Summary Outcomes of the ODIN Project on Food Fortification for Vitamin D Deficiency Prevention
Abstract
1. Introduction
2. The ODIN Project
- What is the prevalence of vitamin D deficiency in Europe?
- What are the dietary requirements for vitamin D during pregnancy, childhood and adolescence to prevent vitamin D deficiency?
- Does ethnicity affect dietary requirements for vitamin D?
- Can fortification/biofortification increase population intakes of vitamin D by enough to reduce the prevalence of inadequate intakes safely?
3. Results
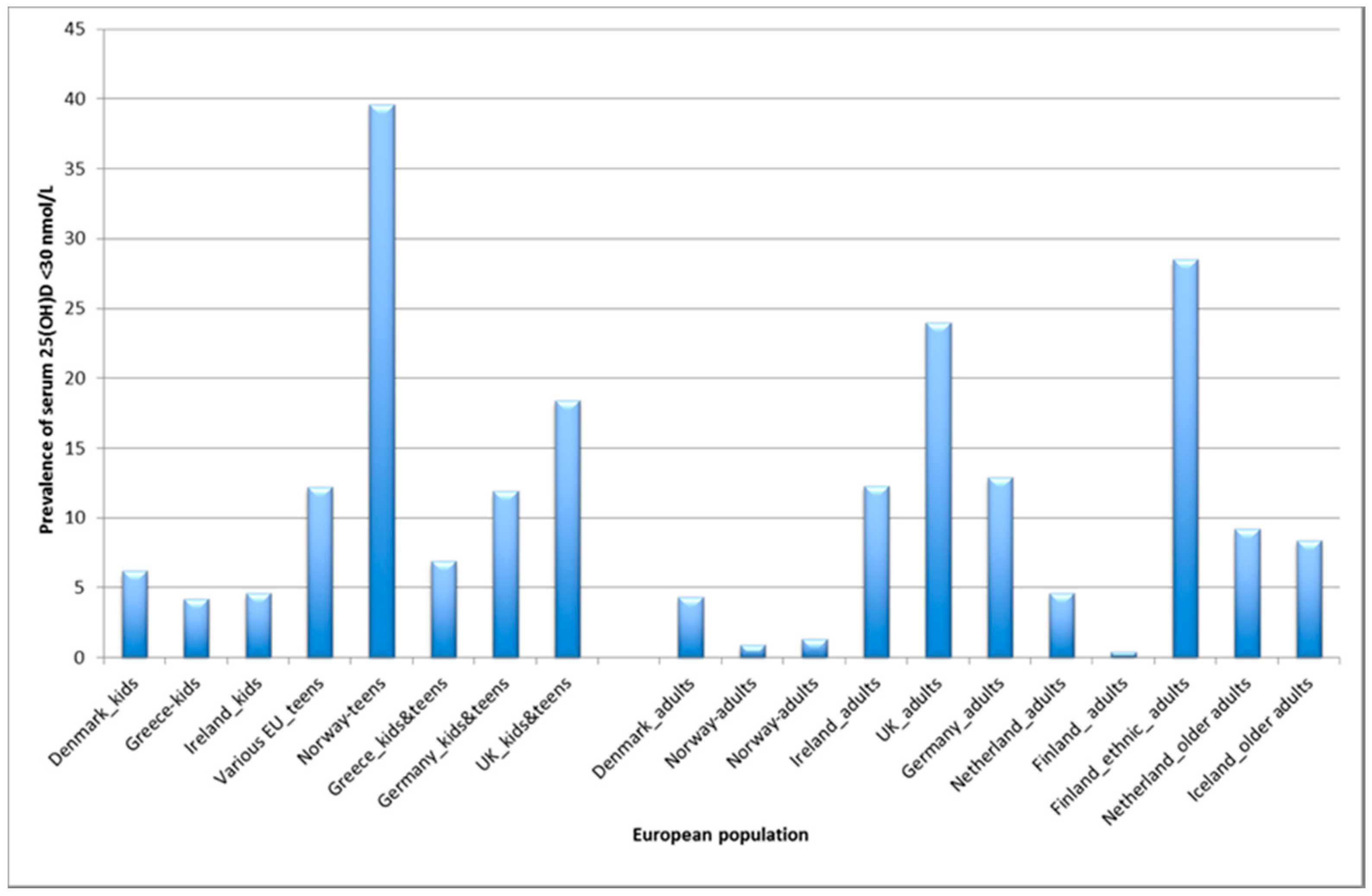
3.1. What is the Prevalence of Vitamin D Deficiency in Europe?
3.2. Dietary Requirements for Vitamin D: Children, Adolescents, Pregnant Women and Persons of Ethnic Minority
3.2.1. Children and Adolescents
- (1)
- For white 4–8 year-old children, 8 µg/day of vitamin D3 prevented 25(OH)D falling below 30 nmol/L during wintertime and 20 µg/day maintained 97.5% ≥50 nmol/L [22];
- (2)
- For white 14–18 year-old adolescents, intakes of 13 µg/day of vitamin D3 were needed to prevent 25(OH)D from dropping <30 nmol/L and ~30 µg/day was required to achieve ≥50 nmol/L during winter [23].
3.2.2. Women of Ethnic Minority in Northern Europe
- (3)
- For white women, 8 µg/day vitamin D3 prevented 25(OH)D falling <30 nmol/L in winter, but 18 µg/day was needed for women of East African descent to meet this minimum threshold [25].
3.2.3. Pregnant Women and New-Born Infants
- (4)
- Among white women, 30 μg/d vitamin D3 safely maintained serum 25(OH)D concentrations ≥50 nmol/L during pregnancy at ~50° N, which ensured that 25(OH)D was >25 nmol/L in 99% and ≥30 nmol/L in 95% of umbilical cord sera [26].
3.3. Can Fortification Increase Population Intakes of Vitamin D by Enough to Reduce the Prevalence of Inadequate Intakes Safely?
3.3.1. Vitamin D Intakes
3.3.2. Vitamin D-Specific Issues in Dietary Surveys
