The Role of Vitamin D and Oxidative Stress in Chronic Kidney Disease
Abstract
1. Introduction
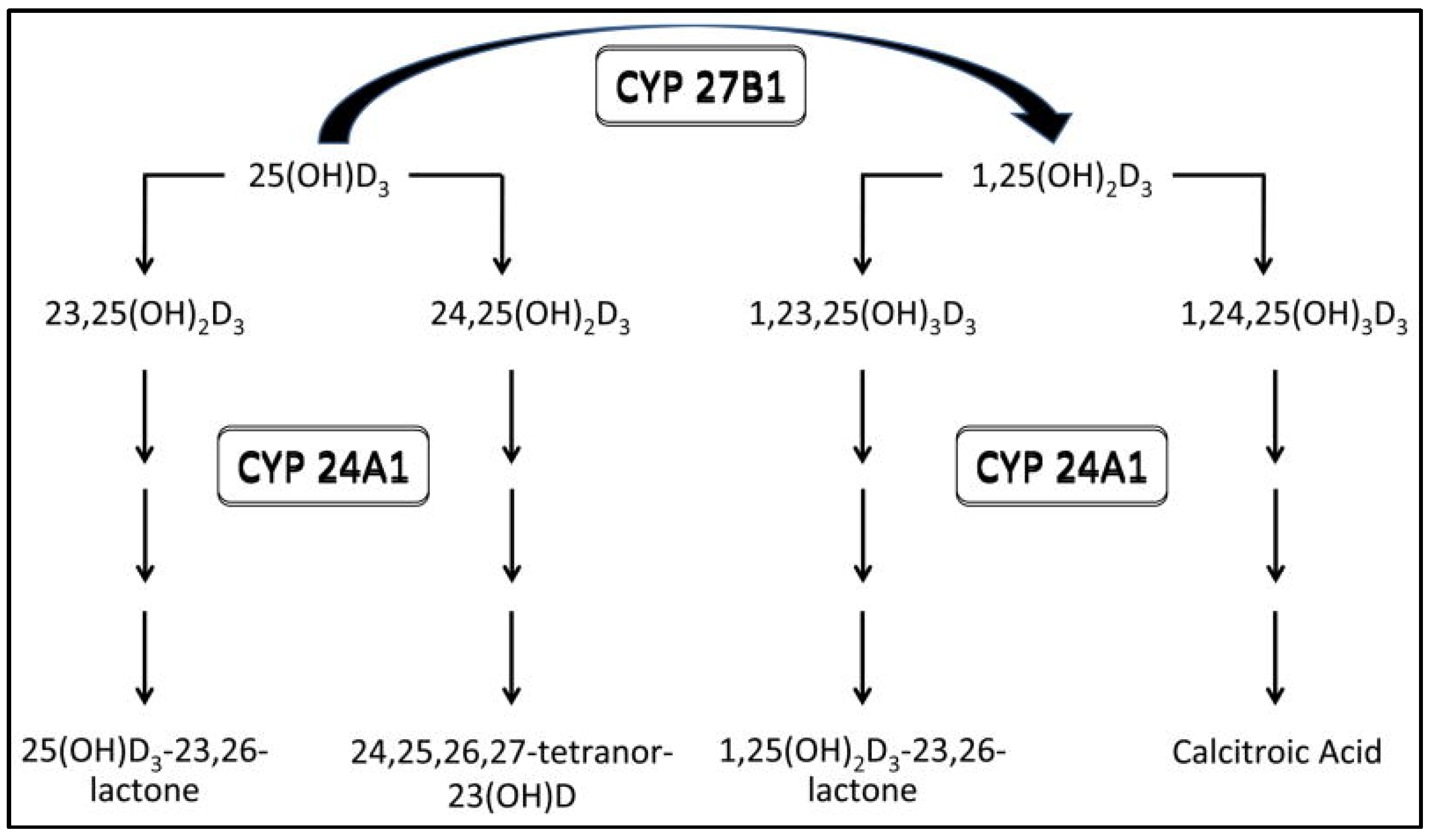
2. Vitamin D Deficiency/Insufficiency in Chronic Kidney Disease
3. Vitamin D, Oxidative Stress and Inflammation
4. Select Pre-Clinical and Clinical Studies
5. Possible Implications for Disparities
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Blair, D.; Byham-Gray, L.; Lewis, E.; McCaffrey, S. Prevalence of vitamin D [25(OH)D] deficiency and effects of supplementation with ergocalciferol (vitamin D2) in stage 5 chronic kidney disease patients. J. Ren. Nutr. 2008, 18, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Mehrotra, R.; Kermah, D.; Budoff, M.; Salusky, I.B.; Mao, S.S.; Gao, Y.L.; Takasu, J.; Adler, S.; Norris, K. Hypovitaminosis D in chronic kidney disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Bakris, G.L.; Molitchm, M.; Smulders, M.; Tian, J.; Williams, L.A.; Andress, D.L. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: Results of the study to evaluate early kidney disease. Kidney Int. 2007, 71, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Broe, K.E.; Chen, T.C.; Weinberg, J.; Bischoff-Ferrari, H.A.; Holick, M.F.; Kiel, D.P. A higher dose of vitamin D reduces the risk of falls in nursing home residents: A randomized, multiple-dose study. J. Am. Geriatr. Soc. 2007, 55, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Garland, C.F.; Garland, F.C.; Gorham, E.D.; Lipkin, M.; Newmark, H.; Mohr, S.B.; Holick, M.F. The role of vitamin D in cancer prevention. Am. J. Public Health 2006, 96, 252–261. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E.; Liu, Y.; Rimm, E.B.; Hollis, B.W.; Fuchs, C.S.; Stampfer, M.J.; Willett, W.C. Prospective study of predictors of vitamin D status and cancer incidence and mortality in men. J. Natl. Cancer Inst. 2006, 98, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D: Importance in the prevention of cancers, type 1 diabetes, heart disease, and osteoporosis. Am. J. Clin. Nutr. 2004, 79, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P. Vitamin D in health and disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1535–1541. [Google Scholar] [CrossRef] [PubMed]
- IOM. Dietary Reference Intakes for Calcium and Vitamin D. Available online: https://www.ncbi.nlm.nih.gov/books/NBK56070/) (accessed on 14 October 2018).
- Holick, M.F.; Binkleym, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine Society. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P. Toward a physiological referent for the vitamin D requirement. J. Endocrinol. Invest. 2014, 37, 1127–1130. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.A.; Gray, R.W.; Lemann, J., Jr. Urinary excretion of 25-hydroxyvitamin D in health and the nephrotic syndrome. J. Lab. Clin. Med. 1982, 99, 325–330. [Google Scholar] [PubMed]
