Prevalence of Vaccine Type Infections in Vaccinated and Non-Vaccinated Young Women: HPV-IMPACT, a Self-Sampling Study
Abstract
1. Introduction
2. Methods
2.1. Study Population and Setting
2.2. Study Procedure
2.3. Data Collection
2.4. Self-Sampling Procedure and Sample Preparation
2.5. Laboratory Analysis
2.6. Study Sample
2.7. Statistical Analyses
2.8. Ethical Approval
3. Results
3.1. Participants’ Socio-Demographic Characteristics
3.2. Vaccination Status
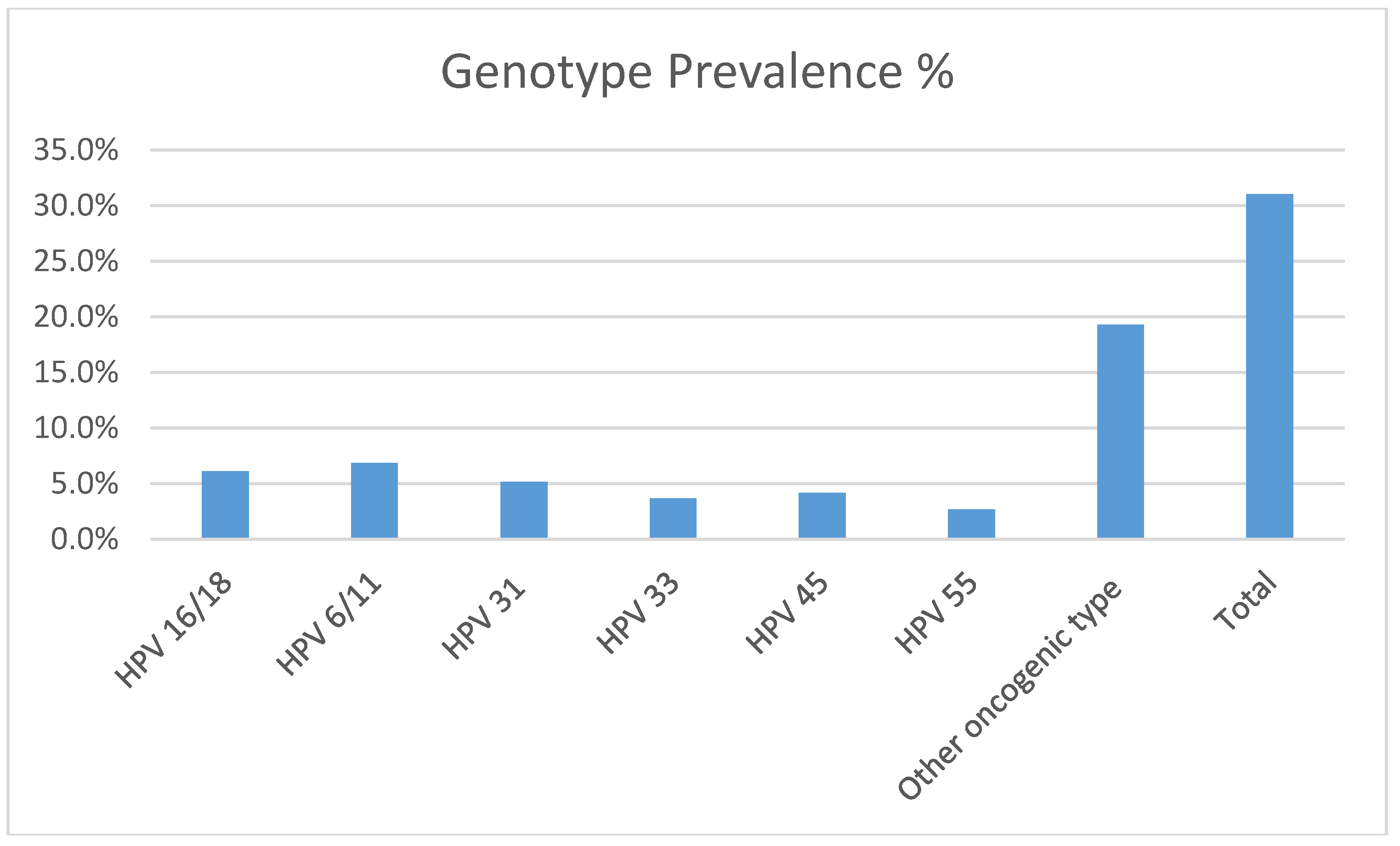
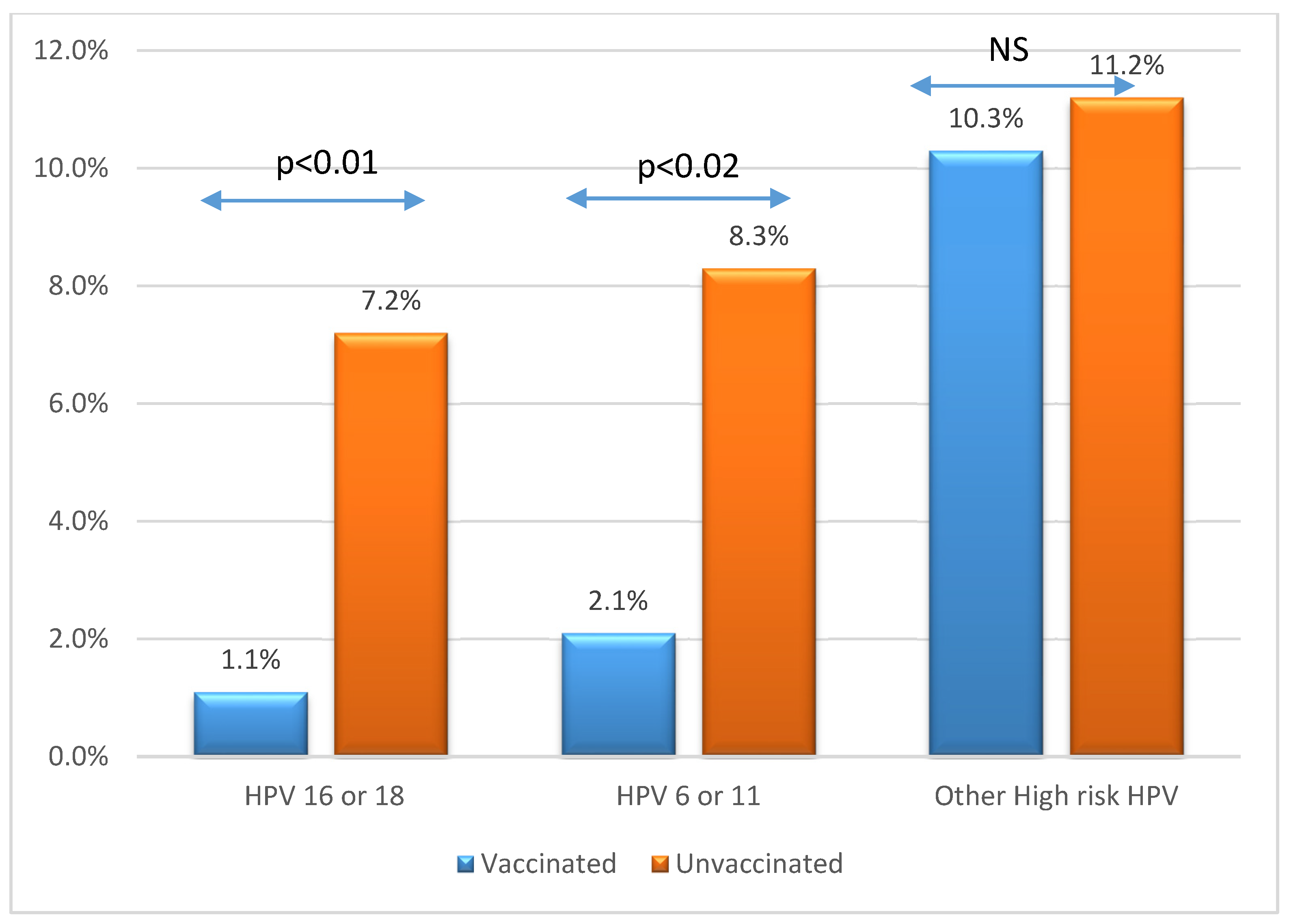
3.3. HPV Genotype Prevalence and Distributions
3.4. Relationship between HPV Infection and Sociodemographic
3.5. Acceptability of Self-Sampling
4. Discussion
5. Strength and Limitations
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sabeena, S.; Bhat, P.V.; Kamath, V.; Arunkumar, G. Global human papilloma virus vaccine implementation: An update. J. Obstet. Gynaecol. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- GLOBOCAN. Estimated Cancer Incidence, Mortality and Prevalence Worldwide. Available online: http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx (accessed on 11 February 2018).
