Legionellosis Associated with Recreational Waters: A Systematic Review of Cases and Outbreaks in Swimming Pools, Spa Pools, and Similar Environments
Abstract
1. Introduction
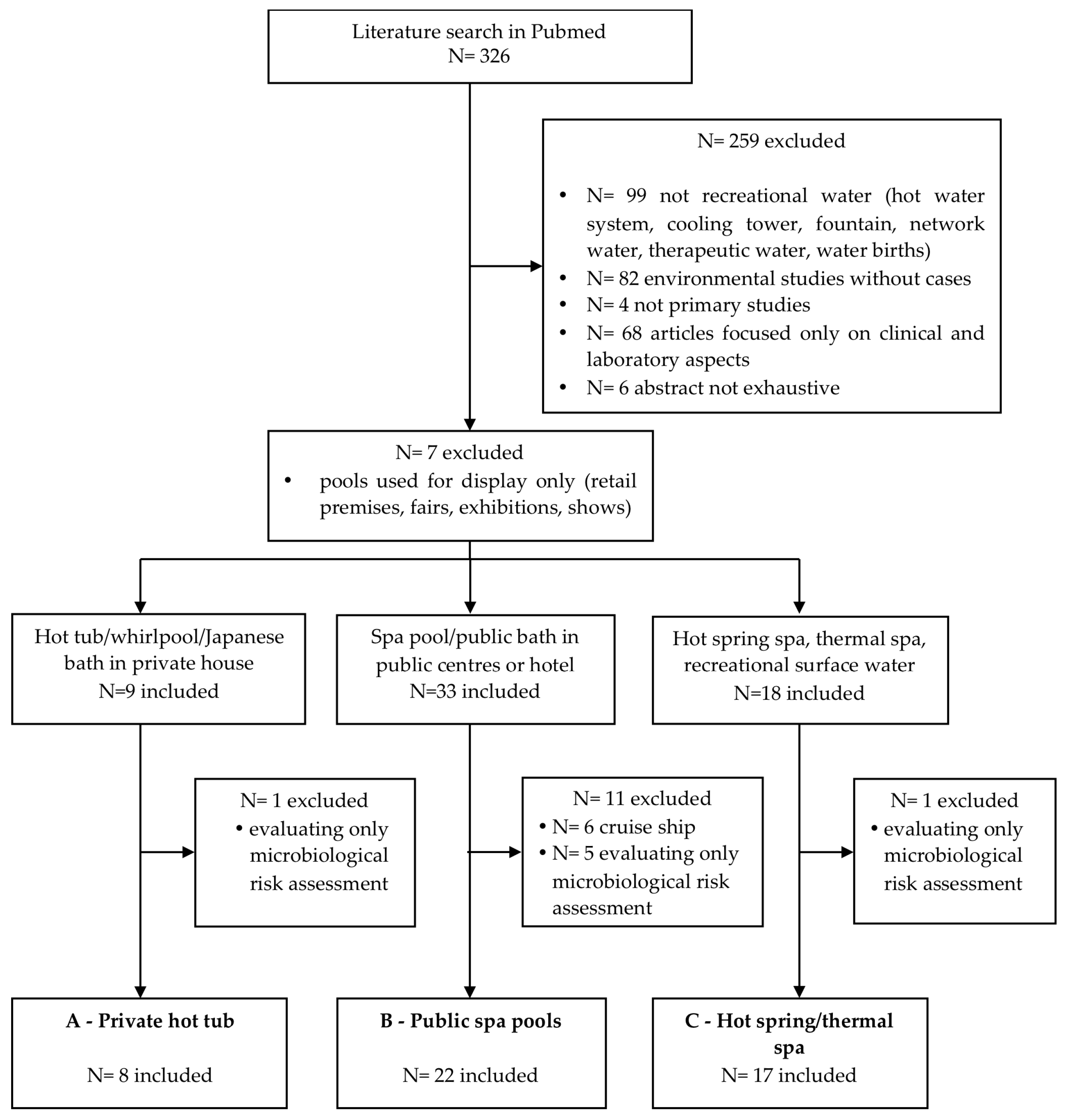
2. Materials and Methods
- Primary studies describing cases/outbreaks of LD or PF originating from recreational water.
- Not recreational water (hot water system, cooling tower, fountain, network water, therapeutic water, water births);
- environmental studies without cases;
- not primary studies;
- articles focused only on clinical and laboratory aspects;
- abstract not available/ not complete or not exhaustive;
- articles focused on pools used for display only (retail premises, fairs, exhibitions, shows);
- articles evaluating only microbiological risk assessment; and
- hot tubs or pools on cruise ships (due to a recently published systematic review) [18].
- (a)
- Private hot tub and similar facilities;
- (b)
- public pools and spas and similar facilities, generally supplied by municipal network water; and
- (c)
- spa facilities supplied by natural water, or hot spring/thermal water. Subsequently, we applied the last two exclusion criteria to each category.
