A Predictive Model Has Identified Tick-Borne Encephalitis High-Risk Areas in Regions Where No Cases Were Reported Previously, Poland, 1999–2012
Abstract
1. Introduction
2. Materials and Methods
2.1. Spatial Resolution
2.2. Study Period and Area
2.3. Selection of Variables for the Model
2.4. Statistical Analysis
2.5. Mapping the Predicted Rates in All Municipalities
2.6. Validation of the Model
2.7. Statistical Software Used
3. Results
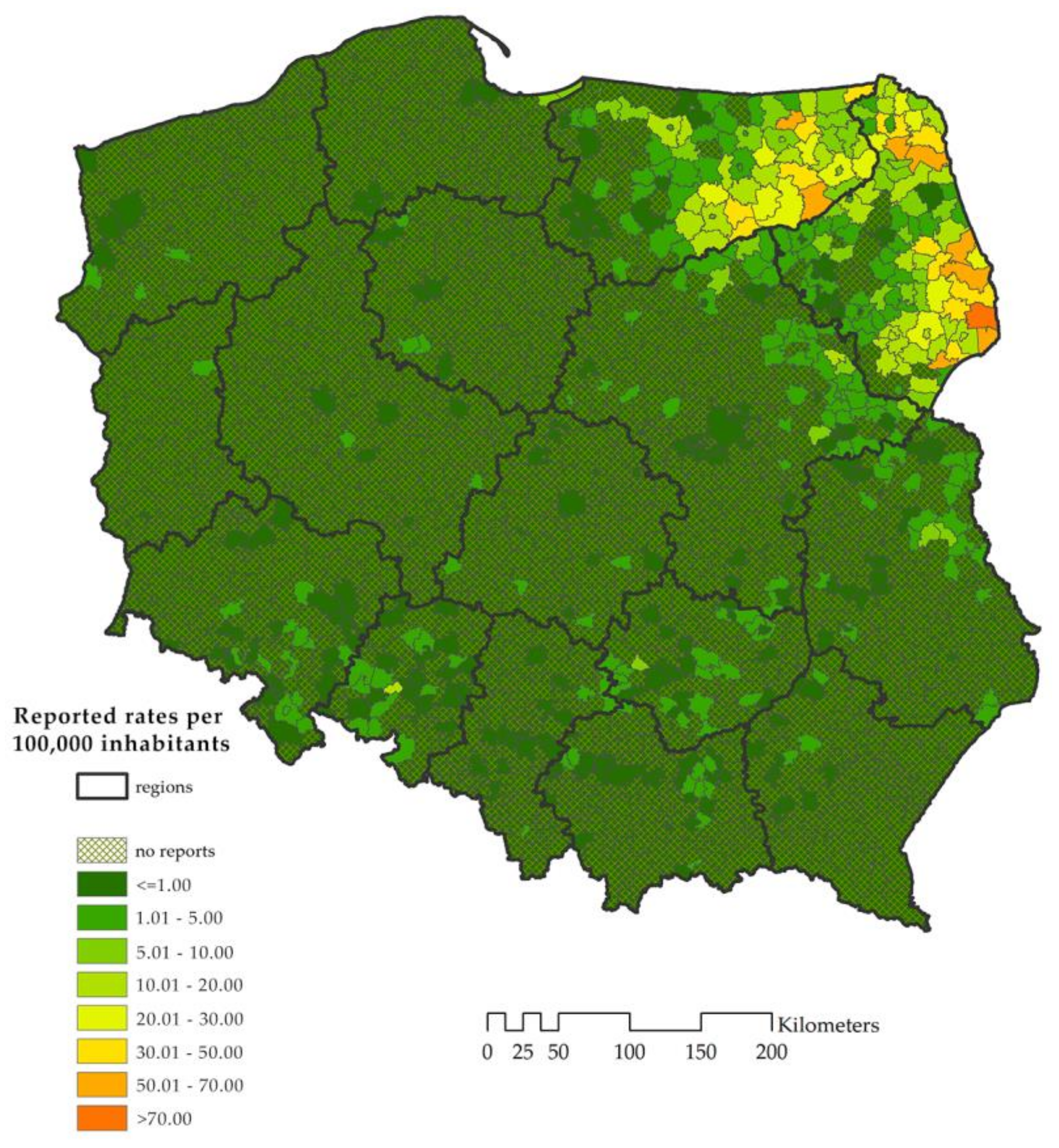
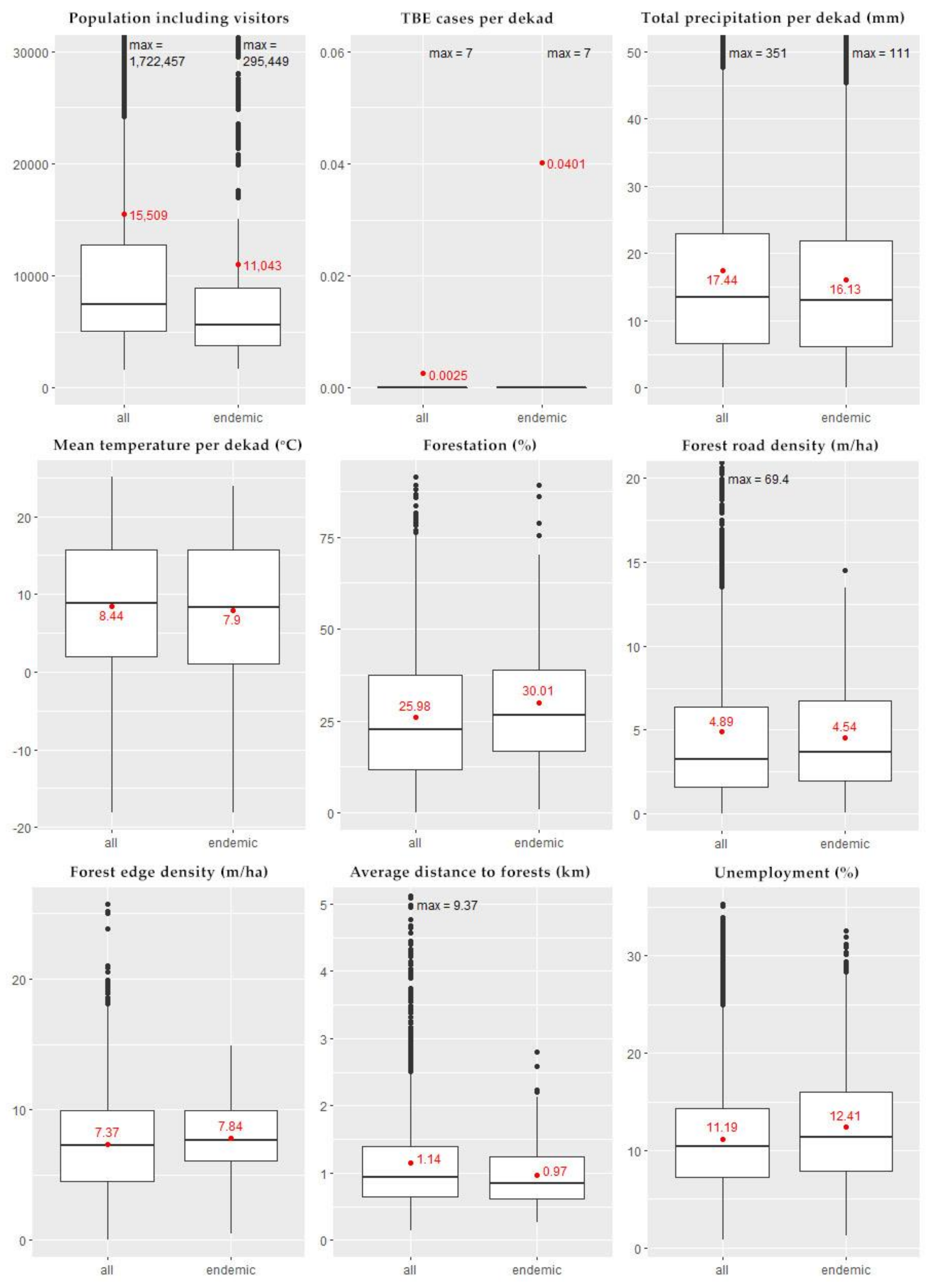