- Garland, S.M.; Hernandez-Avila, M.; Wheeler, C.M.; Perez, G.; Harper, D.M.; Leodolter, S.; Tang, G.W.; Ferris, D.G.; Steben, M.; Bryan, J.; et al. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N. Engl. J. Med. 2007, 356, 1928–1943. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.T.; Simms, K.T.; Lew, J.B.; Smith, M.A.; Saville, M.; Canfell, K. Projected future impact of HPV vaccination and primary HPV screening on cervical cancer rates from 2017–2035: Example from Australia. PLoS ONE 2018, 13, e0185332. [Google Scholar] [CrossRef] [PubMed]
- Hariri, S.; Bennett, N.M.; Niccolai, L.M.; Schafer, S.; Park, I.U.; Bloch, K.C.; Unger, E.R.; Whitney, E.; Julian, P.; Scahill, M.W.; et al. Reduction in HPV 16/18-associated high grade cervical lesions following HPV vaccine introduction in the United States-2008–2012. Vaccine 2015, 33, 1608–1613. [Google Scholar] [CrossRef] [PubMed]
- Mesher, D.; Soldan, K.; Howell-Jones, R.; Panwar, K.; Manyenga, P.; Jit, M.; Beddows, S.; Gill, O.N. Reduction in HPV 16/18 prevalence in sexually active young women following the introduction of HPV immunisation in England. Vaccine 2013, 32, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Garland, S.M.; Cornall, A.M.; Brotherton, J.M.L.; Wark, J.D.; Malloy, M.J.; Tabrizi, S.N.; Group, V.S. Final analysis of a study assessing genital human papillomavirus genoprevalence in young Australian women, following eight years of a national vaccination program. Vaccine 2018, 36, 3221–3230. [Google Scholar] [CrossRef] [PubMed]
- Wymann, M.N.; Zographos, A.S.; Altpeter, E.; Spicher, V.M.; Low, N.; Mausezahl-Feuz, M. Human papillomavirus vaccine uptake in adolescence and adherence to cervical cancer screening in Switzerland: A national cross-sectional survey. Int. J. Public Health 2018, 63, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Jeannot, E.; Petignat, P.; Sudre, P. Successful implementation and results of an HPV vaccination program in Geneva Canton, Switzerland. Public Health Rep. 2015, 130, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, N.; Duckett, K.; Yung, C.F.; Thoon, K.C.; Sidharta, S.; Venkatachalam, I.; Chow, A.; Yoong, J. “I wouldn’t really believe statistics”—Challenges with influenza vaccine acceptance among healthcare workers in Singapore. Vaccine 2018, 36, 1996–2004. [Google Scholar] [CrossRef] [PubMed]
- Karafillakis, E.; Dinca, I.; Apfel, F.; Cecconi, S.; Wurz, A.; Takacs, J.; Suk, J.; Celentano, L.P.; Kramarz, P.; Larson, H.J. Vaccine hesitancy among healthcare workers in Europe: A qualitative study. Vaccine 2016, 34, 5013–5020. [Google Scholar] [CrossRef] [PubMed]
- Ciftci, F.; Sen, E.; Demir, N.; Ciftci, O.; Erol, S.; Kayacan, O. Beliefs, attitudes, and activities of healthcare personnel about influenza and pneumococcal vaccines. Hum. Vaccines Immunother. 2018, 14, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Osborne, S.L.; Tabrizi, S.N.; Brotherton, J.M.; Cornall, A.M.; Wark, J.D.; Wrede, C.D.; Jayasinghe, Y.; Gertig, D.M.; Pitts, M.K.; Garland, S.M. Assessing genital human papillomavirus genoprevalence in young Australian women following the introduction of a national vaccination program. Vaccine 2015, 33, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Kumakech, E.; Berggren, V.; Wabinga, H.; Lillsunde-Larsson, G.; Helenius, G.; Kaliff, M.; Karlsson, M.; Kirimunda, S.; Musubika, C.; Andersson, S. Significantly Reduced Genoprevalence of Vaccine-Type HPV-16/18 Infections among Vaccinated Compared to Non-Vaccinated Young Women 5.5 Years after a Bivalent HPV-16/18 Vaccine (Cervarix(R)) Pilot Project in Uganda. PLoS ONE 2016, 11, e0160099. [Google Scholar] [CrossRef] [PubMed]
