Biomonitoring and Subsequent Risk Assessment of Combined Exposure to Phthalates in Iranian Children and Adolescents
Abstract
1. Introduction
2. Materials and Method
2.1. Study Population and Sample Collection
2.2. Target Compound and Analysis
2.3. Phthalate Metabolite Analysis
2.4. Daily Intake Estimation
2.5. Mixture Risk Assessment
2.6. Statistical Analysis
3. Results
3.1. Urinary Phthalate Metabolite Concentrations
3.2. Daily Intake Estimations and Combined Risk Assessment
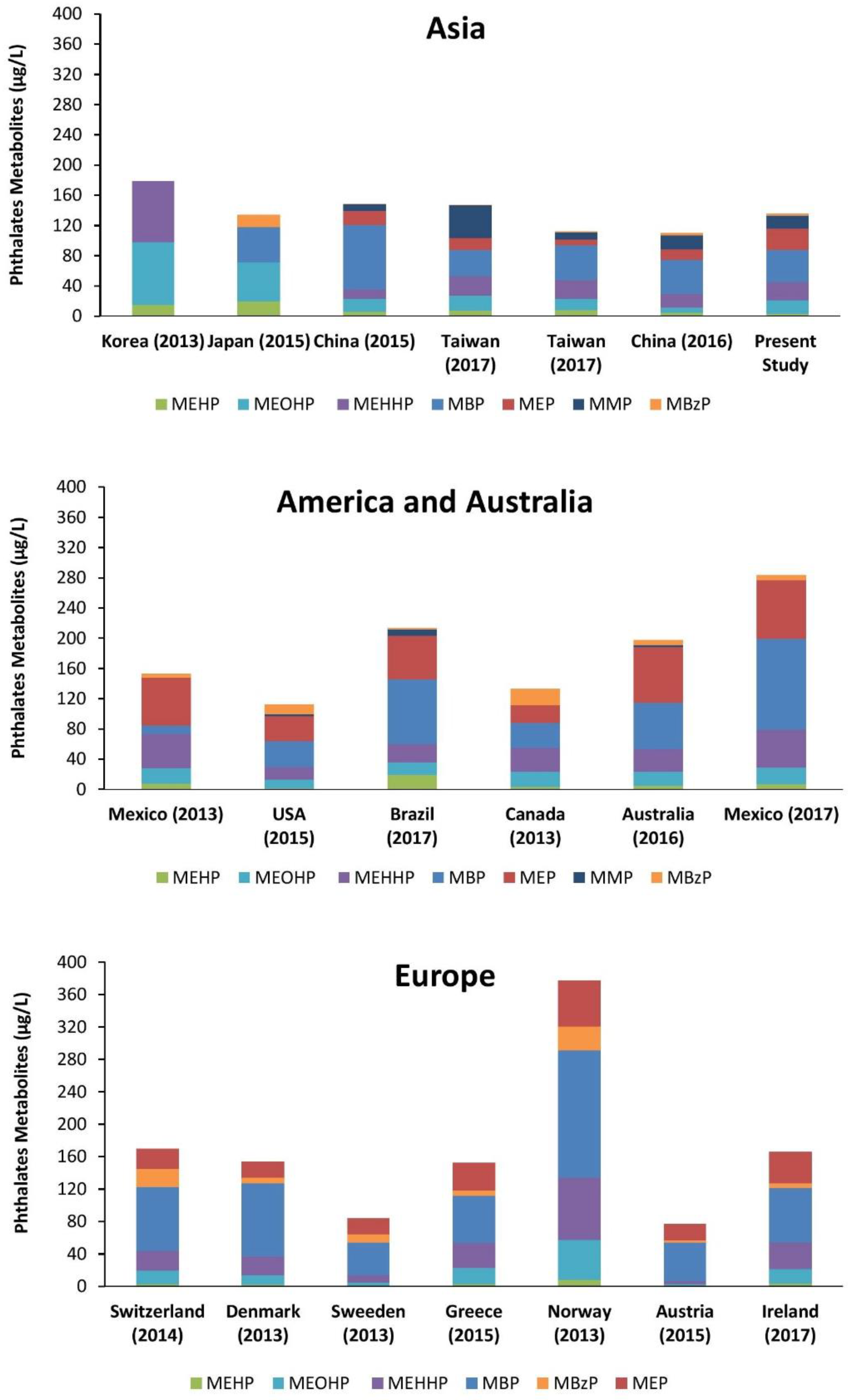
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khetan, S.K. Anti-Androgenic Chemicals. In Endocrine Disruptors in the Environment; John Wiley & Sons: Hoboken, NJ, USA, 2014; pp. 91–110. [Google Scholar]
- Jacobs, M.N.; Marczylo, E.L.; Guerrero-Bosagna, C.; Rüegg, J. Marked for Life: Epigenetic Effects of Endocrine Disrupting Chemicals. Annu. Rev. Environ. Resour. 2017, 42, 105–160. [Google Scholar] [CrossRef]
- EPA, U.S. Environmental Protection Agency, Phthalates Action Plan; Revised 03/14/2012 2012. Available online: https://www.epa.gov/sites/production/files/2015-09/documents/phthalates_actionplan_revised_2012-03-14.pdf (accessed on 14 May 2012).
- European Chemicals Agencyb. SEAC Committee for Risk Assessment (RAC) Committee for Socioeconomic Analysis (SEAC)(2012a): Opinion on an Annex XV Dossier Proposing Restrictions on Four Phthalates. Compiled Version Prepared by the ECHA Secretariat of RAC’s Opinion (Adopted 15 June 2012) and SEAC’s Opinion (Adopted 5 December 2012). Substance Assessment of DIBP as Candidate for Restriction under RoHS. Available online: https://echa.europa.eu/documents/10162/58050be8-f7be-4b55-b106-76dda4989dd6 (accessed on 15 December 2012).
- ECHA. ECHA, European Chemicals Agency. Diethyl Phthalate; European Chemicals Agency: Helsinki, Finland, 2013. [Google Scholar]
- ECB. ECB, European Chemicals Bureau, European Union Risk Assessment Report Bis(2-ethylhexyl)phthalate (DEHP); Office for Official Publications of the European Communities: Luxembourg, 2008. [Google Scholar]
- NRC. Phthalates and Cumulative Risk Assessment: The Task Ahead; The National Academies Press: Washington, DC, USA, 2008.
