Lung Function of Children at Three Sites of Varying Ambient Air Pollution Levels in Uganda: A Cross Sectional Comparative Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Size
2.2. Recruitment
2.3. Data Collection/Procedures
2.4. Data Analysis
2.5. Ethical Considerations
3. Results
3.1. Study Participants Characteristics
3.2. Air Pollution
3.3. Lung Function
3.4. Factors Associated with Lung Function
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thurlbeck, W.M. Postnatal human lung growth. Thorax 1982, 37, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Thurlbeck, W.M. Postnatal Growth and Development of the Lung 1. Am. Rev. Respir. Dis. 1975, 111, 803–844. [Google Scholar] [PubMed]
- Kotecha, S. Lung growth for beginners. Paediatr. Respir. Rev. 2000, 1, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Gray, D.; Willemse, L.; Visagie, A.; Czövek, D.; Nduru, P.; Vanker, A.; Stein, D.J.; Koen, N.; Sly, P.D.; Hantos, Z. Determinants of early-life lung function in African infants. Thorax 2017, 72, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Martinez, F.D. The origins of asthma and chronic obstructive pulmonary disease in early life. Proc. Am. Thorac. Soc. 2009, 6, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Bui, D.S.; Burgess, J.A.; Lowe, A.J.; Perret, J.L.; Lodge, C.J.; Bui, M.; Morrison, S.; Thompson, B.R.; Thomas, P.S.; Giles, G.G.; et al. Childhood Lung Function Predicts Adult Chronic Obstructive Pulmonary Disease and Asthma-Chronic Obstructive Pulmonary Disease Overlap Syndrome. Am. J. Respir. Crit. Care Med. 2017, 196, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Stern, D.A.; Morgan, W.J.; Wright, A.L.; Guerra, S.; Martinez, F.D. Poor airway function in early infancy and lung function by age 22 years: A non-selective longitudinal cohort study. Lancet 2007, 370, 758–764. [Google Scholar] [CrossRef]
- Wong, K.; Rowe, B.; Douwes, J.; Senthilselvan, A. International prevalence of asthma and wheeze in adults: Results from the world health survey. In B47. Asthma Epidemiology: Clinical and Pharmacological Determinants of Asthma Outcomes; Am Thoracic Soc.: New York, NY, USA, 2010; p. A3117. [Google Scholar]
- Salvi, S. Health effects of ambient air pollution in children. Paediatr. Respir. Rev. 2007, 8, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Kajekar, R. Environmental factors and developmental outcomes in the lung. Pharmacol. Ther. 2007, 114, 129–145. [Google Scholar] [CrossRef] [PubMed]
- Bateson, T.F.; Schwartz, J. Children’s response to air pollutants. J. Toxicol. Environ. health A 2008, 71, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Asgari, M.M.; DuBois, A.; Asgari, M.; Gent, J.; Beckett, W.S. Association of ambient air quality with children’s lung function in urban and rural Iran. Arch. Environ. Health 1998, 53, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Priftis, K.N.; Anthracopoulos, M.B.; Paliatsos, A.G.; Tzavelas, G.; Nikolaou-Papanagiotou, A.; Douridas, P.; Nicolaidou, P.; Mantzouranis, E. Different effects of urban and rural environments in the respiratory status of Greek schoolchildren. Respir. Med. 2007, 101, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Sonnappa, S.; Lum, S.; Kirkby, J.; Bonner, R.; Wade, A.; Subramanya, V.; Lakshman, P.T.; Rajan, B.; Nooyi, S.C.; Stocks, J. Disparities in pulmonary function in healthy children across the Indian urban-rural continuum. Am. J. Respir. Crit. Care Med. 2015, 191, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Umoh, V.A.; Peters, E. The relationship between lung function and indoor air pollution among rural women in the Niger Delta region of Nigeria. Lung India Off. Organ Indian Chest Soc. 2014, 31, 110. [Google Scholar] [CrossRef] [PubMed]