- Machalek, D.A.; Garland, S.M.; Brotherton, J.M.L.; Bateson, D.; McNamee, K.; Stewart, M.; Skinner, S.R.; Liu, B.; Cornall, A.M.; Kaldor, J.M.; et al. Very low prevalence of vaccine human papillomavirus (HPV) types among 18 to 35 year old Australian women, nine years following implementation of vaccination. J. Infect. Dis. 2018, 217, 1590–1600. [Google Scholar] [CrossRef] [PubMed]
- Carozzi, F.; Puliti, D.; Ocello, C.; Anastasio, P.S.; Moliterni, E.A.; Perinetti, E.; Serradell, L.; Burroni, E.; Confortini, M.; Mantellini, P.; et al. Monitoring vaccine and non-vaccine HPV type prevalence in the post-vaccination era in women living in the Basilicata region, Italy. BMC Infect. Dis. 2018, 18, 38. [Google Scholar] [CrossRef] [PubMed]
- Woestenberg, P.J.; King, A.J.; van der Sande, M.A.; Donken, R.; Leussink, S.; van der Klis, F.R.; Hoebe, C.J.; Bogaards, J.A.; van Benthem, B.H. No evidence for cross-protection of the HPV-16/18 vaccine against HPV-6/11 positivity in female STI clinic visitors. J. Infect. 2017, 74, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, L.E.; Liu, G.; Hariri, S.; Steinau, M.; Dunne, E.F.; Unger, E.R. Prevalence of HPV After Introduction of the Vaccination Program in the United States. Pediatrics 2016, 137, e20151968. [Google Scholar] [CrossRef] [PubMed]
- Saccucci, M.; Franco, E.L.; Ding, L.; Bernstein, D.I.; Brown, D.; Kahn, J.A. Non-Vaccine-Type Human Papillomavirus Prevalence After Vaccine Introduction: No Evidence for Type Replacement but Evidence for Cross-Protection. Sex. Transm. Dis. 2018, 45, 260–265. [Google Scholar] [PubMed]
- Mesher, D.; Soldan, K.; Lehtinen, M.; Beddows, S.; Brisson, M.; Brotherton, J.M.; Chow, E.P.; Cummings, T.; Drolet, M.; Fairley, C.K.; et al. Population-Level Effects of Human Papillomavirus Vaccination Programs on Infections with Nonvaccine Genotypes. Emerg. Infect. Dis. 2016, 22, 1732–1740. [Google Scholar] [CrossRef] [PubMed]
- Cameron, R.L.; Kavanagh, K.; Pan, J.; Love, J.; Cuschieri, K.; Robertson, C.; Ahmed, S.; Palmer, T.; Pollock, K.G. Human Papillomavirus Prevalence and Herd Immunity after Introduction of Vaccination Program, Scotland, 2009–2013. Emerg. Infect. Dis. 2016, 22, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Arbyn, M.; Verdoodt, F.; Snijders, P.J.; Verhoef, V.M.; Suonio, E.; Dillner, L.; Minozzi, S.; Bellisario, C.; Banzi, R.; Zhao, F.H.; et al. Accuracy of human papillomavirus testing on self-collected versus clinician-collected samples: A meta-analysis. Lancet Oncol. 2014, 15, 172–183. [Google Scholar] [CrossRef]
