Effects of Ventilation Improvement on Measured and Perceived Indoor Air Quality in a School Building with a Hybrid Ventilation System
Abstract
1. Introduction
2. Materials and Methods
2.1. Building Characteristics
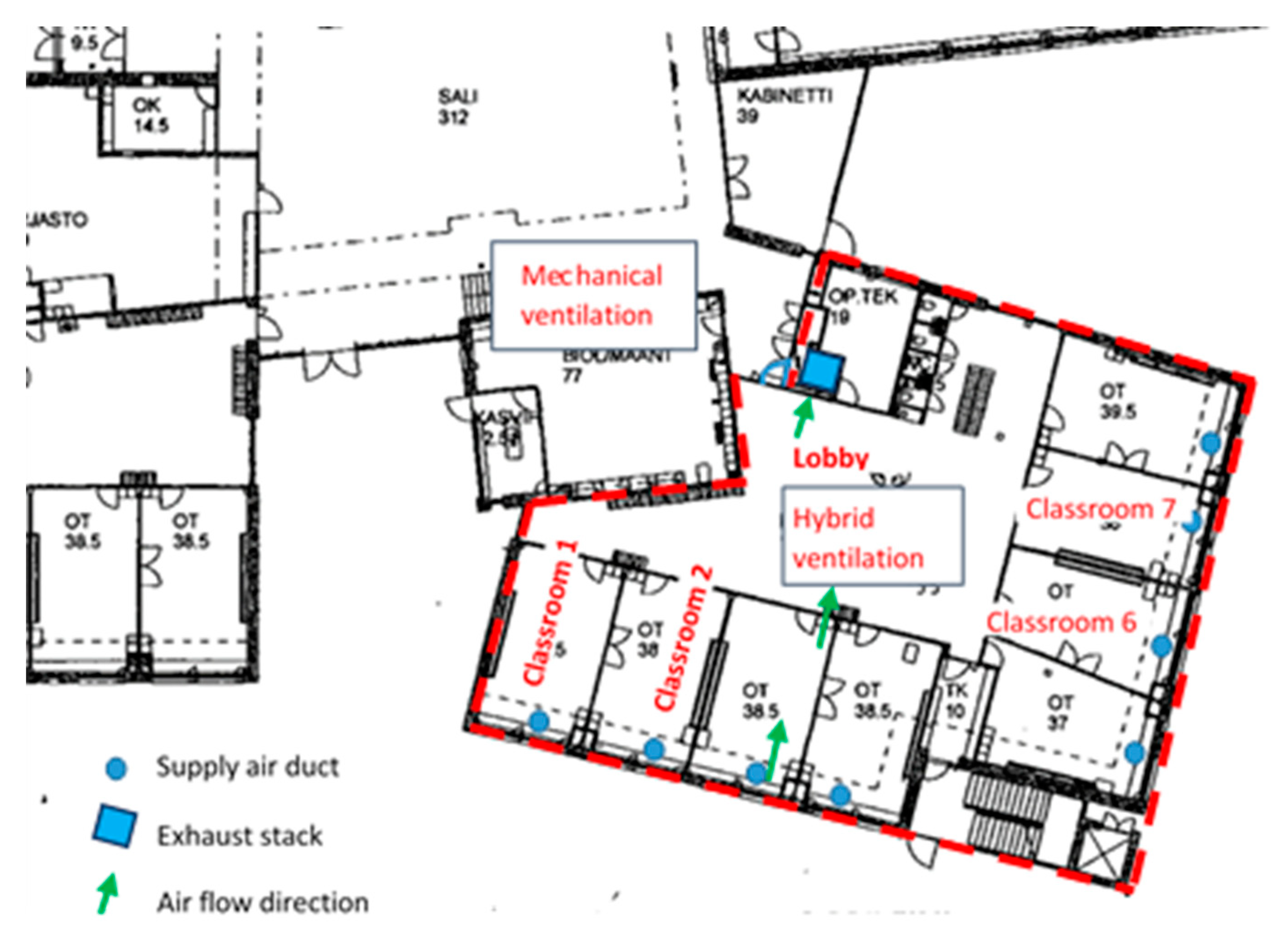
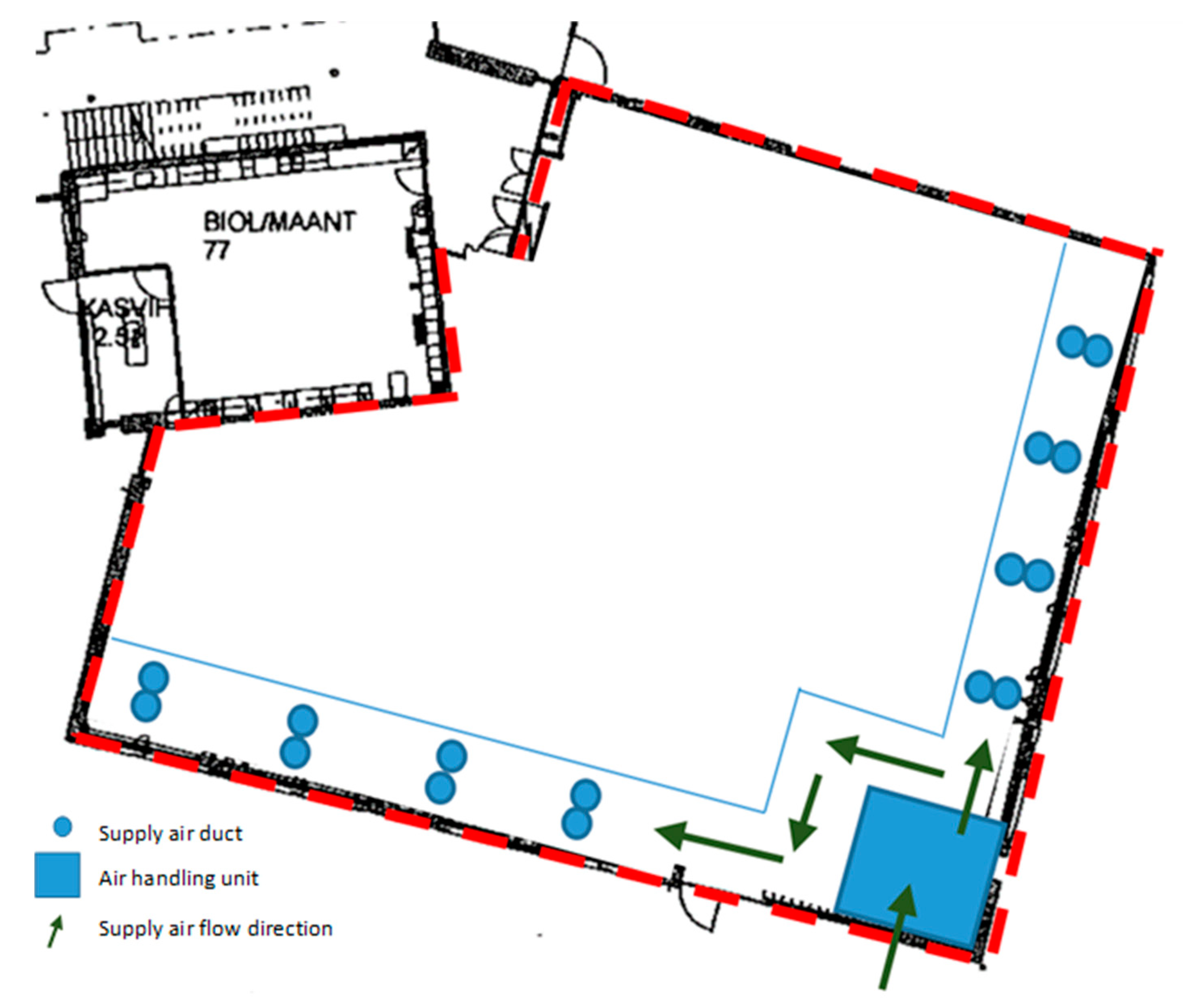
2.2. Functionality of the Hybrid Ventilation System
2.3. Measurements in the School
2.3.1. Air Flow Rates and Pressure Differences across the Building Envelope
2.3.2. Indoor Air Quality (IAQ) Measurements
2.3.3. Characterization of Mycobiota in Indoor Dust
2.3.4. Indoor Air Questionnaire
3. Results and Discussion
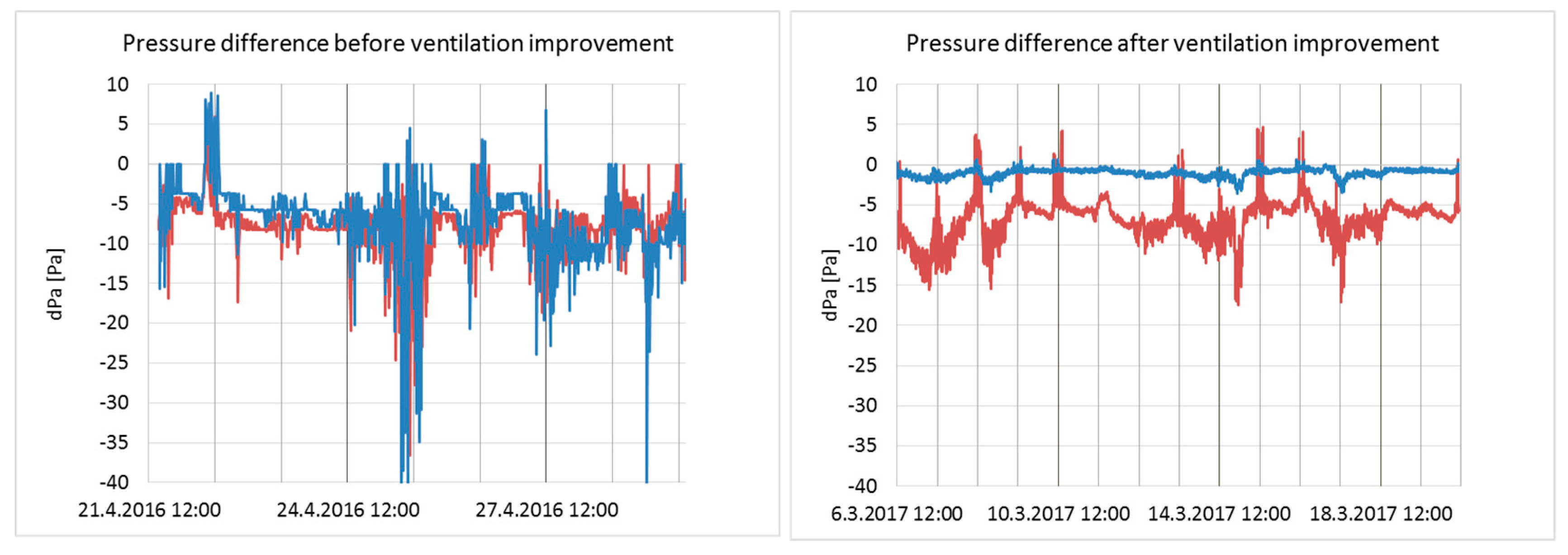
3.1. Air Flow Rates and Pressure Differences across the Building Envelope
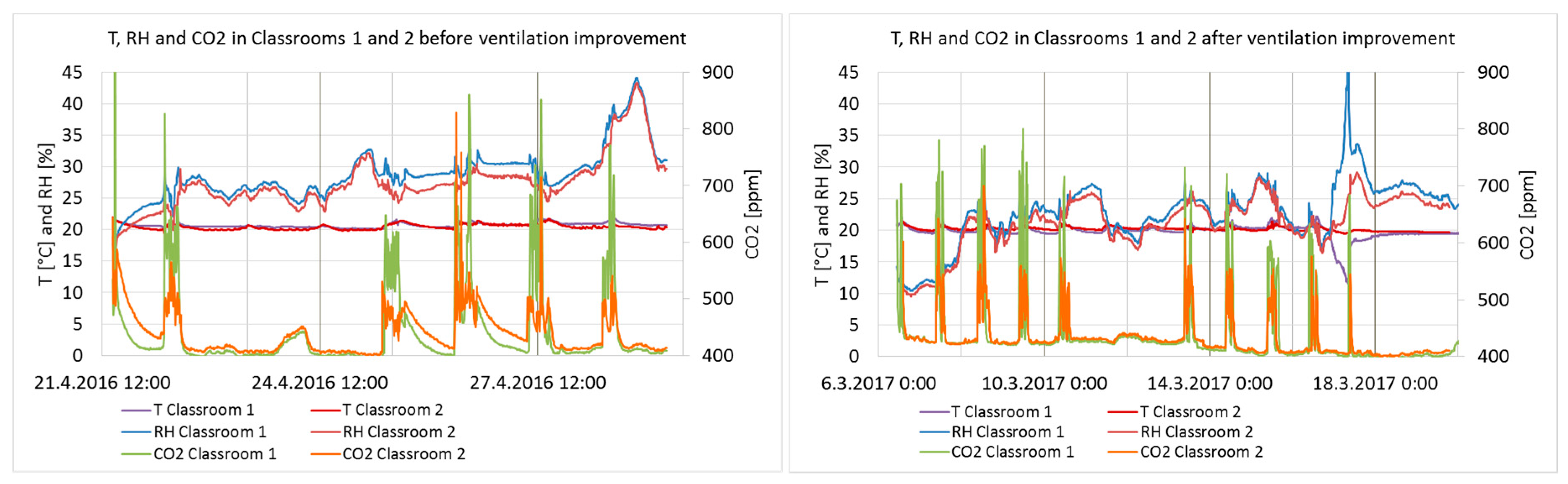
3.2. IAQ Measurements
3.3. Characterization of Mycobiota in Indoor Dust
3.4. Indoor Air Questionnaire
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| IAQ | Indoor air quality |
| TVOC | Total volatile organic compounds |
| PM2.5 | Fine particulate matter, particle size 2.5 μm |
| RH | Relative humidity |
| VOC | Volatile organic compounds |
| T | Temperature |
| CO2 | Carbon dioxide |
| FIOH | Finnish Institute of Occupational Health |
| MEA | Malt extract agar |
| BSMI | Boar sperm motility inhibition |
| ICP | Inhibition of cell proliferation |
| ITS | Internal transcribed spacer |
References
- Reijula, K.; Ahonen, G.; Alenius, H.; Holopainen, R.; Lappalainen, S.; Palomäki, E.; Reiman, M. Rakennusten Kosteus-Ja Homeongelmat; Eduskunnan Tarkastusvaliokunnan Julkaisu 5.12.2012; Eduskunnan Tarkastusvaliokunta: Helsinki, Finland, 2012. (In Finnish) [Google Scholar]
- Toyinbo, O.; Shaughnessy, R.; Turunen, M.; Putus, T.; Metsämuuronen, J.; Kurnitski, J.; Haverinen-Shaughnessy, U. Building Characteristics, Indoor Environmental Quality, and Mathematics Achievement in Finnish Elementary Schools. Build. Environ. 2016, 104, 114–121. [Google Scholar] [CrossRef]
- Salthammer, T.; Uhde, E.; Schripp, T.; Schieweck, A.; Morawska, L.; Mazaheri, M.; Clifford, S.; He, C.; Buonanno, G.; Querol, X. Children’s Well-being at Schools: Impact of Climatic Conditions and Air Pollution. Environ. Int. 2016, 94, 196–210. [Google Scholar] [CrossRef] [PubMed]