3. Results
3.1. Legionellosis in Relation to Recreational Water Source
3.2. Epidemiological Investigations
3.3. Events with Sporadic Cases of Legionellosis
3.4. Outbreaks of Legionellosis
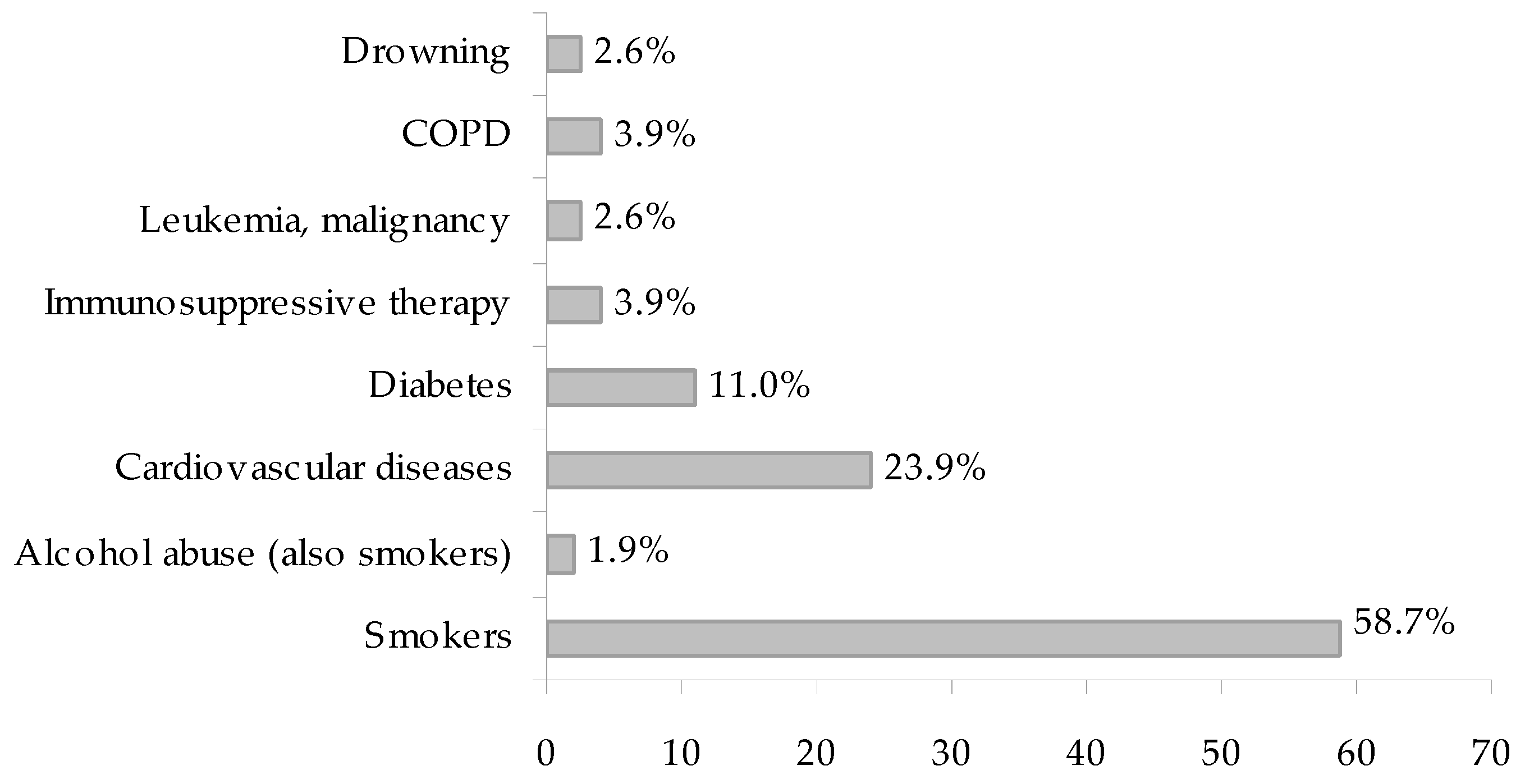
3.5. Patient Contributing Factors
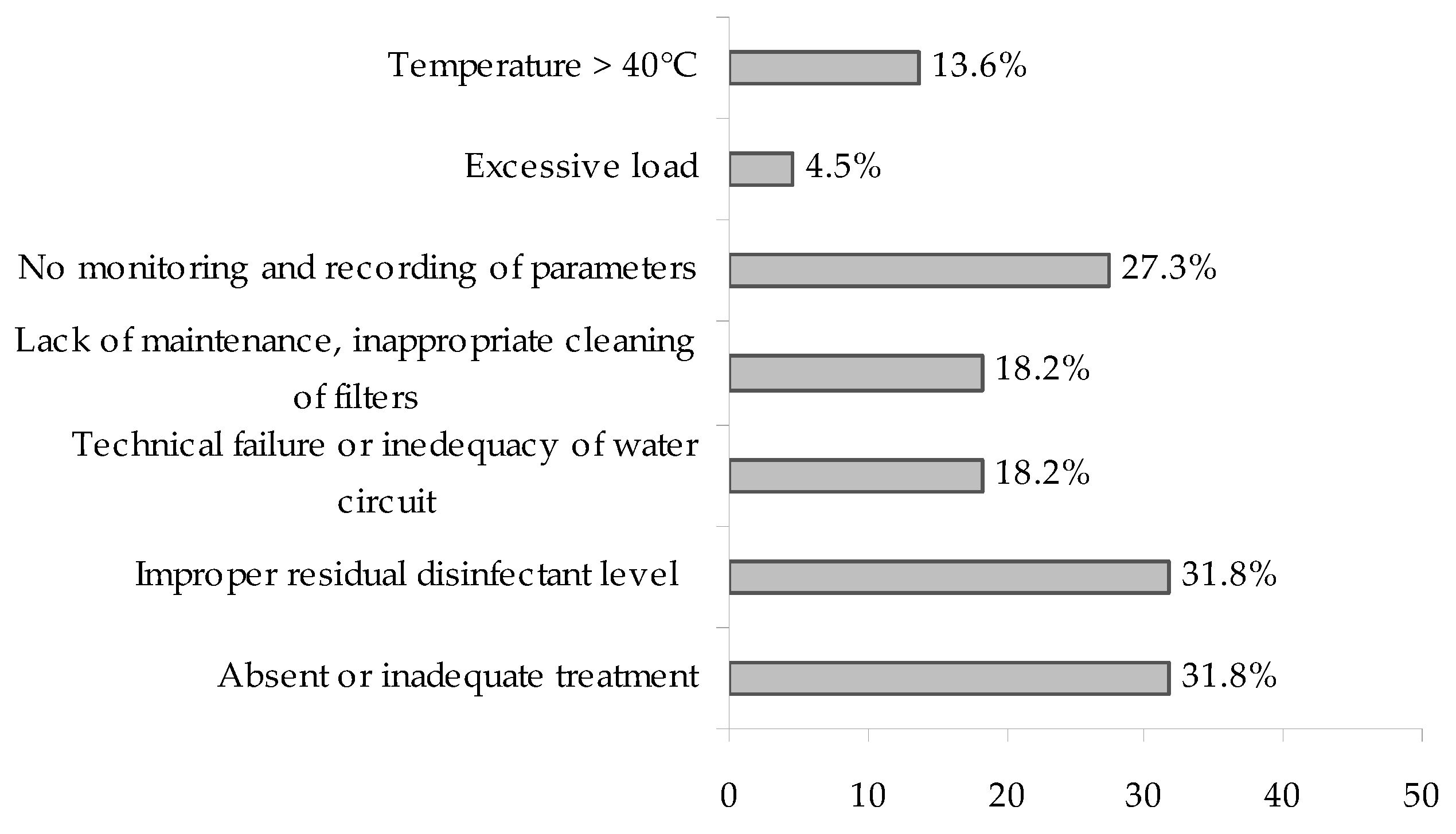
3.6. Environmental Contributing Factors
4. Discussion
4.1. Temporal and Geographical Distribution
4.2. Clinical Features and Laboratory Evidence
4.3. Recreational Water Facilities and Risk Assessment
4.4. Epidemiological Investigation and Strength of Evidence
4.5. Limitations
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| LD | Legionnaires’ Disease |
| PF | Pontiac Fever |
| cfu | colony forming unit |
References
- Muder, R.R.; You, V.L.; Woo, A.H. Mode of transmission of Legionella pneumophila: A critical review. Arch. Int. Med. 1986, 146, 1607–1612. [Google Scholar] [CrossRef]
- Springston, J.P.; Yocavitch, L. Existence and control of Legionella bacteria in building water systems: A review. J. Occup. Environ. Hyg. 2017, 14, 124–134. [Google Scholar] [CrossRef] [PubMed]
- ECDC. European Technical Guidelines for the Prevention, Control and Investigation of Infections Caused by Legionella Species. June 2017. Available online: https://ecdc.europa.eu/sites/portal/files/documents/Legionella%20GuidelinesFinal%20updated%20for%20ECDC%20corrections.pdf (accessed on 9 July 2018).
- Cunha, B.A.; Cunha, C.B. Legionnaire’s Disease and its mimics: A clinical perspective. Infect. Dis. Clin. N. Am. 2017, 31, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Borella, P.; Montagna, M.T.; Stampi, S.; Stancanelli, G.; Romano Spica, V.; Triassi, M.; Marchesi, I.; Bargellini, A.; Tato, D.; Napoli, C.; et al. Legionella contamination in hot water of Italian hotels. Appl. Environ. Microbiol. 2005, 71, 5805–5813. [Google Scholar] [CrossRef] [PubMed]
- Leoni, E.; De Luca, G.; Legnani, P.; Sacchetti, R.; Stampi, S.; Zanetti, F. Legionella waterline colonization: Detection of Legionella species in domestic, hotel and hospital hot water systems. J. Appl. Microbiol. 2005, 98, 373–379. [Google Scholar] [PubMed]