- Schwander, S.; Okello, C.D.; Freers, J.; Chow, J.C.; Watson, J.G.; Corry, M.; Meng, Q. Ambient particulate matter air pollution in Mpererwe District, Kampala, Uganda: A pilot study. J. Environ. Public Health 2014, 763934, 17. [Google Scholar] [CrossRef] [PubMed]
- Wallner, P.; Kundi, M.; Moshammer, H.; Piegler, K.; Hohenblum, P.; Scharf, S.; Fröhlich, M.; Damberger, B.; Tappler, P.; Hutter, H.-P. Indoor air in schools and lung function of Austrian school children. J. Environ. Monit. 2012, 14, 1976–1982. [Google Scholar] [CrossRef] [PubMed]
- Jagger, P.; Shively, G. Land Use Change, Fuel Use and Respiratory Health in Uganda. Energy Policy 2014, 67, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Van Gemert, F.; Kirenga, B.; Chavannes, N.; Kamya, M.; Luzige, S.; Musinguzi, P.; Turyagaruka, J.; Jones, R.; Tsiligianni, I.; Williams, S.; et al. Prevalence of chronic obstructive pulmonary disease and associated risk factors in Uganda (FRESH AIR Uganda): A prospective cross-sectional observational study. Lancet Glob. Health 2015, 3, e44–e51. [Google Scholar] [CrossRef]
- Uganda Bureau of Statistics (UBOS) and ICF. Uganda Demographic and Health Survey 2016: Key Indicators Report; UBOS: Rockville, MD, USA; Kampala, Uganda, 2017. [Google Scholar]
- Banda, H.T.; Mortimer, K.; Bello, G.A.; Mbera, G.B.; Namakhoma, I.; Thomson, R.; Nyirenda, M.J.; Faragher, B.; Madan, J.; Malmborg, R.; et al. Informal Health Provider and Practical Approach to Lung Health interventions to improve the detection of chronic airways disease and tuberculosis at primary care level in Malawi: Study protocol for a randomised controlled trial. Trials 2015, 16, 576. [Google Scholar] [CrossRef] [PubMed]
- Heinzerling, A.P.; Guarnieri, M.J.; Mann, J.K.; Diaz, J.V.; Thompson, L.M.; Diaz, A.; Bruce, N.G.; Smith, K.R.; Balmes, J.R. Lung function in woodsmoke-exposed Guatemalan children following a chimney stove intervention. Thorax 2016. [Google Scholar] [CrossRef] [PubMed]
- Hankinson, J.L.; Odencrantz, J.R.; Fedan, K.B. Spirometric reference values from a sample of the general US population. Am. J. Respir. Crit. Care Med. 1999, 159, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Vyas, S.; Kumaranayake, L. Constructing socio-economic status indices: How to use principal components analysis. Health Policy Plan. 2006, 21, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Gauderman, W.J.; Gilliland, G.F.; Vora, H.; Avol, E.; Stram, D.; McConnell, R.; Thomas, D.; Lurmann, F.; Margolis, H.G.; Rappaport, E.B.; et al. Association between air pollution and lung function growth in southern California children: Results from a second cohort. Am. J. Respir. Crit. Care Med. 2002, 166, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Gauderman, W.J.; Urman, R.; Avol, E.; Berhane, K.; McConnell, R.; Rappaport, E.; Chang, R.; Lurmann, F.; Gilliland, F. Association of improved air quality with lung development in children. N. Engl. J. Med. 2015, 372, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Von Mutius, E.; Martinez, F.D.; Fritzsch, C.; Nicolai, T.; Roell, G.; Thiemann, H.