Effectiveness of a Malaria Surveillance Strategy Based on Active Case Detection during High Transmission Season in the Peruvian Amazon
Abstract
1. Introduction
2. Materials and Methods
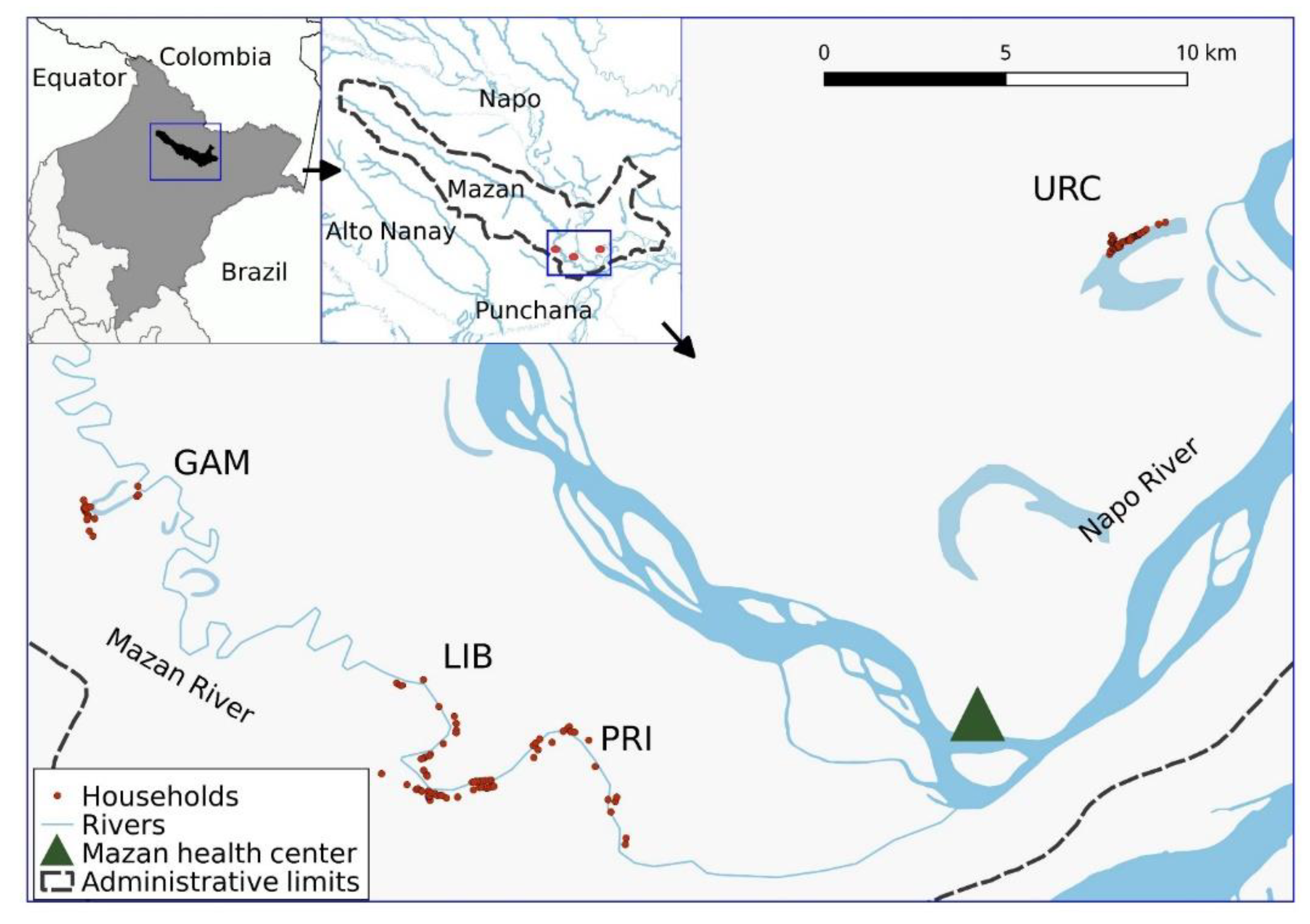
2.1. Study Design and Study Area
2.2. Selection of Communities
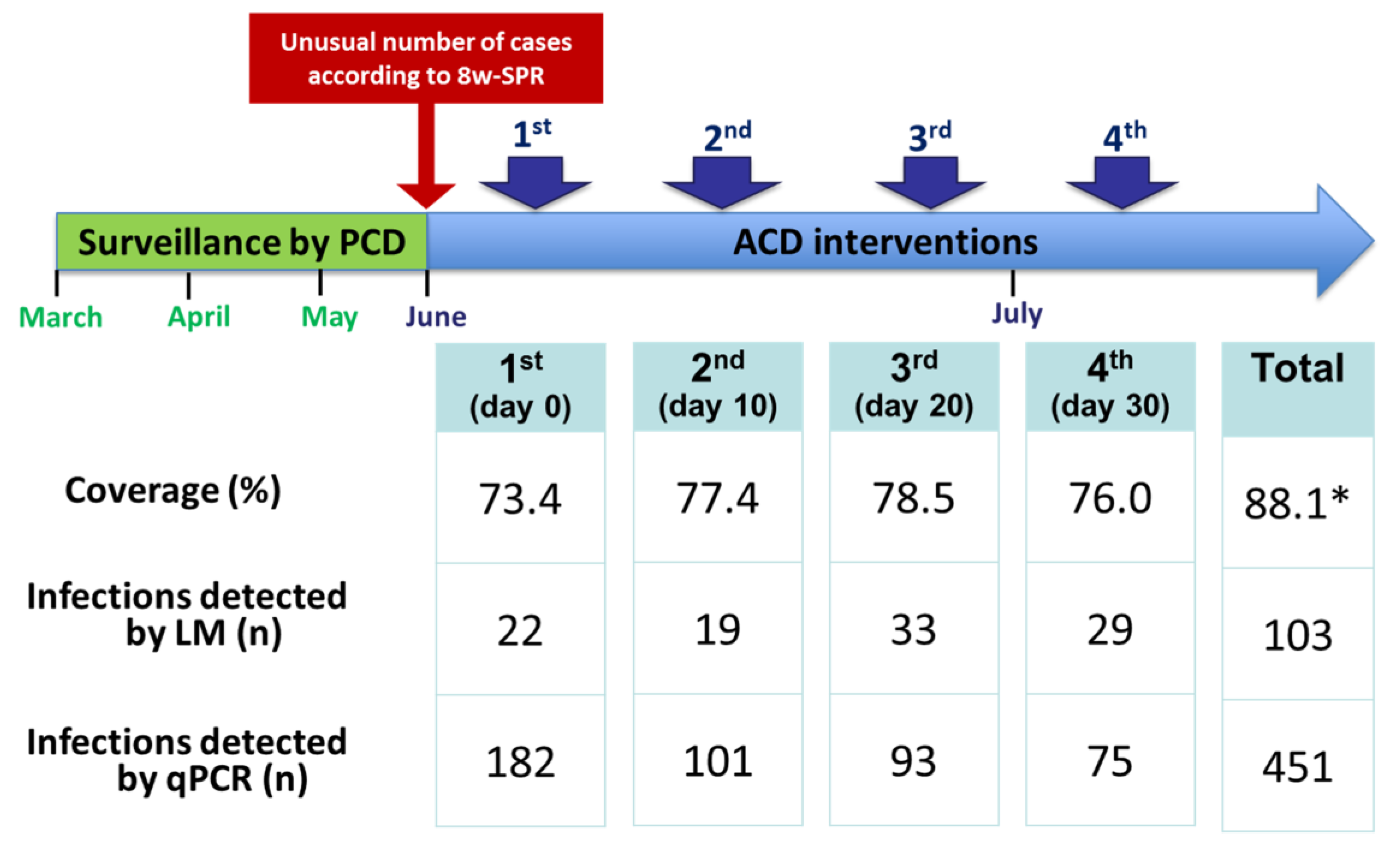
2.3. ACD Interventions and Data Collection
2.4. Laboratory Procedures
2.4.1. Light Microscopy (LM)
2.4.2. Quantitative Real-Time Polymerase Chain Reaction (qPCR)
2.4.3. Multiplex Real Time PCR (mPCR)
2.5. Data analysis
2.6. Ethical Issues
3. Results
3.1. Participants
3.2. Malaria Infections in the Study Period
3.3. Risk Factors by Malaria Species
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. World Malaria Report 2016; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization. Eliminating Malaria; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Ministerio de Salud del Perú-Dirección General de Epidemiología. Análisis de la Situación de Salud del Perú; ASIS 2010; MINSA: Lima, Peru, 2010. [Google Scholar]
- Ministerio de Salud Peru-Centro Nacional de Epidemiología, Prevención y Control de Enfermedades. Sala Situacional Malaria; MINSA: Lima, Peru, 2017. [Google Scholar]
- Proyecto Control de la Malaria en Zonas Fronterizas de la Región Andina: Un enfoque comunitario—PAMAFRO. Compartiendo Lecciones Aprendidas; ORAS-CONHU: Lima, Peru, 2009. [Google Scholar]
- Aramburú Guarda, J.; Ramal Asayag, C.; Witzig, R. Malaria reemergence in the Peruvian Amazon region. Emerg. Infect. Dis. 1999, 5, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Pinkerton, K.; Rom, W. Global Climate Change and Public Health; Springer: New York, NY, USA, 2014. [Google Scholar]
- World Health Organization. Test.Treat.Track. Scaling Up Diagnostic Testing, Treatment and Surveillance for Malaria; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- World Health Organization. Disease Surveillance for Malaria Elimination: Operational Manual; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- World Health Organization. Malaria Elimination. A Field Manual for Low and Moderate Endemic Countries; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Ministerio de Salud del Perú. Norma Técnica Para la Atención de la Malaria y Malaria Severa en el Perú; NTS Nro. 054-MINSA/DGSP-V.01, modificada en Febrero 2015; MINSA: Lima, Peru, 2015.