- Walser, S.M.; Gerstner, D.G.; Brenner, B.; Höller, C.; Liebl, B.; Herr, C.E. Assessing the environmental health relevance of cooling towers—A systematic review of legionellosis outbreaks. Int. J. Hyg. Environ. Health 2014, 217, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Dallolio, L.; Scuderi, A.; Rini, M.S.; Valente, S.; Farruggia, P.; Bucci Sabattini, M.A.; Pasquinelli, G.; Acacci, A.; Roncarati, G.; Leoni, E. Effect of different disinfection protocols on microbial and biofilm contamination of dental unit waterlines in community dental practices. Int. J. Environ. Res. Public Health 2014, 11, 2064–2076. [Google Scholar] [CrossRef] [PubMed]
- Ricci, M.L.; Fontana, S.; Pinci, F.; Fiumana, E.; Pedna, M.F.; Farolfi, P.; Sabattini, M.A.; Scaturro, M. Pneumonia associated with a dental unit waterline. Lancet 2012, 379, 684. [Google Scholar] [CrossRef]
- Donati, M.; Cremonini, E.; Di Francesco, A.; Dallolio, L.; Biondi, R.; Muthusamy, R.; Leoni, E. Prevalence of Simkania negevensis in chlorinated water from spa swimming pools and domestic supplies. J. Appl. Microbiol. 2015, 118, 1076–1082. [Google Scholar] [PubMed]
- Leoni, E.; Legnani, P.; Bucci Sabattini, M.A.; Righi, F. Prevalence of Legionella spp. in swimming pool environment. Water Res. 2001, 35, 3749–3753. [Google Scholar] [CrossRef]
- Leoni, E.; Sacchetti, R.; Zanetti, F.; Legnani, P.P. Control of Legionella pneumophila contamination in a system for respiratory hydrotherapy with sulphurous spa water. Inf. Control Hosp. Epidemiol. 2006, 27, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Leoni, E.; Sanna, T.; Zanetti, F.; Dallolio, L. Controlling Legionella and Pseudomonas aeruginosa re-growth in therapeutic spas: Implementation of physical disinfection treatments, including UV/ultrafiltration, in a respiratory hydrotherapy system. J. Water Health 2015, 13, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Fields, B.S.; Benson, R.F.; Besser, R.E. Legionella and Legionnaire’s disease: 25 year of investigation. Clin. Microbiol. Rev. 2002, 15, 506–526. [Google Scholar] [CrossRef] [PubMed]
- Leoni, E.; Dallolio, L.; Sanna, T.; Stagni, F.; D’Alessandro, G.; Piana, G. Impact of a risk management plan on Legionella contamination of dental unit water. Int. J. Environ. Res. Public Health 2015, 12, 2344–2358. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, K.A.; Prussin, A.J.; Ahmed, W.; Haas, C.N. Outbreaks of Legionnaires’ Disease and Pontiac Fever 2006–2017. Curr. Environ. Health Rep. 2018, 5, 263–271. [Google Scholar]
- WHO. Guidelines for Safe Recreational Water Environments. Volume 2, Swimming Pools and Similar Environments; WHO: Geneva, Switzerland, 2006; Available online: http://www.who.int/water_sanitation_health/bathing/srwe2full.pdf (accessed on 9 July 2018).