-H. Prevalence of asthma and atopy in two areas of West and East Germany. Am. J. Respir. Crit. Care Med. 1994, 149, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Jedrychowski, W.; Flak, E.; Mróz, E. The adverse effect of low levels of ambient air pollutants on lung function growth in preadolescent children. Environ. Health Perspect. 1999, 107, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Churg, A.; Brauer, M.; del Carmen Avila-Casado, M.; Fortoul, T.I.; Wright, J.L. Chronic exposure to high levels of particulate air pollution and small airway remodeling. Environ. Health Perspect. 2003, 111, 714. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.; He, Q.; Kong, L.; Xu, F.; Wei, F.; Chapman, R.; Chen, W.; Edwards, R.; Bascom, R. Respiratory responses to diverse indoor combustion air pollution sources. Indoor Air 2007, 17, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Rao, D.R.; Gaffin, J.M.; Baxi, S.N.; Sheehan, W.J.; Hoffman, E.B.; Phipatanakul, W. The utility of forced expiratory flow between 25% and 75% of vital capacity in predicting childhood asthma morbidity and severity. J. Asthma 2012, 49, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, N.; Siddique, N.; Custovic, A. Allergic disease in urban and rural populations: Increasing prevalence with increasing urbanization. Allergy 2005, 60, 1357–1360. [Google Scholar] [CrossRef] [PubMed]
- Yemaneberhan, H.; Bekele, Z.; Venn, A.; Lewis, S.; Parry, E.; Britton, J. Prevalence of wheeze and asthma and relation to atopy in urban and rural Ethiopia. Lancet 1997, 350, 85–90. [Google Scholar] [CrossRef]
- Souza, M.B.; Saldiva, P.H.; Pope, C.A.; Capelozzi, V.L. Respiratory changes due to long-term exposure to urban levels of air pollution: A histopathologic study in humans. CHEST J. 1998, 113, 1312–1318. [Google Scholar] [CrossRef]
- Abbey, D.E.; Burchette, R.J.; Knutsen, S.F.; McDonnell, W.F.; Lebowitz, M.D.; Enright, P.L. Long-term particulate and other air pollutants and lung function in nonsmokers. Am. J. Respir. Crit. Care Med. 1998, 158, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Balcan, B.; Akan, S.; Ugurlu, A.O.; Handemir, B.O.; Ceyhan, B.B.; Ozkaya, S. Effects of biomass smoke on pulmonary functions: A case control study. Int. J. Chronic Obstr. Pulm. Dis. 2016, 11, 1615. [Google Scholar] [CrossRef] [PubMed]
- Jiwtode, M.T.; Raikar, P.R. Comparison of pulmonary function tests in urban and rural children of Nagpur, Maharashtra, India. Int. J. Res. Med. Sci. 2017, 5, 908–911. [Google Scholar] [CrossRef]
- Kuti, B.P.; Oladimeji, O.I.; Kuti, D.K.; Adeniyi, A.T.; Adeniji, E.O.; Osundare, Y.J. Rural-urban disparity in lung function parameters of Nigerian children: Effects of socio-economic, nutritional and housing factors. Pan Afr. Med. J. 2017, 28. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Zhou, Y.; Jiang, C.; Zhao, Z.; He, F.; Ran, P. Small airway disease: A different phenotype of early stage COPD associated with biomass smoke exposure. Respirology 2018, 23, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Glew, R.; Brock, H.; VanderVoort, J.; Agaba, P.; Harkins, M.; VanderJagta, D. Lung function and nutritional status of semi-nomadic Fulani children and adolescents in northern Nigeria. J. Trop. Pediatr. 2004, 50, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Harikumaran, R.; Nair, C.K.; Shashidhar, S. Spirometric impairments in undernourished children. Indian J. Physiol. Pharmacol. 1999, 43, 467–473. [Google Scholar]
