Short-Term Effects of Tourmaline on Nitrogen Removals and Microbial Communities in a Sequencing Batch Reactor at Low Temperatures
Abstract
1. Introduction
2. Materials and Methods
2.1. Ultrafine Tourmaline Particles
2.2. SBRs with Short-Term Exposure to UTPs at Low Temperatures
2.3. DNA Extraction and PCR
2.4. Illumina MiSeq
2.5. Scanning Electron Microscopy (SEM)
2.6. Analytical Methods
3. Results and Discussion
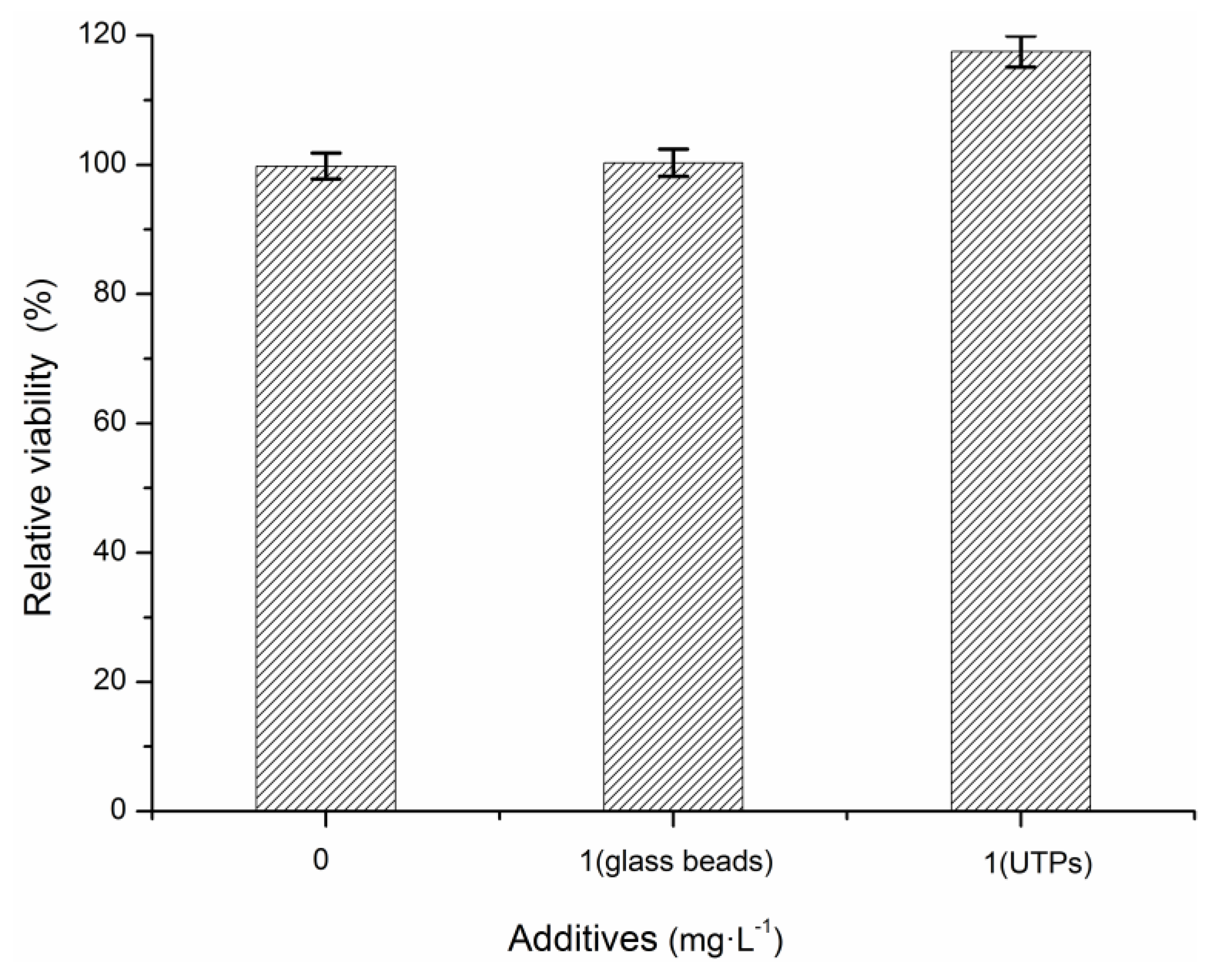
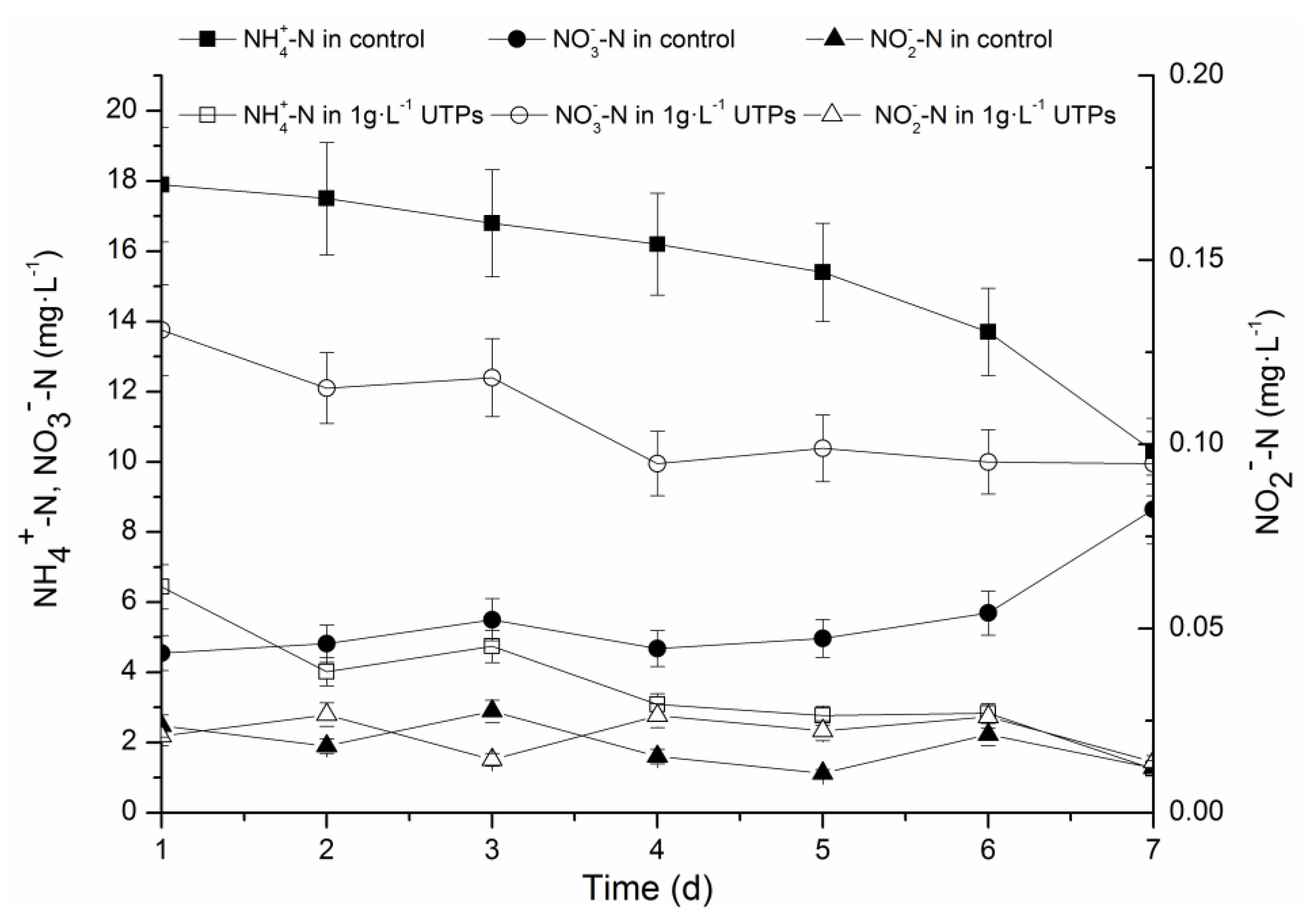
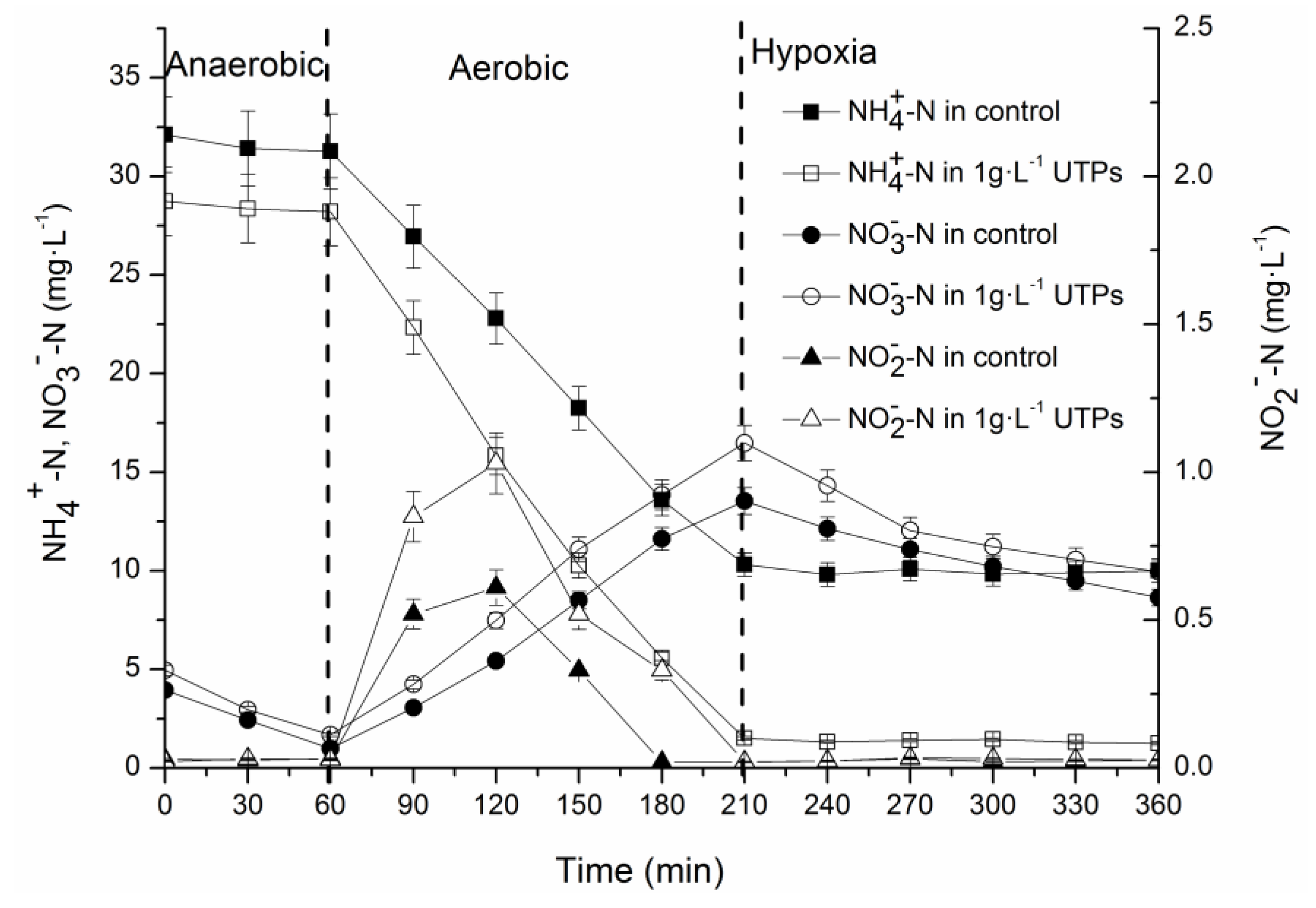
3.1. Effects of UTPs on Effluent at Low Temperatures
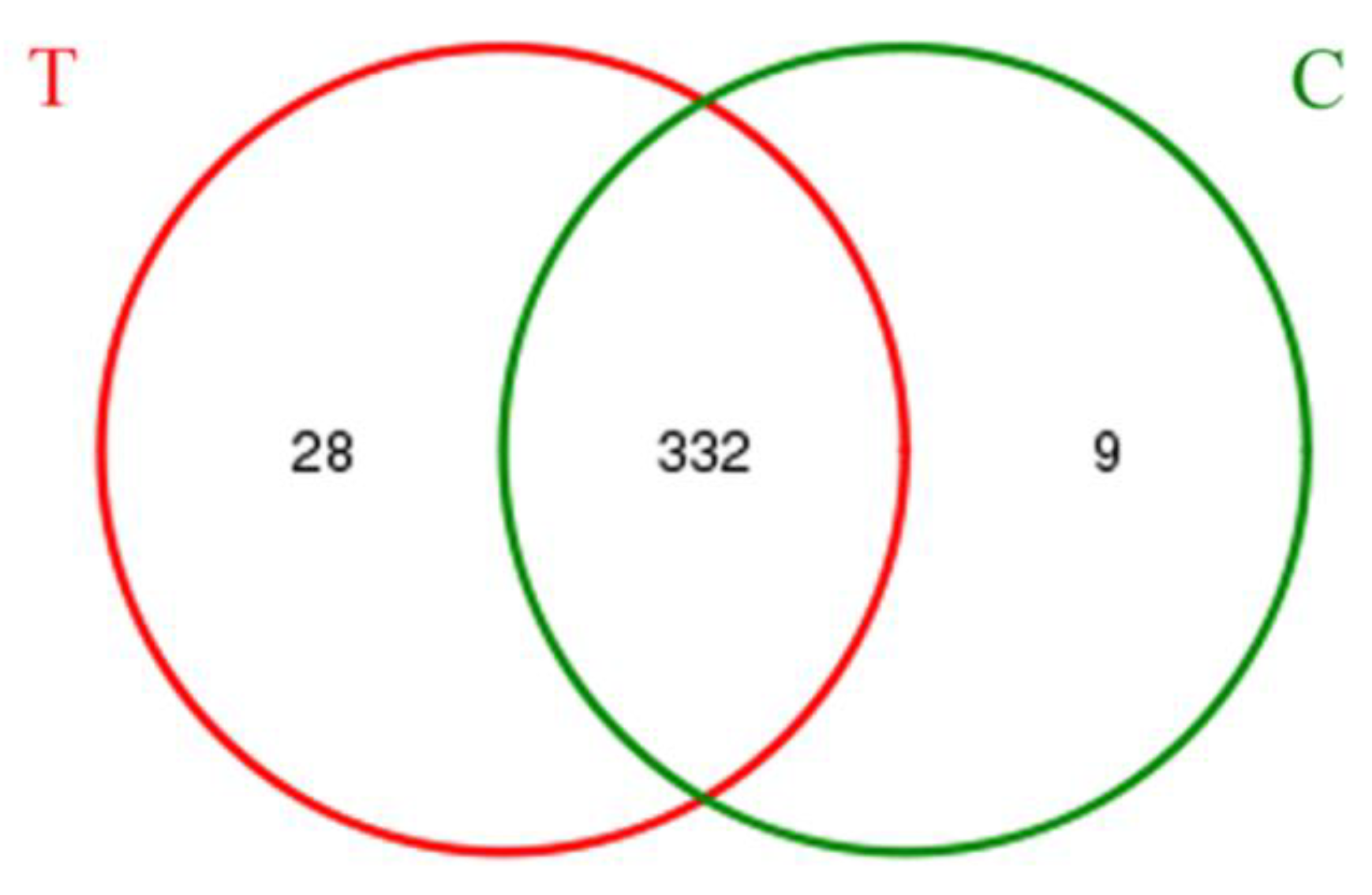
3.2. Bacterial Community Shift in Activated Sludge after Short-Term Exposure to UTPs at Low Temperatures
3.3. Key Functional Species
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Sun, S.; Wei, C.D.; Liu, Y.X. Characterization and water activation behavior of tourmaline nanoparticles. J. Nanosci. Nanotechnol. 2010, 10, 2119–2124. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.S.; Hu, C.H.; Zhang, H.M. Effects of tourmaline addition on the dehydrogenase activity of rhodopseudomonas palustris. Process Biochem. 2006, 41, 221–225. [Google Scholar] [CrossRef]