- Mouchtouri, V.A.; Rudge, J.W. Legionnaires’ Disease in hotels and passenger ships: A systematic review of evidence, sources, and contributing factors. J. Travel Med. 2015, 22, 325–337. [Google Scholar] [PubMed]
- Goldberg, D.J.; Wrench, J.G.; Collier, P.W.; Emslie, J.A.; Fallon, R.J.; Forbes, G.I.; McKay, T.M.; Macpherson, A.C.; Markwick, T.A.; Reid, D. Lochgoilhead fever: Outbreak of non-pneumonic legionellosis due to Legionella micdadei. Lancet 1989, 1, 316–318. [Google Scholar] [CrossRef]
- Faris, B.; Faris, C.; Schousboe, M.; Heath, C.H. Legionellosis from Legionella pneumophila serogroup 13. Emerg. Infect. Dis. 2005, 11, 1405–1409. [Google Scholar] [CrossRef] [PubMed]
- Molmeret, M.; Jarraud, S.; Mori, J.P.; Pernin, P.; Forey, F.; Reyrolle, M.; Vandenesch, F.; Etienne, J.; Farge, P. Different growth rates in amoeba of genotypically related environmental and clinical Legionella pneumophila strains isolated from a thermal spa. Epidemiol. Infect. 2001, 126, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Yabuuchi, E.; Agata, K. An outbreak of legionellosis in a new facility of hot spring bath in Hiuga City. Kansenshogaku Zasshi 2004, 78, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Tolentino, A.; Ahkee, S.; Ramirez, J. Hot tub legionellosis. J. Ky. Med. Assoc. 1996, 94, 393–994. [Google Scholar] [PubMed]
- Chiba, Y.; Okamoto, H.; Nagatomo, A.; Kunikane, H.; Watanabe, K. Legionnaires’ disease diagnosed by bronchoalveolar lavage. Intern. Med. 1998, 37, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, A.; Okada, J.; Kondo, H.; Takayama, Y.; Sunagawa, K.; Enari, T.; Ishii, Y. Legionella pneumonia which occurred in a private whirlpool bath user. Kansenshogaku Zasshi 2004, 78, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Ito, I.; Naito, J.; Kadowaki, S.; Mishima, M.; Ishida, T.; Hongo, T.; Ma, L.; Ishii, Y.; Matsumoto, T.; Yamaguchi, K. Hot spring bath and Legionella pneumonia: An association confirmed by genomic identification. Intern. Med. 2002, 41, 859–863. [Google Scholar] [CrossRef] [PubMed]
- Kuroki, T.; Ishihara, T.; Ito, K.; Kura, F. Bathwater-associated cases of legionellosis in Japan, with a special focus on Legionella concentrations in water. Jpn. J. Infect. Dis. 2009, 62, 201–205. [Google Scholar] [PubMed]
- Kurosawa, H.; Fujita, M.; Kobatake, S.; Kimura, H.; Ohshima, M.; Nagai, A.; Kaneko, S.; Iwasaki, Y.; Kozawa, K. A case of Legionella pneumonia linked to a hot spring facility in Gunma Prefecture, Japan. Jpn. J. Infect. Dis. 2010, 63, 78–79. [Google Scholar] [PubMed]
- Mashiba, K.; Hamamoto, T.; Torikai, K. A case of Legionnaires’ disease due to aspiration of hot spring water and isolation of Legionella pneumophila from hot spring water. Kansenshogaku Zasshi 1993, 67, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.; Fujii, S.; Shiroiwa, R.; Amemura-Maekawa, J.; Chang, B.; Kura, F.; Yamauchi, K. Isolation of Legionella rubrilucens from a pneumonia patient co-infected with Legionella pneumophila. J. Med. Microbiol. 2010, 59, 1242–1246. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, H.; Jitsurong, S.; Shiota, R.; Maruta, K.; Yoshida, S.; Yabuuchi, E. Molecular determination of infection source of a sporadic Legionella pneumonia case associated with a hot spring bath. Microbiol. Immunol. 1997, 41, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Nozue, T.; Chikazawa, H.; Miyanishi, S.; Shimazaki, T.; Oka, R.; Shimazaki, S.; Miyamoto, S. Legionella pneumonia associated with adult respiratory distress syndrome caused by Legionella pneumophila serogroup 3. Intern. Med. 2005, 44, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, E.; Kaida, H.; Izumikawa, K.; Izumikawa, K.; Hara, K.; Hirakata, Y.; Tomono, K.; Kohno, S. Two cases of Legionella pneumophila pneumonia improved by parenteral ciprofloxacin administration. Nihon Kokyuki Gakkai Zasshi 2003, 41, 211–218. [Google Scholar] [PubMed]
- Shimizu, Y.; Nagase, K.; Kadono, K.N.; Funayama, Y.; Tsurushima, Y.; Hibino, T.; Kikuchi, H.; Nagase, S.; Koyama, A. The haemodialysis patient who developed acute respiratory distress syndrome after a trip to a hot spring spa. Nephrol. Dial. Transplant. 1999, 14, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, H.; Yahagi, N.; Kasai, S.; Kitamura, S.; Otsuka, Y. A case of fatal pneumonia caused by Legionella pneumophila serogroup 6 developed after drowning in a public bath. Kansenshogaku Zasshi 1997, 71, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Su, H.P.; Tseng, L.R.; Tzeng, S.C.; Chou, C.Y.; Chung, T.C. A legionellosis case due to contaminated spa water and confirmed by genomic identification in Taiwan. Microbiol. Immunol. 2006, 50, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Shiota, R.; Takeshita, K.; Yamamoto, K.; Imada, K.; Yabuuchi, E.; Wang, L. Legionella pneumophila serogroup 3 isolated from a patient of pneumonia developed after drowning in bathtub of a hot spring spa. Kansenshogaku Zasshi 1995, 69, 1356–1364. [Google Scholar] [PubMed]
