Indoor Air Quality and Potential Health Risk Impacts of Exposure to Antibiotic Resistant Bacteria in an Office Rooms in Southern Poland
Abstract
1. Introduction
2. Materials and Methods
2.1. Characteristics of the Sampling Site and Office Building
2.2. Sample Collection
2.3. Bacterial Strain Selection
2.4. Identification of Selected Bacteria
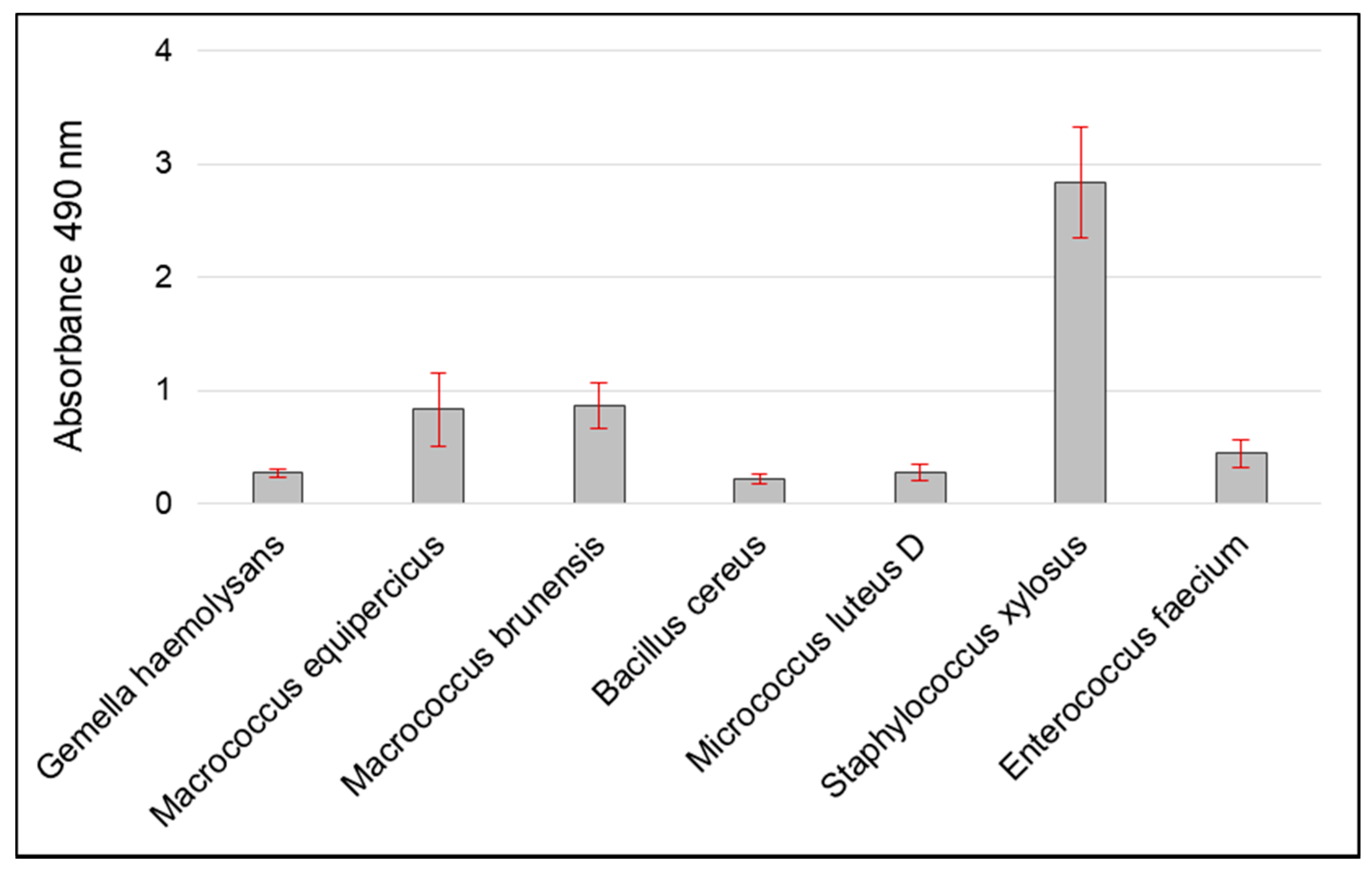
Biofilm Formation Assay
2.5. Antimicrobial Susceptibility Tests and the Detection of Antimicrobial Resistance Genes
Antibiotic Susceptibility Test
2.6. Detection of Antimicrobial Resistance Genes
2.6.1. DNA Isolation
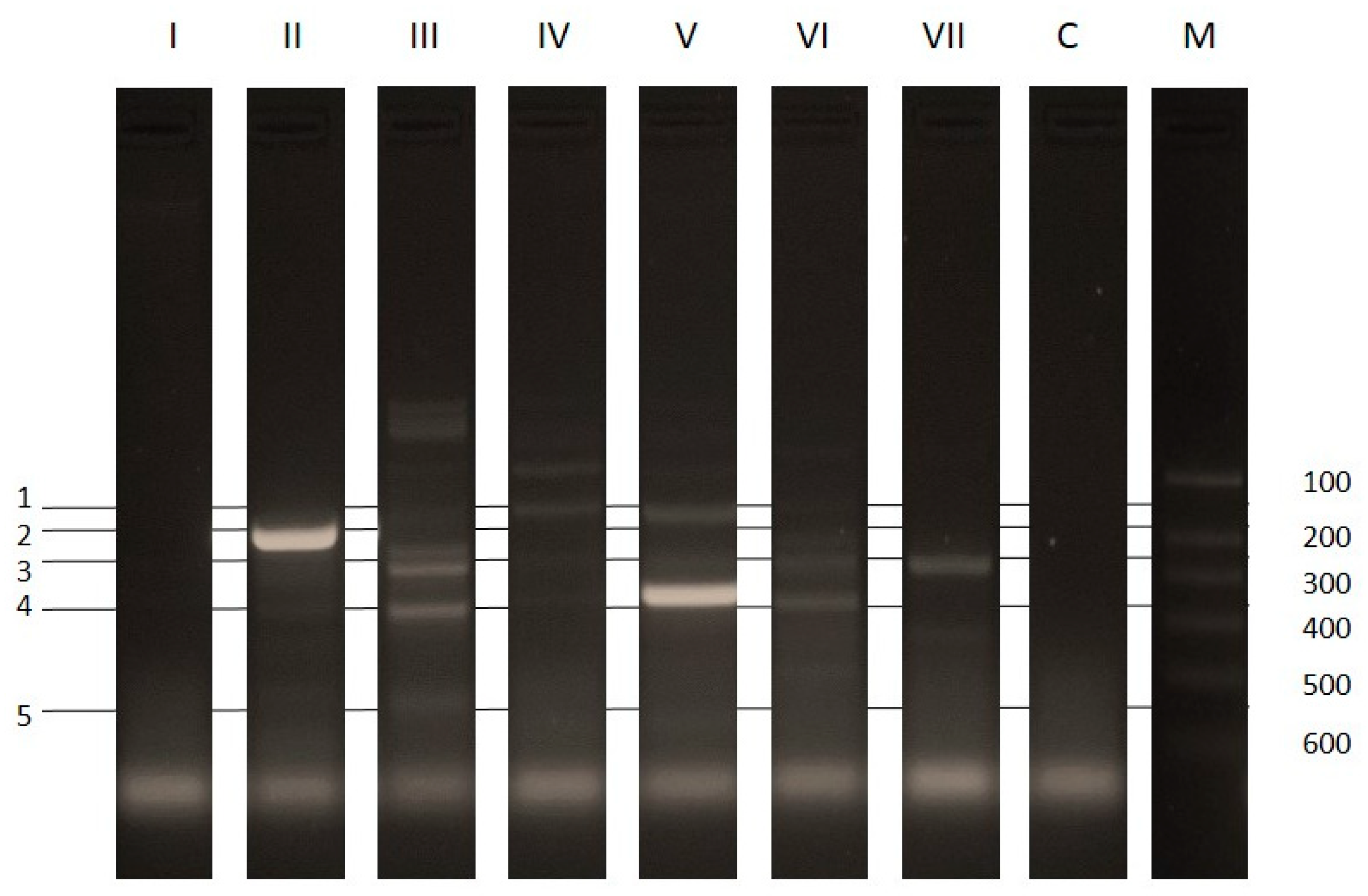
2.6.2. Multiplex PCR for the Detection of Selected Antibiotic Resistance Genes
2.7. Statistical Analysis
3. Results and Discussion
3.1. The Quantity of Culturable Bacterial Aerosol (QCBA)
3.1.1. Concentration of Culturable Bacterial Aerosol (CCBA)
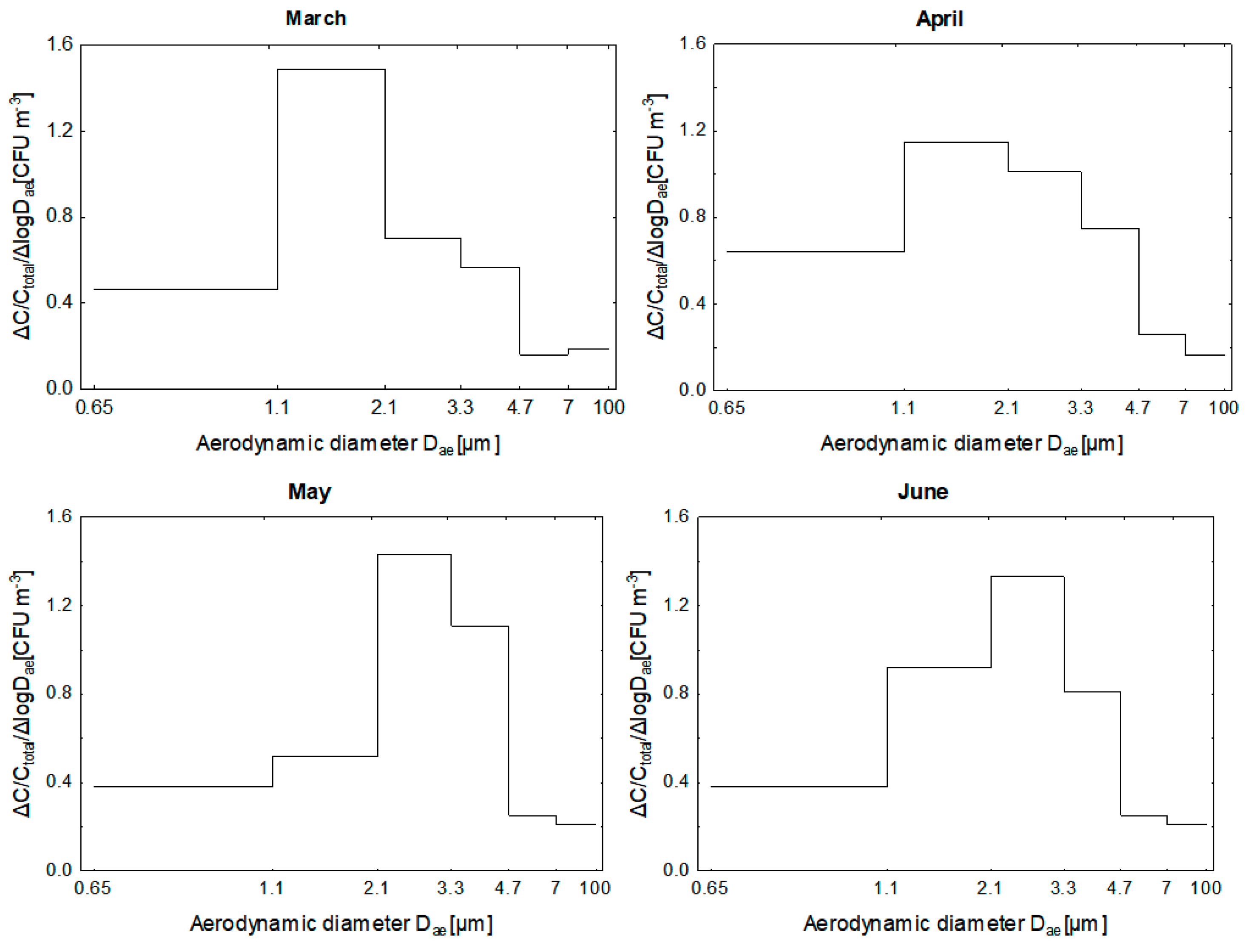
3.1.2. Size Distribution of Bacterial Aerosol
3.1.3. Spring Office Exposure Dose (SPED).
- SPED—is bacterial aerosol exposure dose in office, CFU kg−1
- C—is bacterial aerosol concentration, CFU m−³
- IEF—is indoor exposure fraction: hours spent a day in office, concerning diverse activity patterns hour (in sum on average: eight hours)
- IR—is inhalation rate coefficient characteristic for selected activity levels, m³/day [39]
- BW—is mean body weight, kg.
3.2. The Quality of Culturable Bacterial Aerosol (QlCBA)
Identification of Bacterial Aerosol
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cincinelli, A.; Martellini, T. Indoor Air Quality and Health. Int. J. Environ. Res. Public Health 2017, 14, 1286. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, S.J.; Black, D.W.; Borin, S.S.; Breuer, G.; Burmeister, L.F.; Fuortes, L.J.; Smith, T.F.; Stein, M.A.; Subramanian, P.; Thorne, P.S.; et al. Indoor environmental quality in six commercial office buildings in the midwest United States. Appl. Occup. Environ. Hyg. 2001, 16, 1065–1077. [Google Scholar] [CrossRef] [PubMed]
- Ashmore, M.R.; Dimitroulopoulou, C. Personal exposure of children to air pollution. Atmos. Environ. 2009, 43, 128–141. [Google Scholar] [CrossRef]