- Yamaguchi, S. Surface electric fields of tourmaline. Appl. Phys. A 1983, 31, 183–185. [Google Scholar] [CrossRef]
- Cao, H.; Li, X.; Wu, J.; Yu, K.; Zhang, Y. Simulation of the effects of direct electric current on multispecies biofilms. Process Biochem. 2003, 38, 1139–1145. [Google Scholar] [CrossRef]
- Wei, L.; Ma, F.; Wang, Q.; Wang, H.; Zhang, X. Investigation of the reduction performance of sulfate reducing bacteria enhanced by nano-meter/submicron tourmaline. J. Biotechnol. 2008, 136, S142. [Google Scholar] [CrossRef]
- Xia, M.S.; Hu, C.H.; Zhang, H.M.; Xiong, L.; Xu, Z.R. Effects of tourmaline-treated-water on the growth and the activity of alkaline phosphatase of caco-2 cell. Chin. J. Cell Biol. 2005, 27, 358–362. [Google Scholar]
- Qiu, S.; Ma, F.; Wo, Y.; Xu, S. Study on the biological effect of tourmaline on the cell membrane of E. Coli. Surf. Interface Anal. 2011, 43, 1069–1073. [Google Scholar] [CrossRef]
- Tan, C.; Cui, D.; Liu, Y.; Ji, Y. Influence of tourmaline on the anaerobic ammonium oxidation process in sequencing batch reactors. J. Environ. Eng. 2017, 143, 04017053. [Google Scholar] [CrossRef]