- Navarro, R.; Pérez Perrino, M.; Gómez Tardajos, M.; Reinecke, H. Phthalate plasticizers covalently bound to PVC: Plasticization with suppressed migration. Macromolecules 2010, 43, 2377–2381. [Google Scholar] [CrossRef]
- CDC. Centers for Disease Control Prevention, Fourth National Report on Human Exposure to Environmental Chemicals; Department of Health and Human Services: Atlanta, GA, USA, 2009.
- Bruinen de Bruin, Y.; Lahaniatis, M.; Papameletiou, D.; Del Pozo, C.; Reina, V.; Van Engelen, J.; Heinemeyer, G.; Viso, A.C.; Rodriguez, C.; Jantunen, M. Risk management measures for chemicals in consumer products: Documentation, assessment, and communication across the supply chain. J. Expos. Sci. Environ. Epidemiol. 2007, 17, S55–S66. [Google Scholar] [CrossRef] [PubMed]
- Andrade, A.J.; Grande, S.W.; Talsness, C.E.; Grote, K.; Chahoud, I. A dose–response study following in utero and lactational exposure to di-(2-ethylhexyl)-phthalate (DEHP): Non-monotonic dose–response and low dose effects on rat brain aromatase activity. Toxicology 2006, 227, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Borch, J.; Ladefoged, O.; Hass, U.; Vinggaard, A.M. Steroidogenesis in fetal male rats is reduced by DEHP and DINP, but endocrine effects of DEHP are not modulated by DEHA in fetal, prepubertal and adult male rats. Reprod. Toxicol. 2004, 18, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Okubo, T.; Suzuki, T.; Yokoyama, Y.; Kano, K.; Kano, I. Estimation of estrogenic and anti-estrogenic activities of some phthalate diesters and monoesters by MCF-7 cell proliferation assay in vitro. Biol. Pharm. Bull. 2003, 26, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Cook, T.J.; Knipp, G.T. Effects of di-(2-ethylhexyl)-phthalate (DEHP) and its metabolites on fatty acid homeostasis regulating proteins in rat placental HRP-1 trophoblast cells. Toxicol. Sci. 2005, 84, 287–300. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, R.M.; Irvine, D.S. How strong is the evidence of a link between environmental chemicals and adverse effects on human reproductive health? BMJ 2004, 328, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.S. Environmental anti-androgens and male reproductive health: Focus on phthalates and testicular dysgenesis syndrome. Reproduction 2004, 127, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Heudorf, U.; Mersch-Sundermann, V.; Angerer, J. Phthalates: Toxicology and exposure. Int. J. Hyg. Environ. Health 2007, 210, 623–634. [Google Scholar] [CrossRef] [PubMed]
- Jurewicz, J.; Hanke, W. Exposure to phthalates: Reproductive outcome and children health. A review of epidemiological studies. Int. J. Occup. Med. Environ. Health 2011, 24, 115–141. [Google Scholar] [CrossRef] [PubMed]
- EC. Commission Staff Working Paper, 4th Report on the Implementation of the “Community Strategy for Endocrine Disruptors” a Range of Substances Suspected of Interfering with the Hormone Systems of Humans and Wildlife (COM (1999) 706); European Commission: Brussels, Belgium, 2011. [Google Scholar]
- Braun, J.M.; Sathyanarayana, S.; Hauser, R. Phthalate exposure and children’s health. Curr. Opin. Pediatr. 2013, 25, 247. [Google Scholar] [CrossRef] [PubMed]
- Johns, L.E.; Cooper, G.S.; Galizia, A.; Meeker, J.D. Exposure assessment issues in epidemiology studies of phthalates. Environ. Int. 2015, 85, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Esters, L.-C.P. State of the Science Report Phthalates Substance Grouping Long-Chain Phthalate Esters; Environment Canada: Toronto, ON, USA, 2015.
- Hauser, R.; Calafat, A. Phthalates and human health. Occup. Environ. Med. 2005, 62, 806–818. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, S.; Masai, E.; Kamimura, N.; Takahashi, K.; Anderson, R.C.; Faisal, P.A. Phthalates impact human health: Epidemiological evidences and plausible mechanism of action. J. Hazard. Mater. 2017, 340, 360–383. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, F.W.; Castorina, R.; Maddalena, R.L.; Nishioka, M.G.; McKone, T.E.; Bradman, A. Phthalate exposure and risk assessment in California child care facilities. Environ. Sci. Technol. 2014, 48, 7593–7601. [Google Scholar] [CrossRef] [PubMed]
- Saravanabhavan, G.; Guay, M.; Langlois, É.; Giroux, S.; Murray, J.; Haines, D. Biomonitoring of phthalate metabolites in the Canadian population through the Canadian Health Measures Survey (2007–2009). Int. J. Hyg. Environ. Health 2013, 216, 652–661. [Google Scholar] [CrossRef] [PubMed]
- CDC. Fourth National Report on Human Exposure to Environmental Chemicals. Updated Tables; 2012. Available online: http://www.cdc.gov/exposurereport/pdf/FourthReport_UpdatedTables _Feb2015.pdf (accessed on 1 February 2015).