- Smedje, G.; Norbäck, D. Asthmatic Symptoms in School Children in Relation to Building Dampness and Atopy. Indoor Built Environ. 2003, 12, 249–250. [Google Scholar] [CrossRef]
- De Gennaro, G.; Dambruoso, P.; Loiotile, A.; Di Gilio, A.; Giungato, P.; Tutino, M. Indoor Air Quality in Schools. Environ. Chem. Lett. 2014, 12, 467–482. [Google Scholar] [CrossRef]
- Claudio, L.; Rivera, G.A.; Ramirez, O.F. Association between Markers of Classroom Environmental Conditions and Teachers’ Respiratory Health. J. Sch. Health 2016, 86, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Kielb, C.; Lin, S.; Muscatiello, N.; Hord, W.; Rogers-Harrington, J.; Healy, J. Building-related Health Symptoms and Classroom Indoor Air Quality: A Survey of School Teachers in New York State. Indoor Air 2015, 25, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Magnavita, N. Work-Related Symptoms in Indoor Environments: A Puzzling Problem for the Occupational Physician. Int. Arch. Occup. Environ. Health 2015, 88, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Muscatiello, N.; McCarthy, A.; Kielb, C.; Hsu, W.; Hwang, S.; Lin, S. Classroom Conditions and CO2 Concentrations and Teacher Health Symptom Reporting in 10 New York State Schools. Indoor Air 2015, 25, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Carrer, P.; Wargocki, P.; Fanetti, A.; Bischof, W.; Fernandes, E.D.O.; Hartmann, T.; Kephalopoulos, S.; Palkonen, S.; Seppänen, O. What does the Scientific Literature Tell Us about the Ventilation–health Relationship in Public and Residential Buildings? Build. Environ. 2015, 94, 273–286. [Google Scholar] [CrossRef]
- Sundell, J.; Levin, H.; Nazaroff, W.W.; Cain, W.S.; Fisk, W.J.; Grimsrud, D.T.; Gyntelberg, F.; Li, Y.; Persily, A.; Pickering, A. Ventilation Rates and Health: Multidisciplinary Review of the Scientific Literature. Indoor Air 2011, 21, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Ervasti, J.; Kivimäki, M.; Kawachi, I.; Subramanian, S.; Pentti, J.; Oksanen, T.; Puusniekka, R.; Pohjonen, T.; Vahtera, J.; Virtanen, M. School Environment as Predictor of Teacher Sick Leave: Data-Linked Prospective Cohort Study. BMC Public Health 2012, 12, 770. [Google Scholar] [CrossRef] [PubMed]