- Tan, C.; Xu, H.; Cui, D.; Zuo, J.; Li, J.; Ji, Y.; Qiu, S.; Yao, L.; Chen, Y.; Liu, Y. Effects of tourmaline on nitrogen removal performance and biofilm structures in the sequencing batch biofilm reactor. J. Environ. Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Liang, D.; Lian, T. Comparison of denitrification performance by bacterium achromobacter sp. A14 under different electron donor conditions. Chem. Eng. J. 2018, 333, 320–326. [Google Scholar] [CrossRef]
- He, S.; Ding, L.; Xu, K.; Geng, J.; Ren, H. Effect of low temperature on highly unsaturated fatty acid biosynthesis in activated sludge. Bioresour. Technol. 2016, 211, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Alejandro, G.M.; Barbara, M.P.; Alejandro, R.S.; Paula, M.M.; Anna, M.; Jesus, G.L.; Riku, V. Start-up and operation of an aerobic granular sludge system under low working temperature inoculated with cold-adapted activated sludge from finland. Bioresour. Technol. 2017, 239, 180–189. [Google Scholar]
- Sun, N.; Ge, C.; Ahmad, H.A.; Gao, B.; Ni, S.Q. Realization of microbial community stratification for single-stage nitrogen removal in a sequencing batch biofilter granular reactor. Bioresour. Technol. 