- Obaseki, D.O.; Erhabor, G.E.; Awopeju, O.F.; Adewole, O.O.; Adeniyi, B.O.; Buist, E.A.S.; Burney, P.G. Reduced Forced Vital Capacity in an African Population. Prevalence and Risk Factors. Ann. Am. Thorac. Soc. 2017, 14, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Langkulsen, U.; Jinsart, W.; Karita, K.; Yano, E. Respiratory symptoms and lung function in Bangkok school children. Eur. J. Public Health 2006, 16, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Bremner, P.R.; de Klerk, N.H.; Ryan, G.F.; James, A.L.; Musk, M.; Murray, C.; Le Söuef, P.N.; Young, S.; Spargo, R.; William Musk, A. Respiratory symptoms and lung function in aborigines from tropical Western Australia. Am. J. Respir. Crit. Care Med. 1998, 158, 1724–1729. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Wang, S.; Liu, Y.; He, Q. Physical activity and lung function growth in a cohort of Chinese school children: A prospective study. PLoS ONE 2013, 8, e66098. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Feng, K.; Chen, L.; Zu, S.; Han, S.; Zhu, G. Relation between fat mass, fat free mass and ventilatory function in children and adolescents. Sheng li xue bao [Acta Physiol. Sin.] 2010, 62, 455–464. [Google Scholar] [PubMed]
- Joens-Matre, R.R.; Welk, G.J.; Calabro, M.A.; Russell, D.W.; Nicklay, E.; Hensley, L.D. Rural–urban differences in physical activity, physical fitness, and overweight prevalence of children. J. Rural Health 2008, 24, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Özdirenç, M.; Özcan, A.; Akin, F.; Gelecek, N. Physical fitness in rural children compared with urban children in Turkey. Pediatr. Int. 2005, 47, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Micklesfield, L.K.; Pedro, T.M.; Kahn, K.; Kinsman, J.; Pettifor, J.M.; Tollman, S.; Norris, S.A. Physical activity and sedentary behavior among adolescents in rural South Africa: Levels, patterns and correlates. BMC Public Health 2014, 14, 40. [Google Scholar] [CrossRef] [PubMed]
- Khalequzzaman, M.; Kamijima, M.; Sakai, K.; Ebara, T.; Hoque, B.A.; Nakajima, T. Indoor air pollution and health of children in biomass fuel-using households of Bangladesh: Comparison between urban and rural areas. Environ. Health Prev. Med. 2011, 16, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Fuel Choice, Acute Respiratory Infection and Child Growth in Uganda. Available online: http://docs.lib.purdue.edu/dissertations/AAI1597565/ (accessed on 5 October 2018).
- Sanbata, H.; Asfaw, A.; Kumie, A. Association of biomass fuel use with acute respiratory infections among under-five children in a slum urban of Addis Ababa, Ethiopia. BMC Public Health 2014, 14, 1122. [Google Scholar] [CrossRef] [PubMed]
- Hartley, D. Rural health disparities, population health, and rural culture. Am. J. Public Health 2004, 94, 1675–1678. [Google Scholar] [CrossRef] [PubMed]
- Mbonye, A.K. Risk factors for diarrhoea and upper respiratory tract infections among children in a rural area of Uganda. J. Health Popul. Nutr. 2004, 22, 52–58. [Google Scholar] [PubMed]
- Uganda Demographic and Health Survey 2011. Available online: http://dhsprogram.com/pubs/pdf/PR18/PR18.pdf (accessed on 13 October 2018).
- Child Anthropometrics and Malnutrition in Uganda. Available online: http://siteresources.worldbank.org/INTSURAGRI/Resources/7420178-1294259038276/UG_Anthro_Brief.pdf (accessed on 13 October 2018).