- Wargocki, P.; Wyon, D.P. Ten Questions Concerning Thermal and Indoor Air Quality Effects on the Performance of Office Work and Schoolwork. Build. Environ. 2017, 112, 359–366. [Google Scholar] [CrossRef]
- Lübeck, M.; Poulsen, S.K.; Lübeck, P.S.; Jensen, D.F.; Thrane, U. Identification of Trichoderma Strains from Building Materials by ITS1 Ribotyping, UP-PCR Fingerprinting and UP-PCR Cross Hybridization. FEMS Microbiol. Lett. 2000, 185, 129–134. [Google Scholar] [CrossRef]
- McMullin, D.R.; Renaud, J.B.; Barasubiye, T.; Sumarah, M.W.; Miller, J.D. Metabolites of Trichoderma Species Isolated from Damp Building Materials. Can. J. Microbiol. 2017, 63, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Mikkola, R.; Andersson, M.A.; Kredics, L.; Grigoriev, P.A.; Sundell, N.; Salkinoja-Salonen, M.S. 20-Residue and 11-residue Peptaibols from the Fungus Trichoderma longibrachiatum are Synergistic in Forming Na+/K+-permeable Channels and Adverse Action Towards Mammalian Cells. FEBS J. 2012, 279, 4172–4190. [Google Scholar] [CrossRef] [PubMed]
- Kuhls, K.; Lieckfeldt, E.; Börner, T.; Guého, E. Molecular Reidentification of Human Pathogenic Trichoderma Isolates as Trichoderma longibrachiatum and Trichoderma citrinoviride. Med. Mycol. 1999, 37, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Vornanen-Winqvist, C.; Järvi, K.; Toomla, S.; Ahmed, K.; Andersson, M.A.; Mikkola, R.; Marik, T.; Kredics, L.; Salonen, H.; Kurnitski, J. Ventilation Positive Pressure Intervention Effect on Indoor Air Quality in a School Building with Moisture Problems. Int. J. Environ. Res. Public Health 2018, 15, 230. [Google Scholar] [CrossRef] [PubMed]
- Leppänen, H. Assessing Microbial Exposure in Indoor Environments by Using House Dust Samples; University of Eastern Finland: Kuopio, Finland, 2017. [Google Scholar]