2017, 241, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Yu, Y.; Zhu, S.; Bian, D.; Huo, M.; Zhou, D.; Huo, H. Characterization of a novel micro-pressure swirl reactor for removal of chemical oxygen demand and total nitrogen from domestic wastewater at low temperature. Biodegradation 2017, 28, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Sundaresan, N.; Philip, L. Performance evaluation of various aerobic biological systems for the treatment of domestic wastewater at low temperatures. Water Sci. Technol. 2008, 58, 819–830. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Shang, Y.; Wang, H.; Yang, K. Rapid formation and pollutant removal ability of aerobic granules in a sequencing batch airlift reactor at low temperature. Environ. Technol. 2016, 37, 3078–3085. [Google Scholar] [CrossRef] [PubMed]
- Yahong, H.; Shan, Q.; Fang, M.; Jue, W. Short-term and long-term effects of tourmaline on activated sludge viability and performance. Water Air Soil Pollut. 2017, 228, 395. [Google Scholar] [CrossRef]
- Jing, X.; Sanders, N.J.; Shi, Y.; Chu, H.; Classen, A.T.; Zhao, K.; Chen, L.; Shi, Y.; Jiang, Y.; He, J.S. The links between ecosystem multifunctionality and above- and belowground biodiversity are mediated by climate. Nat. Commun. 2015, 6, 8159. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. Uparse: Highly accurate otu sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–1031. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Su, Y.; Chen, Y. Acute and chronic responses of activated sludge viability and performance to silica nanoparticles. Environ. Sci. Technol. 2012, 46, 7182–7188. [Google Scholar] [CrossRef] [PubMed]
- APHA. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Rout, P.R.; Bhunia, P.; Dash, R.R. Simultaneous removal of nitrogen and phosphorous from domestic wastewater using bacillus cereus gs-5 strain exhibiting heterotrophic nitrification, aerobic denitrification and denitrifying phosphorous removal. Bioresour. Technol. 2017, 244, 484–495. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Sun, H.; Peng, Y.; Wang, S.; Ma, J. Achieving nitritation at low temperatures using free ammonia inhibition on nitrobacter and real-time control in an sbr treating landfill leachate. J. Environ. Sci. 2015, 30, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Jiang, H.; Zhu, G.; Song, X.; Liu, X.; Qiao, Y. Effect of tourmaline on denitrification characteristics of hydrogenotrophic bacteria. Environ. Sci. Pollut. Res. 2016, 23, 4868–4875. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, Y.; Quan, X. Electricity assisted anaerobic treatment of salinity wastewater and its effects on microbial communities. Water Res. 2012, 46, 3535–3543. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; He, Q.; Chen, D.; Wei, L.; Zou, Z.; Zhou, J.; Yang, K.; Zhang, H. Microbial community in a hydrogenotrophic denitrification reactor based on pyrosequencing. Appl. Microbiol. Biotechnol. 2015, 99, 10829–10837. [Google Scholar] [CrossRef] [PubMed]
- Jena, J.; Kumar, R.; Saifuddin, M.; Dixit, A.; Das, T. Anoxic-aerobic SBR system for nitrate, phosphate and cod removal from high-strength wastewater and diversity study of microbial communities. Biochem. Eng. J. 2016, 105, 80–89. [Google Scholar] [CrossRef]
- Park, S.; Bae, W.; Rittmann, B.E. Operational boundaries for nitrite accumulation in nitrification based on minimum/maximum substrate concentrations that include effects of oxygen limitation, pH, and free ammonia and free nitrous acid inhibition. Environ. Sci. Technol. 2010, 44, 335–342. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Gao, S.; Zhang, S.; Zhang, W.; Wang, H. Chronic responses of aerobic granules to zinc oxide nanoparticles in a sequencing batch reactor performing simultaneous nitrification, denitrification and phosphorus removal. Bioresour. Technol. 