- Kikafunda, J.K.; Walker, A.F.; Collett, D.; Tumwine, J.K. Risk factors for early childhood malnutrition in Uganda. Pediatrics 1998, 102, e45. [Google Scholar] [CrossRef] [PubMed]
- Vella, V.; Tomkins, A.; Borghesi, A.; Migliori, G.B.; Adriko, B.; Crevatin, E. Determinants of child nutrition and mortality in north-west Uganda. Bull. World Health Organ. 1992, 70, 637. [Google Scholar] [PubMed]
| Characteristic | Kampala City (n = 185) | Jinja Municipality (n = 151) | Buwenge Sub-County (n = 201) | p-Value |
|---|---|---|---|---|
| Socio-demographics | ||||
| Age, Mean (SD) | 11.3 (1.3) | 11.0 (1.3) | 11.0 (1.3) | 0.2375 |
| Gender | ||||
| Boys, (%) | 44.3 | 33.1 | 54.2 | 0.001 |
| Occupation of father | 0.000 | |||
| Professional | 36.8 | 32.9 | 11.4 | |
| Unemployed | 2.7 | 4.1 | 8.3 | |
| Peasant farmer | 7.0 | 9.6 | 47.7 | |
| Market vendor | 4.9 | 8.2 | 7.3 | |
| Builder | 4.9 | 6.8 | 7.8 | |
| Clerical worker (sales clerk, secretaries, driver etc.) | 4.9 | 15.7 | 5.7 | |
| Other | 38.9 | 22.6 | 11.9 | |
| Anthropometry | ||||
| Height (cm), mean (SD) | 145.5 (10.0) | 144.8 (8.6) | 139.5 (11.6) | 0.0000 |
| Weight (kg), mean (SD) | 36.7 (8.7) | 35.7 (8.4) | 32.1 (9.8) | 0.0000 |
| BMI categories | 0.000 | |||
| Underweight (BMI < 5%), % | 10.9 | 17.3 | 31.5 | |
| Normal (BMI: 5–85%), % | 80.4 | 76.7 | 62.4 | |
| Overweight (BMI: 85–95%), % | 8.2 | 2.7 | 5.6 | |
| Obese (BMI > 95%), % | 0.5 | 3.3 | 0.5 | |
| Respiratory symptoms | ||||
| Currently coughs several times a day, % (95% CI) | 46.0 (38.7–53.2) | 51.7 (43.5–59.9) | 61.0 (54.2–67.9) | 0.012 |
| Wheeze% (95% CI) | 23.2 (17.1–29.4) | 30.7 (23.3–38.1) | 21.6 (15.7–27.5) | 0.131 |
| Shortness of breath (get out of breath more easily than others), % (95% CI) | 26.1 (19.7–32.5) | 24.4 (17.0–31.8) | 28.6 (22.0–35.2) | 0.703 |
| Has at least one respiratory symptom | 54.1 (46.8–61.3) | 62.9 (55.2–70.7) | 66.7 (60.1–73.2) | 0.035 |
| Characteristic | Kampala City (n = 185) | Jinja Municipality (n = 151) | Buwenge Sub-County (n = 201) | p-Value |
|---|---|---|---|---|
| Particulate air pollutant levels in surveyed schools | ||||
| PM2.5 particle levels (Minimum/Maximum) | 62.5/424 | 30.0/236.5 | 16/101.5 | 0.0000 |
| PM2.5 particle levels 24 h average, mean (SD), μg/m3 | 177.5 (43.9) | 96.3(9.5) | 31.4 (12.2) | 0.0000 |