- World Health Organization. Malaria Light Microscopy: Creating a Culture of Quality; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Branch, O.; Casapia, W.M.; Gamboa, D.V.; Hernandez, J.N.; Alava, F.F.; Roncal, N.; Alvarez, E.; Perez, E.J.; Gotuzzo, E. Clustered local transmission and asymptomatic Plasmodium falciparum and Plasmodium vivax malaria infections in a recently emerged, hypoendemic Peruvian Amazon community. Malar. J. 2005, 23, 27. [Google Scholar] [CrossRef] [PubMed]
- Roshanravan, B.; Kari, E.; Gilman, R.H.; Cabrera, L.; Lee, E.; Metcalfe, J.; Calderon, M.; Lescano, A.G.; Montenegro, S.H.; Calampa, C.; et al. Endemic malaria in the Peruvian Amazon region of Iquitos. Am. J. Trop. Med. Hyg. 2003, 69, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Aguirre, A.; Speybroeck, N.; Llanos-Cuentas, A.; Rosanas-Urgell, A.; Carrasco-Escobar, G.; Rodriguez, H.; Gamboa, D.; Contreras-Mancilla, J.; Alava, F.; Soares, I.S.; et al. Hotspots of Malaria Transmission in the Peruvian Amazon: Rapid Assessment through a Parasitological and Serological Survey. PLoS ONE. 2015, 10, e0137458. [Google Scholar] [CrossRef] [PubMed]
- Moonen, B.; Cohen, J.M.; Snow, R.W.; Slutsker, L.; Drakeley, C.; Smith, D.L.; Abeyasinghe, R.R.; Rodriguez, M.H.; Maharaj, R.; Tanner, M.; et al. Operational strategies to achieve and maintain malaria elimination. The Lancet 2010, 376, 1592–1603. [Google Scholar] [CrossRef]
- Sturrock, H.J.W.; Hsiang, M.S.; Cohen, J.M.; Smith, D.L.; Greenhouse, B.; Bousema, T.; Gosling, R.D. Targeting asymptomatic malaria infections: Active surveillance in control and elimination. PLoS Med. 2013, 10. [Google Scholar] [CrossRef] [PubMed]
- Macauley, C. Aggressive active case detection: A malaria control strategy based on the Brazilian model. Soc. Sci. Med. 2005, 60, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Stresman, G.H.; Kamanga, A.; Moono, P.; Hamapumbu, H.; Mharakurwa, S.; Kobayashi, T.; Moss, W.J.; Shiff, C. A method of active case detection to target reservoirs of asymptomatic malaria and gametocyte carriers in a rural area in Southern Province, Zambia. Malar. J. 2010, 9, 265. [Google Scholar] [PubMed]
- Smith Gueye, C.; Sanders, K.C.; Galappaththy, G.N.L.; Rundi, C.; Tobgay, T.; Sovannaroth, S.; Gao, Q.; Surya, A.; Thakur, G.D.; Baquilod, M.; et al. Active case detection for malaria elimination: A survey among Asia Pacific countries. Malar. J. 2013, 9, 358. [Google Scholar] [CrossRef] [PubMed]
- Dormond, L.; Jaton-Ogay, K.; de Vallière, S.; Genton, B.; Bille, J.; Greub, G. Multiplex real-time PCR for the diagnosis of malaria: Correlation with microscopy. Clin. Microbiol. Infect. 2011, 17, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Salud Perú. Dirección Regional de Salud Loreto. Boletín Epidemiológico SE 52-2015 [Internet]. 2016. Available online: http://www.diresaloreto.gob.pe/epidemiologia/boletines-epidemiologicos-2015/ (accessed on 19 June 2017).
- PERÚ Instituto Nacional de Estadística e Informática [Internet]. Available online: http://www.inei.gob.pe/ (accessed on 7 March 2017).
- Boletín Epidemiológico Perú [Internet]. Available online: http://www.dge.gob.pe/portal/docs/vigilancia/boletines/2013/52.pdf (accessed on 19 September 2018).
- Parker, B.S.; Paredes Olortegui, M.; Peñataro Yori, P.; Escobedo, K.; Florin, D.; Rengifo Pinedo, S.; Greffa, R.C.; Vega, L.C.; Ferrucci, H.R.; Pan, W.K.; et al. Hyperendemic malaria transmission in areas of occupation-related travel in the Peruvian Amazon. Malar. J. 2013, 12, 178. [Google Scholar] [CrossRef] [PubMed]
- Llanos-Cuentas, A.; Lacerda, M.V.; Rueangweerayut, R.; Krudsood, S.; Gupta, S.K.; Kochar, S.K.; Arthur, P.; Chuenchom, N.; Möhrle, J.J.; Duparc, S.; et al. Tafenoquine plus chloroquine for the treatment and relapse prevention of Plasmodium vivax malaria (DETECTIVE): A multicentre, double-blind, randomised, phase 2b dose-selection study. Lancet Lond. Engl. 2014, 383, 1049–1058. [Google Scholar] [CrossRef]
- Marquiño, W.; Huilca, M.; Calampa, C.; Falconí, E.; Cabezas, C.; Naupay, R.; RUEBUSH, T.K., II. Efficacy of mefloquine and a mefloquine-artesunate combination therapy for the treatment of uncomplicated Plasmodium falciparum malaria in the Amazon Basin of Peru. Am. J. Trop. Med. Hyg. 2003, 68, 608–612. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Hu, W.; Liu, H.; Xiao, Y.; Guo, Y.; Chen, S.; Zhao, L.; Tong, S. Can slide positivity rates predict malaria transmission? Malar. J. 2012, 11, 117. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Salud del Perú. Manual de Procedimientos de Laboratorio Para el Diagnóstico de Malaria. Serie de Normas Técnicas N° 39. [Internet]. 2003. Available online: http://www.ins.gob.pe/repositorioaps/0/4/jer/-1/Antimalaricos/manualMALARIA.pdf (accessed on 6 June 2017).