- Vesper, S.J.; McKinstry, C.; Yang, C.; Haugland, R.A.; Kercsmar, C.M.; Yike, I.; Schluchter, M.D.; Kirchner, H.L.; Sobolewski, J.; Allan, T.M.; et al. Specific Molds Associated with Asthma in Water-Damaged Homes. J. Occup. Environ. Med. 2006, 48, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Pakpour, S.; Klironomos, J.; Li, D. Microfungi in indoor environments: What is known and what is not. In Biology of Microfungi; Springer: Cham, Switzerland, 2016; pp. 373–412. [Google Scholar]
- Heiselberg, P. Design Principles for Natural and Hybrid Ventilation; Aalborg University, Indoor Environmental Engineering: Aalborg, Denmark, 2000. [Google Scholar]
- Chenari, B.; Carrilho, J.D.; da Silva, M.G. Towards Sustainable, Energy-Efficient and Healthy Ventilation Strategies in Buildings: A Review. Renew. Sustain. Energy Rev. 2016, 59, 1426–1447. [Google Scholar] [CrossRef]
- Becker, R.; Goldberger, I.; Paciuk, M. Improving Energy Performance of School Buildings while Ensuring Indoor Air Quality Ventilation. Build. Environ. 2007, 42, 3261–3276. [Google Scholar] [CrossRef]
- Emmerich, S.J. Simulated Performance of Natural and Hybrid Ventilation Systems in an Office Building. HVAC R Res. 2006, 12, 975–1004. [Google Scholar] [CrossRef]
- Lomas, K.J.; Cook, M.J.; Fiala, D. Low Energy Architecture for a Severe US Climate: Design and Evaluation of a Hybrid Ventilation Strategy. Energy Build. 2007, 39, 32–44. [Google Scholar] [CrossRef]
- Vornanen-Winqvist, C.; Ahmed, K.; Toomla, S.; Kurnitski, J.; Mikkola, R.; Salonen, H. Ventilation and Indoor Air Quality in Newly Built School with Hybrid Ventilation. In Proceedings of the Healthy Buildings 2017 Europe, Conference Proceedings, Lublin University of Technology, Lublin, Poland, 2–5 July 2017. [Google Scholar]
- Verriele, M.; Schoemaecker, C.; Hanoune, B.; Leclerc, N.; Germain, S.; Gaudion, V.; Locoge, N. The MERMAID Study: Indoor and Outdoor Average Pollutant Concentrations in 10 Low-energy School Buildings in France. Indoor Air 2016, 26, 702–713. [Google Scholar] [CrossRef] [PubMed]
- ISO 16000-6:2011. Indoor Air—Part 6: Determination of Volatile Organic Compounds in Indoor and Test Chamber Air by Active Sampling on Tenax TA Sorbent, Thermal Desorption and Gas Chromatography Using MS or MS-FID; ISO: Geneva, Switzerland, 2011. [Google Scholar]