- Koch, H.M.; Wittassek, M.; Brüning, T.; Angerer, J.; Heudorf, U. Exposure to phthalates in 5–6 years old primary school starters in Germany—A human biomonitoring study and a cumulative risk assessment. Int. J. Hyg. Environ. Health 2011, 214, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, C.; Uhl, M.; Weiss, S.; Scharf, S.; König, J. Austrian reference values for phthalate metabolite exposure in children/adolescents and adults. Int. J. Hyg. Environ. Health 2018, 111, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, G.; Ginsberg, J.; Foos, B. Approaches to children’s exposure assessment: Case study with diethylhexylphthalate (DEHP). Int. J. Environ. Res. Public Health 2016, 13, 670. [Google Scholar] [CrossRef] [PubMed]
- Zota, A.R.; Calafat, A.M.; Woodruff, T.J. Temporal trends in phthalate exposures: Findings from the National Health and Nutrition Examination Survey, 2001–2010. Environ. Health Perspect. 2014, 122, 235. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, H.; Nielsen, J.K.S.; Mørck, T.A.; Hansen, P.W.; Jensen, J.F.; Nielsen, O.; Andersson, A.-M.; Knudsen, L.E. Urinary excretion of phthalate metabolites, phenols and parabens in rural and urban Danish mother–child pairs. Int. J. Hyg. Environ. Health 2013, 216, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Colacino, J.A.; Harris, T.R.; Schecter, A. Dietary intake is associated with phthalate body burden in a nationally representative sample. Environ. Health Perspect. 2010, 118, 998–1003. [Google Scholar] [CrossRef] [PubMed]
- Calafat, A.M.; McKee, R.H. Integrating biomonitoring exposure data into the risk assessment process: Phthalates [diethyl phthalate and di (2-ethylhexyl) phthalate] as a case study. Environ. Health Perspect. 2006, 114, 1783. [Google Scholar] [CrossRef] [PubMed]
- Anand-Ivell, R.; Cohen, A.; Nørgaard-Pedersen, B.; Jönsson, B.A.; Bonde, J.-P.; Hougaard, D.M.; Lindh, C.H.; Toft, G.; Lindhard, M.S.; Ivell, R. Amniotic Fluid INSL3 Measured During the Critical Time Window in Human Pregnancy Relates to Cryptorchidism, Hypospadias, and Phthalate Load: A Large Case–Control Study. Front. Physiol. 2018, 9, 406. [Google Scholar] [CrossRef] [PubMed]
- Hines, E.P.; Calafat, A.M.; Silva, M.J.; Mendola, P.; Fenton, S.E. Concentrations of phthalate metabolites in milk, urine, saliva, and serum of lactating North Carolina women. Environ. Health Perspect. 2008, 117, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Main, K.M.; Mortensen, G.K.; Kaleva, M.M.; Boisen, K.A.; Damgaard, I.N.; Chellakooty, M.; Schmidt, I.M.; Suomi, A.-M.; Virtanen, H.E.; Petersen, J.H. Human breast milk contamination with phthalates and alterations of endogenous reproductive hormones in infants three months of age. Environ. Health Perspect. 2005, 114, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.J.; Reidy, J.A.; Samandar, E.; Herbert, A.R.; Needham, L.L.; Calafat, A.M. Detection of phthalate metabolites in human saliva. Arch. Toxicol. 2005, 79, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Calafat, A.M.; Slakman, A.R.; Silva, M.J.; Herbert, A.R.; Needham, L.L. Automated solid phase extraction and quantitative analysis of human milk for 13 phthalate metabolites. J. Chromatogr. B 2004, 805, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Gurusankar, R.; Yenugadhati, N.; Krishnan, K.; Hays, S.; Haines, D.; Zidek, A.; Kuchta, S.; Kinniburgh, D.; Gabos, S.; Mattison, D. The role of human biological monitoring in health risk assessment. Int. J. Risk Assess. Manag. 2017, 20, 136–197. [Google Scholar] [CrossRef]
- Council, N.R. Phthalates and Cumulative Risk Assessment: The Tasks Ahead; National Academies Press: Washington, DC, USA, 2009. [Google Scholar]
- Kumar, A.R.; Sivaperumal, P. Analytical methods for the determination of biomarkers of exposure to phthalates in human urine samples. TrAC Trends Anal. Chem. 2016, 75, 151–161. [Google Scholar] [CrossRef]
- Christensen, K.L.; Makris, S.L.; Lorber, M. Generation of hazard indices for cumulative exposure to phthalates for use in cumulative risk assessment. Regul. Toxicol. Pharmacol. 2014, 69, 380–389. [Google Scholar] [CrossRef] [PubMed]
- McLanahan, E.; Stanek, J.; Davis, A. RATE Exposure Assessment Modules—EXA 408, EXA 409; U.S. EPA Office of Research and Development: Washington, DC, USA, 2013.
- Kortenkamp, A. Low dose mixture effects of endocrine disrupters and their implications for regulatory thresholds in chemical risk assessment. Curr. Opin. Pharmacol. 2014, 19, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Howdeshell, K.L.; Furr, J.; Lambright, C.R.; Rider, C.V.; Wilson, V.S.; Gray, L.E. Cumulative effects of dibutyl phthalate and diethylhexyl phthalate on male rat reproductive tract development: Altered fetal steroid hormones and genes. Toxicol. Sci. 2007, 99, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, C.; Uhl, M.; Weiss, S.; Koch, H.M.; Scharf, S.; König, J. Human biomonitoring of phthalate exposure in Austrian children and adults and cumulative risk assessment. Int. J. Hyg. Environ. Health 2015, 218, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; Chen, M.; Kransler, K.M.; Zaleski, R.T. Assessment of chemical coexposure patterns based upon phthalate biomonitoring data within the 2007/2008 National Health and Nutrition Examination Survey. J. Expos. Sci. Environ. Epidemiol. 2015, 25, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Rastkari, N.; Ahmadkhaniha, R. Magnetic solid-phase extraction based on magnetic multi-walled carbon nanotubes for the determination of phthalate monoesters in urine samples. J. Chromatogr. A 2013, 1286, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Dirtu, A.C.; Geens, T.; Dirinck, E.; Malarvannan, G.; Neels, H.; Van Gaal, L.; Jorens, P.G.; Covaci, A. Phthalate metabolites in obese individuals undergoing weight loss: Urinary levels and estimation of the phthalates daily intake. Environ. Int. 2013, 59, 344–353. [Google Scholar] [CrossRef] [PubMed]
- CDC. The Centers for Disease Control and Prevention (CDC) Laboratory Procedure Manual, Phthalate Metabolites in Urine. Method No: 6306.03; 26 March 2012. Available online: https://www.cdc.gov/nchs/data/nhanes/nhanes_09_10/PHTHTE_F_met.pdf (accessed on 23 October 2018).