2017, 238, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Knowles, G.; Downing, A.L.; Barrett, M.J. Determination of kinetic constants for nitrifying bacteria in mixed culture, with the aid of an electronic computer. J. Gen. Microbiol. 1965, 38, 263. [Google Scholar] [CrossRef] [PubMed]

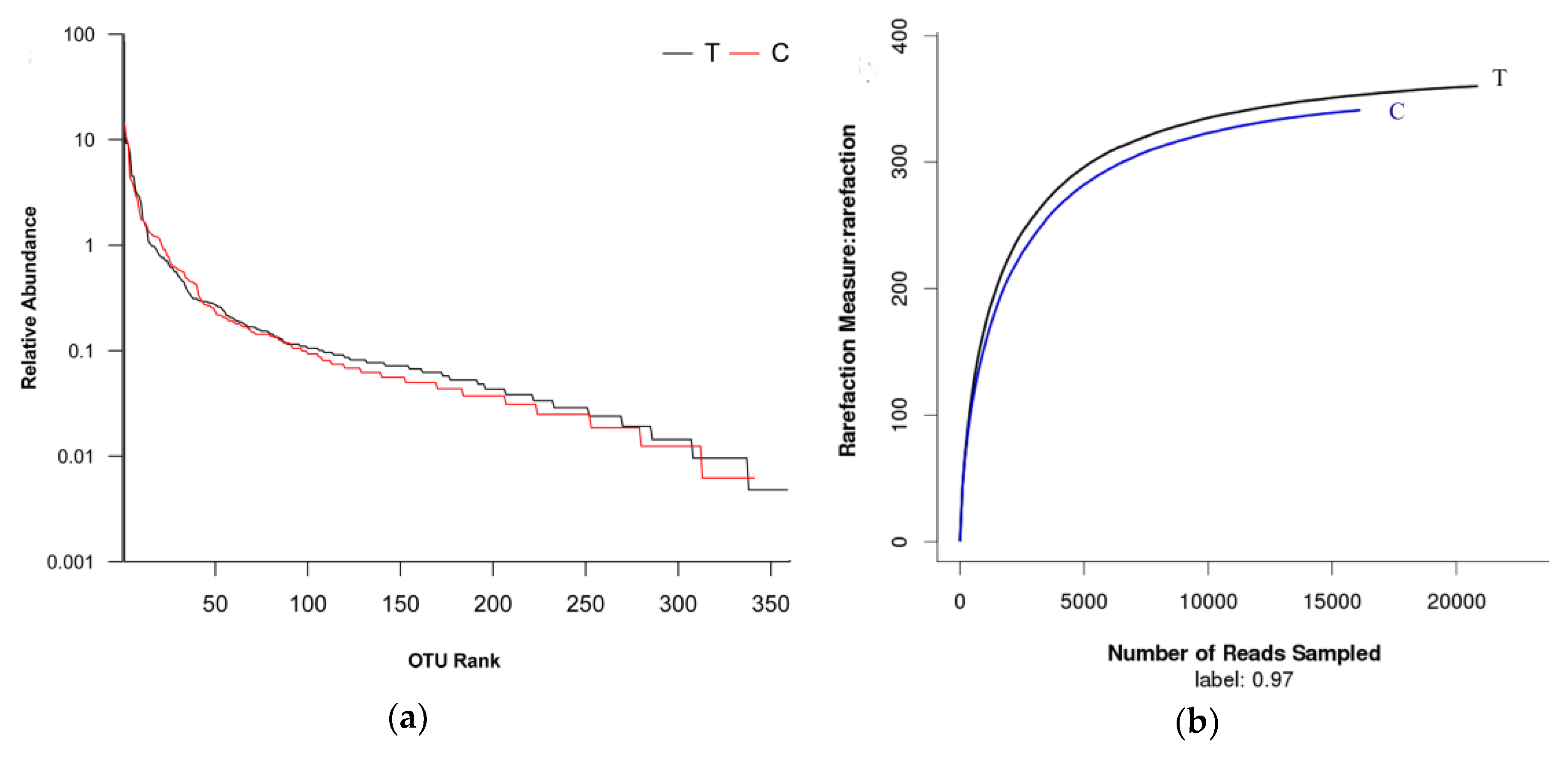
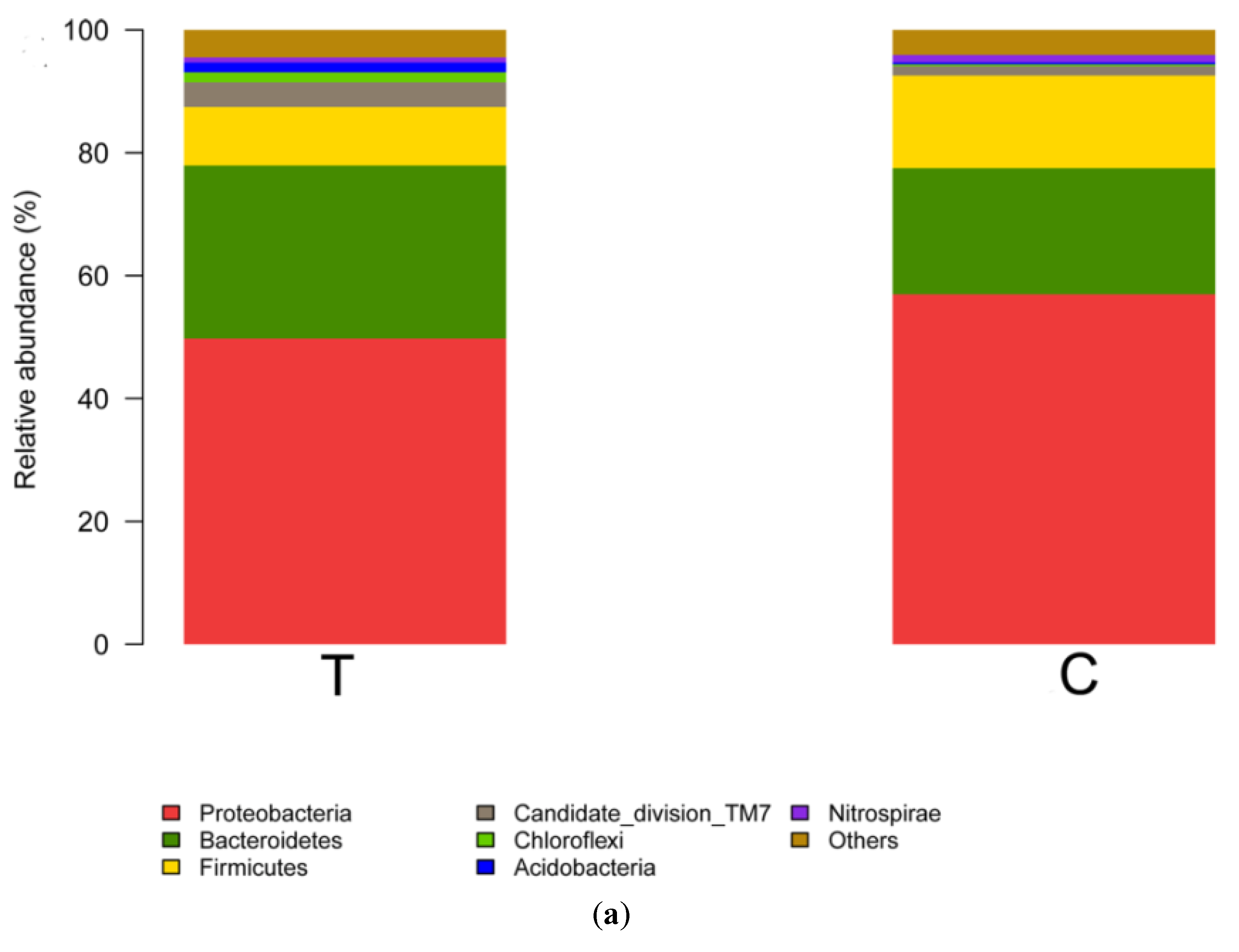
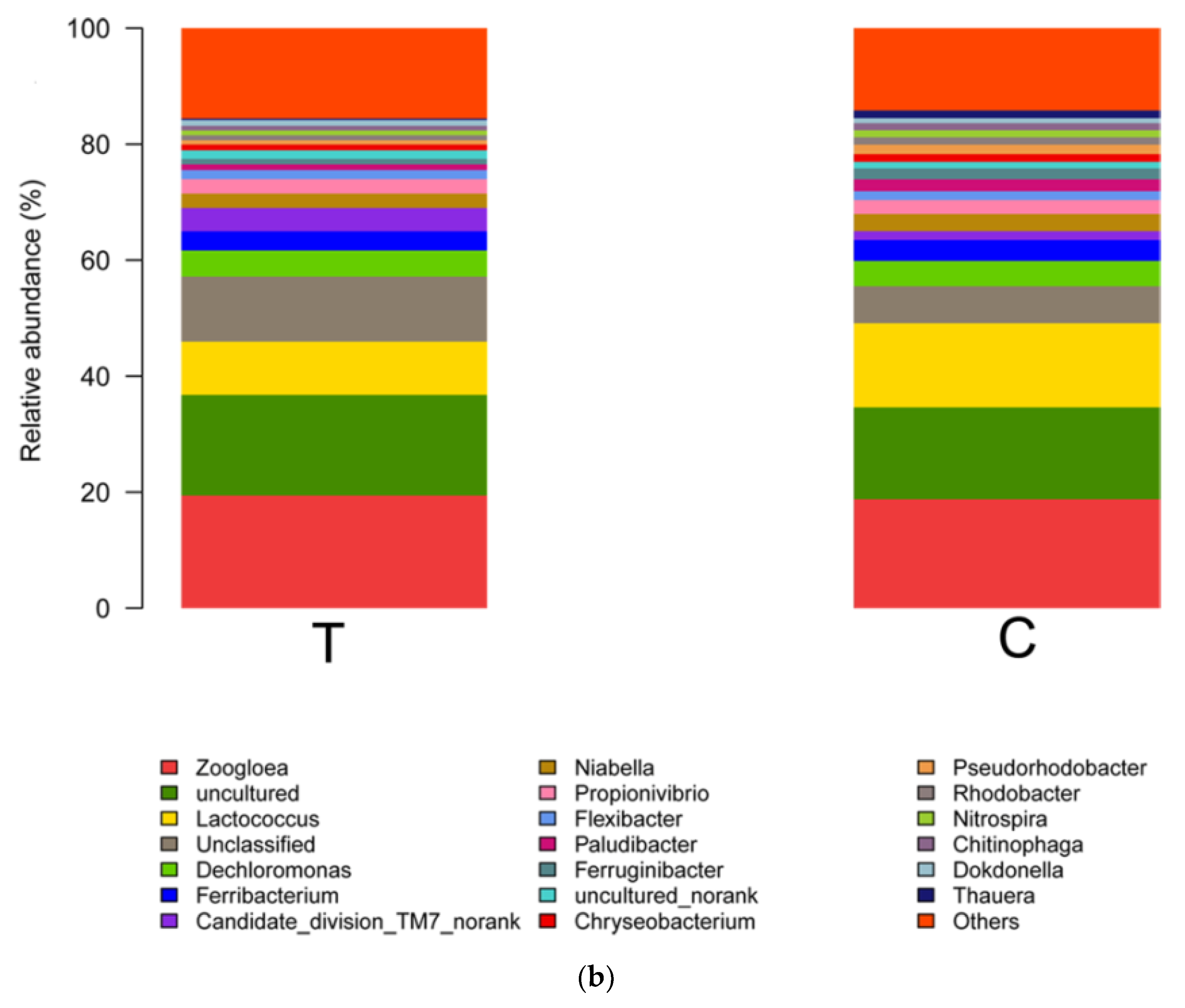
| Samples ID | Reads | OTU | ACE | Chao | Coverage | Shannon | Simpson |
|---|---|---|---|---|---|---|---|
| The test group | 20,839 | 360 | 369 (364, 381) | 368 (363, 383) | 0.998896 | 4.07 (4.05, 4.09) | 0.0433 (0.0422, 0.0444) |
| The control group | 16,092 | 341 | 353 (346, 366) | 353 (346, 371) | 0.998198 | 3.97 (3.94, 4) | 0.0501 (0.0485, 0.0517) |
| Functions | Taxa | The Test Group | The Control Group |
|---|---|---|---|
| AOB | Nitrosomonas | 0.19% | 0.19% |
| NOB | Nitrospira | 0.83% | 1.22% |
| DNB | Zoogloea | 19.41% | 18.77% |
| Thauera | 0.26% | 1.30% | |
| Rhodobacter | 0.85% | 1.26% | |
| Arcobacter | 0.19% | 0.61% | |
| Acinetobacter | 0.56% | 0.34% | |
| Flavobacterium | 0.20% | 0.42% | |
| Comamonas | 0.03% | 0.06% | |