3.1. Descriptive Statistics
3.2. Time Series Analysis
3.3. Variable Type Selection
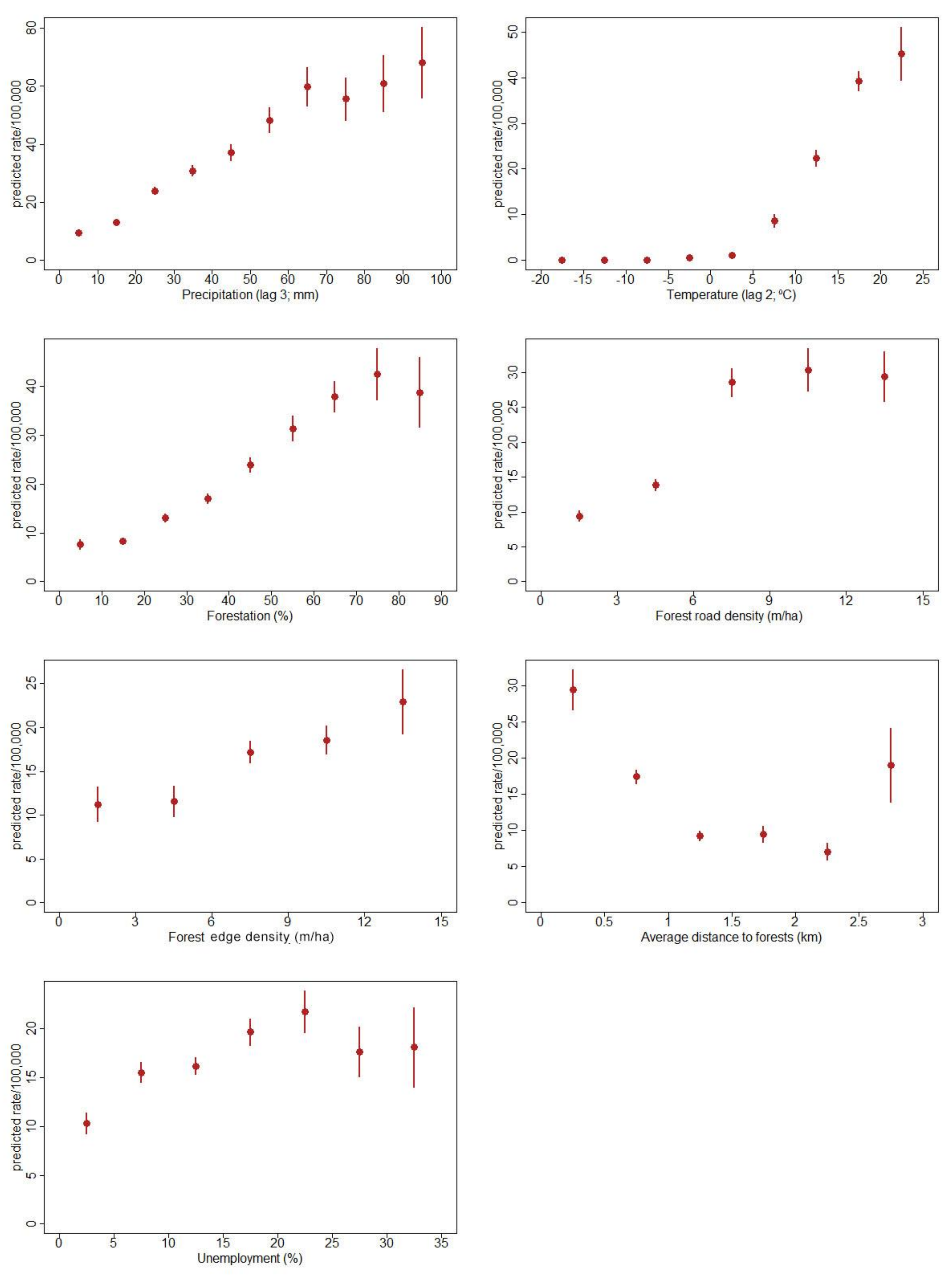
3.4. The Final Model
- Number of TBE cases in the previous dekad (first-order autocorrelation term);
- Sum of precipitation three dekads before (in mm);
- Temperature index: a binary variable indicating whether the mean air temperature two dekads before was above 0;
- Mean air temperature two dekads before (in °C);
- An interaction term between the above binary variable with the mean air temperature recorded two dekads before;
- Forestation (forested area divided by municipality area, in %);
- Forest edge density (length of forest edge divided by municipality area), categorized: 0–3 m/ha, 3–6, 6–9, 9–12, 12 and more (the most numerous category 6–9 km selected as reference level);
- Forest road density (length of forest roads divided by the municipality area, in m/ha);
- Average distance from settlements to forests (in kilometres); and
- Unemployment (number of unemployed divided by population in working age, in %).