- Anderson, W.A.; Castle, L.; Hird, S.; Jeffery, J.; Scotter, M.J. A twenty-volunteer study using deuterium labelling to determine the kinetics and fractional excretion of primary and secondary urinary metabolites of di-2-ethylhexylphthalate and di-iso-nonylphthalate. Food Chem. Toxicol. 2011, 49, 2022–2029. [Google Scholar] [CrossRef] [PubMed]
- Koch, H.; Christensen, K.; Harth, V.; Lorber, M.; Brüning, T. Di-n-butyl phthalate (DnBP) and diisobutyl phthalate (DiBP) metabolism in a human volunteer after single oral doses. Arch. Toxicol. 2012, 86, 1829–1839. [Google Scholar] [CrossRef] [PubMed]
- Anderson, W.; Castle, L.; Scotter, M.; Massey, R.; Springall, C. A biomarker approach to measuring human dietary exposure to certain phthalate diesters. Food Addit. Contam. 2001, 18, 1068–1074. [Google Scholar] [CrossRef] [PubMed]
- Koch, H.M.; Angerer, J. Phthalates: Biomarkers and human biomonitoring. Biomark. Hum. Biomonit. 2011, 1, 179–233. [Google Scholar]
- Albro, P.W.; Corbett, J.T.; Schroeder, J.L.; Jordan, S.; Matthews, H.B. Pharmacokinetics, interactions with macromolecules and species differences in metabolism of DEHP. Environ. Health Perspect. 1982, 45, 19. [Google Scholar] [CrossRef] [PubMed]
- Tranfo, G.; Papaleo, B.; Caporossi, L.; Capanna, S.; De Rosa, M.; Pigini, D.; Corsetti, F.; Paci, E. Urinary metabolite concentrations of phthalate metabolites in Central Italy healthy volunteers determined by a validated HPLC/MS/MS analytical method. Int. J. Hyg. Environ. Health 2013, 216, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Lessmann, F.; Schütze, A.; Weiss, T.; Brüning, T.; Koch, H.M. Determination of metabolites of di (2-ethylhexyl) terephthalate (DEHTP) in human urine by HPLC-MS/MS with on-line clean-up. J. Chromatogr. B 2016, 1011, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Mage, D.T.; Allen, R.H.; Kodali, A. Creatinine corrections for estimating children’s and adult’s pesticide intake doses in equilibrium with urinary pesticide and creatinine concentrations. J. Expos. Sci. Environ. Epidemiol. 2008, 18, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Koch, H.M.; Bolt, H.M.; Preuss, R.; Angerer, J. New metabolites of di (2-ethylhexyl) phthalate (DEHP) in human urine and serum after single oral doses of deuterium-labelled DEHP. Arch. Toxicol. 2005, 79, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Song, N.R.; On, J.-W.; Lee, J.; Park, J.-D.; Kwon, H.-J.; Yoon, H.J.; Pyo, H. Biomonitoring of urinary di(2-ethylhexyl) phthalate metabolites of mother and child pairs in South Korea. Environ. Int. 2013, 54, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Kortenkamp, A.; Faust, M. Combined exposures to anti-androgenic chemicals: Steps towards cumulative risk assessment. Int. J. Androl. 2010, 33, 463–474. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food (AFC) on a request from the Commission related to Di-Butylphthalate (DBP) for use in food contact materials. EFSA J. 2005, 242, 1–14. [Google Scholar]
- EFSA. Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food (AFC) on a request from the Commission related to Butylbenzylphthalate (BBP) for use in food contact materials. EFSA J. 2005, 241, 1–14. [Google Scholar]
- EFSA. Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food (AFC) on a request from the Commission related to Bis(2-ethylhexyl)phthalate (DEHP) for use in food contact materials. EFSA J. 2005, 243, 1–20. [Google Scholar]
- IRIS-EPA. IRIS EPA (Integrated Risk Information System) U.S. Environmental Protection Agency, Chemical Assessment Summary, Di(2-ethylhexyl)phthalate (DEHP). CASRN 117-81-7. 01/31/1987a; 1987. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0014_summary.pdf (accessed on 22 January 1986).
- IRIS-EPA. IRIS EPA (Integrated Risk Information System) U.S. Environmental Protection Agency, Chemical Assessment Summary, Dibutyl phthalate. CASRN 84-74-2. 01/31/1987b; 1987. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0038_summary.pdf (accessed on 22 January 1986).
- IRIS-EPA. IRIS EPA (Integrated Risk Information System) U.S. Environmental Protection Agency, Chemical Assessment Summary, Butyl benzyl phthalate. CASRN 85-68-7. 08/22/1988; 1988. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0293_summary.pdf (accessed on 22 January 1986).
- Hertzberg, R.; Mumtaz, M. Component-Based Risk Assessment Approaches with Additivity and Interactions. In Chemical Mixtures and Combined Chemical and Nonchemical Stressors; Springer: Berlin, Germany, 2018; pp. 369–419. [Google Scholar]
- Fox, M.A.; Brewer, L.E.; Martin, L. An Overview of Literature Topics Related to Current Concepts, Methods, Tools, and Applications for Cumulative Risk Assessment (2007–2016). Int. J. Environ. Res. Public Health 2017, 14, 389. [Google Scholar] [CrossRef] [PubMed]
- EC (European Commission); Scientific Committee on Consumer Products (SCCP). Opinion of the SCCP on Phthalates in Cosmetic Products. 2007. Available online: http://ec.europa.eu/health/archive/ph risk/committees/sccp/documents/out168 en.pdf (accessed on 12 March 2014).
- ECHA. European Chemicals Agency, Substance Evaluation Conclusion Document as Required by REACH Article 48 for Diethyl Phthalate (DEP), EC No 201-550-6, CAS No 84-66-2. Available online: https://www.echa.europa.eu/documents/10162/a5c64025-d83a-4952-8476-ac396ee9acc9 (accessed on 28 August 2015).
- Han, X.; Price, P.S. Applying the maximum cumulative ratio methodology to biomonitoring data on dioxin-like compounds in the general public and two occupationally exposed populations. J. Expos. Sci. Environ. Epidemiol. 2013, 23, 343. [Google Scholar] [CrossRef] [PubMed]
- Price, P.S.; Han, X. Maximum cumulative ratio (MCR) as a tool for assessing the value of performing a cumulative risk assessment. Int. J. Environ. Res. Public Health 2011, 8, 2212–2225. [Google Scholar] [CrossRef] [PubMed]
- Reyes, J.M.; Price, P.S. An analysis of cumulative risks based on biomonitoring data for six phthalates using the Maximum Cumulative Ratio. Environ. Int. 2018, 112, 77–84. [Google Scholar] [CrossRef] [PubMed]
- DEMOCOPHES—Belgium. National Report on Human Biomonitoring in Mothers and Children. 2013. Available online: http://www.eu-hbm.info/euresult/democophes-short-technical-report (accessed on 28 February 2013).