3.3.3. Evidence for food fortification
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Autier, P.; Mullie, P.; Macacu, A.; Dragomir, M.; Boniol, M.; Coppens, K.; Pizot, C.; Boniol, M. Effect of vitamin D supplementation on non-skeletal disorders: A systematic review of meta-analyses and randomised trials. Lancet Diabetes Endocrinol. 2017, 5, 986–1004. [Google Scholar] [CrossRef]
- Ebeling, P.; Adler, R.; Jones, G.; Liberman, U.A.; Mazziotti, G.; Minisola, S.; Munns, C.; Napoli, N.; Pittas, A.; Giustina, A.; et al. Management of endocrine disease: Therapeutics of vitamin D. Eur. J. Endocrinol. 2018, 179, R239–R259. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Bassuk, S.S.; Lee, I.M.; Cook, N.R.; Albert, M.A.; Gordon, D.; Zaharris, E.; Macfadyen, J.G.; Danielson, E.; Lin, J.; et al. The vitamin D and OmegA-3 TriaL (VITAL): Rationale and design of a large randomized controlled trial of vitamin D and marine omega-3 fatty acid supplements for the primary prevention of cancer and cardiovascular disease. Contemp. Clin. Trials 2012, 33, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, A.D.; Manson, J.E. Update on the vitamin D and OmegA-3 trial (VITAL). J. Steroid Biochem. Mol. Biol. 2016, 155, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; Ross, A.C., Taylor, C.L., Yaktine, A.L., Del Valle, H.B., Eds.; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Scientific Advisory Committee on Nutrition Report on Vitamin D and Health. Available online: http://www.sacn.gov.uk/pdfs/sacn_vitamin D_and_health_report_web.pdf (accessed on 1 July 2016).
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific opinion on dietary reference values for vitamin D. EFSA J. 2016, 14, 4547. [Google Scholar]
- NORDEN Nordic Nutrition Recommendations, 5th Edition (NNR5)—Vitamin D. Available online: http://www.slv.se/en-gb/Startpage-NNR/Public-consultation11/ (accessed on 28 August 2013).