- Andersson, M.A.; Jääskeläinen, E.L.; Shaheen, R.; Pirhonen, T.; Wijnands, L.M.; Salkinoja-Salonen, M.S. Sperm Bioassay for Rapid Detection of Cereulide-Producing Bacillus Cereus in Food and Related Environments. Int. J. Food Microbiol. 2004, 94, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Bencsik, O.; Papp, T.; Berta, M.; Zana, A.; Forgó, P.; Dombi, G.; Andersson, M.A.; Salkinoja-Salonen, M.; Vágvölgyi, C.; Szekeres, A. Ophiobolin A from Bipolaris Oryzae Perturbs Motility and Membrane Integrities of Porcine Sperm and Induces Cell Death on Mammalian Somatic Cell Lines. Toxins 2014, 6, 2857–2871. [Google Scholar] [CrossRef] [PubMed]
- Samson, R.A.; Hoekstra, E.S.; Frisvad, J.C. Introduction to Food-and Airborne Fungi; Centraalbureau voor Schimmelcultures (CBS): Utrecht, The Netherlands, 2004. [Google Scholar]
- Andersson, M.A.; Mikkola, R.; Raulio, M.; Kredics, L.; Maijala, P.; Salkinoja-Salonen, M.S. Acrebol, a Novel Toxic Peptaibol Produced by an Acremonium Exuviarum Indoor Isolate. J. Appl. Microbiol. 2009, 106, 909–923. [Google Scholar] [CrossRef] [PubMed]
- Andersson, K. Epidemiological Approach to Indoor Air Problems. Indoor Air 1998, 8, 32–39. [Google Scholar] [CrossRef]
- Reijula, K.; Sundman Digert, C. Assessment of Indoor Air Problems at Work with a Questionnaire. Occup. Environ. Med. 2004, 61, 33–38. [Google Scholar] [PubMed]
- Hellgren, U.; Hyvärinen, M.; Holopainen, R.; Reijula, K. Perceived Indoor Air Quality, Air-Related Symptoms and Ventilation in Finnish Hospitals. Int. J. Occup. Med. Environ. Health 2011, 24, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Finnish Ministry of the Environment. 1009/2017 Ympäristöministeriön Asetus Uuden Rakennuksen Sisäilmastosta ja Ilmanvaihdosta; Finnish Ministry of the Environment: Helsinki, Finland, 2018. (In Finnish) [Google Scholar]
- Classification of Indoor Climate 2018. RT 07-11299 Target Values, Design Guidance and Product Requirements. 2018. Available online: https://www.rakennustieto.fi/kortistot/rt/kortit/11299.html.stx (accessed on 18 June 2018). (In Finnish).