- Huang, P.-C.; Tsai, C.-H.; Liang, W.-Y.; Li, S.-S.; Pan, W.-H.; Chiang, H.-C. Age and gender differences in urinary levels of eleven phthalate metabolites in general Taiwanese population after a DEHP episode. PLoS ONE 2015, 10, e0133782. [Google Scholar] [CrossRef] [PubMed]
- Kasper-Sonnenberg, M.; Koch, H.M.; Wittsiepe, J.; Brüning, T.; Wilhelm, M. Phthalate metabolites and bisphenol A in urines from German school-aged children: Results of the Duisburg birth cohort and Bochum cohort studies. Int. J. Hyg. Environ. Health 2014, 217, 830–838. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific repoert of EFSA, Overview of the procedures currently used at EFSA for the assessment of dietary exposure to different chemical substances. European Food Safety Authority. EFSA J. 2011, 9, 2490. [Google Scholar] [CrossRef]
- Davies, N. Chemicals of Future Concern. AATCC Rev. 2015, 15, 32–37. [Google Scholar] [CrossRef]
- Mieritz, M.G.; Frederiksen, H.; Sørensen, K.; Aksglaede, L.; Mouritsen, A.; Hagen, C.P.; Skakkebaek, N.E.; Andersson, A.M.; Juul, A. Urinary phthalate excretion in 555 healthy Danish boys with and without pubertal gynaecomastia. Int. J. Androl. 2012, 35, 227–235. [Google Scholar] [CrossRef] [PubMed]
- CDC. Fourth National Report on Human Exposure to Environmental Chemicals. Updated Tables; March 2018; Volume 1. Available online: https://www.cdc.gov/exposurereport/pdf/FourthReport_UpdatedTables_Volume1_Mar2018.pdf (accessed on 31 March 2018).
- Wang, B.; Wang, H.; Zhou, W.; Chen, Y.; Zhou, Y.; Jiang, Q. Urinary excretion of phthalate metabolites in school children of China: Implication for cumulative risk assessment of phthalate exposure. Environ. Sci. Technol. 2014, 49, 1120–1129. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Shi, H.; Zhang, Y.; Cao, Y. Dietary intake and phthalates body burden in boys and girls. Arch. Public Health 2015, 73, 1. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Zhang, Z.; Liu, L.; Li, Y.; Ren, N.; Kannan, K. Occurrence and profiles of phthalates in foodstuffs from China and their implications for human exposure. J. Agric. Food Chem. 2012, 60, 6913–6919. [Google Scholar] [CrossRef] [PubMed]
- Wormuth, M.; Scheringer, M.; Vollenweider, M.; Hungerbühler, K. What are the sources of exposure to eight frequently used phthalic acid esters in Europeans? Risk Anal. 2006, 26, 803–824. [Google Scholar] [CrossRef] [PubMed]
- Bamai, Y.A.; Araki, A.; Kawai, T.; Tsuboi, T.; Yoshioka, E.; Kanazawa, A.; Cong, S.; Kishi, R. Comparisons of urinary phthalate metabolites and daily phthalate intakes among Japanese families. Int. J. Hyg. Environ. Health 2015, 218, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, G.; Christie, P.; Zhang, M.; Luo, Y.; Teng, Y. Occurrence and risk assessment of phthalate esters (PAEs) in vegetables and soils of suburban plastic film greenhouses. Sci. Total Environ. 2015, 523, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.-C.; Tsai, C.-H.; Chen, C.-C.; Wu, M.-T.; Chen, M.-L.; Wang, S.-L.; Chen, B.-H.; Lee, C.-C.; Jaakkola, J.J.; Wu, W.-C. Intellectual evaluation of children exposed to phthalate-tainted products after the 2011 Taiwan phthalate episode. Environ. Res. 2017, 156, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Zhou, F.; Wang, Y.; Ning, Y.; Yang, J.-Y.; Zhou, Y.-K. Exposure to phthalates in children aged 5–7 years: Associations with thyroid function and insulin-like growth factors. Sci. Total Environ. 2017, 579, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-C.; Wang, Y.-H.; Wang, S.-L.; Huang, P.-C.; Chuang, S.-C.; Chen, M.-H.; Chen, B.-H.; Sun, C.-W.; Fu, H.-C.; Lee, C.-C. Exposure sources and their relative contributions to urinary phthalate metabolites among children in Taiwan. Int. J. Hyg. Environ. Health 2017, 220, 869–879. [Google Scholar] [CrossRef] [PubMed]
- Larsson, K.; Björklund, K.L.; Palm, B.; Wennberg, M.; Kaj, L.; Lindh, C.H.; Jönsson, B.A.; Berglund, M. Exposure determinants of phthalates, parabens, bisphenol A and triclosan in Swedish mothers and their children. Environ. Int. 2014, 73, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Carlstedt, F.; Jönsson, B.; Bornehag, C.G. PVC flooring is related to human uptake of phthalates in infants. Indoor Air 2013, 23, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Myridakis, A.; Chalkiadaki, G.; Fotou, M.; Kogevinas, M.; Chatzi, L.; Stephanou, E.G. Exposure of preschool-age Greek children (RHEA Cohort) to bisphenol A, parabens, phthalates, and organophosphates. Environ. Sci. Technol. 2015, 50, 932–941. [Google Scholar] [CrossRef] [PubMed]
- Barr, D.B.; Silva, M.J.; Kato, K.; Reidy, J.A.; Malek, N.A.; Hurtz, D.; Sadowski, M.; Needham, L.L.; Calafat, A.M. Assessing human exposure to phthalates using monoesters and their oxidized metabolites as biomarkers. Environ. Health Perspect. 2003, 111, 1148. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.N.; Grice, J.; Cullen, A.; Faustman, E.M. A toxicological framework for the prioritization of children’s safe product act data. Int. J. Environ. Res. Public Health 2016, 13, 431. [Google Scholar] [CrossRef] [PubMed]
- Koch, H.M.; Becker, K.; Wittassek, M.; Seiwert, M.; Angerer, J.; Kolossa-Gehring, M. Di-n-butylphthalate and butylbenzylphthalate—Urinary metabolite levels and estimated daily intakes: Pilot study for the German Environmental Survey on children. J. Expos. Sci. Environ. Epidemiol. 2007, 17, 378–387. [Google Scholar] [CrossRef] [PubMed]
- CDC. Fourth National Report on Human Exposure to Environmental Chemicals. Updated Tables; September 2013. Available online: http://www.cdc.gov/exposurereport/pdf/FourthReport_UpdatedTables_Sep2013.pdf (accessed on 30 September 2013).