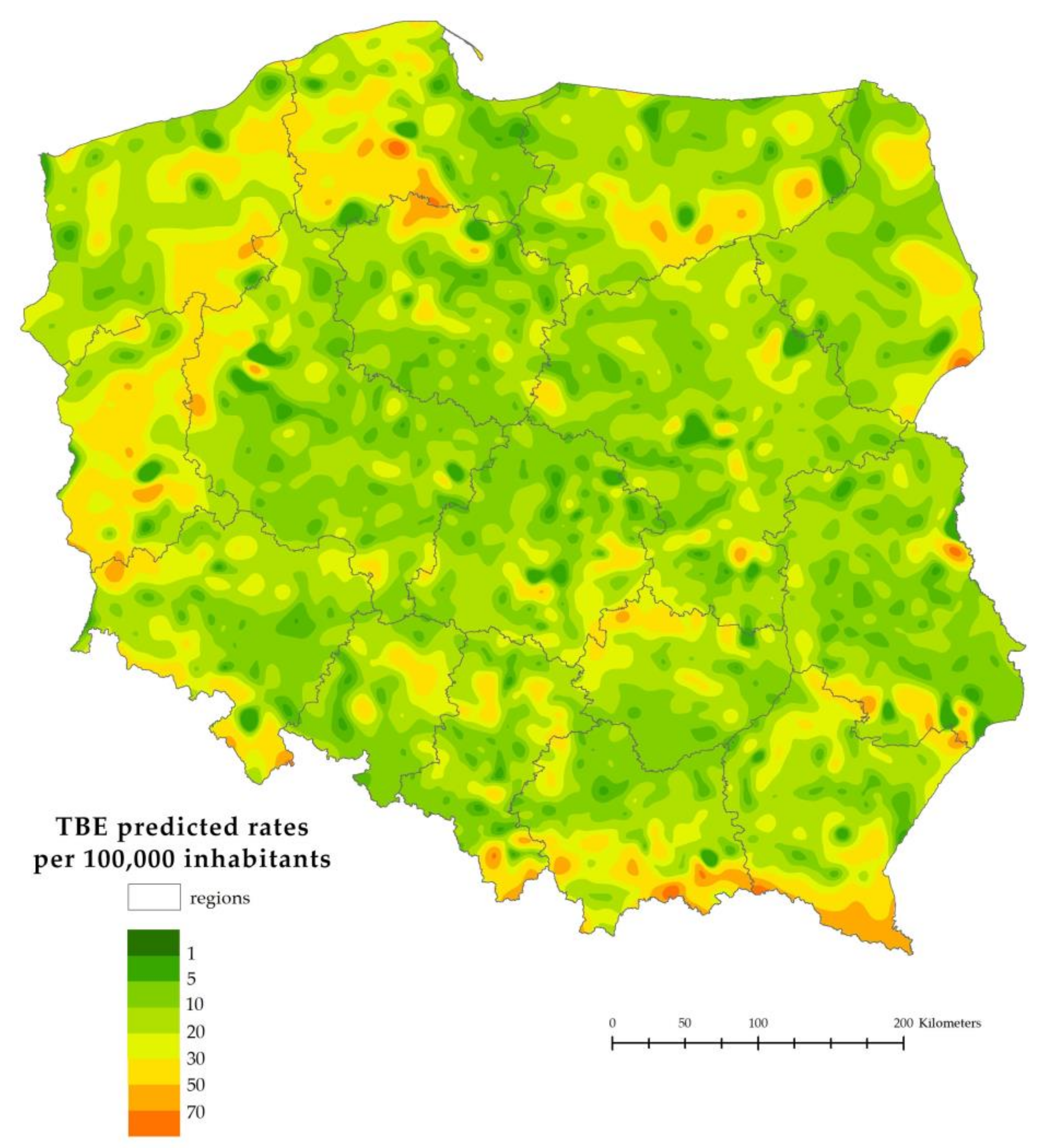
3.5. Validation of the Model
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Randolph, S.E. Tick-borne encephalitis virus, ticks and humans: Short-term and long-term dynamics. Curr. Opin. Infect. Dis. 2008, 21, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Dobler, G.; Hufert, F.T.; Pfeffer, M.; Essbauer, S. Tick-borne encephalitis: From microfocus to human disease. In Progress in Parasitology; Mehlhorn, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 1, pp. 323–331. [Google Scholar]
- Lindquist, L.; Vapalahti, O. Tick-borne encephalitis. Lancet 2008, 371, 1861–1871. [Google Scholar] [CrossRef]
- Rizzoli, A.; Hauffe, H.C.; Tagliapietra, V.; Neteler, M.; Rosà, R. Forest Structure and Roe Deer Abundance Predict Tick-Borne Encephalitis Risk in Italy. PLoS ONE 2009, 4, e4336. [Google Scholar] [CrossRef] [PubMed]
- Burri, C.; Bastic, V.; Maeder, G.; Patalas, E.; Gern, L. Microclimate and the Zoonotic Cycle of Tick-Borne Encephalitis Virus in Switzerland. J. Med. Entomol. 2011, 48, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Lambin, E.F.; Tran, A.; Vanwambeke, S.O.; Linard, C.; Soti, V. Pathogenic landscapes: Interactions between land, people, disease vectors, and their animal hosts. Int. J. Health Geogr. 2010, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Zeman, P.; Pazdiora, P.; Benes, C. Spatio-temporal variation of tick-borne encephalitis (TBE) incidence in the Czech Republic: Is the current explanation of the disease's rise satisfactory? Ticks Tick-Borne Dis. 2010, 1, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Daniel, M.; Kriz, B.; Danielova, V.; Valter, J.; Benes, C. Changes of meteorological factors and tick-borne encephalitis incidence in the Czech Republic. Epidemiol. Mikrobiol. Imunol. 2009, 58, 179–187. [Google Scholar] [PubMed]
- Vanwambeke, S.O.; Šumilo, D.; Bormane, A.; Lambin, E.F.; Randolph, S.E. Landscape Predictors of Tick-Borne Encephalitis in Latvia: Land Cover, Land Use, and Land Ownership. Vector-Borne Zoonotic Dis. 2010, 10, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Zeimes, C.B.; Olsson, G.E.; Hjertqvist, M.; Vanwambeke, S.O. Shaping zoonosis risk: Landscape ecology vs. landscape attractiveness for people, the case of tick-borne encephalitis in Sweden. Parasit Vectors 2014, 7, 370. [Google Scholar] [CrossRef] [PubMed]
- Šumilo, D.; Bormane, A.; Asokliene, L.; Vasilenko, V.; Golovljova, I.; Avsic Zupanc, T.; Hubalek, Z.; Randolph, S.E. Socio-economic factors in the differential upsurge of tick-borne encephalitis in central and Eastern Europe. Rev. Med. Virol. 2008, 18, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Kiffner, C.; Zucchini, W.; Schomaker, P.; Vor, T.; Hagedorn, P.; Niedrig, M.; Rühe, F. Determinants of tick-borne encephalitis in counties of southern Germany, 2001–2008. Int. J. Health Geogr. 2010, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Peña, A.; Gray, J.S.; Kahl, O.; Lane, R.S.; Nijhof, A.M. Research on the ecology of ticks and tick-borne pathogens--methodological principles and caveats. Front. Cell. Infect. Microbiol. 2013, 3, 29. [Google Scholar] [CrossRef] [PubMed]