- Wichmann, J.; Lind, T.; Nilsson, M.A.M.; Bellander, T. PM2.5, soot and NO2 indoor-outdoor relationships at homes, pre-schools and schools in Stockholm, Sweden. Atmos. Environ. 2010, 44, 4536–4544. [Google Scholar] [CrossRef]
- Wang, Y.F.; Wang, C.H.; Hsu, K.L. Size and seasonal distributions of airborne bioaerosols in commuting trains. Atmos. Environ. 2010, 44, 4331–4338. [Google Scholar] [CrossRef]
- Nasir, Z.A.; Colbeck, I.; Sultan, S.; Ahmed, S. Bioaerosols in residential micro-environments in low income countries: A case study from Pakistan. Environ. Pollut. 2012, 168, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Gołofit-Szymczak, M.; Górny, R.L. Bacterial and fungal aerosols in air-conditioned office buildings in Warsaw, Poland-the winter season. Int. J. Occup. Saf. Ergon. 2010, 16, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Brągoszewska, E.; Pastuszka, J.S. Influence of meteorological factors on the level and characteristics of culturable bacteria in the air in Gliwice, Upper Silesia (Poland). Aerobiologia 2018. [Google Scholar] [CrossRef] [PubMed]
- Pastuszka, J.S. Exposure of the General Population Living in Upper Silesia Industrial Zone to the Particulate, Fibrous and Biological (Bacteria and Fungi) Aerosols; Wroclaw Technical University: Wroclaw, Poland, 2001. (in Polish) [Google Scholar]
- Gołofit-Szymczak, M.; Górny, R.L. Microbiological Air Quality in Office Buildings Equipped with Different Ventilation Systems. Indoor Air 2018, 28, 792–805. [Google Scholar] [CrossRef] [PubMed]
- Douwes, J.; Thorne, P.; Pearce, N.; Heederik, D. Bioaerosol health effects and exposure assessment: Progress and prospects. Ann. Occup. Hyg. 2003, 47, 187–200. [Google Scholar] [PubMed]
- Di Giulio, M.; Grande, R.; Di Campli, E.; Di Bartolomeo, S.; Cellini, L. Indoor air quality in university environments. Environ. Monit. Assess. 2010, 170, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, K.M.; Gerba, C.P.; Maxwell, S.L.; Kelley, S.T. Office space bacterial abundance and diversity in three metropolitan areas. PLoS ONE 2012. [Google Scholar] [CrossRef] [PubMed]
- Nazaroff, W.W. Indoor bioaerosol dynamics. Indoor Air 2016, 26, 61–78. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.C.; Kung, P.Y.; Wu, T.N.; Shen, Y.H. Characterization of indoor-air bioaerosols in Southern Taiwan. Aerosol Air Qual. Res. 2012, 12, 651–661. [Google Scholar] [CrossRef]