- Watson, A.M.; Boyce, T.G.; Wylam, M.E. Legionella pneumonia: Infection during immunosuppressive therapy for idiopathic pulmonary hemosiderosis. Pediatr. Infect. Dis. J. 2004, 23, 82–84. [Google Scholar] [CrossRef] [PubMed]
- Spitalny, K.C.; Vogt, R.L.; Orciari, L.A.; Witherell, L.E.; Etkind, P.; Novick, L.F. Pontiac fever associated with a whirlpool spa. Am. J. Epidemiol. 1984, 120, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Mangione, E.J.; Remis, R.S.; Tait, K.A.; McGee, H.B.; Gorman, G.W.; Wentworth, B.B.; Baron, P.A.; Hightower, A.W.; Barbaree, J.M.; Broome, C.V. An outbreak of Pontiac fever related to whirlpool use, Michigan 1982. JAMA 1985, 253, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.A.; Beebe, J.L.; Butler, J.C.; Martin, W.; Benson, R.; Hoffman, R.E.; Fields, B.S. Use of polymerase chain reaction in an epidemiologic investigation of Pontiac fever. J. Infect. Dis. 1993, 168, 769–772. [Google Scholar] [CrossRef] [PubMed]
- Lüttichau, H.R.; Vinther, C.; Uldum, S.A.; Møller, J.; Faber, M.; Jensen, J.S. An outbreak of Pontiac fever among children following use of a whirlpool. Clin. Infect. Dis. 1998, 26, 1374–1378. [Google Scholar] [PubMed]
- Fields, B.S.; Haupt, T.; Davis, J.P.; Arduino, M.J.; Miller, P.H.; Butler, J.C. Pontiac fever due to Legionella micdadei from a whirlpool spa: Possible role of bacterial endotoxin. J. Infect. Dis. 2001, 184, 1289–1292. [Google Scholar] [CrossRef] [PubMed]
- Götz, H.M.; Tegnell, A.; De Jong, B.; Broholm, K.A.; Kuusi, M.; Kallings, I.; Ekdahl, K.A. Whirlpool associated outbreak of Pontiac fever at a hotel in Northern Sweden. Epidemiol. Infect. 2001, 126, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Modi, A.; Gardner, J.; Lighton, L.; Coetzee, N. Pontiac fever outbreak associated with a spa-pool, United Kingdom, April 2008. Euro Surveill. 2008, 13, 18934. [Google Scholar] [PubMed]
- Vogt, R.L.; Hudson, P.J.; Orciari, L.; Heun, E.M.; Woods, T.C. Legionnaires’ disease and whirlpool-spa. Ann. Intern. Med. 1987, 107, 596. [Google Scholar] [CrossRef] [PubMed]
- Den Boer, J.W.; Yzerman, E.; Van Belkum, A.; Vlaspolder, F.; Van Breukelen, F.J. Legionnaire’s disease and saunas. Lancet 1998, 351, 114. [Google Scholar] [CrossRef]
- Nakadate, T.; Yamauchi, K.; Inoue, H. An outbreak of Legionnaire’s disease associated with a Japanese spa. Nihon Kokyuki Gakkai Zasshi 1999, 37, 601–607. [Google Scholar] [PubMed]
- Nakamura, H.; Yagyu, H.; Kishi, K.; Tsuchida, F.; Oh-Ishi, S.; Yamaguchi, K.; Matsuoka, T. A large outbreak of Legionnaires’ disease due to an inadequate circulating and filtration system for bath water, epidemiologic manifestations. Intern. Med. 2003, 42, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Yagyu, H.; Tsuchida, F.; Sudou, A.; Watanabe, O.; Kioi, K.; Kishi, K.; Oh-ishi, S.; Kiguchi, T.; Yamaguchi, K.; et al. A major outbreak of Legionnaire’s disease due to a public bathhouse, clinical examination. Nihon Kokyuki Gakkai Zasshi 2003, 41, 325–330. [Google Scholar] [PubMed]
- Kawano, K.; Okada, M.; Kura, F.; Amemura-Maekawa, J.; Watanabe, H. Largest outbreak of legionellosis associated with spa baths, comparison of diagnostic tests. Kansenshogaku Zasshi 2007, 81, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, N.; Matsumoto, Y.; Ashitani, J.; Katoh, S.; Nakazato, M. An outbreak of Legionnaires’ disease associated with a circulating bath water system at a public bathhouse. Nihon Kokyuki Gakkai Zasshi 2004, 42, 75–79. [Google Scholar] [PubMed]
- Matsumoto, N.; Sasaki, T.; Nakao, H.; Katoh, T.; Fukuda, Y.; Nakazato, M.; Okayama, A. An outbreak of Legionnaires’ disease associated with a circulating bathwater system at a public bathhouse. II: Radiological findings of pneumonia. J. Infect. Chemother. 2008, 14, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Okada, M.; Kawano, K.; Kura, F.; Amemura-Maekawa, J.; Watanabe, H.; Yagita, K.; Endo, T.; Suzuki, S. The largest outbreak of legionellosis in Japan associated with spa baths, epidemic curve and environmental investigation. Kansenshogaku Zasshi 2005, 79, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Matsumoto, N.; Nakao, H.; Katoh, T.; Fukuda, Y.; Nakazato, M.; Okayama, A. An outbreak of Legionnaires’ disease associated with a circulating bathwater system at a public bathhouse. I: A clinical analysis. J. Infect. Chemother. 2008, 14, 117–122. [Google Scholar] [PubMed]
- Campese, C.; Roche, D.; Clément, C.; Fierobe, F.; Jarraud, S.; de Waelle, P.; Perrin, H.; Che, D. Cluster of Legionnaires’ disease associated with a public whirlpool spa, France, April–May 2010. Euro Surveill. 2010, 15, 19602. [Google Scholar] [PubMed]