- Wang, S.; Ang, H.; Tade, M. Volatile Organic Compounds in Indoor Environment and Photocatalytic Oxidation: State of the Art. Environ. Int. 2007, 33, 694–705. [Google Scholar] [CrossRef] [PubMed]
- Salonen, H. Indoor Air Contaminants in Office Buildings; Finnish Institute of Occupational Health: Helsinki, Finland, 2009. [Google Scholar]
- Ministry of Social Affairs and Health, Finland. Decree of the Ministry of Social Affairs and Health on Health-Related Conditions of Housing and Other Residential Buildings and Qualification Requirements for Third-Party Experts (545/2015); Ministry of Social Affairs and Health: Helsinki, Finland, 2015.
- Finnish Institute of Occupational Health. Threshold Values for Indoor Environment 27 February 2017. Available online: https://www.ttl.fi/wp-content/uploads/2016/09/sisaympariston-viitearvoja.pdf (accessed on 28 February 2018). (In Finnish).
- Hatvani, L.; Manczinger, L.; Vágvölgyi, C.; Kredics, L. Trichoderma as a Human Pathogen. In Trichoderma: Biology and Applications; Mukherjee, P.K., Horwitz, B.A., Singh, U.S., Mukherjee, M., Schmoll, M., Eds.; CAB International: Wallingford, UK, 2013; pp. 292–313. [Google Scholar]
- Howell, C. Mechanisms Employed by Trichoderma Species in the Biological Control of Plant Diseases: The History and Evolution of Current Concepts. Plant Dis. 2003, 87, 4–10. [Google Scholar] [CrossRef]
- Salonen, H.; Duchaine, C.; Mazaheri, M.; Clifford, S.; Lappalainen, S.; Reijula, K.; Morawska, L. Airborne Viable Fungi in School Environments in Different Climatic regions—A Review. Atmos. Environ. 2015, 104, 186–194. [Google Scholar] [CrossRef]
- Peltola, J.; Ritieni, A.; Mikkola, R.; Grigoriev, P.A.; Pocsfalvi, G.; Andersson, M.A.; Salkinoja-Salonen, M.S. Biological Effects of Trichoderma Harzianum Peptaibols on Mammalian Cells. Appl. Environ. Microbiol. 2004, 70, 4996–5004. [Google Scholar] [CrossRef] [PubMed]
- Jaakkola, M.; Laitinen, S.; Piipari, R.; Uitti, J.; Nordman, H.; Haapala, A.; Jaakkola, J. Immunoglobulin G Antibodies Against Indoor Dampness-related Microbes and Adult-onset Asthma: A Population-based Incident Case–control Study. Clin. Exp. Immunol. 2002, 129, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Hyvarinen, A.; Reponen, T.; Husman, T.; Nevalainen, A. Comparison of the Indoor Air Quality in Mould Damaged and Reference Buildings in a Subarctic Climate. Cent. Eur. J. Public Health 2001, 9, 133–139. [Google Scholar] [PubMed]
- Frontczak, M.; Wargocki, P. Literature Survey on how Different Factors Influence Human Comfort in Indoor Environments. Build. Environ. 2011, 46, 922–937. [Google Scholar] [CrossRef]
- Brauer, C.; Mikkelsen, S. The Influence of Individual and Contextual Psychosocial Work Factors on the Perception of the Indoor Environment at Work: A Multilevel Analysis. Int. Arch. Occup. Environ. Health 2010, 83, 639–651. [Google Scholar] [CrossRef] [PubMed]