- Aydogdu, H.; Asan, A.; Tatman Otkun, M. Indoor and outdoor airborne bacteria in child day-care centers in Edirne City (Turkey), seasonal distribution and influence of meteorological factors. Environ. Monit. Assess. 2010, 164, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Moon, K.W.; Huh, E.H.; Jeong, H.C. Seasonal evaluation of bioaerosols from indoor air of residential apartments within the metropolitan area in South Korea. Environ. Monit. Assess. 2014, 186, 2111–2120. [Google Scholar] [CrossRef] [PubMed]
- Soto, T.; Garcia Murcia, R.M.; Franco, A.; Vicente-Soler, J.; Cansado, J.; Gacto, M. Indoor airborne microbial load in a Spanish university (University of Murcia, Spain). An. Boil. 2009, 31, 109–115. [Google Scholar]
- Latif, M.T.; Yong, S.M.; Saad, A.; Mohamad, N.; Baharudin, N.H.; Mokhtar, M. Bin; Tahir, N.M. Composition of heavy metals in indoor dust and their possible exposure: A case study of preschool children in Malaysia. Air Qual. Atmos. Health 2014, 7, 181–193. [Google Scholar] [CrossRef]
- Watson, A.Y.; Bates, R.R.; Kennedy, D. Assessment of Human Exposure to Air Pollution: Methods, Measurements, and Models. In Air Pollution, the Automobile, and Public Health; National Academies Press: Washington, DC, USA, 1988. [Google Scholar]
- Douglas, P.; Robertson, S.; Gay, R.; Hansell, A.L.; Gant, T.W. A systematic review of the public health risks of bioaerosols from intensive farming. Int. J. Hyg. Environ. Health 2018, 221, 134–173. [Google Scholar] [CrossRef] [PubMed]
- Committee on Damp Indoor Spaces and Health; Board on Health Promotion and Disease Prevention; Institute of Medicine. Damp indoor spaces and health; National Academies Press: Washington, DC, USA, 2004. [Google Scholar]
- Brągoszewska, E.; Biedroń, I.; Kozielska, B.; Pastuszka, J.S. Microbiological indoor air quality in an office building in Gliwice, Poland: Analysis of the case study. Air Qual. Atmos. Health 2018, 11, 729–740. [Google Scholar] [CrossRef]
- Brągoszewska, E.; Mainka, A.; Pastuszka, J.; Lizończyk, K.; Desta, Y. Assessment of Bacterial Aerosol in a Preschool, Primary School and High School in Poland. Atmosphere 2018, 9, 87. [Google Scholar] [CrossRef]