- Holick, M.F. Vitamin D for health and in chronic kidney disease. Semin. Dial. 2005, 18, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Jacob, A.I.; Sallman, A.; Santiz, Z.; Hollis, B.W. Defective photoproduction of cholecalciferol in normal and uremic humans. J. Nutr. 1984, 114, 1313–1319. [Google Scholar] [CrossRef] [PubMed]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2017, 7, 1–59. [Google Scholar] [CrossRef]
- Bosworth, C.; de Boer, I.H. Impaired vitamin D metabolism in CKD. Semin. Nephrol. 2013, 33, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2009. [Google Scholar] [CrossRef]
- Jones, D.P. Redefining oxidative stress. Antioxid. Redox Signal. 2006, 8, 1865–1879. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. Vitamin D deficiency accelerates ageing and age-related diseases: A novel hypothesis. J. Physiol. 2017, 595, 6825–6836. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. Vitamin D: A custodian of cell signalling stability in health and disease. Biochem. Soc. Trans. 2015, 43, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Pedruzzi, L.M.; Stockler-Pinto, M.B.; Leite, M., Jr.; Mafra, D. Nrf2-keap1 system versus NF-kappaB: The good and the evil in chronic kidney disease? Biochimie 2012, 94, 2461–2466. [Google Scholar] [CrossRef] [PubMed]
- Nakai, K.; Fujii, H.; Kono, K.; Goto, S.; Kitazawa, R.; Kitazawa, S.; Hirata, M.; Shinohara, M.; Fukagawa, M.; Nishi, S. Vitamin D activates the Nrf2-Keap1 antioxidant pathway and ameliorates nephropathy in diabetic rats. Am. J. Hypertens. 2014, 27, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, T.M.; da Costa, D.C.; Resende, A.C.; Soulage, C.O.; Bezerra, F.F.; Daleprane, J.B. Activation of Nrf2-Antioxidant Signaling by 1,25-Dihydroxycholecalciferol Prevents Leptin-Induced Oxidative Stress and Inflammation in Human Endothelial Cells. J. Nutr. 2017, 147, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, H.; Sun, X.; Ren, L. The protective role of vitamin D3 in a murine model of asthma via the suppression of TGF-beta/Smad signaling and activation of the Nrf2/HO-1 pathway. Mol. Med. Rep. 2016, 14, 2389–2396. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.G.; Liu, Y.X.; Wang, H.; Wang, B.P.; Qu, H.Q.; Wang, B.L.; Zhu, M. Active form of vitamin D ameliorates non-alcoholic fatty liver disease by alleviating oxidative stress in a high-fat diet rat model. Endocr. J. 2017, 64, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Akbari, M.; Ostadmohammadi, V.; Lankarani, K.B.; Tabrizi, R.; Kolahdooz, F.; Heydari, S.T.; Kavari, S.H.; Mirhosseini, N.; Mafi, A.; Dastorani, M.; et al. The effects of vitamin D supplementation on biomarkers of inflammation and oxidative stress among women with polycystic ovary syndrome: A systematic review and meta-analysis of randomized controlled trials. Horm. Metab. Res. 2018, 50, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Thethi, T.K.; Bajwa, M.A.; Ghanim, H.; Jo, C.; Weir, M.; Goldfine, A.B.; Umpierrez, G.; Desouza, C.; Dandona, P.; Fang-Hollingsworth, Y.; et al. Effect of paricalcitol on endothelial function and inflammation in type 2 diabetes and chronic kidney disease. J. Diabetes Complications 2015, 29, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Alborzi, P.; Patel, N.A.; Peterson, C.; Bills, J.E.; Bekele, D.M.; Bunaye, Z.; Light, R.P.; Agarwal, R. Paricalcitol reduces albuminuria and inflammation in chronic kidney disease: A randomized double-blind pilot trial. Hypertension 2008, 52, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Coyne, D.W.; Goldberg, S.; Faber, M.; Ghossein, C.; Sprague, S.M. A randomized multicenter trial of paricalcitol versus calcitriol for secondary hyperparathyroidism in stages 3-4 CKD. Clin. J. Am. Soc. Nephrol. 2014, 9, 1620–1626. [Google Scholar] [CrossRef] [PubMed]
- Thadhani, R.; Appelbaum, E.; Pritchett, Y.; Chang, Y.; Wenger, J.; Tamez, H.; Bhan, I.; Agarwal, R.; Zoccali, C.; Wanner, C. Vitamin D therapy and cardiac structure and function in patients with chronic kidney disease: The PRIMO randomized controlled trial. JAMA 2012, 307, 674–684. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.Y.; Fang, F.; Chan, J.; Wen, Y.Y.; Qing, S.; Chan, I.H.; Lo, G.; Lai, K.N.; Lo, W.K.; Lam, C.W.; et al. Effect of paricalcitol on left ventricular mass and function in CKD—The OPERA trial. J. Am. Soc. Nephrol. 2014, 25, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Martins, D.; Wolf, M.; Pan, D.; Zadshir, A.; Tareen, N.; Thadhani, R.; Felsenfeld, A.; Levine, B.; Mehrotra, R.; Norris, K. Prevalence of cardiovascular risk factors and the serum levels of 25-hydroxyvitamin D in the United States: Data from the Third National Health and Nutrition Examination Survey. Arch. Intern. Med. 2007, 167, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- Artaza, J.N.; Contreras, S.; Garcia, L.A.; Mehrotra, R.; Gibbons, G.; Shohet, R.; Martins, D.; Norris, K.C. Vitamin D and cardiovascular disease: Potential role in health disparities. J. Health Care Poor Underserved 2011, 22, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Ginde, A.A.; Liu, M.C.; Camargo, C.A., Jr. Demographic differences and trends of vitamin D insufficiency in the U.S. population, 1988–2004. Arch. Intern. Med. 2009, 169, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Saran, R.; Robinson, B.; Abbott, K.C.; Agodoa, L.Y.C.; Bhave, N.; Bragg-Gresham, J.; Balkrishnan, R.; Dietrich, X.; Eckard, A.; Eggers, P.W.; et al. U.S. renal data system 2017 annual data report: Epidemiology of kidney disease in the United States. Am. J. Kidney Dis. 2018, 71, A7. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.; Betancourt, J.; Chang, Y.; Shah, A.; Teng, M.; Tamez, H.; Gutierrez, O.; Camargo, C.A., Jr.; Melamed, M.; Norris, K.; et al. Impact of activated vitamin D and race on survival among hemodialysis patients. J. Am. Soc. Nephrol. 2008, 19, 1379–1388. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Miller, J.E.; Kovesdy, C.P.; Mehrotra, R.; Lukowsky, L.R.; Streja, E.; Ricks, J.; Jing, J.; Nissenson, A.R.; Greenland, S.; et al. Impact of race on hyperparathyroidism, mineral disarrays, administered vitamin D mimetic, and survival in hemodialysis patients. J. Bone Miner. Res. 2010, 25, 2724–2734. [Google Scholar] [CrossRef] [PubMed]