| Exposure to tobacco smoke | ||||
| Parental current smoker (%) | 4.9 | 0.0 | 4.0 | 0.031 |
| Stay/live with smoker (%) | 30.9 | 14.6 | 30.9 | 0.001 |
| Exposure to biomass smoke | ||||
| Exposed to biomass used indoors (%) | 83.6 | 92.7 | 94.6 | 0.002 |
| Use wood for cooking/lighting (%) | 16.7 | 51.9 | 97.8 | 0.000 |
| Use charcoal for cooking/lighting (%) | 96.1 | 97.9 | 67.9 | 0.000 |
| Use kerosene for cooking/lighting (%) | 23.6 | 30.4 | 36.2 | 0.221 |
| Outdoor rubbish burning near home (%) | 48.6 | 90.8 | 77.7 | 0.000 |
| Exposed to bush burning (self/others) (%) | 13.8 | 48.9 | 55.6 | 0.000 |
| Lives within 500 m of industry (%) | 16.1 | 48.9 | 40.7 | 0.000 |
| Lives within 500 m of road used by cars (%) | 89.7 | 79.5 | 73.1 | 0.000 |
| Characteristic | By Site | |||
|---|---|---|---|---|
| Kampala City (n = 185) | Jinja Municipality (n = 151) | Buwenge Sub-County (n = 201) | p-Value | |
| Actual lung function parameters of children unadjusted for their age, sex, and height | ||||
| FVC (L), mean (SD) | 2.2 (0.5) | 2.1 (0.4) | 2.0 (0.5) | 0.0001 |
| FEV1, (L), mean (SD) | 2.0 (0.5) | 1.9 (0.5) | 1.8 (0.4) | 0.0002 |
| FEV1/FVC ratio, mean (SD) | 0.90 (0.09) | 0.91 (0.06 | 0.91 (0.05) | 0.000 |
| FEF25–75 (mL), mean (SD) | 2.6 (0.7) | 2.5 (0.7) | 2.5 (0.7) | 0.3704 |
| Percentage of predicted lung function | ||||
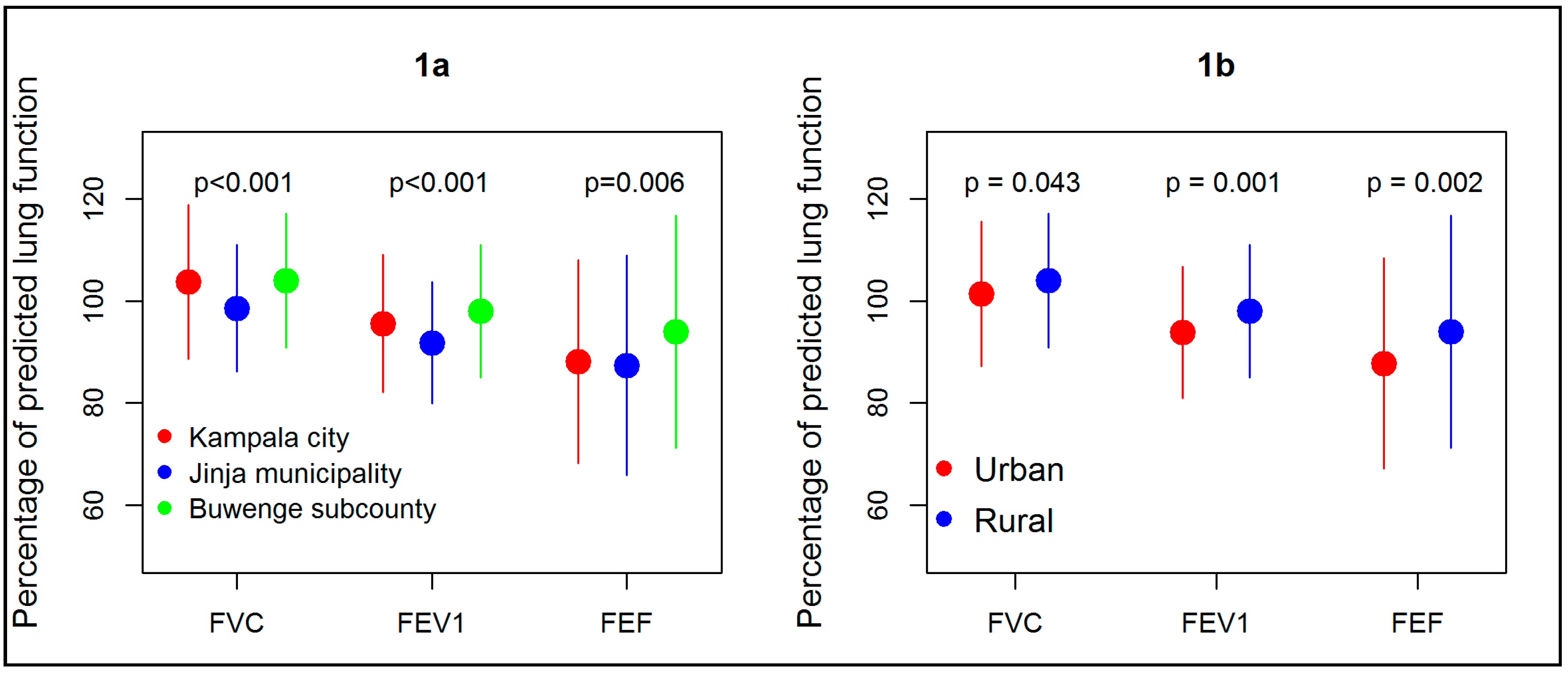
| FVC% predicted, mean (SD) | 103.7 (15.1) | 98.6 (12.4) | 104.0 (13.3) | 0.034 |
| FEV1% predicted, mean (SD) | 95.6 (13.5) | 91.8 (11.9) | 98.0 (13.0) | 0.000 |
| FEF25–75 predicted, mean (SD) | 88.1 (19.9) | 87.4 (21.5) | 94.0 (22.8) | 0.006 |
| FVC < 80%, n (%) | 5 (2.8) | 6 (4.1) | 6 (3.1) | 0.777 |
| FEV1 < 80%, n (%) | 21 (11.6) | 18 (12.4) | 11 (5.8) | 0.068 |
| FEF25–75 < 80%, n (%) | 65 (35.9) | 58 (40.0) | 56 (29.3) | 0.113 |
| FEV1/FVC ratio < 0.7, n (%) | 1 (0.6) | 0 (0.0) | 0 (0.0) | 0.629 |
| Characteristic | Urban (n = 336) | Rural (n = 201) | p-Value |
|---|---|---|---|
| Actual lung of children unadjusted for their age, sex, and height | |||
| FVC (L), mean (SD) | 2.2 (0.5) | 2.0 (0.5) | 0.0006 |
| FEV1, (L), mean (SD) | 2.0 (0.4) | 1.8 (0.4) | 0.001 |
| FEV1/FVC ratio, mean (SD) | 0.90 (0.05) | 0.91 (0.05) | 0.039 |