- Randolph, S.E.; Green, R.M.; Peacey, M.F.; Rogers, D.J. Seasonal synchrony: The key to tick-borne encephalitis foci identified by satellite data. Parasitology 2000, 121, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Wroblewska, Z.; Dobrzynski, L.; Olkowska, D.; Magdzik, W.; Zaleska, H. A serologic survey of the healthy population of Poland for encephalitis arboviruses in the years 1965–1967. Przegl. Epidemiol. 1968, 22, 293–307. [Google Scholar]
- Wroblewska, Z.; Zabicka, J.; Nawrocka, E.; Olkowska, D.; Taytsch-Kapulkin, F.Z. Occurrence of arbovirus antibodies in foresters in Poland in 1971–1972. Acta Microbiol. Pol. 1973, 22, 123–130. [Google Scholar]
- Stefanoff, P.; Siennicka, J.; Kaba, J.; Nowicki, M.; Ferenczi, E.; Gut, W. Identification of new endemic tick-borne encephalitis foci in Poland—A pilot seroprevalence study in selected regions. Int. J. Med. Microbiol. 2008, 298, 102–107. [Google Scholar] [CrossRef]
- Stefanoff, P.; Rogalska, J.; Kaba, J.; Nowicki, M.; Gut, W. Prevalence of anti-TBEV antibodies in goats as a tool to validate high-risk areas for tick-borne encephalitis in Poland. In Proceedings of the The European Scientific Conference on Applied Infectious Disease Epidemiology, Berlin, Germany, 19–21 November 2008; p. 42. [Google Scholar]
- Stefanoff, P.; Rogalska, J.; Zajkowska, J.; Czerska, M.; Seroka, W.; Czarkowski, M.P. Surveillance of aseptic central nervous system infections in Poland: Is it meeting its objectives? Eurosurveillance 2011, 16. [Google Scholar] [CrossRef]
- Stefanoff, P.; Zielicka-Hardy, A.; Hlebowicz, M.; Konior, R.; Lipowski, D.; Szenborn, L.; Siennicka, J.; Orliková, H. TBE Enhanced Surveillance Working Group. New endemic foci of tick-borne encephalitis (TBE) identified in districts where testing for TBE was not available before 2009 in Poland. Parasit Vectors 2013, 6, 180. [Google Scholar] [CrossRef] [PubMed]
- Tveito, O.E.; Wegehenkel, M.; van der Wel, F.; Dobesch, H. The Use of Geographic Information Systems in Climatology and Meteorology; Office for Official Publications of the European Communities: Luxembourg, 2008; pp. 48–53. [Google Scholar]
- Hofstra, N.; Haylock, M.; New, M.; Jones, P.; Frei, C. Comparison of six methods for the interpolation of daily, European climate data. J. Geophys. Res: Atmos. 2008, 113, D05109. [Google Scholar] [CrossRef]
- Ustrnul, Z.; Czekierda, D. Application of GIS for the development of climatological air temperature maps: An example from Poland. Meteorol. Appl. 2005, 12, 43–50. [Google Scholar] [CrossRef]
- Lupikasza, E.; Ustrnul, Z.; Czekierda, D. The role of explanatory variables in spatial interpolation of selected climate elements. Ann. Geomat. 2007, 7, 55–68. [Google Scholar]
- Randolph, S.E.; Asokliene, L.; Avsic Zupanc, T.; Bormane, A.; Burri, C.; Gern, L.; Golovljova, I.; Hubalek, Z.; Knap, N.; Kondrusik, M.; et al. Variable spikes in tick-borne encephalitis incidence in 2006 independent of variable tick abundance but related to weather. Parasit Vectors 2008, 1, 44. [Google Scholar] [CrossRef] [PubMed]
- Stefanoff, P.; Rosińska, M.; Samuels, S.; White, D.J.; Morse, D.L.; Randolph, S.E. A national case-control study identifies human socio-economic status and activities as risk factors for tick-borne encephalitis in Poland. PLoS ONE 2012, 7, e45511. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.E.; Hilborn, E.D.; Thomas, J.C. Towards landscape design guidelines for reducing Lyme disease risk. Int. J. Epidemiol. 2006, 35, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Zamba, G.K.D.; Cavanaugh, J.E. Markov regression models for count time series with excess zeros: A partial likelihood approach. Stat. Methodol. 2013, 14, 26–38. [Google Scholar] [CrossRef]