- Menteşe, S.; Arisoy, M.; Rad, A.Y.; Güllü, G. Bacteria and fungi levels in various indoor and outdoor environments in Ankara, Turkey. Clean Soil Air Water 2009, 37, 487–493. [Google Scholar] [CrossRef]
- Kim, K.Y.; Kim, C.N. Airborne microbiological characteristics in public buildings of Korea. Build. Environ. 2007, 42, 2188–2196. [Google Scholar] [CrossRef]
- Dutkiewicz, J.; Górny, R.L. Biologic factors hazardous to health: Classification and criteria of exposure assessment. Med. Pr. 2002, 53, 29–39. [Google Scholar] [PubMed]
- Nevalainen, A.; Pastuszka, J.; Liebhaber, F.; Willeke, K. Performance of bioaerosol samplers: Collection characteristics and sampler design considerations. Atmos. Environ. 1992, 26, 531–540. [Google Scholar] [CrossRef]
- Nevalainen, A.; Willeke, K.; Liebhaber, F.; Pastuszka, J.S.; Burge, H.; Henningson, E. Bioaerosol Sampling. In Aerosol Measurement: Principles, Techniques and Applications; Willeke, K., Baron, P., Eds.; Van Nostrand Reinhold: New York, NY, USA, 1993; pp. 471–492. [Google Scholar]
- Brągoszewska, E.; Mainka, A.; Pastuszka, J. Bacterial and Fungal Aerosols in Rural Nursery Schools in Southern Poland. Atmosphere 2016, 7, 142. [Google Scholar] [CrossRef]
- Bragoszewska, E.; Mainka, A.; Pastuszka, J.S. Concentration and size distribution of culturable bacteria in ambient air during spring and winter in Gliwice: A typical urban area. Atmosphere 2017, 8, 239. [Google Scholar] [CrossRef]
- Srednik, M.E.; Tremblay, Y.D.N.; Labrie, J.; Archambault, M.; Jacques, M.; Cirelli, A.F.; Gentilini, E.R. Biofilm formation and antimicrobial resistance genes of coagulase-negative staphylococci isolated from cows with mastitis in Argentina. FEMS Microbiol. Lett. 2017. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Xia, J.; Xu, Y. Biological features of biofilm-forming ability of Acinetobacter baumannii strains derived from 121 elderly patients with hospital-acquired pneumonia. Clin. Exp. Med. 2016, 16, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Hudzicki, J. Kirby-Bauer Disk Diffusion Susceptibility Test Protocol. Available online: http://www.asmscience.org/content/education/protocol/protocol.3189 (accessed on 21 November 2018).