- Mangold, K.A.; Manson, R.U.; Koay, E.S.C.; Stephens, L.; Regner, M.; Thomson, R.B.; Peterson, L.R.; Kaul, K.L. Real-time PCR for detection and identification of Plasmodium spp. J. Clin. Microbiol. 2005, 43, 2435–2440. [Google Scholar] [CrossRef] [PubMed]
- Cnops, L.; Jacobs, J.; Van Esbroeck, M. Validation of a four-primer real-time PCR as a diagnostic tool for single and mixed Plasmodium infections. Clin. Microbiol. Infect. 2011, 17, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO malaria Terminology; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Antinori, S.; Galimberti, L.; Milazzo, L.; Corbellino, M. Biology of Human Malaria Plasmodia Including Plasmodium Knowlesi. Mediterr. J. Hematol. Infect. Dis. 2012, 4. [Google Scholar] [CrossRef] [PubMed]
- Parekh, F.K.; Hernandez, J.N.; Krogstad, D.J.; Casapia, W.M.; Branch, O.H. Prevalence and risk of Plasmodium falciparum and P. vivax malaria among pregnant women living in the hypoendemic communities of the Peruvian Amazon. Am. J. Trop. Med. Hyg. 2007, 77, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Aguirre, A.; Guzman-Guzman, M.; Gamboa, D.; Chuquiyauri, R.; Ramirez, R.; Manrique, P.; Carrasco-Escobar, G.; Puemape, C.; Llanos-Cuentas, A.; Vinetz, J.M. Micro-heterogeneity of malaria transmission in the Peruvian Amazon: A baseline assessment underlying a population-based cohort study. Malar. J. 2017, 16, 312. [Google Scholar] [CrossRef] [PubMed]
- Rovira-Vallbona, E.; Contreras-Mancilla, J.J.; Ramirez, R.; Guzmán-Guzmán, M.; Carrasco-Escobar, G.; Llanos-Cuentas, A.; Vinetz, J.M.; Gamboa, D.; Rosanas-Urgell, A. Predominance of asymptomatic and sub-microscopic infections characterizes the Plasmodium gametocyte reservoir in the Peruvian Amazon. PLoS Negl. Trop. Dis. 2017, 11, e0005674. [Google Scholar] [CrossRef] [PubMed]
- Bousema, T.; Drakeley, C. Epidemiology and Infectivity of Plasmodium falciparum and Plasmodium vivax Gametocytes in Relation to Malaria Control and Elimination. Clin. Microbiol. Rev. 2011, 24, 377–410. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Meeting Report of the WHO Evidence Review Group on Low-Density Malaria Infections; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Nguyen, T.-N.; von Seidlein, L.; Nguyen, T.-V.; Truong, P.-N.; Hung, S.D.; Pham, H.-T.; Nguyen, T.U.; Le, T.D.; Mukaka, M.; Day, N.P.; et al. The persistence and oscillations of submicroscopic Plasmodium falciparum and Plasmodium vivax infections over time in Vietnam: An open cohort study. Lancet Infect. Dis. 2018, 18, 565–572. [Google Scholar] [CrossRef]
- Da Silva-Nunes, M.; Ferreira, M.U. Clinical spectrum of uncomplicated malaria in semi-immune Amazonians: Beyond the “symptomatic“ vs “asymptomatic“ dichotomy. Mem. Inst. Oswaldo Cruz. 2007, 102, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Thanh, P.V.; Van Hong, N.; Van Van, N.; Van Malderen, C.; Obsomer, V.; Rosanas-Urgell, A.; Grietens, K.P.; Xa, N.X.; Bancone, G.; Chowwiwat, N.; et al. Epidemiology of forest malaria in Central Vietnam: The hidden parasite reservoir. Malar. J. 2015, 14. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.C.G.; Kuehn, A.; Castro, A.J.M.; Vitor-Silva, S.; Figueiredo, E.F.G.; Brasil, L.W.; Brito, M.A.; Sampaio, V.S.; Bassat, Q.; Felger, I.; et al. High proportions of asymptomatic and submicroscopic Plasmodium vivax infections in a peri-urban area of low transmission in the Brazilian Amazon. Parasit. Vectors. 2018, 11, 194. [Google Scholar] [CrossRef] [PubMed]
- Mueller, I.; Galinski, M.R.; Tsuboi, T.; Arevalo-Herrera, M.; Collins, W.E.; King, C.L. Natural acquisition of immunity to Plasmodium vivax: Epidemiological observations and potential targets. Adv. Parasitol. 2013, 81, 77–131. [Google Scholar] [CrossRef] [PubMed]