- German Nutrition Society. New reference values for vitamin D. Ann. Nutr. Metab. 2012, 60, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Kiely, M. Towards prevention of vitamin D deficiency and beyond: Knowledge gaps and research needs in vitamin D nutrition and public health. Br. J. Nutr. 2011, 106, 1617–1627. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Kiely, M. Recommended dietary intakes for vitamin D: Where do they come from, what do they achieve and how can we meet them? J. Hum. Nut. Diet. 2014, 27, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Kiely, M.; Cashman, K.D. The ODIN project: Development of food-based approaches for prevention of vitamin D deficiency throughout life. Nutr. Bull. 2015, 40, 235–246. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Molgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Sarafin, K.; Durazo-Arvizu, R.; Tian, L.; Phinney, K.W.; Tai, S.; Camara, J.E.; Merkel, J.; Green, E.; Sempos, C.T.; Brooks, S.P. Standardizing 25-hydroxyvitamin D values from the Canadian Health Measures Survey. Am. J. Clin. Nutr. 2015, 102, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Schleicher, R.L.; Sternberg, M.R.; Looker, A.C.; Yetley, E.A.; Lacher, D.A.; Sempos, C.T.; Taylor, C.L.; Durazo-Arvizu, R.A.; Maw, K.L.; Chaudhary-Webb, M.; et al. National estimates of serum total 25-Hydroxyvitamin D and metabolite concentrations measured by Liquid Chromatography-Tandem Mass Spectrometry in the US population during 2007-2010. J. Nutr. 2016, 146, 1051–1061. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Worldwide Prevalence of Anaemia 1993–2005: WHO Global Database on Anaemia; De Benoist, B., McLean, E., Egli, I., Cogswell, M., Eds.; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Sempos, C.T.; Vesper, H.W.; Phinney, K.W.; Thienpont, L.M.; Coates, P.M. Vitamin D status as an international issue: National surveys and the problem of standardization. Scand. J. Clin. Lab. Invest. Suppl. 2012, 234, 32–40. [Google Scholar] [CrossRef]
- Durazo-Arvizu, R.A.; Tian, L.; Brooks, S.P.J.; Sarafin, K.; Cashman, K.D.; Kiely, M.; Merkel, J.; Myers, G.L.; Coates, P.M.; Sempos, C.T. The Vitamin D Standardization Program (VDSP) manual for retrospective laboratory standardization of serum 25-hydroxyvitamin D data. J. AOAC Int. 2017, 100, 1234–1243. [Google Scholar] [CrossRef] [PubMed]
- Quraishi, S.A.; Camargo, C.A.; Manson, J.E. Low vitamin D status in Europe: Moving from evidence to sound public health policies. Am. J. Clin. Nutr. 2016, 103, 957–958. [Google Scholar] [CrossRef] [PubMed]
- Manios, Y.; Moschonis, G.; Lambrinou, C.P.; Tsoutsoulopoulou, K.; Binou, P.; Karachaliou, A.; Breidenassel, C.; Gonzalez-Gross, M.; Kiely, M.; Cashman, K.D. A systematic review of vitamin D status in southern European countries. Eur. J. Nutr. 2018, 57, 2001–2036. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, C.; Damsgaard, C.T.; Hauger, H.; Ritz, C.; Lanham-New, S.A.; Smith, T.J.; Hennessy, Á.; Dowling, K.; Cashman, K.D.; Kiely, M.; et al. Estimation of the dietary requirement for vitamin D in white children aged 4–8 y: A randomized, controlled, dose-response trial. Am. J. Clin. Nutr. 2016, 104, 1310–1317. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Tripkovic, L.; Damsgaard, C.T.; Molgaard, C.; Ritz, C.; Wilson-Barnes, S.L.; Dowling, K.G.; Hennessy, Á.; Cashman, K.D.; Kiely, M.; et al. Estimation of the dietary requirement for vitamin D in adolescents aged 14–18 y: A dose-response, double-blind, randomized placebo-controlled trial. Am. J. Clin. Nutr. 2016, 104, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Adebayo, F.A.; Itkonen, S.T.; Ohman, T.; Skaffari, E.; Saarnio, E.M.; Erkkola, M.; Cashman, K.D.; Lamberg-Allardt, C. Vitamin D intake, serum 25-hydroxyvitamin D status and response to moderate vitamin D3 supplementation: A randomised controlled trial in East African and Finnish women. Br. J. Nutr. 2018, 119, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Ritz, C.; Adebayo, F.A.; Dowling, K.G.; Itkonen, S.T.; Öhman, T.; Skaffari, E.; Saarnio, E.M.; Kiely, M.; Lamberg-Allardt, C. Differences in the dietary requirement for vitamin D among Caucasian and East African women at Northern latitude. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, K.M.; Hennessy, Á.; Hull, G.L.; Healy, K.; Ritz, C.; Kenny, L.C.; Cashman, K.D.; Kiely, M.E. Estimation of the maternal vitamin D intake that maintains circulating 25-hydroxyvitamin D in late gestation at a concentration sufficient to keep umbilical cord sera ≥25–30 nmol/L: A dose-response, double-blind, randomized placebo-controlled trial in pregnant women at northern latitude. Am. J. Clin. Nutr. 2018, 108, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Ohlund, I.; Lind, T.; Hernell, O.; Silfverdal, S.A.; Karlsland Åkeson, P. Increased vitamin D intake differentiated according to skin color is needed to meet requirements in young Swedish children during winter: A double-blind randomized clinical trial. Am. J. Clin. Nutr. 2017, 106, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, J.C.; Jindal, P.S.; Smith, L.M. Vitamin D supplementation in young White and African American women. J. Bone. Miner. Res. 2014, 29, 173–181. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, K.M.; Kiely, M.E. Ethnic disparities in the dietary requirement for vitamin D during pregnancy: Considerations for nutrition policy and research. Proc. Nutr. Soc. 2018, 77, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Kiely, M.; Hemmingway, A.; O’Callaghan, K.M. Vitamin D in pregnancy: Current perspectives and future directions. Ther. Adv. Musculoskelet. Dis. 2017, 9, 145–154. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, K.M.; Kiely, M. Systematic review of vitamin D and hypertensive disorders of pregnancy. Nutrients 2018, 10, 294. [Google Scholar] [CrossRef] [PubMed]
- Munns, C.F.; Shaw, N.; Kiely, M.; Specker, B.L.; Thacher, T.D.; Ozono, K.; Michigami, T.; Tiosano, D.; Mughal, M.Z.; Mäkitie, O.; et al. Global consensus recommendations on prevention and management of nutritional rickets. J. Clin. Endocrinol. Metab. 2016, 101, 394–415. [Google Scholar] [CrossRef] [PubMed]
- Kiely, M.; O’Donovan, S.M.; Kenny, L.C.; Hourihane, J.O.; Irvine, A.D.; Murray, D.M. Vitamin D metabolite concentrations in umbilical cord blood serum and associations with clinical characteristics in a large prospective mother-infant cohort in Ireland. J. Steroid Biochem. Mol. Biol. 2017, 167, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Ritz, C.; Kiely, M. Improved dietary guidelines for vitamin D: Application of Individual Participant Data (IPD)-level meta-regression analyses. Nutrients 2017, 9, 469. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, C.M.; Kazantzidis, A.; Ryan, M.J.; Barber, N.; Sempos, C.T.; Durazo-Arvizu, R.A.; Jorde, R.; Grimnes, G.; Eiriksdottir, G.; Gudnason, V.; et al. Seasonal changes in vitamin D-effective UVB availability in Europe and associations with population serum 25-Hydroxyvitamin D. Nutrients 2016, 8, 533. [Google Scholar] [CrossRef] [PubMed]
- Roseland, J.M.; Phillipis, K.M.; Patterson, K.Y.; Pehrsson, P.R.; Taylor, C.L. Vitamin D in foods: An evolution of knowledge. In Vitamin D, 4th ed.; Feldman, D., Pike, J.W., Bouillon, R., Giovannucci, E., Goltzman, D., Hewison, M., Eds.; Elsevier Academic Press: London, UK, 2018; pp. 41–77. [Google Scholar]