- Becker, K.; Güen, T.; Seiwert, M.; Conrad, A.; Pick-Fuß, H.; Müller, J.; Wittassek, M.; Schulz, C.; Kolossa-Gehring, M. GerES IV: Phthalate metabolites and bisphenol A in urine of German children. Int. J. Hyg. Environ. Health 2009, 212, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Wittassek, M.; Heger, W.; Koch, H.M.; Becker, K.; Angerer, J.; Kolossa-Gehring, M. Daily intake of di (2-ethylhexyl) phthalate (DEHP) by German children–A comparison of two estimation models based on urinary DEHP metabolite levels. Int. J. Hyg. Environ. Health 2007, 210, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kang, S.; Lee, G.; Lee, S.; Jo, A.; Kwak, K.; Kim, D.; Koh, D.; Kho, Y.L.; Kim, S.; et al. Urinary phthalate metabolites among elementary school children of Korea: Sources, risks, and their association with oxidative stress marker. Sci. Total Environ. 2014, 472, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Søeborg, T.; Frederiksen, H.; Andersson, A.-M. Cumulative risk assessment of phthalate exposure of Danish children and adolescents using the hazard index approach. Int. J. Androl. 2012, 35, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Dewalque, L.; Charlier, C.; Pirard, C. Estimated daily intake and cumulative risk assessment of phthalate diesters in a Belgian general population. Toxicol. Lett. 2014, 231, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.; Cerejeira, M.J. Concentration addition-based approach for aquatic risk assessment of realistic pesticide mixtures in Portuguese river basins. Environ. Sci. Pollut. Res. 2015, 22, 6756–6765. [Google Scholar] [CrossRef] [PubMed]
- Vallotton, N.; Price, P.S. Use of the maximum cumulative ratio as an approach for prioritizing aquatic coexposure to plant protection products: A case study of a large surface water monitoring database. Environ. Sci. Technol. 2016, 50, 5286–5293. [Google Scholar] [CrossRef] [PubMed]
- De Brouwere, K.; Cornelis, C.; Arvanitis, A.; Brown, T.; Crump, D.; Harrison, P.; Jantunen, M.; Price, P.; Torfs, R. Application of the maximum cumulative ratio (MCR) as a screening tool for the evaluation of mixtures in residential indoor air. Sci. Total Environ. 2014, 479, 267–276. [Google Scholar] [CrossRef] [PubMed]
| Parent Phthalate Compound | Phthalate Monoester Metabolite | Limit of Detection (LOD) (µg/L) | Molecular Weight of Diester Parent Compound, g/mole (MWparent) | Molecular Weight of Metabolite, g/mole (MWmetabolite) | Urinary Excretion Fraction (FUE, 24-h), Expressed as Percent (%) * |
|---|---|---|---|---|---|
| Di-2-ethylhexyl phthalate (DEHP) | Mono-(2-ethylhexyl) phthalate (MEHP) | 0.050 | 390.56 | 278.34 | 6.2 |
| Mono-(2-ethyl-5-hydroxyhexyl) phthalate (MEHHP) | 0.050 | 390.56 | 294.35 | 14.9 | |
| Mono-(2-ethyl-5-oxohexyl) phthalate (MEOHP) | 0.050 | 390.56 | 292.33 | 10.9 | |
| Dibutyl phthalate (DBP) | Monobutyl phthalate (MBP) | 0.040 | 278.34 | 222.24 | 84 |
| Butylbenzyl phthalate (BBP) | Mono-benzyl phthalate (MBzP) | 0.050 | 312.36 | 256.25 | 73 |
| Diethyl phthalate (DEP) | Monoethyl phthalate (MEP) | 0.025 | 222 | 194 | 69 |
| Dimethyl phthalate (DMP) | Monomethyl phthalate (MMP) | 0.030 | 194 | 180 | 69 |
| Phthalate | Toxicological Target | Toxicity Health-Based Guidance Values | ||||
|---|---|---|---|---|---|---|
| EFSA TDI | U.S. EPA RfD | RfD-AA | EFSA TDI (µg/kg bw/day) 1 | U.S. EPA RfD (µg/kg bw/day) 2 | RfD-AA (µg/kg bw/day) 3 | |
| DBP | Germ cell development and mammary gland changes | Increased mortality | Suppression of testicular testosterone production | 10 | 100 | 100 |
| BBP | Anogenital distance change | Increased liver-to-body weight and liver-to-brain weight ratios | Suppression of testicular testosterone production | 500 | 200 | 330 |
| DEHP | Testicular toxicity and developmental toxicity | Increased relative liver weight | Nipple retention | 50 | 20 | 30 |
| Phthalate Metabolites | Group | n | GM | 5th | 50th | 95th | Max | p-Value a | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MEHP | All | 56 | 2.81 | (2.82) | 0.48 | (0.53) | 3.3 | (3.3) | 9.1 | (11.5) | 11.6 (13.2) | 0.406 | (0.579) | |
| ≥6~<12 | 22 | 2.9 | (3.02) | 0.54 | (0.55) | 4.0 | (3.6) | 7.83 | (8.72) | 9.1 (11.5) | ||||