- White, M.T.; Karl, S.; Battle, K.E.; Hay, S.I.; Mueller, I.; Ghani, A.C. Modelling the contribution of the hypnozoite reservoir to Plasmodium vivax transmission. eLife 2014, 3, e04692. [Google Scholar] [CrossRef] [PubMed]
- Kerlin, D.H.; Gatton, M.L. A simulation model of the within-host dynamics of Plasmodium vivax infection. Malar. J. 2015, 14, 51. [Google Scholar] [CrossRef] [PubMed]
- White, N.J.; Imwong, M. Relapse. In Advances in Parasitology; Hay, S.I., Price, R., Baird, J.K., Eds.; Academic Press: Cambridge, MA, USA, 2012; pp. 113–150. [Google Scholar]
- Delgado-Ratto, C.; Soto-Calle, V.E.; Van den Eede, P.; Gamboa, D.; Rosas, A.; Abatih, E.N.; Ferrucci, H.R.; Llanos-Cuentas, A.; Van Geertruyden, J.P.; Erhart, A.; et al. Population structure and spatio-temporal transmission dynamics of Plasmodium vivax after radical cure treatment in a rural village of the Peruvian Amazon. Malar. J. 2014, 13, 8. [Google Scholar] [CrossRef] [PubMed]
- Kerlin, D.H.; Gatton, M.L. Preferential Invasion by Plasmodium Merozoites and the Self-Regulation of Parasite Burden. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Escobar, G.; Gamboa, D.; Castro, M.C.; Bangdiwala, S.I.; Rodriguez, H.; Contreras-Mancilla, J.; Alava, F.; Speybroeck, N.; Lescano, A.G.; Vinetz, J.M.; et al. Micro-epidemiology and spatial heterogeneity of P. vivax parasitaemia in riverine communities of the Peruvian Amazon: A multilevel analysis. Sci. Rep. 2017, 7, 8082. [Google Scholar] [CrossRef] [PubMed]
- Adekunle, A.I.; Pinkevych, M.; McGready, R.; Luxemburger, C.; White, L.J.; Nosten, F.; Cromer, D.; Davenport, M.P. Modeling the dynamics of Plasmodium vivax infection and hypnozoite reactivation in vivo. PLoS Negl. Trop. Dis. 2015, 9, e0003595. [Google Scholar] [CrossRef] [PubMed]
- Robinson, L.J.; Wampfler, R.; Betuela, I.; Karl, S.; White, M.T.; Suen, C.S.L.W.; Hofmann, N.E.; Kinboro, B.; Waltmann, A.; Brewster, J.; et al. Strategies for understanding and reducing the Plasmodium vivax and Plasmodium ovale hypnozoite reservoir in Papua New Guinean children: A randomised placebo-controlled trial and mathematical model. PLoS Med. 2015, 12, e1001891. [Google Scholar] [CrossRef] [PubMed]
- Chuquiyauri, R.; Peñataro, P.; Brouwer, K.C.; Fasabi, M.; Calderon, M.; Torres, S.; Gilman, R.H.; Kosek, M.; Vinetz, J.M. Microgeographical differences of Plasmodium vivax relapse and re-infection in the Peruvian Amazon. Am. J. Trop. Med. Hyg. 2013, 89, 326–338. [Google Scholar] [CrossRef] [PubMed]
- Miguel, R.B.; Coura, J.R.; Samudio, F.; Suárez-Mutis, M.C. Evaluation of three different DNA extraction methods from blood samples collected in dried filter paper in Plasmodium subpatent infections from the Amazon region in Brazil. Rev. Inst. Med. Trop. Sao Paulo 2013, 55. [Google Scholar] [CrossRef] [PubMed]
- Mixson-Hayden, T.; Lucchi, N.W.; Udhayakumar, V. Evaluation of three PCR-based diagnostic assays for detecting mixed Plasmodium infection. BMC Res. Notes 2010, 3, 88. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Escobar, G.; Miranda-Alban, J.; Fernandez-Miñope, C.; Brouwer, K.C.; Torres, K.; Calderon, M.; Gamboa, D.; Llanos-Cuentas, A.; Vinetz, J.M. High prevalence of very-low Plasmodium falciparum and Plasmodium vivax parasitaemia carriers in the Peruvian Amazon: Insights into local and occupational mobility-related transmission. Malar. J. 2017, 16, 415. [Google Scholar] [CrossRef] [PubMed]
| Community | ||||||
|---|---|---|---|---|---|---|
| GAM | LIB | PRI | URC | Total | ||
| Households | ||||||
| In census | N | 21 | 58 | 20 | 59 | 158 |
| With at least one ACD visit | n (%) | 19 (90.5) | 56 (96.6) | 20 (100) | 59 (100) | 154 (97.5) |
| Individuals | ||||||
| In census | N | 101 | 347 | 121 | 364 | 933 |
| With at least one ACD visit | n (%) | 92 (91.1) | 299 (86.2) | 100 (82.6) | 331 (90.9) | 822 (88.1) |
| With one visit | n (%) | 11 (10.9) | 25 (7.2) | 4 (3.3) | 4 (1.1) | 44 (4.7) |