- Wójcik-Fatla, A.; Zając, V.; Sawczyn, A.; Sroka, J.; Cisak, E.; Dutkiewicz, J. Infections and mixed infections with the selected species of Borrelia burgdorferi sensu lato complex in Ixodes ricinus ticks collected in eastern Poland: A significant increase in the course of 5 years. Exp. Appl. Acarol. 2016, 68, 197–212. [Google Scholar] [CrossRef] [PubMed]
- Biernat, B.; Stańczak, J.; Michalik, J.; Sikora, B.; Wierzbicka, A. Prevalence of infection with Rickettsia helvetica in Ixodes ricinus ticks feeding on non-rickettsiemic rodent hosts in sylvatic habitats of west-central Poland. Ticks Tick-Borne Dis. 2016, 7, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Stefanoff, P.; Pfeffer, M.; Hellenbrand, W.; Rogalska, J.; Rühe, F.; Makówka, A.; Michalik, J.; Wodecka, B.; Rymaszewska, A.; Kiewra, D.; et al. Virus detection in questing ticks is not a sensitive indicator for risk assessment of tick-borne encephalitis in humans. Zoonoses Public Health 2013, 60, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Drelich, A.; Andreassen, Å.; Vainio, K.; Kruszyński, P.; Wąsik, T.J. Prevalence of tick-borne encephalitis virus in a highly urbanized and low risk area in Southern Poland. Ticks Tick-Borne Dis. 2014, 5, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Kiewra, D.; Stefańska-Krzaczek, E.; Szymanowski, M.; Szczepańska, A. Local-scale spatio-temporal distribution of questing Ixodes ricinus L. (Acari: Ixodidae)-A case study from a riparian urban forest in Wrocław, SW Poland. Ticks Tick-Borne Dis. 2017, 8, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Stefanoff, P.; Orliková, H.; Príkazský, V.; Bene, C.; Rosińska, M. Cross-border surveillance differences: Tick-borne encephalitis and lyme borreliosis in the Czech Republic and Poland, 1999-2008. Cent. Eur. J. Public Health 2014, 22, 54–59. [Google Scholar] [PubMed]
- Randolph, S. Predicting the risk of tick-borne diseases. Int. J. Med. Microbiol. 2002, 291, 6–10. [Google Scholar] [CrossRef]

| Variable Description | Granularity | Unit | Source | Data Processing |
|---|---|---|---|---|
| Number of TBE cases | By dekaddekad of onset | Count | National Institute of Public Health | We assigned notified cases to their municipality of exposure, by dekaddekad of onset. |
| Population denominator | By year | Count | Central Statistical Office | For each municipality, we obtained the population estimates on the 30 June of each year. Since we assigned cases by municipality of exposure, the numerator included both residents and tourists. Therefore, we added to the denominator the estimated number of visiting tourists (Polish nationals), based on the Central Statistical Office estimate of the number of bed-days occupied by visitors, divided the number of days in a year. For 1999–2003, we imputed the proportions of municipality population increases. |