- Whiting, S.J.; Calvo, M.S. Vitamin D fortification and supplementation policies to correct vitamin D insufficiency/deficiency globally. In Vitamin D, 4th ed.; Feldman, D., Pike, J.W., Bouillon, R., Giovannucci, E., Goltzman, D., Hewison, M., Eds.; Elsevier Academic Press: London, UK, 2018; pp. 91–108. [Google Scholar]
- Roman Viñas, B.; Ribas Barba, L.; Ngo, J.; Gurinovic, M.; Novakovic, R.; Cavelaars, A.; de Groot, L.C.; van’t Veer, P.; Matthys, C.; Serra Majem, L. Projected prevalence of inadequate nutrient intakes in Europe. Ann. Nutr. Metab. 2011, 59, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Black, L.J.; Walton, J.; Flynn, A.; Cashman, K.D.; Kiely, M. Small increments in vitamin D intake by Irish adults over a decade sShow that strategic initiatives to fortify the food supply are needed. J. Nutr. 2015, 145, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Whiting, S.J.; Langlois, K.A.; Vatanparast, H.; Greene-Finestone, L.S. The vitamin D status of Canadians relative to the 2011 Dietary Reference Intakes: An examination in children and adults with and without supplement use. Am. J. Clin. Nutr. 2011, 94, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Fulgoni, V.L., 3rd; Keast, D.R.; Bailey, R.L.; Dwyer, J. Foods, fortificants, and supplements: Where do Americans get their nutrients? J. Nutr. 2011, 141, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Kiely, M.; Black, L.J. Dietary strategies to maintain adequacy of circulating 25-hydroxyvitamin D concentrations. Scand. J. Clin. Lab. Invest. Suppl. 2012, 243, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Kiely, M. Vitamin D and food fortification. In Vitamin D, 4th ed.; Feldman, D., Pike, J.W., Bouillon, R., Giovannucci, E., Goltzman, D., Hewison, M., Eds.; Elsevier Academic Press: London, UK, 2018; pp. 109–127. [Google Scholar]
- Brett, N.R.; Gharibeh, N.; Weiler, H.A. Effect of vitamin D supplementation, food fortification, or bolus injection on vitamin D status in children aged 2–18 years: A meta-analysis. Adv. Nutr. 2018, 9, 454–464. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, S.; Cranney, A.; Horsley, T.; Weiler, H.A.; Atkinson, S.A.; Hanley, D.A.; Ooi, D.S.; Ward, L.; Barrowman, N.; Fang, M.; et al. Efficacy of food fortification on serum 25-hydroxyvitamin D concentrations: Systematic review. Am. J. Clin. Nutr. 2008, 88, 1528–1534. [Google Scholar] [CrossRef] [PubMed]
- Black, L.J.; Seamans, K.M.; Cashman, K.D.; Kiely, M. An updated systematic review and meta-analysis of the efficacy of vitamin D food fortification. J. Nutr. 2012, 142, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Jääskeläinen, T.; Itkonen, S.T.; Lundqvist, A.; Erkkola, M.; Koskela, T.; Lakkala, K.; Dowling, K.G.; Hull, G.L.; Kröger, H.; Karppinen, J.; et al. The positive impact of general vitamin D food fortification policy on vitamin D status in a representative adult Finnish population: Evidence from an 11-y follow-up based on standardized 25-hydroxyvitamin D data. Am. J. Clin. Nutr. 2017, 105, 1512–1520. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Kiely, M. Tackling inadequate vitamin D intakes within the population: Fortification of dairy products with vitamin D may not be enough. Endocrine 2016, 51, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Calvo, M.S.; Whiting, S.J. Public health strategies to overcome barriers to optimal vitamin D status in populations with special needs. J. Nutr. 2006, 136, 1135–1139. [Google Scholar] [CrossRef] [PubMed]