| With two visits | n (%) | 25 (24.8) | 24 (6.9) | 10 (8.3) | 15 (4.1) | 74 (7.9) |
| With three visits | n (%) | 12 (11.9) | 63 (18.2) | 32 (26.4) | 53 (14.6) | 160 (17.1) |
| With four visits | n (%) | 44 (43.6) | 187 (53.9) | 54 (44.6) | 259 (71.2) | 544 (58.3) |
| In the 1st visit | n (%) | 64 (63.4) | 242 (69.7) | 67 (55.4) | 312 (85.7) | 685 (73.4) |
| In the 2nd visit | n (%) | 63 (62.4) | 255 (73.5) | 95 (78.5) | 309 (84.9) | 722 (77.4) |
| In the 3rd visit | n (%) | 73 (72.3) | 258 (74.4) | 87 (71.9) | 314 (86.3) | 732 (78.5) |
| In the 4th visit | n (%) | 73 (72.3) | 255 (73.5) | 87 (71.9) | 294 (80.8) | 709 (76.0) |
| Characteristics | Gamitanacocha (GAM) | Libertad (LIB) | Primero de Enero (PRI) | Urco Miraño (URC) | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| N = 92 | N = 299 | N = 100 | N = 331 | N = 822 | |||||||
| n | % | n | % | n | % | n | % | n | % | ||
| Gender | |||||||||||
| Male | 43 | 46.7 | 159 | 53.2 | 55 | 55.0 | 174 | 52.6 | 431 | 52.4 | |
| Age (years) | |||||||||||
| <5 | 18 | 19.6 | 56 | 18.7 | 16 | 16.0 | 56 | 16.9 | 146 | 17.8 | |
| 5–14 | 23 | 25.0 | 72 | 24.1 | 34 | 34.0 | 113 | 34.1 | 242 | 29.4 | |
| 15–39 | 32 | 34.8 | 101 | 33.8 | 32 | 32.0 | 82 | 24.8 | 247 | 30.1 | |
| ≥40 | 19 | 20.7 | 70 | 23.4 | 18 | 18.0 | 80 | 24.2 | 187 | 22.8 | |
| Education (≥18 years) * n = 391 | |||||||||||
| None | 3 | 6.7 | 24 | 15.8 | 5 | 10.6 | 20 | 14.7 | 52 | 13.7 | |
| Primary school | 33 | 73.3 | 102 | 67.1 | 33 | 70.2 | 70 | 51.5 | 238 | 62.6 | |
| Secondary school or higher | 9 | 20.0 | 26 | 17.1 | 9 | 19.2 | 46 | 33.8 | 90 | 23.7 | |
| Missing | 8 | 3 | 11 | ||||||||
| Main occupation (≥15 years) * | |||||||||||
| None or others | 10 | 19.6 | 49 | 28.7 | 11 | 22.0 | 60 | 37.0 | 130 | 30.0 | |
| Farmer | 41 | 80.4 | 122 | 71.4 | 39 | 78.0 | 102 | 63.0 | 304 | 70.1 | |
| Malaria episodes (previous 12 months) | |||||||||||
| 0 | 65 | 71.4 | 229 | 76.9 | 80 | 80.0 | 259 | 78.3 | 633 | 77.2 | |
| ≥1 | 26 | 28.6 | 69 | 23.2 | 20 | 20.0 | 72 | 21.8 | 187 | 22.8 | |
| Missing | 1 | 1 | 2 | ||||||||
| Travel (previous month) * | |||||||||||
| No | 80 | 87.0 | 269 | 90.0 | 81 | 81.0 | 306 | 92.5 | 736 | 89.5 | |
| Yes | 12 | 13.0 | 30 | 10.0 | 19 | 19.0 | 25 | 7.6 | 86 | 10.5 | |
| Housing structure (number of walls) * | |||||||||||
| 0 | 10 | 11.0 | 42 | 14.2 | 16 | 16.0 | 44 | 13.3 | 112 | 13.7 | |
| 1–3 | 53 | 58.2 | 207 | 70.2 | 79 | 79.0 | 188 | 56.8 | 527 | 64.5 | |
| 4 | 28 | 30.8 | 46 | 15.6 | 5 | 5.0 | 99 | 29.9 | 178 | 21.8 | |
| Missing | 1 | 4 | 5 | ||||||||
| Indoor residual spraying (previous 12 months) * | |||||||||||
| No | 14 | 15.4 | 28 | 9.49 | 10 | 10.0 | 1 | 0.31 | 53 | 6.5 | |
| Yes | 77 | 84.6 | 267 | 90.5 | 90 | 90.0 | 325 | 99.7 | 759 | 93.5 | |
| Missing | 1 | 4 | 5 | 10 | |||||||
| Electricity available * | |||||||||||
| No | 67 | 73.6 | 215 | 72.9 | 69 | 69.0 | 62 | 18.7 | 413 | 50.6 | |
| Yes | 24 | 26.4 | 80 | 27.1 | 31 | 31.0 | 269 | 81.3 | 404 | 49.5 | |
| Missing | 1 | 4 | 5 | ||||||||
| Time of Detection of Malaria Infections by qPCR | Time of Detection of Malaria Infections by the Local Strategy Using LM | ||||||
|---|---|---|---|---|---|---|---|
| Same Visit | Next Visit (10-days Later) | After 2 Visits (20-days Later) | After 3 Visits (30-days Later) | Non Detected | Total | ||
| n (%) | n (%) | n (%) | n (%) | n (%) | N | ||
| Overall | |||||||
| 1st visit | 22 (12.1) | 9 (4.9) | 5 (2.7) | 8 (4.4) | 138 (75.8) | 182 | |
| 2nd visit | 10 (9.9) | 6 (5.9) | 4 (4.0) | ND | 81 (80.2) | 101 | |
| 3rd visit | 22 (23.7) | 4 (4.3) | ND | ND | 67 (72.0) | 93 | |
| 4th visit | 13 (17.3) | ND | ND | ND | 62 (82.7) | 75 | |
| Total | 67 (14.9) | 19 (4.2) | 9 (2.0) | 8 (1.8) | 348 (77.2) | 451 | |
| P. vivax | |||||||
| 1st visit | 20 (14.5) | 9 (6.5) | 4 (2.9) | 7 (5.1) | 98 (71.0) | 138 | |