| Mean temperature | By dekaddekad | °C | Institute of Meteorology | We used mean daily air temperature measurements from 54 synoptic weather stations evenly distributed in Poland. To assign measurements to each municipality, we used residual kriging—a spatial interpolation method [21,22] previously validated for use with Polish meteorological data [23]. For each dekaddekad, we calculated the mean temperature and created a raster map at 250 m spatial resolution, including the values interpolated from the 54 stations. Then we used a vector map of NUTS-5 boundaries to assign the average value to each municipality. |
| Sum of precipitation | By dekaddekad | Mm | Institute of Meteorology | We used daily sum of precipitation measurements from 54 meteorological stations. To assign measurements to each municipality, we used co-kriging, recommended when spatial correlation is found between covariables and the variable of interest and when the covariables are oversampled with respect to the primary variable [21]. The method was previously validated with Polish data [24]. For each dekaddekad, we calculated the total precipitation and created a raster map at 250 m spatial resolution, including the values interpolated from the 54 stations. Then we used a vector map of NUTS-5 boundaries to assign the average value to each municipality. |
| Unemployed | By year | Count | Central Statistical Office | Data at the municipality level on the number of registered unemployed were available for 2003–2012. For 1999–2002, we imputed these numbers to each municipality based on the numbers recorded in districts (NUTS-4), according to the proportional distribution between municipalities forming each district, as observed during 2003–2012. |
| Forested area | Calculated once for study period | Ha | CORINE Land Cover 2006 NUTS-5 boundaries | We merged all polygons representing forest classes (CLC code 3.1). We intersected the forest layer with the map of NUTS-5 administrative boundaries to obtain the area of forests contained in each municipality. |
| Length of forest edge | Calculated once for study period | Km | CORINE Land Cover 2006 NUTS-5 boundaries | Using the above described forest layer, we converted the forest polygons to lines. Then we intersected the resulting layer with the NUTS-5 administrative boundaries. We excluded segments overlaying with the municipality boundaries or located within a 50 m buffer, to account for the results of the intersection between forest edges with administrative boundaries. Then we computed the remaining length of lines for each municipality in km. |
| Average distance from settlements to forests | Calculated once for study period | Km | CORINE Land Cover 2006 IMAGIS settlement map NUTS-5 boundaries | We used the proximity (raster distance) function of QGIS to calculate the distances between forests (from CORINE CLC 3.1) at 100 m resolution. Then we converted the data raster into a polygon distance layer, where each 100 × 100 m polygon had an attribute describing the distance from the nearest forest. Next, we intersected the above described distance polygon layer with two complementary maps: the polygon CORINE map (CLC code 1.1 urban fabric), containing more precise information on urban settlements and a more detailed point map of smaller settlements (after deleting points overlapping with urban fabric polygons). We intersected both maps with the polygon distance layer, and calculated the average distance from settlements to forests, using the mean of both values for each municipality. |