| FEF25–75 (mL), mean (SD) | 2.6 (0.7) | 2.5 (0.7) | 0.269 |
| FVC% predicted, mean (SD) | 101.4 (14.2) | 104.0 (13.3) | 0.043 |
| FEV1% predicted, mean (SD) | 93.9 (12.9) | 98.0 (13.0) | 0.001 |
| FEF25–75 predicted, mean (SD) | 87.8 (20.6) | 94.0 (22.8) | 0.002 |
| FVC < 80%, n (%) | 11 (3.4) | 6 (3.1) | 0.878 |
| FEV1 < 80%, n (%) | 39 (12.0) | 11 (5.8) | 0.021 |
| FEF25–75 < 80%, n (%) | 123 (37.7) | 56 (29.3) | 0.052 |
| FEV1/FVC ratio < 0.7, n (%) | 1 (0.3) | 0 (0.0) | 0.629 |
| Characteristic | Kampala City (n = 185) | Jinja Municipality (n = 151) | p-Value |
|---|---|---|---|
| Actual lung of children unadjusted for their age, sex, and height | |||
| FVC (L), mean (SD) | 2.2 (0.5) | 2.1 (0.4) | 0.004 |
| FEV1, (L), mean (SD) | 2.0 (0.5) | 1.9 (0.5) | 0.011 |
| FEV1/FVC ratio, mean (SD) | 0.90 (0.09) | 0.91 (0.06 | 0.059 |
| FEF25–75 (mL), mean (SD) | 2.6 (0.7) | 2.5 (0.7) | 0.389 |
| FVC% predicted, mean (SD) | 103.7 (15.1) | 98.6 (12.4) | 0.002 |
| FEV1% predicted, mean (SD) | 95.6 (13.5) | 91.8 (11.9) | 0.008 |
| FEF25–75 predicted, mean (SD) | 88.1 (19.9) | 87.4 (21.5) | 0.779 |
| FVC < 80%, n (%) | 5 (2.8) | 6 (4.1) | 0.494 |
| FEV1 < 80%, n (%) | 21 (11.6) | 18 (12.4) | 0.822 |
| FEF25–75 < 80%, n (%) | 65 (35.9) | 58 (40.0) | 0.449 |
| FEV1/FVC ratio < 0.7, n (%) | 1 (0.6) | 0.0 | 0.555 |
| Factor | Coefficient | 95% CI | p-Value |
|---|---|---|---|
| FVC% | |||
| Residence (ref: Urban) | |||
| Rural | 3.87 | 1.25–6.50 | 0.004 |
| Lives within 500 m of industry | −1.28 | −3.90–1.35 | 0.340 |
| BMI (ref. normal): | |||
| Under-weight | −6.62 | −9.74–−3.50 | 0.000 |
| Over-weight | 11.15 | 5.55–16.75 | 0.000 |
| Obese | 10.44 | −1.46–22.35 | 0.085 |
| Current coughs several times a day | −2.65 | −5.28–−0.02 | 0.048 |
| Wheeze | 0.09 | −2.98–3.16 | 0.953 |
| FEV1% | |||
| Residence (ref: Urban) | |||
| Rural | 5.46 | −0.10–11.02 | 0.054 |
| PM2.5 particle levels 24 h average, μg/m3 | −0.01 | −0.05–0.02 | 0.491 |
| Lives within 500 m of industry | −2.49 | −5.73–0.75 | 0.132 |
| BMI (ref. normal): | |||
| Under-weight | −6.54 | −10.26–−2.82 | 0.001 |
| Over-weight | 4.84 | −1.91–11.56 | 0.160 |
| Obese | 9.53 | −5.05–24.11 | 0.200 |
| SES (ref. high): | |||
| Low | −0.96 | −6.46–4.54 | 0.732 |
| Middle | −3.28 | −7.19–0.63 | 0.100 |