- Nelson, E.J.; Maynard, B.R.; Loux, T.; Fatla, J.; Gordon, R.; Arnold, L.D. The acceptability of self-sampled screening for HPV DNA: A systematic review and meta-analysis. Sex. Transm. Infect. 2017, 93, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Goggin, P.; Sauvageau, C.; Gilca, V.; Defay, F.; Lambert, G.; Mathieu, C.S.; Guenoun, J.; Comete, E.; Coutlee, F. Low prevalence of vaccine-type HPV infections in young women following the implementation of a school-based and catch-up vaccination in Quebec, Canada. Hum. Vaccines Immunother. 2018, 14, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.U.H.; Rebolj, M.; Ejegod, D.M.; Pedersen, H.; Rygaard, C.; Lynge, E.; Harder, E.; Thomsen, L.T.; Kjaer, S.K.; Bonde, J. Prevalence of Human Papillomavirus in Self-Taken Samples from Screening Nonattenders. J. Clin. Microbiol. 2017, 55, 2913–2923. [Google Scholar] [CrossRef] [PubMed]
- Riesen, M.; Garcia, V.; Low, N.; Althaus, C.L. Modeling the consequences of regional heterogeneity in human papillomavirus (HPV) vaccination uptake on transmission in Switzerland. Vaccine 2017, 35, 7312–7321. [Google Scholar] [CrossRef] [PubMed]
- Jacot-Guillarmod, M.; Pasquier, J.; Greub, G.; Bongiovanni, M.; Achtari, C.; Sahli, R. Impact of HPV vaccination with Gardasil(R) in Switzerland. BMC Infect. Dis. 2017, 17, 790. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Study Population (n = 409) | |||
|---|---|---|---|---|
| n | % or Mean | 95% Confidence Interval | ||
| Age (mean years) | 24 | 21.1 | 27.2 | |
| Recruitment site | ||||
| Faculty of medicine | 225 | 55.0% | 50.2 | 59.8 |
| School of health sciences | 184 | 45.0% | 40.2 | 49.8 |
| Country of birth | ||||
| Switzerland | 365 | 89.2% | 85.9 | 92 |
| France | 30 | 7.3% | 5.1 | 10.2 |
| Other European country | 10 | 2.4% | 1.2 | 4.3 |
| other country | 4 | 1.0% | 0.3 | 2.3 |
| Tobacco smoking | ||||
| Yes, every day | 35 | 8.6% | 6.1 | 11.6 |
| Yes, but not every day | 45 | 11.0% | 8.2 | 14.3 |
| No, never | 329 | 80.4% | 76.4 | 84 |
| Have you ever had sexual intercourse | ||||
| Yes | 399 | 97.6% | 95.7 | 98.7 |
| No | 10 | 2.4% | 1.2 | 4.3 |
| Your age at your first sexual intercourse (mean years) | ||||
| Average | 17 | 16.7 | 17.2 | |
| How many sexual partners did you have in your life (mean number of partner) | ||||
| Average | 5.3 | 4.5 | 6.2 | |
| Do you use condoms as a means of protection/contraception | ||||
| Never | 120 | 29.3% | 22.7 | 31.4 |
| Sometimes | 90 | 22.0% | 18.2 | 26.2 |
| Often | 100 | 24.4% | 20.5 | 28.8 |
| Always | 99 | 24.2% | 20.2 | 28.4 |
| Have you been vaccinated against HPV | ||||
| Yes | 284 | 69.4% | 64.8 | 73.7 |
| No | 125 | 30.6% | 26.4 | 35.2 |
| How many doses of HPV vaccine have you received (only for vaccinated women n = 284) | ||||
| One | 20 | 7.0% | 7.4 | 14.5 |
| two | 60 | 21.1% | 16.7 | 26.2 |
| Three | 204 | 71.8% | 66.4 | 76.8 |
| Your age when you receive the first dose of the HPV vaccine (mean years) | ||||
| Average | 14.8 | 14.2 | 15.6 | |
| Have you checked your vaccination record to answer previous questions | ||||
| Yes | 220 | 53.8% | 49 | 58.6 |
| No | 189 | 46.2% | 41.4 | 51.1 |
| In general, do you think that HPV vaccination is a vaccination: | ||||
| More important than others | 14 | 3.4% | 1.9 | 5.5 |
| Less important than others | 74 | 18.1% | 14.6 | 22 |
| As important as the others | 321 | 78.5% | 74.3 | 82.3 |