- Sánchez-Busó, L.; Guiral, S.; Crespi, S.; Moya, V.; Camaró, M.L.; Olmos, M.P.; Adrián, F.; Morera, V.; González-Morán, F.; Vanaclocha, H.; et al. Genomic investigation of a Legionellosis outbreak in a persistently colonized hotel. Front. Microbiol. 2016, 6, 1556. [Google Scholar] [CrossRef] [PubMed]
- Kuroki, T.; Amemura-Maekawa, J.; Ohya, H.; Furukawa, I.; Suzuki, M.; Masaoka, T.; Aikawa, K.; Hibi, K.; Morita, M.; Lee, K.I.; et al. Outbreak of Legionnaire’s Disease caused by Legionella pneumophila serogroups 1 and 13. Emerg. Infect. Dis. 2017, 23, 349–351. [Google Scholar] [CrossRef] [PubMed]
- Fallon, R.J.; Rowbotham, T.J. Microbiological investigations into an outbreak of Pontiac fever due to Legionella micdadei associated with use of a whirlpool. J. Clin. Pathol. 1990, 43, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.L.; Mundy, L.M.; Tucer, P.C. Hot tub legionellosis. Legionnaires’ Disease and Pontiac Fever after a point-source exposure to Legionella pneumophila. Arch. Intern. Med. 1993, 153, 2597–2599. [Google Scholar] [CrossRef] [PubMed]
- Benin, A.L.; Benson, R.F.; Arnold, K.E.; Fiore, A.E.; Cook, P.G.; Williams, L.K.; Fields, B.; Besser, R.E. An outbreak of travel-associated Legionnaires disease and Pontiac fever, the need for enhanced surveillance of travel-associated legionellosis in the United States. J. Infect. Dis. 2002, 185, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Huhn, G.D.; Adam, B.; Ruden, R.; Hilliard, L.; Kirkpatrick, P.; Todd, J.; Crafts, W.; Passaro, D.; Dworkin, M.S. Outbreak of travel-related Pontiac fever among hotel guests illustrating the need for better diagnostic tests. J. Travel Med. 2005, 12, 173–179. [Google Scholar] [PubMed]
- Burnsed, L.J.; Hicks, L.A.; Smithee, L.M.; Fields, B.S.; Bradley, K.K.; Pascoe, N.; Richards, S.M.; Mallonee, S.; Littrell, L.; Benson, R.F.; et al. Legionellosis outbreak investigation team. A large, travel-associated outbreak of legionellosis among hotel guests, utility of the urine antigen assay in confirming Pontiac fever. Clin. Infect. Dis. 2007, 44, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Foster, K.; Gorton, R.; Waller, J. Outbreak of legionellosis associated with a spa pool, United Kingdom. Euro Surveill. 2006, 11, E060921.2. [Google Scholar] [CrossRef]
- Euser, S.M.; Pelgrim, M.; den Boer, J.W. Legionnaires’ disease and Pontiac fever after using a private outdoor whirlpool spa. Scand. J. Infect. Dis. 2010, 42, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Josef, C.A.; Lee, J.; van Wijngaarden, J.; Draser, V.; Castellanis Pastoris, M. European Guidelines for Control and Prevention of Travel Associated Legionnaires’ Disease. The European Working Group for Legionella Infections (EWGLI), 2005. Available online: http://www.legionellaonline.it/linee-guidaEWGLI_gen2005.pdf (accessed on 9 July 2018).
- WHO. Legionella and the Prevention of Legionellosis; WHO: Geneva, Switzerland, 2007; Available online: http://www.who.int/water_sanitation_health/emerging/legionella.pdf (accessed on 9 July 2018).
- Bouwknegt, M.; Schijven, J.F.; Schalk, J.A.; de Roda Husman, A.M. Quantitative risk estimation for a Legionella pneumophila infection due to whirlpool use. Risk Anal. 2013, 33, 1228–1236. [Google Scholar] [PubMed]
- Azuma, K.; Uchiyama, I.; Okumura, J. Assessing the risk of Legionnaires’ disease: The inhalation exposure model and the estimated risk in residential bathrooms. Regul. Toxicol. Pharmacol. 2013, 65, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Press, E. The health hazards of saunas and spas and how to minimize them. Am. J. Public Health 1991, 81, 1034–1037. [Google Scholar] [CrossRef] [PubMed]
- Coetzee, N.; Duggal, H.; Hawker, J.; Ibbotson, S.; Harrison, T.G.; Phin, N.; Laza-Stanca, V.; Johnston, R.; Iqbal, Z.; Rehman, Y.; et al. An outbreak of Legionnaires’ disease associated with a display spa pool in retail premises, Stoke-on-Trent, United Kingdom, July 2012. Euro Surveill. 2012, 17, 20271. [Google Scholar] [PubMed]
- Den Boer, J.W.; Yzerman, E.P.; Schellekens, J.; Lettinga, K.D.; Boshuizen, H.C.; Van Steenbergen, J.E.; Bosman, A.; Van den Hof, S.; Van Vliet, H.A.; Peeters, M.F.; et al. A large outbreak of Legionnaires’ disease at a flower show, the Netherland, 1999. Emerg. Infect. Dis. 2002, 8, 37–43. [Google Scholar] [CrossRef] [PubMed]
| Strength of Evidence | Epidemiological and Microbiological Criteria |
|---|---|
| Strong |
|
| Or | |
| |
| Probable |
|
| Or | |
| |
| Possible |
|
| Characteristics of the Events | Hot Tub/Whirlpool/Japanese Bath in Private House (8 Events) | Spa Pools/Public Baths in Public Centres or Hotels (22 Events) | Hot Spring Spa, Thermal Spa, Recreational Surface Water (12 Events) | Total Recreational Waters (42 Events) |