- Bródka, K.; Sowiak, M.; Kozajda, A.; Cyprowski, M.; Szadkowska-Stańczyk, I. Czynniki biologiczne wpływające na jakość powietrza w pomieszczeniach biurowych. Med. Pr. 2012, 63, 303–315. [Google Scholar] [PubMed]
- Mui, K.W.; Wong, L.T.; Hui, P.S. Risks of unsatisfactory airborne bacteria level in air-conditioned offices of subtropical climates. Build. Environ. 2008, 43, 475–479. [Google Scholar] [CrossRef]
- Gizaw, Z.; Gebrehiwot, M.; Yenew, C. High bacterial load of indoor air in hospital wards: The case of University of Gondar teaching hospital, Northwest Ethiopia. Multidiscip. Respir. Med. 2016, 11, 24. [Google Scholar] [CrossRef] [PubMed]
- WHO. Health Risk Assessment of Air Pollution—General Principles; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Environmental Protection Agency (EPA). Exposure Factors Handbook; EPA: Washington, DC, USA, 2011.
- Brągoszewska, E. Bacterial Aerosol Occuring in the Atmospheric Air in Gliwice and Its Share of the Total Human Exposure to the Bacteria Absorbed by Inhalation. Ph.D. Thesis, Silesian University of Technology, Gliwice, Poland, 2014. [Google Scholar]
- Brągoszewska, E.; Mainka, A.; Pastuszka, J.S. Bacterial aerosols in an urban nursery school in Gliwice, Poland: A case study. Aerobiologia 2016, 32, 469–480. [Google Scholar] [CrossRef]
- Kim, N.Y.; Kim, Y.R.; Kim, M.K.; Cho, D.W.; Kim, J. Isolation and characterization of airborne bacteria and fungi in indoor environment of elementary schools. Korean J. Microbiol. 2007, 43, 193–200. [Google Scholar]
- Stryjakowska-Sekulska, M.; Piotraszewska-Pajak, A.; Szyszka, A.; Nowicki, M.; Filipiak, M. Microbiological quality of indoor air in university rooms. Pol. J. Environ. Stud. 2007, 16, 623–632. [Google Scholar]
- Daisey, J.M.; Angell, W.J.; Apte, M.G. Indoor air quality, ventilation and health symptoms in schools: An analysis of existing information. Indoor Air 2003, 13, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Urban, C.; Rubin, D.; Segal-Maurer, S. Subacute endocarditis caused by Gemella haemolysans and a review of the literature. Scand. J. Infect. Dis. 2004, 36, 885–888. [Google Scholar] [CrossRef] [PubMed]
- Vandeplassche, E.; Coenye, T.; Crabbé, A. Developing selective media for quantification of multispecies biofilms following antibiotic treatment. PLoS ONE 2017, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Guay, G.G.; Rothstein, D.M. Expression of the tetK gene from Staphylococcus aureus in Escherichia coli: Comparison of substrate specificities of tetA(B), tetA(C), and tetK efflux proteins. Antimicrob. Agents Chemother. 1993, 37, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Miranda, C.D.; Kehrenberg, C.; Ulep, C.; Schwarz, S.; Roberts, M.C. Diversity of Tetracycline Resistance Genes in Bacteria from Chilean Salmon Farms. Antimicrob. Agents Chemother. 2003, 47, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Wuertz, S.; Okabe, S.; Hausner, M. Microbial communities and their interactions in biofilm systems: An overview. Water Sci. Technol. 2004, 49, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Houry, A.; Briandet, R.; Aymerlch, S.; Gohar, M. Involvement of motility and flagella in Bacillus cereus biofilm formation. Microbiology 2010, 156, 1009–1018. [Google Scholar] [CrossRef] [PubMed]
- Wijman, J.G.E.; De Leeuw, P.P.L.A.; Moezelaar, R.; Zwietering, M.H.; Abee, T. Air-liquid interface biofilms of Bacillus cereus: Formation, sporulation, and dispersion. Appl. Environ. Microbiol. 2007, 73, 1481–1488. [Google Scholar] [CrossRef] [PubMed]
- Górny, R.; Lis, D.; Wlazło, A.; Łudzeń-Izbińska, B.; Sokal, J. Increase of microbial air contamination in surgical drape and coat factory as a result of improper design of ventilation system. Indoor Air 2005, 2, 1460–1463. [Google Scholar]