| ≥12~≤18 | 34 | 2.8 | (2.7) | 0.485 | (0.53) | 3.2 | (3.3) | 9.3 | (12.1) | 11.6 (13.2) | ||||
| Boys | 24 | 3.70 | (3.61) | 1.0 | (0.65) | 4.9 | (4.1) | 9.1 | (12.1) | 11.6 (13.2) | 0.213 | (0.127) | ||
| Girls | 32 | 2.35 | (2.33) | 0.32 | (0.32) | 2.8 | (2.6) | 7.83 | (7.2) | 9.3 (9.5) | ||||
| MEOHP | All | 56 | 18.14 | (18.12) | 3.62 | (3.6) | 17.5 | (19.3) | 65.1 | (73.8) | 79.43 | (99.4) | 0.470 | (0.233) |
| ≥6~<12 | 22 | 19.5 | (20.3) | 4.0 | (3.7) | 21.43 | (24.2) | 58.3 | (46.2) | 63.7 | (121.1) | |||
| ≥12~≤18 | 34 | 17.3 | (16.9) | 3.6 | (2.2) | 16.62 | (18.0) | 69.33 | (82.7) | 79.43 | (99.4) | |||
| Boys | 24 | 19.32 | (19.1) | 4.0 | (3.7) | 19.9 | (20.4) | 60.64 | (82.74) | 79.43 | (99.4) | 0.823 | (0.881) | |
| Girls | 32 | 17.3 | (17.43) | 3.62 | (2.2) | 16.74 | (18.9) | 65.1 | (53.3) | 69.33 | (64.4) | |||
| MEHHP | All | 56 | 26.73 | (26.7) | 6.4 | (3.91) | 24.1 | (28.6) | 80.5 | (100.24) | 129.85 | (135.3) | 0.387 | (0.227) |
| ≥6~<12 | 22 | 28.9 | (30.02) | 14.31 | (13.64) | 27.03 | (35.54) | 74.8 | (60.5) | 95.7 | (68.6) | |||
| ≥12~≤18 | 34 | 25.4 | (24.8) | 6.4 | (3.9) | 21.92 | (25.3) | 80.5 | (100.25) | 129.85 | (33.8) | |||
| Boys | 24 | 29.03 | (28.7) | 10.61 | (8.9) | 30.9 | (31.05) | 95.7 | (121.13) | 129.85 | (135.3) | 0.602 | (0.740) | |
| Girls | 32 | 25.12 | (25.3) | 6.40 | (3.91) | 22.24 | (26.8) | 78.82 | (61.9) | 80.3 | (78.03) | |||
| MEP | All | 56 | 27.5 | (27.4) | 13.1 | (9.6) | 28.2 | (30.4) | 53.5 | (62.4) | 62.1 | (68.6) | 0.491 | (0.579) |
| ≥6~<12 | 22 | 30.2 | (31.4) | 18.7 | (17.7) | 28.7 | (29.9) | 53.5 | (66.1) | 62.1 | (68.6) | |||
| ≥12~≤18 | 34 | 25.8 | (25.1) | 6.3 | (3.4) | 26.9 | (31.6) | 51.2 | (59.2) | 54.9 | (62.4) | |||
| Boys | 24 | 23.5 | (23.3) | 13.1 | (9.7) | 25.5 | (26.5) | 53.5 | (47.0) | 54.9 | (62.4) | 0.012 | (0.025) | |
| Girls | 32 | 30.8 | (31.1) | 13.6 | (9.6) | 35.4 | (34.2) | 51.2 | (66.1) | 62.1 | (68.6) | |||
| MBP | All | 56 | 36.4 | (36.4) | 12.8 | (9.7) | 42.9 | (38.7) | 70.3 | (84.2) | 72.3 | (105.9) | 0.502 (0.880) | |
| ≥6~<12 | 22 | 34.8 | (36.2) | 14.4 | (13.1) | 42.9 | (41.6) | 61.2 | (71.3) | 64.1 | (72.0) | |||
| ≥12~≤18 | 34 | 37.5 | (36.5) | 12.8 | (9.8) | 45.0 | (36.9) | 71.9 | (98.1) | 72.3 | (105.9) | |||
| Boys | 24 | 37.2 | (36.8) | 14.1 | (9.8) | 47.3 | (39.2) | 71.9 | (98.1) | 72.3 | (105.9) | 0.6429 (0.868) | ||
| Girls | 32 | 35.9 | (36.2) | 12.8 | (13.5) | 38.5 | (38.7) | 69.7 | (78.2) | 70.3 | (84.0) | |||
| MMP | All | 56 | 15.9 | (15.8) | 4.3 | (4.3) | 17.4 | (15.8) | 42.8 | (48.2) | 43.4 | (53.8) | 0.356 (0.737) | |
| ≥6~<12 | 22 | 14.5 | (15.1) | 5.2 | (4.5) | 15.2 | (15.7) | 34.2 | (40.3) | 40.0 | (41.6) | |||
| ≥12~≤18 | 34 | 16.8 | (16.4) | 4.3 | (4.3) | 19.3 | (16.0) | 43.3 | (51.5) | 43.4 | (53.8) | |||
| Boys | 24 | 15.8 | (15.6) | 4.3 | (3.8) | 19.1 | (16.7) | 40.0 | (42.6) | 42.7 | (53.8) | 0.8946 (0.973) | ||
| Girls | 32 | 15.9 | (16.1) | 4.9 | (4.5) | 15.1 | (15.2) | 43.3 | (48.2) | 43.4 | (51.5) | |||
| MBzP | All | 56 | 2.0 | (2.0) | <LOQ | 2.2 | (2.3) | 5.0 | (5.4) | 5.1 | (6.2) | 0.063 (0.043) | ||
| ≥6~<12 | 22 | 2.4 | (2.5) | 0.7 (0.6) | 3.4 | (3.1) | 5.0 | (6.0) | 5.1 | (6.2) | ||||
| ≥12~≤18 | 34 | 1.8 | (1.7) | <LOQ | 2.1 | (2.0) | 4.4 | (5.2) | 5.1 | (5.4) | ||||
| Boys | 24 | 1.8 | (1.7) | 0.7 (0.6) | 2.0 | (2.0) | 4.5 | (4.1) | 5.1 | (4.6) | 0.0354 (0.043) | |||
| Girls | 32 | 2.2 | (2.2) | <LOQ | 3.3 | (2.8) | 5.0 | (6.0) | 5.1 | (6.2) | ||||
| Phthalate Metabolites | MBP | MEP | MMP | MEHP | MEOHP | MEHHP |
|---|---|---|---|---|---|---|
| MBzP | 0.263 * | 0.540 * | 0.025 | −0.003 | 0.127 | 0.021 |
| MBP | 0.277 * | 0.625 * | −0.063 | −0.000 | 0.043 | |
| MEP | 0.006 | 0.187 | 0.163 | 0.119 | ||
| MMP | 0.026 | −0.024 | 0.007 | |||
| MEHP | 0.731 * | 0.798 * | ||||
| MEOHP | 0.896 * |
| Phthalate Metabolites | Group | n | GM | 5th | 50th | 95th | Max |
|---|---|---|---|---|---|---|---|
| DEHP | All | 56 | 3.47 | 0.58 | 3.41 | 15.03 | 17.85 |
| ≥6~<12 | 22 | 3.67 | 0.63 | 4.08 | 16.57 | 17.85 | |
| ≥12~≤18 | 34 | 3.34 | 0.49 | 3.39 | 15.16 | 16.06 | |
| Boys | 24 | 3.66 | 0.66 | 3.78 | 17.40 | 17.85 | |
| Girls | 32 | 3.33 | 0.47 | 3.39 | 11.25 | 11.74 | |
| DEP | All | 56 | 0.78 | 0.23 | 0.88 | 1.85 | 2.56 |
| ≥6~<12 | 22 | 0.84 | 0.40 | 0.86 | 2.45 | 2.56 | |