| 2nd visit | 10 (12.8) | 6 (7.7) | 4 (5.1) | ND | 58 (74.4) | 78 | |
| 3rd visit | 22 (26.5) | 4 (4.8) | ND | ND | 57 (68.7) | 83 | |
| 4th visit | 13 (21.3) | ND | ND | ND | 48 (78.7) | 61 | |
| Total | 65 (18.1) | 19 (5.3) | 8 (2.2) | 7 (1.9) | 261 (72.5) | 360 | |
| P. falciparum | |||||||
| 1st visit | 2 (6.7) | 0 (0) | 1 (3.3) | 0 (0) | 27 (90.0) | 30 | |
| 2nd visit | 0 (0) | 0 (0) | 0 (0) | ND | 17 (100.0) | 17 | |
| 3rd visit | 0 (0) | 0 (0) | ND | ND | 8 (100.0) | 8 | |
| 4th visit | 0 (0) | ND | ND | ND | 14 (100.0) | 14 | |
| Total | 2 (2.9) | 0 (0) | 1 (1.4) | 0 (0) | 66 (95.7) | 69 | |
| Co-infection * | |||||||
| 1st visit | 0 (0) | 0 (0) | 0 (0) | 1 (7.1) | 13 (92.9) | 14 | |
| 2nd visit | 0 (0) | 0 (0) | 0 (0) | ND | 6 (100.0) | 6 | |
| 3rd visit | 0 (0) | 0 (0) | ND | ND | 2 (100.0) | 2 | |
| 4th visit | 0 (0) | ND | ND | ND | 0 (0) | 0 | |
| Total | 0 (0) | 0 (0) | 0 (0) | 1 (4.5) | 21 (95.5) | 22 | |
| Parasite Density Level (Plus System Scale) | Time of Detection by the Local Strategy Using Light Microscopy (LM) in Comparison with the Time of Detection by Quantitative Real-Time Polymerase Chain Reaction (qPCR) | ||
|---|---|---|---|
| Same Visit | Following Visits (10–30 days Later) | Total | |
| n (%) | n (%) | N (%) | |
| P. vivax | |||
| <1/2+ | 24 (36.9) | 16 (50.0) | 40 (41.2) |
| 1/2+ | 13 (20.0) | 5 (15.6) | 18 (18.6) |
| ≥1+ | 28 (43.1) | 11 (34.4) | 39 (40.2) |
| Total | 65 (100) | 32 (100) | 97 (100) |
| P. falciparum | |||
| <1/2+ | 0 (0) | 0 (0) | 0 (0) |
| 1/2+ | 0 (0) | 0 (0) | 0 (0) |
| ≥1+ | 2 (100) | 1 (100) | 3 (100) |
| Total | 2 (100) | 1 (100) | 3 (100) |
| Co-infection * | |||
| <1/2+ | 0 (0) | 1 (100) | 1 (100) |
| 1/2+ | 0 (0) | 0 (0) | 0 (0) |
| ≥1+ | 0 (0) | 0 (0) | 0 (0) |
| Total | 0 (0) | 1 (100) | 1 (100) |
| Time of Detection of Malaria Infections by qPCR | All qPCR-Positive Malaria Infections | ||||||
|---|---|---|---|---|---|---|---|
| Symptomatic | Asymptomatic | Total | |||||
| In the Same Visit | In Following Visit (10-days Later) | After 2 Visits (20-days Later) | After 3 Visits (30-days Later) | ||||
| n (%) | n (%) | n (%) | n (%) | n (%) | N | ||
| Overall | |||||||
| 1st visit | 76 (42.5) | 18 (10.1) | 10 (5.6) | 2 (1.1) | 73 (40.8) | 179 | |
| 2nd visit | 20 (19.8) | 12 (11.9) | 12 (11.9) | ND | 57 (56.4) | 101 | |
| 3rd visit | 22 (23.7) | 11 (11.8) | ND | ND | 60 (64.5) | 93 | |
| 4th visit | 10 (13.3) | ND | ND | ND | 65 (86.7) | 75 | |
| Total | 128 (28.6) | 41 (9.2) | 22 (4.9) | 2 (0.4) | 255 (56.9) | 448 | |
| P. vivax | |||||||
| 1st visit | 60 (44.1) | 13 (9.6) | 8 (5.9) | 1 (0.7) | 54 (39.7) | 136 | |
| 2nd visit | 18 (23.1) | 10 (12.8) | 8 (10.3) | ND | 42 (53.8) | 78 | |
| 3rd visit | 22 (26.5) | 10 (12.0) | ND | ND | 51 (61.4) | 83 | |
| 4th visit | 8 (13.1) | ND | ND | ND | 53 (86.9) | 61 | |
| Total | 108 (30.2) | 33 (9.2) | 16 (4.5) | 1 (0.3) | 200 (55.9) | 358 | |
| P. falciparum | |||||||
| 1st visit | 12 (41.4) | 1 (3.4) | 0 (0) | 1 (3.4) | 15 (51.7) | 29 | |
| 2nd visit | 2 (11.8) | 0 (0) | 4 (23.5) | ND | 11 (64.7) | 17 | |
| 3rd visit | 0 (0) | 1 (12.5) | ND | ND | 7 (87.5) | 8 | |
| 4th visit | 2 (14.3) | ND | ND | ND | 12 (85.7) | 14 | |
| Total | 16 (23.5) | 2 (2.9) | 4 (5.9) | 1 (1.5) | 45 (66.2) | 68 | |
| Co-infection * | |||||||
| 1st visit | 4 (28.6) | 4 (28.6) | 2 (14.3) | 0 (0) | 4 (28.6) | 14 | |
| 2nd visit | 0 (0) | 2 (33.3) | 0 (0) | ND | 4 (66.7) | 6 | |
| 3rd visit | 0 (0) | 0 (0) | ND | ND | 2 (100) | 2 | |
| 4th visit | 0 (0) | ND | ND | ND | 0 (0) | 0 | |
| Total | 4 (18.2) | 6 (27.3) | 2 (9.1) | 0 (0) | 10 (45.5) | 22 | |
| Characteristics | % | P. vivax | P. falciparum | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n/N | Univariate | Multivariate | % | n/N | Univariate | Multivariate | |||||||
| OR | [95% CI] | AOR | [95% CI] | OR | [95% CI] | AOR | [95% CI] | ||||||