- Carretto, E.; Barbarini, D.; Couto, I.; De Vitis, D.; Marone, P.; Verhoef, J.; De Lencastre, H.; Brisse, S. Identification of coagulase-negative staphylococci other than Staphylococcus epidermidis by automated ribotyping. Clin. Microbiol. Infect. 2005, 11, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Fox, L.K.; Seo, K.S.; McGuire, M.A.; Park, Y.H.; Rurangirwa, F.R.; Sischo, W.M.; Bohach, G.A. Comparison of phenotypic and genotypic methods for the species identification of coagulase-negative staphylococcal isolates from bovine intramammary infections. Vet. Microbiol. 2011, 147, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Boerlin, P.; Kuhnert, P.; Hüssy, D.; Schaellibaum, M. Methods for identification of Staphylococcus aureus isolates in cases of bovine mastitis. J. Clin. Microbiol. 2003, 41, 767–771. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, C.C.; Pizauro, L.J.L.; Soltes, G.A.; Slavic, D.; De Ávila, F.A.; Pizauro, J.M.; MacInnes, J.I. Some coagulase negative Staphylococcus spp. isolated from buffalo can be misidentified as Staphylococcus aureus by phenotypic and Sa442 PCR methods. BMC Res. Notes 2018, 11, 346. [Google Scholar] [CrossRef] [PubMed]
- Nováková, D.; Sedláček, I.; Pantůček, R.; Štětina, V.; Švec, P.; Petráš, P. Staphylococcus equorum and Staphylococcus succinus isolated from human clinical specimens. J. Med Microbiol. 2006, 55, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Drosinos, E.H.; Paramithiotis, S.; Kolovos, G.; Tsikouras, I.; Metaxopoulos, I. Phenotypic and technological diversity of lactic acid bacteria and staphylococci isolated from traditionally fermented sausages in Southern Greece. Food Microbiol. 2007, 24, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Blaiotta, G.; Fusco, V.; Ercolini, D.; Pepe, O.; Coppola, S. Diversity of Staphylococcus species strains based on partial kat (catalase) gene sequences and design of a PCR-restriction fragment length polymorphism assay for identification and differentiation of coagulase-positive species (S. aureus, S. delphini, S. hyicus, S. intermedius, S. pseudintermedius, and S. schleiferi subsp. coagulans). J. Clin. Microbiol. 2010, 48, 192–201. [Google Scholar] [PubMed]
- Rossi, F.; Tofalo, R.; Torriani, S.; Suzzi, G. Identification by 16S-23S rDNA intergenic region amplification, genotypic and phenotypic clustering of Staphylococcus xylosus strains from dry sausages. J. Appl. Microbiol. 2001, 90, 365–371. [Google Scholar] [CrossRef] [PubMed]
- García-Mena, J.; Murugesan, S.; Pérez-Muñoz, A.A.; García-Espitia, M.; Maya, O.; Jacinto-Montiel, M.; Monsalvo-Ponce, G.; Piña-Escobedo, A.; Domínguez-Malfavón, L.; Gómez-Ramírez, M.; et al. Airborne Bacterial Diversity from the Low Atmosphere of Greater Mexico City. Microb. Ecol. 2016, 72, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Planchon, S.; Gaillard-Martinie, B.; Dordet-Frisoni, E.; Bellon-Fontaine, M.N.; Leroy, S.; Labadie, J.; Hébraud, M.; Talon, R. Formation of biofilm by Staphylococcus xylosus. Int. J. Food Microbiol. 2006, 109, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Li, H.; Zhang, C.; Liang, B.; Li, J.; Wang, L.; Du, X.; Liu, X.; Qiu, S.; Song, H. Relationship between antibiotic resistance, biofilm formation, and biofilm-specific resistance in Acinetobacter baumannii. Front. Microbiol. 2016, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bouillard, L.; Michel, O.; Devleeschouwer, M. Bacterial contamination of indoor air, surfaces, and settled dust, and related dust endotoxin concentrations in healthy office buildings. Ann. Agric. Environ. Med. 2005, 12, 187–192. [Google Scholar] [PubMed]
- Duprè, I.; Zanetti, S.; Schito, A.M.; Fadda, G.; Sechi, L.A. Incidence of virulence determinants in clinical Enterococcus faecium and Enterococcus faecalis isolates collected in Sardinia (Italy). J. Med Microbiol. 2003, 52, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.; Spiegelman, G.B.; Yim, G. The world of subinhibitory antibiotic concentrations. Curr. Opin. Microbiol. 2006, 9, 445–553. [Google Scholar] [CrossRef] [PubMed]
- Holmes, A.H.; Moore, L.S.P.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J.V. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 387, 176–187. [Google Scholar] [CrossRef]