|---|---|---|---|---|
| Number of events with single cases | 5 | 2 | 10 | 17 |
| Number of outbreaks or events with repeated cases a | 3 | 20 | 2 | 25 |
| Number of total cases | 28 | 744 | 307 | 1079 |
| Median number of cases per outbreak (range) | 6 (4–13) | 23.5 (3–170) | 148.5 (2–295) | 23 (2–295) |
| Total number of PF cases (fatal cases) | 22 (0) | 598 (0) | 0 | 620 (0) |
| Total number of LD cases (fatal cases) | 6 (1) | 146 (16) | 307 (12) | 459 (29) |
| Fatality rate on total cases (on LD cases) | 3.6% (16.7%) | 2.2% (11.0%) | 3.9% (3.9%) | 2.7% (6.3%) |
| Analytical epidemiology in outbreak investigation (% of total outbreaks) | 0 (0%) | 8 (40.0%) | 1 (50.0%) | 9 (36.0%) |
| Events with environmental investigation (% of total events) | 6 (75.0%) | 20 (90.9%) | 9 (75.0%) | 35 (83.3%) |
| Legionella spp. detected in environmental water samples (% of total events) | 4 (50.0%) | 20 (90.9%) | 8 (66.7%) | 32 (76.2%) |
| Identical Legionella genotype in clinical and environmental isolates (% of total events) | 1 (12.5%) | 6 (27.3%) | 7 (58.3%) | 14 (33.3%) |
| Strength of evidence | ||||
| Strong (%) | 1 (12.5%) | 15 (68.2%) | 7 (58.3%) | 23 (52.4%) |
| Probable (%) | 3 (37.5%) | 5 (22.7%) | 1 (8.3%) | 9 (21.4%) |
| Possible (%) | 4 (50.0%) | 2 (9.1%) | 4 (33.3%) | 10 (23.9%) |
| Pontiac Fever (1 Event) a | Legionnaires’ Disease (16 Events) b | |
|---|---|---|
| Number of cases (fatal cases) | 1 (0) | 16 (5) |
| Gender | ||
| Males | 9 | |
| Females | 6 | |
| Not reported | 1 | 1 |
| Median age (range) | 37 | 56.5 (10–88) |
| Confirmation by culture in clinical specimen | 0 | 12 (75.0%) |
| Legionella species and serogroup | ||
| L. pneumophila SG 1 | 0 | 3 (18.7%) |
| L. pneumophila SG 2 | 0 | 1 (6.2%) |
| L. pneumophila SG 3 | 0 | 2 (12.5%) |
| L. pneumophila SG 4 | 0 | 1 (6.2%) |
| L. pneumophila SG 6 | 0 | 5 (31.2%) |
| L. pneumophila SG 13 | 0 | 2 (12.5%) |
| L. pneumophila (SG not reported) | 1 (100%) | 1 (6.2%) |
| L. rubrilucens | 0 | 1 (6.2%) |
| Environmental source | ||
| Private hot tub | 1 | 4 (25.0%) |
| Public and hotel spa | 0 | 2 (12.5%) |
| Hot spring/thermal spa | 0 | 9 (56.2%) |
| Estuarine water | 0 | 1 (6.2%) |
| Legionella colonization | ||
| <1000 cfu/L | 0 | 2 (12.5%) |
| 1000–10,000 cfu/L | 0 | 2 (12.5%) |
| >10,000 cfu/L | 0 | 2 (12.5%) |
| Not reported | 1 (100%) | 11 (68.7%) |
| Identical Legionella genotype in clinical and environmental isolates | 0 | 7 (43.7%) |
| Strength of evidence | ||
| Strong (%) | 0 | 7 (43.7%) |
| Probable (%) | 1 (100%) | 2 (12.5%) |
| Possible (%) | 0 | 7 (43.7%) |
| Event No. Country, Year (Reference) | Water System | Legionella spp. (Confirmed Diagnosis Based on) | No. of Cases (Fatal Cases) | Attack Rate | Proportion of Males | Median Age (Range) | Environmental Isolates (cfu/L) | Strength of Evidence |
|---|---|---|---|---|---|---|---|---|
| 1 Vermont, US, 1981 [39] | Inn whirlpool spa | L. pneumophila SG 6 (antibody titre) | 34 (0) | 45.9% | 53.0% | 27.9 | L. pneumophila SG 1,6 L. dumoffii | Strong |
| 2 Michigan, US, 1982 [40] | Public whirlpool spa (women’s pool) | L. pneumophila SG 6 (antibody titre) | 14 (0) | 29.8% | 0 | 32 (25–39) | L. pneumophila SG 6 | Strong |
| 3 Colorado, US, 1992 [41] | Resort indoor whirlpool | L. pneumophila SG 6 (antibody titre) | 13 (0) | 38.0% | na | na | L. pneumophila SG 6 (>1,000,000) | Strong |
| 4 Denmark, 1995 [42] | Private summerhouse whirlpool | L. pneumophila SG 1 (culture, antibody titre) L. micdadei (antibody titre) | 13 (0) | 86.7% | na | na | negative samples (after whirlpool cleaning) | Possible |
| 5 Wisconsin, US, 1998 [43] | Hotel whirlpool spa | L. micdadei (antibody titre) | 45 (0) | whirlpool area: 66.0% whirlpool users: 71.0% | na | na | L. micdadei (90,000/L) | Strong |
| 6 Sweden, 1999 [44] | Hotel whirlpool spa | L. micdadei (antibody titre) | 29 (0) | whirlpool area: 71.0% whirlpool users: 88.9% | 37.9% | 41 (21–57) | negative samples | Probable |
| 7 England, 2008 [45] | Resort whirlpool spa | L. pneumophila SG 1 (antibody titre, urinary antigen) | 6 (0) | 86.0% | 0 | (24–37) | Legionella non pneumophila (100/L) | Probable |
| Event No. Country, Year (Reference) | Water System | Legionella spp. (Diagnosis Based on) | Number of Cases (Fatal Cases) | Attack Rate | Proportion of Males | Median Age (Range) | Environmental Isolates (cfu/L) | Strength of Evidence |
|---|---|---|---|---|---|---|---|---|
| 1 Vermont, US, 1987 [46] | Inn whirlpool spa | L. pneumophila SG 1 (culture, antibody titre) | 3 (0) | na | na | na | L. pneumophila SG 1,4 | Strong |
| 2 Netherlands 1992–96 [47] | Public spa sauna’s footbath | L. pneumophila SG 1 (culture) | 6 repeated cases (2) | na | 83.3% | males: 50 females: 28 | L. pneumophila SG 1 | Strong |