- Madsen, K.H.; Rasmussen, L.B.; Andersen, R.; Molgaard, C.; Jakobsen, J.; Bjerrum, P.J.; Andersen, E.W.; Mejborn, H.; Tetens, I. Randomized controlled trial of the effects of vitamin D-fortified milk and bread on serum 25-hydroxyvitamin D concentrations in families in Denmark during winter: The vitamin D study. Am. J. Clin. Nutr. 2013, 98, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Milešević, J.; Samaniego, L.; Kiely, M.; Glibetić, M.; Roe, M.; Finglas, P. Specialized food composition dataset for vitamin D content in foods based on European standards: Application to dietary intake assessment. Food Chem. 2018, 240, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Manios, Y.; Moschonis, G.; Mavrogianni, C.; van den Heuvel, E.; Singh-Povel, C.M.; Kiely, M.; Cashman, K.D. Reduced-fat Gouda-type cheese enriched with vitamin D3 effectively prevents vitamin D deficiency during winter months in postmenopausal women in Greece. Eur. J. Nutr. 2017, 56, 2367–2377. [Google Scholar] [CrossRef] [PubMed]
- Hayes, A.; Duffy, S.; O’Grady, M.; Jakobsen, J.; Galvin, K.; Teahan-Dillon, J.; Kerry, J.; Kelly, A.; O’Doherty, J.; Higgins, S.; et al. Vitamin D-enhanced eggs are protective of wintertime serum 25-hydroxyvitamin D in a randomized controlled trial of adults. Am. J. Clin. Nutr. 2016, 104, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Kiely, M.; Seamans, K.M.; Urbain, P. Effect of ultraviolet light-exposed mushrooms on vitamin D status: Liquid Chromatography-Tandem Mass Spectrometry Reanalysis of biobanked sera from a randomized controlled trial and a systematic review plus Meta-Analysis. J. Nutr. 2016, 146, 565–575. [Google Scholar] [CrossRef] [PubMed]
- Duffy, S.K.; Kelly, A.K.; Rajauria, G.; Jakobsen, J.; Clarke, L.C.; Monahan, F.J.; Dowling, K.G.; Hull, G.; Galvin, K.; Cashman, K.D.; et al. The use of synthetic and natural vitamin D sources in pig diets to improve meat quality and vitamin D content. Meat Sci. 2018, 143, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Duffy, S.K.; O’ Doherty, J.V.; Rajauria, G.; Clarke, L.C.; Hayes, A.; Dowling, K.G.; O’Grady, M.N.; Kerry, J.P.; Jakobsen, J.; Cashman, K.D.; et al. Vitamin D-biofortified beef: A comparison of cholecalciferol with synthetic versus UVB-mushroom-derived ergosterol as feed source. Food Chem. 2018, 256, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Duffy, S.K.; O’ Doherty, J.V.; Rajauria, G.; Clarke, L.C.; Cashman, K.D.; Hayes, A.; O’Grady, M.N.; Kerry, J.P.; Kelly, A.K. Cholecalciferol supplementation of heifer diets increases beef vitamin D concentration and improves beef tenderness. Meat Sci. 2017, 134, 103–110. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kiely, M.; Cashman, K.D. Summary Outcomes of the ODIN Project on Food Fortification for Vitamin D Deficiency Prevention. Int. J. Environ. Res. Public Health 2018, 15, 2342. https://doi.org/10.3390/ijerph15112342
Kiely M, Cashman KD. Summary Outcomes of the ODIN Project on Food Fortification for Vitamin D Deficiency Prevention. International Journal of Environmental Research and Public Health. 2018; 15(11):2342. https://doi.org/10.3390/ijerph15112342
Chicago/Turabian StyleKiely, Mairead, and Kevin D. Cashman. 2018. "Summary Outcomes of the ODIN Project on Food Fortification for Vitamin D Deficiency Prevention" International Journal of Environmental Research and Public Health 15, no. 11: 2342. https://doi.org/10.3390/ijerph15112342
APA StyleKiely, M., & Cashman, K. D. (2018). Summary Outcomes of the ODIN Project on Food Fortification for Vitamin D Deficiency Prevention. International Journal of Environmental Research and Public Health, 15(11), 2342. https://doi.org/10.3390/ijerph15112342