- Zhang, X.-X.; Zhang, T.; Fang, H.H.P. Antibiotic resistance genes in water environment. Appl. Microbiol. Biotechnol. 2009, 82, 397–414. [Google Scholar] [CrossRef] [PubMed]
- Stalder, T.; Barraud, O.; Casellas, M.; Dagot, C.; Ploy, M.-C. Integron Involvement in Environmental Spread of Antibiotic Resistance. Front. Microbiol. 2012, 3, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Skariyachan, S.; Mahajanakatti, A.B. Environmental monitoring of bacterial contamination and antibiotic resistance patterns of the fecal coliforms isolated from Cauvery River, a major drinking water source in Karnataka, India. Environ. Monit. Assess. 2015, 187, 279. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Li, S.; Zhang, S.; Yu, X. Reduction in horizontal transfer of conjugative plasmid by UV irradiation and low-level chlorination. Water Res. 2016, 91, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Bridier, A.; Briandet, R.; Thomas, V.; Briandet, R.; Dubois-Brissonnet, F. Resistance of bacterial biofilms to disinfectants: A review. Biofouling 2011, 29, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Ouyang, W.; Qian, Y.; Su, C.; Su, J.; Chen, H. High-throughput profiling of antibiotic resistance genes in drinking water treatment plants and distribution systems. Environ. Pollut. 2016, 213, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Lehtola, M.J.; Miettinen, I.T.; Keinänen, M.M.; Kekki, T.K.; Laine, O.; Hirvonen, A.; Vartiainen, T.; Martikainen, P.J. Microbiology, chemistry and biofilm development in a pilot drinking water distribution system with copper and plastic pipes. Water Res. 2004, 38, 3769–3779. [Google Scholar] [CrossRef] [PubMed]
- Melo, L.F.; Mergulh, F.J. Escherichia coli adhesion, biofilm development and antibiotic susceptibility on biomedical materials. J. Biomed. Mater. Res. Part A 2014, 2011, 1414–1423. [Google Scholar]
- McKinney, C.W.; Pruden, A. Ultraviolet disinfection of antibiotic resistant bacteria and their antibiotic resistance genes in water and wastewater. Environ. Sci. Technol. 2012, 46, 13393–13400. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, L.; Zhang, X.; Xu, C.; Dong, L.; Yao, M. Bioaerosol emissions and detection of airborne antibiotic resistance genes from a wastewater treatment plant. Atmos. Environ. 2016, 124, 404–412. [Google Scholar] [CrossRef]
- Just, N.A.; Létourneau, V.; Kirychuk, S.P.; Singh, B.; Duchaine, C. Potentially pathogenic bacteria and antimicrobial resistance in bioaerosols from cage-housed and floor-housed poultry operations. Ann. Occup. Hyg. 2012, 56, 440–449. [Google Scholar] [PubMed]
- Li, J.; Cao, J.; Zhu, Y.; Chen, Q.; Shen, F.; Wu, Y.; Xu, S.; Fan, H.; Da, G.; Huang, R.; et al. Global Survey of Antibiotic Resistance Genes in Air. Environ. Sci. Technol. 2018, 52, 10975–10984. [Google Scholar] [CrossRef] [PubMed]

| Environmental Parameters, Mean ± SD 1 | ||||
|---|---|---|---|---|
| Month | Indoor Temperature, °C | Indoor Relative Humidity (RH), % | Outdoor Temperature, °C | Outdoor Relative Humidity (RH), % |
| March | 19.2 ± 4.1 | 31.2 ± 2.1 | 14.1 ± 1.1 | 76.0 ± 12.7 |
| April | 19.8 ± 6.1 | 34.8 ± 3.2 | 16.4 ± 2.1 | 77.8 ± 11.2 |
| May | 23.2 ± 6.4 | 32.4 ± 4.0 | 19.1 ± 3.2 | 72.6 ± 12.6 |
| June | 24.1 ± 6.2 | 30.2 ± 2.2 | 22.2 ± 2.4 | 71.6 ± 8.1 |
| Group of Antibiotics | Antibiotic | Dose (mg) |
|---|---|---|
| Aminocoumarin | Novobiocin | 30 |
| Amikacin | 30 | |
| Gentamicin | 120 | |
| Gentamicin | 200 | |
| Neomycin | 30 | |
| Netilmicin | 30 | |
| Tobramycin | 30 | |
| Carbapenems | Doripenem | 10 |
| Ertapenem | 10 | |
| Imipenem | 10 | |
| Cephalosporins | Cefaclor | 30 |
| Cefadroxil | 30 | |
| Cefepime | 30 | |
| Cefoxitin | 30 | |
| Ceftaroline | 5 | |
| Ceftazidime | 30 | |
| Drugs against mycobacteria | Rifampicin | 30 |
| Glycopeptides | Teicolpanin | 30 |
| Vancomycin | 30 | |
| Macrolides | Azithromycin | 15 |
| Erythromycin | 30 | |
| Monobactams | Aztreonam | 30 |
| Nitrofurans | Nitrofurantoin | 300 |
| others | Metronidazole | 50 |
| Mupirocin | 200 | |
| Trimethoprim | 5 | |
| Penicillins | Amoxycillin | 30 |
| Ampicillin | 25 | |
| Piperacillin | 100 | |