| Gender | |||||||||||||
| Female | 46.8 | 183/391 | Ref | 9.21 | 36/391 | Ref | |||||||
| Male | 45.0 | 194/431 | 0.9 | [0.7; 1.2] | 12.8 | 55/431 | 1.5 | [0.9; 2.4] | 1.5 | [0.9; 2.3] ° | |||
| Age (years) | |||||||||||||
| <5 | 37.0 | 54/146 | Ref | Ref | 7.53 | 11/146 | Ref | ||||||
| 5–14 | 50.0 | 121/242 | 1.7 | [1.1; 2.6] * | 1.7 | [1.1; 2.6] * | 11.6 | 28/242 | 1.8 | [0.8; 3.9] | 1.7 | [0.8; 3.7] | |
| 15–39 | 47.8 | 118/247 | 1.6 | [1.0; 2.4] * | 1.2 | [0.7; 2.0] | 10.9 | 27/247 | 1.6 | [0.7; 3.5] | 1.7 | [0.7; 3.9] | |
| ≥40 | 44.9 | 84/187 | 1.4 | [0.9; 2.1] | 0.9 | [0.5; 1.7] | 13.4 | 25/187 | 2.1 | [1.0; 4.7] ° | 2.2 | [0.9; 5.4] ° | |
| Main occupation | |||||||||||||
| None or others | 43.86 | 225/513 | Ref | Ref | 10.5 | 54/513 | Ref | ||||||
| Farmer | 49.2 | 152/309 | 1.3 | [1.0; 1.7] ° | 1.6 | [1.0; 2.4] * | 12.0 | 37/309 | 1.2 | [0.8; 1.9] | 0.8 | [0.4; 1.6] | |
| Malaria episodes (previous 12 months) | |||||||||||||
| 0 | 45.7 | 289/633 | Ref | 11.1 | 70/633 | Ref | |||||||
| ≥1 | 46.5 | 87/187 | 1.1 | [0.8; 1.5] | 11.23 | 21/187 | 1.0 | [0.6; 1.7] | |||||
| Travel (previos month) | |||||||||||||
| No | 46.3 | 341/736 | Ref | 11.1 | 82/736 | Ref | |||||||
| Yes | 41.9 | 36/86 | 0.9 | [0.6; 1.4] | 10.5 | 9/86 | 0.9 | [0.4; 2.1] | |||||
| Housing structure (number of walls) | |||||||||||||
| 0 | 47.3 | 53/112 | Ref | 10.7 | 12/112 | Ref | |||||||
| 1–3 | 46.3 | 244/527 | 1.0 | [0.6; 1.5] | 10.4 | 55/527 | 1.0 | [0.4; 2.3] | |||||
| 4 | 44.4 | 79/178 | 0.9 | [0.5; 1.4] | 12.9 | 23/178 | 1.4 | [0.5; 3.5] | |||||
| Indoor residual spraying (previous 12 months) | |||||||||||||
| No | 35.9 | 19/53 | Ref | 13.21 | 7/53 | Ref | |||||||
| Yes | 46.5 | 353/759 | 1.4 | [0.7; 2.5] | 10.9 | 83/759 | 0.8 | [0.3; 2.3] | |||||
| Electricity available | |||||||||||||
| No | 44.3 | 183/413 | Ref | 11.1 | 46/413 | Ref | |||||||
| Yes | 47.8 | 193/404 | 1.0 | [0.7; 1.4] | 10.9 | 44/404 | 0.9 | [0.5; 1.6] | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreno-Gutierrez, D.; Llanos-Cuentas, A.; Luis Barboza, J.; Contreras-Mancilla, J.; Gamboa, D.; Rodriguez, H.; Carrasco-Escobar, G.; Boreux, R.; Hayette, M.-P.; Beutels, P.; et al. Effectiveness of a Malaria Surveillance Strategy Based on Active Case Detection during High Transmission Season in the Peruvian Amazon. Int. J. Environ. Res. Public Health 2018, 15, 2670. https://doi.org/10.3390/ijerph15122670
Moreno-Gutierrez D, Llanos-Cuentas A, Luis Barboza J, Contreras-Mancilla J, Gamboa D, Rodriguez H, Carrasco-Escobar G, Boreux R, Hayette M-P, Beutels P, et al. Effectiveness of a Malaria Surveillance Strategy Based on Active Case Detection during High Transmission Season in the Peruvian Amazon. International Journal of Environmental Research and Public Health. 2018; 15(12):2670. https://doi.org/10.3390/ijerph15122670
Chicago/Turabian StyleMoreno-Gutierrez, Diamantina, Alejandro Llanos-Cuentas, José Luis Barboza, Juan Contreras-Mancilla, Dionicia Gamboa, Hugo Rodriguez, Gabriel Carrasco-Escobar, Raphaël Boreux, Marie-Pierre Hayette, Philippe Beutels, and et al. 2018. "Effectiveness of a Malaria Surveillance Strategy Based on Active Case Detection during High Transmission Season in the Peruvian Amazon" International Journal of Environmental Research and Public Health 15, no. 12: 2670. https://doi.org/10.3390/ijerph15122670
APA StyleMoreno-Gutierrez, D., Llanos-Cuentas, A., Luis Barboza, J., Contreras-Mancilla, J., Gamboa, D., Rodriguez, H., Carrasco-Escobar, G., Boreux, R., Hayette, M.-P., Beutels, P., Speybroeck, N., & Rosas-Aguirre, A. (2018). Effectiveness of a Malaria Surveillance Strategy Based on Active Case Detection during High Transmission Season in the Peruvian Amazon. International Journal of Environmental Research and Public Health, 15(12), 2670. https://doi.org/10.3390/ijerph15122670