| Hyphomicrobium | 0.04% | 0.02% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, Y.; Qiu, S.; Zeng, H.; Ma, F.; Wang, J.; Qiu, Y.; An, X. Short-Term Effects of Tourmaline on Nitrogen Removals and Microbial Communities in a Sequencing Batch Reactor at Low Temperatures. Int. J. Environ. Res. Public Health 2018, 15, 1280. https://doi.org/10.3390/ijerph15061280
Han Y, Qiu S, Zeng H, Ma F, Wang J, Qiu Y, An X. Short-Term Effects of Tourmaline on Nitrogen Removals and Microbial Communities in a Sequencing Batch Reactor at Low Temperatures. International Journal of Environmental Research and Public Health. 2018; 15(6):1280. https://doi.org/10.3390/ijerph15061280
Chicago/Turabian StyleHan, Yahong, Shan Qiu, Hongyun Zeng, Fang Ma, Jue Wang, Yilun Qiu, and Xuedi An. 2018. "Short-Term Effects of Tourmaline on Nitrogen Removals and Microbial Communities in a Sequencing Batch Reactor at Low Temperatures" International Journal of Environmental Research and Public Health 15, no. 6: 1280. https://doi.org/10.3390/ijerph15061280
APA StyleHan, Y., Qiu, S., Zeng, H., Ma, F., Wang, J., Qiu, Y., & An, X. (2018). Short-Term Effects of Tourmaline on Nitrogen Removals and Microbial Communities in a Sequencing Batch Reactor at Low Temperatures. International Journal of Environmental Research and Public Health, 15(6), 1280. https://doi.org/10.3390/ijerph15061280