| ≥12~≤18 | 34 | 0.74 | 0.09 | 0.89 | 1.86 | 1.93 | |
| Boys | 24 | 0.64 | 0.20 | 0.66 | 1.78 | 1.93 | |
| Girls | 32 | 0.90 | 0.20 | 0.97 | 2.11 | 2.56 | |
| DBP | All | 56 | 0.95 | 0.23 | 1. 1 | 2.47 | 3.10 |
| ≥6~<12 | 22 | 0.87 | 0.20 | 1.11 | 2.10 | 2.16 | |
| ≥12~≤18 | 34 | 0.97 | 0.23 | 1.00 | 2.73 | 3.09 | |
| Boys | 24 | 0.91 | 0.19 | 0.97 | 2.97 | 3.09 | |
| Girls | 32 | 0.94 | 0.28 | 1.11 | 2.24 | 1.11 | |
| DMP | All | 56 | 0.42 | 0.11 | 0.77 | 1.40 | 0.23 |
| ≥6~<12 | 22 | 0.38 | 0.08 | 0.38 | 1.23 | 1.24 | |
| ≥12~≤18 | 34 | 0.45 | 0.11 | 0.44 | 1.86 | 1.5 | |
| Boys | 24 | 0.40 | 0.08 | 0.40 | 1.40 | 1.41 | |
| Girls | 32 | 0.44 | 0.12 | 0.42 | 1.44 | 1.54 | |
| BBP | All | 56 | 0.06 | 0.008 | 0.06 | 0.16 | 0.23 |
| ≥6~<12 | 22 | 0.07 | 0.09 | 0.07 | 0.22 | 0.23 | |
| ≥12~≤18 | 34 | 0.05 | 0.007 | 0.06 | 0.162 | 0.17 | |
| Boys | 24 | 0.05 | 0.012 | 0.06 | 0.11 | 0.112 | |
| Girls | 32 | 0.07 | 0.007 | 0.08 | 0.19 | 0.23 |
| Phthalates | Group | HQ TDI | HQ RfD-AA | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Min | Median | 95p | Max | n > 1 | Min | Median | 95p | Max | n > 1 | ||
| DBP | All | 0.02 | 0.11 | 0.25 | 0.31 | 0 | 0.001 | 0.01 | 0.02 | 0.03 | 0 |
| ≥6~<12 | 0.02 | 0.11 | 0.21 | 0.22 | 0 | 0.002 | 0.01 | 0.02 | 0.02 | 0 | |
| ≥12~≤18 | 0.02 | 0.10 | 0.27 | 0.31 | 0 | 0.002 | 0.01 | 0.02 | 0.02 | 0 | |
| Boys | 0.02 | 0.10 | 0.30 | 0.31 | 0 | 0.002 | 0.01 | 0.03 | 0.03 | 0 | |
| Girls | 0.03 | 0.11 | 0.22 | 0.11 | 0 | 0.003 | 0.01 | 0.022 | 0.01 | ||
| BBP | All | 0.00002 | 0.0001 | 0.0003 | 0.0005 | 0 | 0.00002 | 0.0002 | 0.0004 | 0.0007 | 0 |
| ≥6~<12 | 0.0002 | 0.0001 | 0.0004 | 0.0005 | 0 | 0.00002 | 0.0002 | 0.0006 | 0.0007 | 0 | |
| ≥12~≤18 | 0.00001 | 0.0001 | 0.0003 | 0.0003 | 0 | 0.00002 | 0.0002 | 0.0005 | 0.0005 | 0 | |
| Boys | 0.00002 | 0.0001 | 0.0002 | 0.0002 | 0 | 0.00004 | 0.0002 | 0.0003 | 0.0003 | 0 | |
| Girls | 0.00001 | 0.0002 | 0.0004 | 0.0005 | 0 | 0.00002 | 0.0002 | 0.0006 | 0.0007 | 0 | |
| DEHP | All | 0.01 | 0.10 | 0.31 | 0.40 | 0 | 0.02 | 0.11 | 0.50 | 0.60 | 0 |
| ≥6~<12 | 0.01 | 0.10 | 0.33 | 0.36 | 0 | 0.02 | 0.14 | 0.55 | 0.60 | 0 | |
| ≥12~≤18 | 0.01 | 0.11 | 0.30 | 0.32 | 0 | 0.016 | 0.11 | 0.51 | 0.54 | 0 | |
| Boys | 0.01 | 0.10 | 0.35 | 0.40 | 0 | 0.02 | 0.13 | 0.58 | 0.60 | 0 | |
| Girls | 0.01 | 0.11 | 0.22 | 0.23 | 0 | 0.015 | 0.11 | 0.37 | 0.40 | 0 | |
| HI | All | 0.03 | 0.20 | 0.56 | 0.70 | 0 | 0.02 | 0.13 | 0.52 | 0.62 | 0 |
| ≥6~<12 | 0.03 | 0.20 | 0.54 | 0.60 | 0 | 0.03 | 0.14 | 0.57 | 0.62 | 0 | |
| ≥12~≤18 | 0.03 | 0.20 | 0.57 | 0.63 | 0 | 0.20 | 0.12 | 0.50 | 0.55 | 0 | |
| Boys | 0.03 | 0.20 | 0.65 | 0.70 | 0 | 0.03 | 0.13 | 0.60 | 0.62 | 0 | |
| Girls | 0.04 | 0.20 | 0.44 | 0.35 | 0 | 0.02 | 0.12 | 0.40 | 0.40 | 0 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zare Jeddi, M.; Eshaghi Gorji, M.; Rietjens, I.M.C.M.; Louisse, J.; Bruinen de Bruin, Y.; Liska, R. Biomonitoring and Subsequent Risk Assessment of Combined Exposure to Phthalates in Iranian Children and Adolescents. Int. J. Environ. Res. Public Health 2018, 15, 2336. https://doi.org/10.3390/ijerph15112336
Zare Jeddi M, Eshaghi Gorji M, Rietjens IMCM, Louisse J, Bruinen de Bruin Y, Liska R. Biomonitoring and Subsequent Risk Assessment of Combined Exposure to Phthalates in Iranian Children and Adolescents. International Journal of Environmental Research and Public Health. 2018; 15(11):2336. https://doi.org/10.3390/ijerph15112336
Chicago/Turabian StyleZare Jeddi, Maryam, Mohamad Eshaghi Gorji, Ivonne M. C. M. Rietjens, Jochem Louisse, Yuri Bruinen de Bruin, and Roman Liska. 2018. "Biomonitoring and Subsequent Risk Assessment of Combined Exposure to Phthalates in Iranian Children and Adolescents" International Journal of Environmental Research and Public Health 15, no. 11: 2336. https://doi.org/10.3390/ijerph15112336
APA StyleZare Jeddi, M., Eshaghi Gorji, M., Rietjens, I. M. C. M., Louisse, J., Bruinen de Bruin, Y., & Liska, R. (2018). Biomonitoring and Subsequent Risk Assessment of Combined Exposure to Phthalates in Iranian Children and Adolescents. International Journal of Environmental Research and Public Health, 15(11), 2336. https://doi.org/10.3390/ijerph15112336