| 3 France 1994–97 [21] | Thermal spa | L. pneumophila SG 1 (culture) | 2 repeated cases (1) | na | 50% | 54.5 (40–69) | L. pneumophila SG 1,2,3,6,9,13 L. dumoffii | Strong |
| 4 Japan, 1996 [48] | Public Japanese spa | L. pneumophila SG 1 (antibody titre) | 3 (0) | na | na | na | L. pneumophila SG 1 | Probable |
| 5 Japan, 2000 [27] | Public bath house | L. pneumophila SG 1,6 (culture, antibody titre, urinary antigen) | 23 (2) | 0.13% | 91.3% | 67 (50–86) | L. pneumophila SG 1 (880,000) | Strong |
| 6 Japan, 2000 [49,50] | Public bath house | L. pneumophila SG 1 (culture, antibody titre, urinary antigen) | 34 (20 confirmed) (3) | 0.20% | 65.0% (only confirmed) | 62.2 (27–85) | L. pneumophila SG 1,3,5,6 (11400–84200) | Strong |
| 7 Japan, 2002 [22,51,52,53,54,55] | Hot spring bath | L. pneumophila SG 1 (culture, antibody titre, urinary antigen) | 295 including suspected cases (7) | 1.5% | 64.5% (of 76 examined) | 65 (9–95) | L. pneumophila SG 1,8 (1,600,000) L. dumoffii (5,200,000) L. londiniensis (15,000,000) | Strong |
| 8 Japan, 2003 [27] | Public bath house | L. pneumophila SG 1 (culture) | 9 (1) | 0.13% | na | 65 (52–82) | L. pneumophila SG 1 (1,300,000) | Probable |
| 9 France, 2010 [56] | Public whirlpool spa | L. pneumophila SG 1 (culture, urinary antigen) | 3 (1) | na | 33.3% | 50 (30–70) | L. pneumophila SG 1 (150,000) | Strong |
| 10 Spain, 2011–12 [57] | Hotel spa pool | L. pneumophila SG 1 (culture) | Total: 44 (6) Cluster1: 21 Cluster2: 2 Cluster3: 3 Cluster4: 18 | na | na | tourists: 71.5 hotel workers: 49.5 | L. pneumophila SG 1 L. micdadei | Strong |
| 11 Japan, 2015 [58] | Spa house (men’s pool) | L. pneumophila SG 1,13 (culture) | 7 (0) | na | 100% | 66.3 | L. pneumophila SG 1,13 | Strong |
| Event No. Country, Year (Reference) | Water System | Legionella spp. (Diagnosis Based on) | Number of Cases PF + LD (Fatal Cases) | Attack Rate | Proportion of Males | Median Age (Range) | Environmental Isolates (cfu/L) | Strength of Evidence |
|---|---|---|---|---|---|---|---|---|
| 1 Scotland, 1987–88 [19,59] | Hotel whirlpool spa | L. micdadei (antibody titre) | 169 + 1 (0) | 90.9% (LD: 0.5%) | 48.8% | 32 (2–72) | L. micdadei | Probable |
| 2 Vermont US, 1991 [60] | Private hot tub in holiday home | L. pneumophila SG 1 (antibody titre) | 5 + 1 (0) | na | na | na | not investigated | Possible |
| 3 Georgia US, 1999 [61] | Hotel whirlpool spa | L. pneumophila SG 6 (culture, antibody titre, urinary antigen) | 22 + 2 (0) | 22.0% (LD: 1.8%) | na | PF: 12 (5–31) LD: 66 (61–71) | L. pneumophila SG 6 | Strong |
| 4 Illinois US, 2002 [62] | Hotel spa area | L. micdadei L. maceachernii (antibody titre) | 49 + 1 (0) | 62.7% (LD: 1.2%) | 46% | 20 (2–58) | L. micdadei L. maceachernii L. dumoffii | Strong |
| 5 Oklaoma US, 2004 [63] | Hotel pool and hot tub area | L. pneumophila SG 1 (antibody titre, urinary antigen) | 101 + 6 (0) | 33.7% (LD: 1.9%) | PF: 43.6% LD: 100% | PF: 15 (2–65) LD: 6.5 (2–44) | L. pneumophila SG 1 | Strong |
| 6 England, 2006 [64] | Leisure club spa pool | L. pneumophila SG 1 (antibody titre, urinary antigen) | 116 + 2 (0) | na | PF: 41.4% LD: 100% | (18–85) | L. pneumophila SG 1 | Probable |
| 7 Netherlands, 2009 [65] | Private outdoor whirlpool spa | L. pneumophila SG 1 (antibody titre, urinary antigen) | 3 + 1 (1 LD) | na | PF: 66.7% LD: 0%) | PF: 54 (52–83) LD: 78 | L. pneumophila SG 1 | Probable |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leoni, E.; Catalani, F.; Marini, S.; Dallolio, L. Legionellosis Associated with Recreational Waters: A Systematic Review of Cases and Outbreaks in Swimming Pools, Spa Pools, and Similar Environments. Int. J. Environ. Res. Public Health 2018, 15, 1612. https://doi.org/10.3390/ijerph15081612
Leoni E, Catalani F, Marini S, Dallolio L. Legionellosis Associated with Recreational Waters: A Systematic Review of Cases and Outbreaks in Swimming Pools, Spa Pools, and Similar Environments. International Journal of Environmental Research and Public Health. 2018; 15(8):1612. https://doi.org/10.3390/ijerph15081612
Chicago/Turabian StyleLeoni, Erica, Federica Catalani, Sofia Marini, and Laura Dallolio. 2018. "Legionellosis Associated with Recreational Waters: A Systematic Review of Cases and Outbreaks in Swimming Pools, Spa Pools, and Similar Environments" International Journal of Environmental Research and Public Health 15, no. 8: 1612. https://doi.org/10.3390/ijerph15081612
APA StyleLeoni, E., Catalani, F., Marini, S., & Dallolio, L. (2018). Legionellosis Associated with Recreational Waters: A Systematic Review of Cases and Outbreaks in Swimming Pools, Spa Pools, and Similar Environments. International Journal of Environmental Research and Public Health, 15(8), 1612. https://doi.org/10.3390/ijerph15081612