- Haghighat, F.; Donnini, G. Impact of Psycho-Social Factors on Perception of the Indoor Air Environment Studies in 12 Office Buildings. Build. Environ. 1999, 34, 479–503. [Google Scholar] [CrossRef]
- Lahtinen, M.; Huuhtanen, P.; Kähkönen, E.; Reijula, K. Psychosocial Dimensions of Solving an Indoor Air Problem. Indoor Air 2002, 12, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Lampi, J. Developing Indoor Air Questionnaire for Primary School Pupils: Test-Retest Repeatability of Self-and Parent-Administered Questionnaires. Eur. Respir. J. 2015, 46, PA3402. [Google Scholar] [CrossRef]


| Measured Factor | Device | Accuracy | Place | Time |
|---|---|---|---|---|
| Supply air flow rate | SWEMA 3000 md | ±0.3% read value, lowest ±0.3 Pa | All classrooms | 60 s average |
| Pressure difference across the envelope | KIMO CP101, logger Grant 1000 | 1.5% of reading ±3 Pa | Classrooms 1 and 2 | Continuous, 1 week (May 2016) |
| Envic dp-101s-pd2, logger Grant 1000 | 3% of reading ±0.2 m/s | Classrooms 1 and 2 | Continuous, 2 weeks (March 2017) | |
| Temperature (T) | Rotronic CL11 | ±0.3 °C | Classrooms 1 and 2 | Continuous (1–2 weeks) |
| Relative humidity (RH) | Rotronic CL11 | ±3% (10 ... 95%) | Classrooms 1 and 2 | Continuous (1–2 weeks) |
| Carbon dioxide (CO2) | Rotronic CL11 | ±(30 ppm + 5% of reading) | Classrooms 1 and 2 | Continuous (1–2 weeks) |
| Formaldehyde | FM-801 | ±10 ppb at 40, 80, 160 ppb | Classroom 2 | Continuous, 1 week |
| Particulate matter 2.5 μm (PM2.5) | MIE pDR-1500 | ±5% | Classroom 2 | Continuous, 1 week |
| Volatile organic compounds (VOCs) | Tenax TA, TD-GC-MS | ±20% (average) | Classroom 2 | 40 min |
| Mycobiota of settled dust | Plastic bag, MEA plates, BSMI, ICP, ITS | Classrooms 1 and 2, lobby | Cultivated for 4 weeks | |
| Perceived indoor air quality | Örebro (MM40)-questionnaire (Finnish Institute of Occupational Health (FIOH)) | Personnel of the whole building | 2-week response time | |
| Classroom 1 | Classroom 2 | Outdoor, Per Month * | ||||||
|---|---|---|---|---|---|---|---|---|
| RH (%) | T (°C) | CO2 (ppm) | RH (%) | T (°C) | CO2 (ppm) | T (°C) | ||
| Before | Min | 18 | 20 | 394 | 16 | 20 | 402 | −1.4 |
| Max | 40 | 22 | 1431 | 38 | 22 | 829 | 13.7 | |
| Average | 29 | 21 | 488 | 27 | 21 | 458 | 4.9 | |
| After | Min | 11 | 12 | 394 | 10 | 20 | 400 | −7.8 |
| Max | 46 | 22 | 801 | 29 | 21 | 700 | 11.9 | |
| Average | 23 | 20 | 464 | 22 | 20 | 450 | 1.0 | |
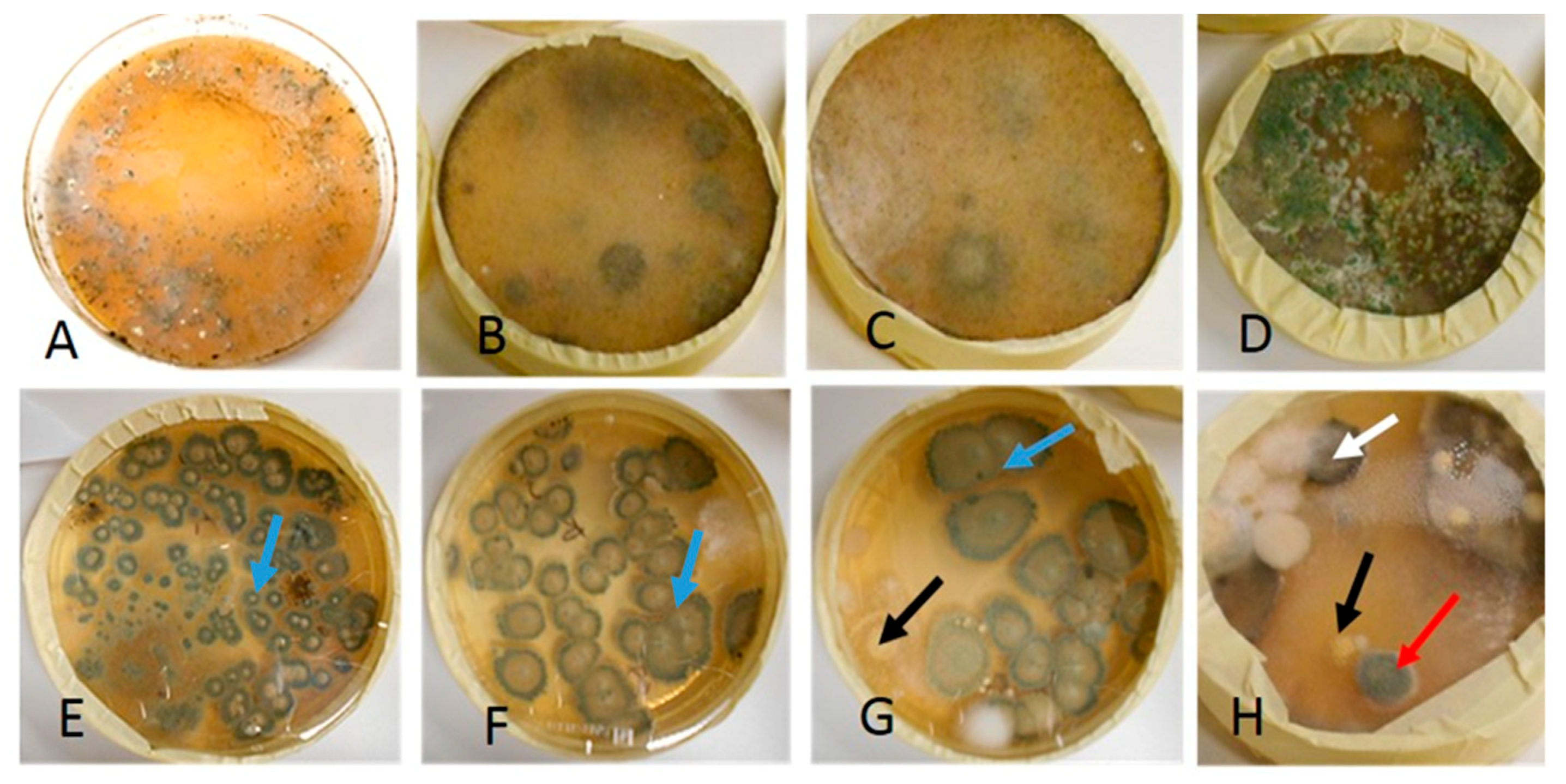
| Toxicity | Colony Color | Size of Conidia/Spores | Morphology under Light Microscope | |||
|---|---|---|---|---|---|---|
| Growth at 37 °C | BSMI | ICP | MEA | (μm) | ||
| Aspergillus section Nigri 1 strain | + | - | + | Black | 3.5–5 |  |
