. Sera for examination of MuV antibody titers (GMT) were collected a few days before and one month after the second vaccination and 10 years later when the children were 14–16 years of age. In response to the second vaccination the GMT of neutralizing antibodies to 2 different MuV viruses significantly increased by 2.6-fold and 2.0-fold, respectively. 10 years later level of neutralizing antibodies was similar to that observed before the second MMR application. | ||||
| Lewnard et al., 2017 [14] | In a meta-analysis it was calculated that immunity after receipt of any mumps vaccine persists on average 27.4 years (CI 16.7–51.1 years). Among vaccinated individuals 25% may lose protection within 7.9 years (CI: 4.7–14.7 years), 50% within 19.0 years (11.2–35.4 years), and 75% within 38.0 years (22.4–70.8 years). | ||||
| Davidkin et al., 2008 [39] | The authors examined children after receipt of the second MMR. 20 years after vaccination only 40% of the children exhibited an antibody level interpreted as positive, 34% were equivocal and 26% were negative. Within 8 and 15 years after vaccination GMT decreased from 1:2491 to 1:767 to 1:597, respectively. The decline was more pronounced in males than in females. | ||||
| Cortese et al., 2008 [40] | In an outbreak at a college in Kansas (USA) students vaccinated twice had been examined. Those who had received their second dose 10 years before the outbreak or earlier more likely got affected (OR 2.46, 95% CI: 1.25–4.82) | ||||
| Seagle et al., 2018 [43] | From children having received the second MMR GMT were determined in an observation period of up to 12 years. Decline of GMT was 9.2% per year. | ||||
| LeBaron et al., 2009 [44] | Children received the second MMR either at kindergarten or middle-school entry. Although the response to the vaccine was vigorous 12 years later antibody titers were similar to those measured before the second MMR. | ||||
| Briss et al., 1994 [45] | In an outbreak at a high school in Tennessee RR was 2.9 (95% CI: 0.7–11.6) for students vaccinated before 1988 in comparison to those vaccinated later. | ||||
| Hersh et al., 1991 [46] | Students vaccinated 4 years before an outbreak in Kansas (USA) had a higher attack rate than those vaccinated more recently (RR = 5.2, 95% CI: 0.6–30). | ||||
| Schwarz et al., 2010 [47] | In an outbreak in Moldovia VE of 1 dose vaccination declined from 91% (95% CI: 88–92%) in 2-year-olds to 72% (95% CI: 70–74%) in 15- to 19-year-olds. | ||||
| Vygen et al., 2016 [48] | In various mumps clusters in France the odds of mumps significantly increased for individuals vaccinated twice by 10% for every year that had passed since the second dose (aOR 1.10; 95% CI: 1.02–1.19). | ||||
| Cohen et al., 2007 [49] | In England VE of 1/2 doses vaccination decreased with older age of children. VE of 1 dose vaccination in 2-year-olds: 96% (95% CI: 81–99%) VE of 1 dose vaccination in 11–12-year olds: 66% (95% CI: 30–83%) VE of 2 doses vaccination in 5–6-year-olds: 99% (95% CI: 97–99.5%) VE of 2 doses vaccination in 11–12-year olds: 86% (95% CI: 74–93%) | ||||
| Braeye et al., 2014 [50] | In an outbreak at a university in Flanders (Belgium) risk of students vaccinated twice was examined. Those who had been vaccinated 10 years ago or less had a lower risk than students vaccinated more than 10 years ago (RR: 0.33, 95% CI: 0.10–1.02). | ||||
| Man et al., 2012 [51] | VE for a single dose of mumps vaccine was 65% (95% CI: 19–85%) when applied within the past 3 years 15% (95% CI: −2–25%) when applied 3–6 years before an outbreak in Chinese schools. | ||||
| Castilla et al., 2009 [52] | In an outbreak in Spain affecting children older than 15 months risk of children who had received 2 vaccine doses was higher for children who had received the second dose 3 or more years before study enrollment (OR = 10.2, 95% CI: 1.5–70.7). | ||||
| Cardemil et al., 2017 [53] | Attack rates of students during a mumps outbreak vaccinated twice depended on the time since the second vaccination: | ||||
| Time passed since the 2nd vaccination (years) | ≤2 | 3–12 | 13–15 | 16–23 | |
| Attack rate per 1000 population | 1.6 | 3.9 | 11.3 | 17.6 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beleni, A.-I.; Borgmann, S. Mumps in the Vaccination Age: Global Epidemiology and the Situation in Germany. Int. J. Environ. Res. Public Health 2018, 15, 1618. https://doi.org/10.3390/ijerph15081618
Beleni A-I, Borgmann S. Mumps in the Vaccination Age: Global Epidemiology and the Situation in Germany. International Journal of Environmental Research and Public Health. 2018; 15(8):1618. https://doi.org/10.3390/ijerph15081618
Chicago/Turabian StyleBeleni, Andrea-Ioana, and Stefan Borgmann. 2018. "Mumps in the Vaccination Age: Global Epidemiology and the Situation in Germany" International Journal of Environmental Research and Public Health 15, no. 8: 1618. https://doi.org/10.3390/ijerph15081618
APA StyleBeleni, A.-I., & Borgmann, S. (2018). Mumps in the Vaccination Age: Global Epidemiology and the Situation in Germany. International Journal of Environmental Research and Public Health, 15(8), 1618. https://doi.org/10.3390/ijerph15081618