| Would you recommend to your family/friends this self-sampling as monitoring vaccination | ||||
| Yes | 367 | 89.7% | 86.5 | 92.4 |
| No | 42 | 10.3% | 7.6 | 13.5 |
| In case of positivity of your self-collection, we authorize you to contact you again | ||||
| Yes | 405 | 99.0% | 97.6 | 99.7 |
| No | 4 | 1.0% | 0.3 | 2.3 |
| OR | 95% CI | Adjusted OR | 95% CI | |
|---|---|---|---|---|
| Recruitment site | ||||
| Faculty of medicine | 1 | - | 1 | - |
| School of health sciences | 1.3 | 0.9–1.7 | 1.4 | 0.9–1.8 |
| Country of birth | ||||
| Switzerland | 1 | - | 1 | - |
| France | 1.2 | 0.5–1.9 | 0.9 | 0.3–1.6 |
| Other European country | 3.4 | 2.1–3.7 | 3.8 | 2.4–4.1 |
| other country | 4.9 | 1.7–8.1 | 4.4 | 1.3–7.6 |
| Tobacco smoking | ||||
| yes, every day | 1.7 | 0.7–2.7 | 1.5 | 0.5–3.1 |
| yes, but not every day | 1.5 | 0.8–2.7 | 1.7 | 0.9–2.8 |
| No, never | 1 | - | 1 | - |
| Have you ever had sexual intercourse | ||||
| Yes | 7.8 | 6.7–8.9 | 7.2 | 6.2–8.5 |
| No | 1 | - | 1 | - |
| How many sexual partners did you have in your life (mean number of partner) | ||||
| 0 | 0.2 | 0.01–0.35 | 0.3 | 0.01–0.37 |
| 1 | 1 | - | 1 | - |
| 2–5 | 3.6 | 2.1–6 | 3.3 | 2.3–6.3 |
| >5 | 9.8 | 5.4–14.2 | 7.8 | 2.4–12.2 |
| Do you use condoms as a means of protection/contraception | ||||
| Never | 6.3 | 4.7–7.9 | 6.6 | 4.8–8.2 |
| Sometimes | 7.9 | 6.8–9 | 7.5 | 6.3–8.7 |
| Often | 2.6 | 0.9–5.2 | 2.5 | 0.9–5.2 |
| Always | 1 | - | 1 | - |
| Have you been vaccinated against HPV | ||||
| Yes | 11.2 | 7.1–15.2 | 8.9 | 5.9–13.2 |
| No | 1 | - | 1 | - |
| How many doses of HPV vaccine have you received | ||||
| One | 1.4 | 0.7–2.1 | 1.5 | 0.7–2.1 |
| two | 1.1 | 0.3–1.9 | 1.3 | 0.5–2.2 |
| Three | 1 | - | 1 | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeannot, E.; Viviano, M.; De Pree, C.; Amadane, M.; Kabengele, E.; Vassilakos, P.; Petignat, P. Prevalence of Vaccine Type Infections in Vaccinated and Non-Vaccinated Young Women: HPV-IMPACT, a Self-Sampling Study. Int. J. Environ. Res. Public Health 2018, 15, 1447. https://doi.org/10.3390/ijerph15071447
Jeannot E, Viviano M, De Pree C, Amadane M, Kabengele E, Vassilakos P, Petignat P. Prevalence of Vaccine Type Infections in Vaccinated and Non-Vaccinated Young Women: HPV-IMPACT, a Self-Sampling Study. International Journal of Environmental Research and Public Health. 2018; 15(7):1447. https://doi.org/10.3390/ijerph15071447
Chicago/Turabian StyleJeannot, Emilien, Manuella Viviano, Charlotte De Pree, Mona Amadane, Emmanuel Kabengele, Pierre Vassilakos, and Patrick Petignat. 2018. "Prevalence of Vaccine Type Infections in Vaccinated and Non-Vaccinated Young Women: HPV-IMPACT, a Self-Sampling Study" International Journal of Environmental Research and Public Health 15, no. 7: 1447. https://doi.org/10.3390/ijerph15071447
APA StyleJeannot, E., Viviano, M., De Pree, C., Amadane, M., Kabengele, E., Vassilakos, P., & Petignat, P. (2018). Prevalence of Vaccine Type Infections in Vaccinated and Non-Vaccinated Young Women: HPV-IMPACT, a Self-Sampling Study. International Journal of Environmental Research and Public Health, 15(7), 1447. https://doi.org/10.3390/ijerph15071447