| FEF25–75% | |||
| Residence (ref: Urban) | |||
| Rural | 8.67 | 0.8–16.49 | 0.030 |
| PM2.5 particle levels 24 h average †, μg/m3 | 0.01 | −0.04–0.06 | 0.709 |
| Rubbish/bush burning | 4.21 | −0.42–8.85 | 0.075 |
| Exposure to biomass smoke | −7.48 | −14.09–−0.86 | 0.027 |
| Wheeze | 4.52 | −0.08–9.12 | 0.054 |
| BMI (ref. normal): | |||
| Under-weight | −3.71 | −8.76–1.34 | 0.149 |
| Over-weight | 0.28 | −8.71–9.28 | 0.951 |
| Obese | 6.55 | −12.96–26.07 | 0.510 |
| SES (ref. high): | |||
| Low | 1.03 | −6.47–8.54 | 0.787 |
| Middle | −3.33 | −8.47–1.81 | 0.204 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirenga, B.J.; Nantanda, R.; De Jong, C.; Mugenyi, L.; Meng, Q.; Aniku, G.; Williams, S.; Aanyu-Tukamuhebwa, H.; Kamya, M.; Schwander, S.; et al. Lung Function of Children at Three Sites of Varying Ambient Air Pollution Levels in Uganda: A Cross Sectional Comparative Study. Int. J. Environ. Res. Public Health 2018, 15, 2653. https://doi.org/10.3390/ijerph15122653
Kirenga BJ, Nantanda R, De Jong C, Mugenyi L, Meng Q, Aniku G, Williams S, Aanyu-Tukamuhebwa H, Kamya M, Schwander S, et al. Lung Function of Children at Three Sites of Varying Ambient Air Pollution Levels in Uganda: A Cross Sectional Comparative Study. International Journal of Environmental Research and Public Health. 2018; 15(12):2653. https://doi.org/10.3390/ijerph15122653
Chicago/Turabian StyleKirenga, Bruce J., Rebecca Nantanda, Corina De Jong, Levicatus Mugenyi, Qingyu Meng, Gilbert Aniku, Sian Williams, Hellen Aanyu-Tukamuhebwa, Moses Kamya, Stephan Schwander, and et al. 2018. "Lung Function of Children at Three Sites of Varying Ambient Air Pollution Levels in Uganda: A Cross Sectional Comparative Study" International Journal of Environmental Research and Public Health 15, no. 12: 2653. https://doi.org/10.3390/ijerph15122653
APA StyleKirenga, B. J., Nantanda, R., De Jong, C., Mugenyi, L., Meng, Q., Aniku, G., Williams, S., Aanyu-Tukamuhebwa, H., Kamya, M., Schwander, S., Van der Molen, T., & Mohsenin, V. (2018). Lung Function of Children at Three Sites of Varying Ambient Air Pollution Levels in Uganda: A Cross Sectional Comparative Study. International Journal of Environmental Research and Public Health, 15(12), 2653. https://doi.org/10.3390/ijerph15122653