| Length of forest roads | Calculated once for study period | Km | CORINE Land Cover 2006 www.geofabrik.de/ NUTS-5 boundaries | We intersected the layer containing the road network with the CORINE map of forests (CLC 3.1) and with the NUTS-5 boundaries. We extracted all types of roads crossing the forests polygons. We calculated the total length in km in each municipality. |
| Variable | Coefficient | Level of Significance |
|---|---|---|
| LOG-LINEAR PART | ||
| Number of TBE cases (−1 dekad) | 0.215 | *** |
| Sum of precipitation (−3 dekads) | 0.009 | *** |
| Temperature index (if > 0 °C) | 2.071 | *** |
| Mean temperature (−2 dekads) | −0.227 | NS |
| Interaction (temp. index × mean temp.) | 0.203 | NS |
| Forestation | 0.036 | *** |
| Forest edge density (ref: 6–9 m/ha) | - | - |
| 0–3 | 0.321 | NS |
| 3–6 | −0.505 | *** |
| 9–12 | 0.299 | ** |
| >12 | 0.736 | *** |
| Forest road density | −0.059 | ** |
| Average distance to forests | 0.139 | NS |
| Unemployment | 0.047 | *** |
| Constant in the model | −11.197 | *** |
| LOGISTIC PART | ||
| Number of TBE cases (−1 dekad) | −0.839 | *** |
| Sum of precipitation (−3 dekads) | −0.010 | * |
| Temperature index (if >0 °C) | 1.600 | * |
| Mean temperature (−2 dekads) | −0.493 | *** |
| Interaction (temp. index * mean temp.) | 0.219 | NS |
| Forestation | 0.032 | *** |
| Forest edge density (ref: 6–9 m/ha) | - | - |
| 0–3 | 0.148 | NS |
| 3–6 | −0.374 | NS |
| 9–12 | 0.472 | * |
| >12 | 1.074 | *** |
| Forest road density | −0.194 | *** |
| Average distance to forests | 0.568 | NS |
| Unemployment | 0.072 | *** |
| Constant in the model | 0.704 | NS |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stefanoff, P.; Rubikowska, B.; Bratkowski, J.; Ustrnul, Z.; Vanwambeke, S.O.; Rosinska, M. A Predictive Model Has Identified Tick-Borne Encephalitis High-Risk Areas in Regions Where No Cases Were Reported Previously, Poland, 1999–2012. Int. J. Environ. Res. Public Health 2018, 15, 677. https://doi.org/10.3390/ijerph15040677
Stefanoff P, Rubikowska B, Bratkowski J, Ustrnul Z, Vanwambeke SO, Rosinska M. A Predictive Model Has Identified Tick-Borne Encephalitis High-Risk Areas in Regions Where No Cases Were Reported Previously, Poland, 1999–2012. International Journal of Environmental Research and Public Health. 2018; 15(4):677. https://doi.org/10.3390/ijerph15040677
Chicago/Turabian StyleStefanoff, Pawel, Barbara Rubikowska, Jakub Bratkowski, Zbigniew Ustrnul, Sophie O. Vanwambeke, and Magdalena Rosinska. 2018. "A Predictive Model Has Identified Tick-Borne Encephalitis High-Risk Areas in Regions Where No Cases Were Reported Previously, Poland, 1999–2012" International Journal of Environmental Research and Public Health 15, no. 4: 677. https://doi.org/10.3390/ijerph15040677
APA StyleStefanoff, P., Rubikowska, B., Bratkowski, J., Ustrnul, Z., Vanwambeke, S. O., & Rosinska, M. (2018). A Predictive Model Has Identified Tick-Borne Encephalitis High-Risk Areas in Regions Where No Cases Were Reported Previously, Poland, 1999–2012. International Journal of Environmental Research and Public Health, 15(4), 677. https://doi.org/10.3390/ijerph15040677