| Ticarcillin | 75 | |
| Quinolones/Fluoroquinolones | Ciprofloxacin | 10 |
| Nalidixic acid | 30 | |
| Norfloxacin | 10 | |
| Ofloxacin | 5 | |
| Sulfonamides | Trimethoprim/sulph | 25 |
| Tetracyclines | Doxycycline | 30 |
| Minocycline | 30 |
| Target | Primer | Sequence (5’ to 3’) | Amplicon Size (bp) |
|---|---|---|---|
| aacA-aphD | aacA-aphD1 aacA-aphD2 | TAA TCC AAG AGC AAT AAG GGC GCC ACA CTA TCA TAA CCA CTA | 227 |
| ermA | ermA1 ermA2 | AAG CGG TAA ACC CCT CTG A TTC GCA AAT CCC TTC TCA AC | 190 |
| tekK | tekK1 tekK2 | GTA GCG ACA ATA GGT AAT AGT GTA GTG ACA ATA AAC CTC CTA | 360 |
| mecA | mecA1 mecA2 | AAA ATC GAT GGT AAA GGT TGG C AGT TCT GCA GTA CCG GAT TTG C | 532 |
| vatB | varB1 vatB2 | GCT GCG AAT TCA GTT GTT ACA CTG ACC AAT CCC ACC ATT TTA | 136 |
| Month | CCBA | SD 1 | CCBA max | CCBA min |
|---|---|---|---|---|
| March | 786 | 118.14 | 852 | 289 |
| April | 821 | 141.21 | 946 | 324 |
| May | 582 | 117.47 | 774 | 126 |
| June | 424 | 96.24 | 581 | 114 |
| Average spring CCBA | 653.25 | 118.26 | 788.25 | 213.25 |
| Month | SPED—Office Building Exposure Dose (CFU kg−1) |
|---|---|
| March | 210 |
| April | 218 |
| May | 155 |
| June | 113 |
| Average SPED | 174 |
| Species of Isolated Bacteria |
|---|
| Macrococcus equipercicus |
| Macrococcus brunensis |
| Micrococcus luteus D |
| Staphylococcus xylosus |
| Gemella haemolysans |
| Enterococcus faecium |
| Bacillus cereus |
| Bacteria | Antibiotics Resistance |
|---|---|
| Gemella haemolysans | Vancomycin, Azithromycin, Erythromycin, Aztreonam, Nitrofurantoin, Metronidazole, Mupirocin, Ticarcillin, Nalidixic acid, Norfloxacin |
| Macrococcus equipercicus | Neomycin, Tobramycin, Ertapenem, Imipenem, Cefaclor, Cefadroxil, Cefepime, Cefoxitin, Ceftazidime, Teicolpanin, Azithromycin, Erythromycin, Aztreonam, Metronidazole, Trimethoprim, Amoxycillin, Ampicillin, Piperacillin, Ticarcillin, Nalidixic acid |
| Macrococcus brunensis | Amikacin, Gentamicin, Doripenem, Imipenem, Cefaclor, Cefepime, Ceftaroline, Rifampicin, Azithromycin, Erythromycin, Aztreonam, Metronidazole, Amoxycillin, Ampicillin, Ofloxacin |
| Bacillus cereus | Doripenem, Ertapenem, Cefaclor, Cefadroxil, Cefepime, Cefoxitin, Ceftaroline, Ceftazidime, Erythromycin, Aztreonam, Metronidazole, Ciprofloxacin, Nalidixic acid, Doxycycline |
| Micrococcus luteus D | Doripenem, Ertapenem, Cefaclor, Cefadroxil, Cefepime, Cefoxitin, Ceftaroline, Ceftazidime, Teicolpanin, Nitrofurantoin, Metronidazole, Mupirocin, Trimethoprim, Amoxycillin, Ampicillin, Piperacillin, Ticarcillin, Norfloxacin, Trimethoprim/sulph |
| Staphylococcus xylosus | Aztreonam, Metronidazole, Nalidixic acid |
| Enterococcus faecium | Amikacin, Neomycin, Netilmicin, Tobramycin, Ertapenem, Cefadroxil, Cefepime, Cefoxitin, Ceftazidime, Azithromycin, Erythromycin, Aztreonam, Metronidazole |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brągoszewska, E.; Biedroń, I. Indoor Air Quality and Potential Health Risk Impacts of Exposure to Antibiotic Resistant Bacteria in an Office Rooms in Southern Poland. Int. J. Environ. Res. Public Health 2018, 15, 2604. https://doi.org/10.3390/ijerph15112604
Brągoszewska E, Biedroń I. Indoor Air Quality and Potential Health Risk Impacts of Exposure to Antibiotic Resistant Bacteria in an Office Rooms in Southern Poland. International Journal of Environmental Research and Public Health. 2018; 15(11):2604. https://doi.org/10.3390/ijerph15112604
Chicago/Turabian StyleBrągoszewska, Ewa, and Izabela Biedroń. 2018. "Indoor Air Quality and Potential Health Risk Impacts of Exposure to Antibiotic Resistant Bacteria in an Office Rooms in Southern Poland" International Journal of Environmental Research and Public Health 15, no. 11: 2604. https://doi.org/10.3390/ijerph15112604
APA StyleBrągoszewska, E., & Biedroń, I. (2018). Indoor Air Quality and Potential Health Risk Impacts of Exposure to Antibiotic Resistant Bacteria in an Office Rooms in Southern Poland. International Journal of Environmental Research and Public Health, 15(11), 2604. https://doi.org/10.3390/ijerph15112604