- Powe, C.E.; Karumanchi, S.A.; Thadhani, R. Vitamin D-binding protein and vitamin D in blacks and whites. N. Engl. J. Med. 2014, 370, 880–881. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, O.M.; Farwell, W.R.; Kermah, D.; Taylor, E.N. Racial differences in the relationship between vitamin D, bone mineral density, and parathyroid hormone in the National Health and Nutrition Examination Survey. Osteoporos. Int. 2011, 22, 1745–1753. [Google Scholar] [CrossRef] [PubMed]
- Robinson-Cohen, C.; Hoofnagle, A.N.; Ix, J.H.; Sachs, M.C.; Tracy, R.P.; Siscovick, D.S.; Kestenbaum, B.R.; de Boer, I.H. Racial differences in the association of serum 25-hydroxyvitamin D concentration with coronary heart disease events. JAMA 2013, 310, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Norris, K.C.; Williams, S.F. Race/ethnicity, serum 25-hydroxyvitamin D, and heart disease. JAMA 2013, 310, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Berg, A.H.; Powe, C.E.; Evans, M.K.; Wenger, J.; Ortiz, G.; Zonderman, A.B.; Suntharalingam, P.; Lucchesi, K.; Powe, N.R.; Karumanchi, S.A.; et al. 24,25-Dihydroxyvitamin d3 and vitamin D status of community-dwelling black and white Americans. Clin. Chem. 2015, 61, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, O.M.; Isakova, T.; Andress, D.L.; Levin, A.; Wolf, M. Prevalence and severity of disordered mineral metabolism in Blacks with chronic kidney disease. Kidney Int. 2008, 73, 956–962. [Google Scholar] [CrossRef] [PubMed]
- Melamed, M.L.; Chonchol, M.; Gutiérrez, O.M.; Kalantar-Zadeh, K.; Kendrick, J.; Norris, K.; Scialla, J.J.; Thadhani, R. The role of vitamin D in CKD stages 3 to 4: Report of a scientific workshop sponsored by the National Kidney Foundation. Am. J. Kidney Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Cook, N.R.; Lee, I.-M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Norris, K.C.; Olabisi, O.; Barnett, M.E.; Meng, Y.-X.; Martins, D.; Obialo, C.; Lee, J.E.; Nicholas, S.B. The Role of Vitamin D and Oxidative Stress in Chronic Kidney Disease. Int. J. Environ. Res. Public Health 2018, 15, 2701. https://doi.org/10.3390/ijerph15122701
Norris KC, Olabisi O, Barnett ME, Meng Y-X, Martins D, Obialo C, Lee JE, Nicholas SB. The Role of Vitamin D and Oxidative Stress in Chronic Kidney Disease. International Journal of Environmental Research and Public Health. 2018; 15(12):2701. https://doi.org/10.3390/ijerph15122701
Chicago/Turabian StyleNorris, Keith C., Opeyemi Olabisi, M. Edwina Barnett, Yuan-Xiang Meng, David Martins, Chamberlain Obialo, Jae Eun Lee, and Susanne B. Nicholas. 2018. "The Role of Vitamin D and Oxidative Stress in Chronic Kidney Disease" International Journal of Environmental Research and Public Health 15, no. 12: 2701. https://doi.org/10.3390/ijerph15122701
APA StyleNorris, K. C., Olabisi, O., Barnett, M. E., Meng, Y.-X., Martins, D., Obialo, C., Lee, J. E., & Nicholas, S. B. (2018). The Role of Vitamin D and Oxidative Stress in Chronic Kidney Disease. International Journal of Environmental Research and Public Health, 15(12), 2701. https://doi.org/10.3390/ijerph15122701