| Asp. westerdijkiae 2 strains | - | + | + | Yellow | 2.5–3 |  |
| Eurotium sp. 1 strain | + | + | + | Green | 5–7 |  |
| Penicillium sp. 10 strains (Terverticilliate) | Green | 3.4 | 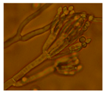 | |||
| Penicillium sp. 3 strains (Monoverticilliate) | Green | 2.3 | 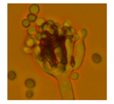 | |||
| Rhizopus sp. 10 strains | - | - | - | Grey | 5–10 | 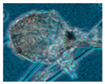 |
| Trichoderma citrinoviride * 10 strains | + | + | + | Green | 1.6 × 3 |  |
| Trichoderma sp. 5 strains | - | + | + | Green | 4 |  |
| School Samples | Settled Dust | ||
|---|---|---|---|
| Before ventilation improvement | Sampled 31 May 2016 | Number of colonies/plate | Number of plates containing a colony morphotype/all plates |
| Locations: Classroom 2 and lobby | Trichoderma citrinoviridea,b | >100 | 1/6 |
| Rhizopus sp. | Plate overgrown | 2/6 | |
| Trichoderma sp. a | >100 | 2/6 | |
| After ventilation improvement | Sampled 6 March 2017 | ||
| Locations: Classrooms 1, 2 and 7 | Penicillium sp. c Penicillium sp. d | >100–120 10 | 3/9 2/9 |
| Aspergillus westerdijkiaea | 2–3 | 2/9 | |
| Asp. Nigera,b | 1–2 | 3/9 | |
| Eurotium sp. a | 1 | 1/9 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vornanen-Winqvist, C.; Salonen, H.; Järvi, K.; Andersson, M.A.; Mikkola, R.; Marik, T.; Kredics, L.; Kurnitski, J. Effects of Ventilation Improvement on Measured and Perceived Indoor Air Quality in a School Building with a Hybrid Ventilation System. Int. J. Environ. Res. Public Health 2018, 15, 1414. https://doi.org/10.3390/ijerph15071414
Vornanen-Winqvist C, Salonen H, Järvi K, Andersson MA, Mikkola R, Marik T, Kredics L, Kurnitski J. Effects of Ventilation Improvement on Measured and Perceived Indoor Air Quality in a School Building with a Hybrid Ventilation System. International Journal of Environmental Research and Public Health. 2018; 15(7):1414. https://doi.org/10.3390/ijerph15071414
Chicago/Turabian StyleVornanen-Winqvist, Camilla, Heidi Salonen, Kati Järvi, Maria A. Andersson, Raimo Mikkola, Tamás Marik, László Kredics, and Jarek Kurnitski. 2018. "Effects of Ventilation Improvement on Measured and Perceived Indoor Air Quality in a School Building with a Hybrid Ventilation System" International Journal of Environmental Research and Public Health 15, no. 7: 1414. https://doi.org/10.3390/ijerph15071414
APA StyleVornanen-Winqvist, C., Salonen, H., Järvi, K., Andersson, M. A., Mikkola, R., Marik, T., Kredics, L., & Kurnitski, J. (2018). Effects of Ventilation Improvement on Measured and Perceived Indoor Air Quality in a School Building with a Hybrid Ventilation System. International Journal of Environmental Research and Public Health, 15(7), 1414. https://doi.org/10.3390/ijerph15071414






