Prevalence and Risk Factors of Human Papillomavirus in Male Patients: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Method
2.1. Search Strategy
2.2. Inclusion/Exclusion Criteria
2.3. Variables and Data Collection
2.4. Selection of Studies, Critical Reading and Level of Evidence
2.5. Data Analysis
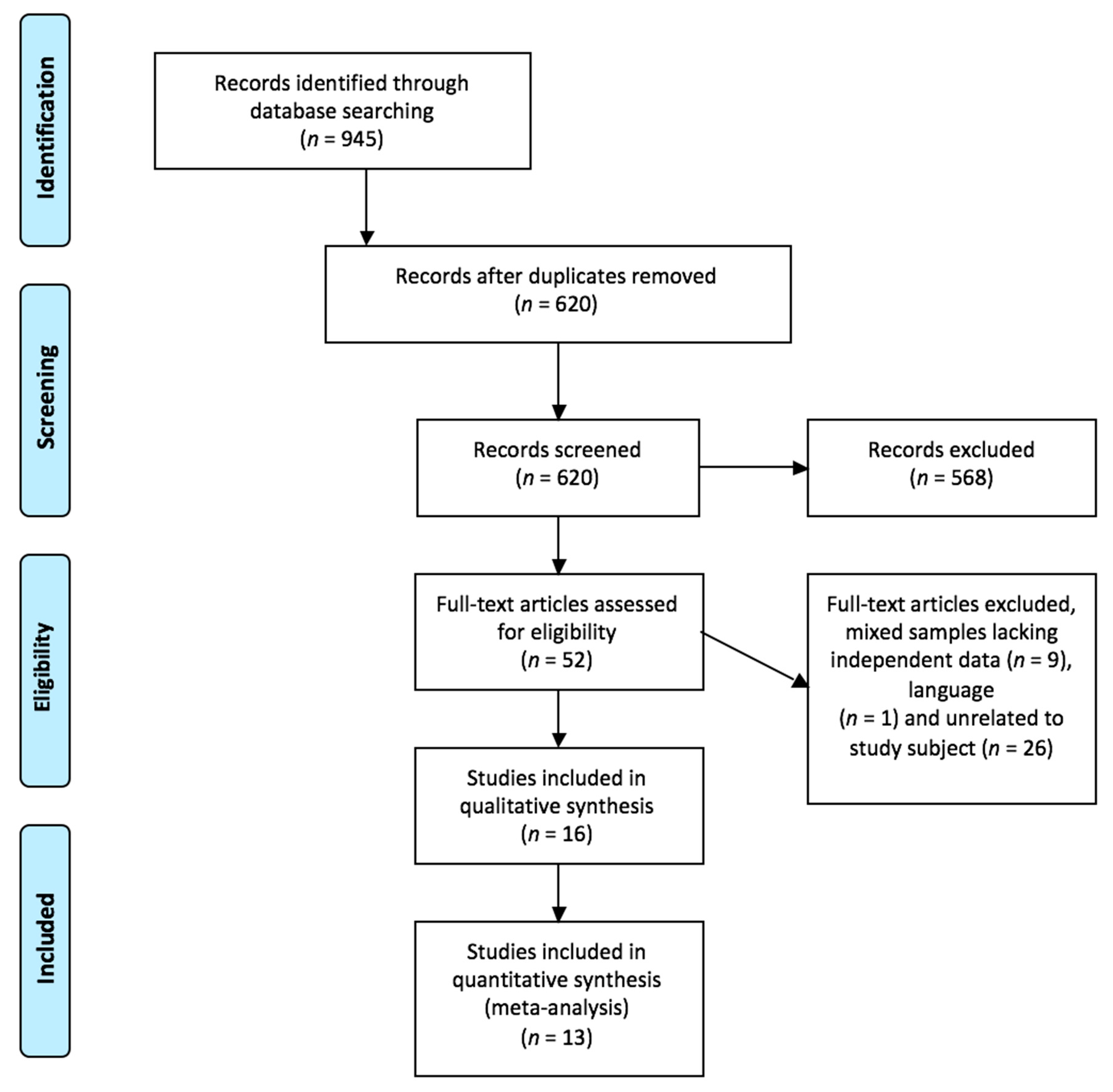
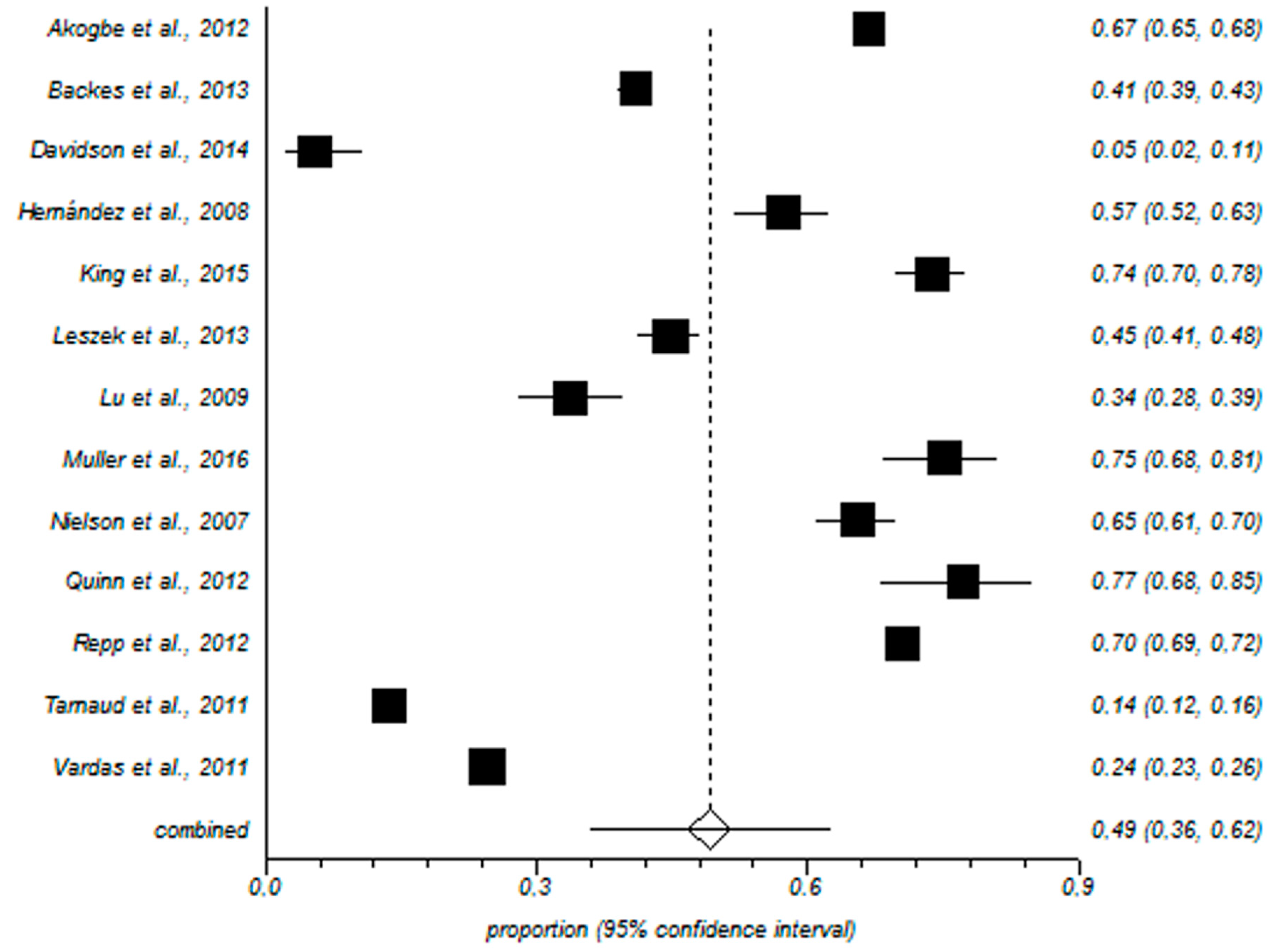
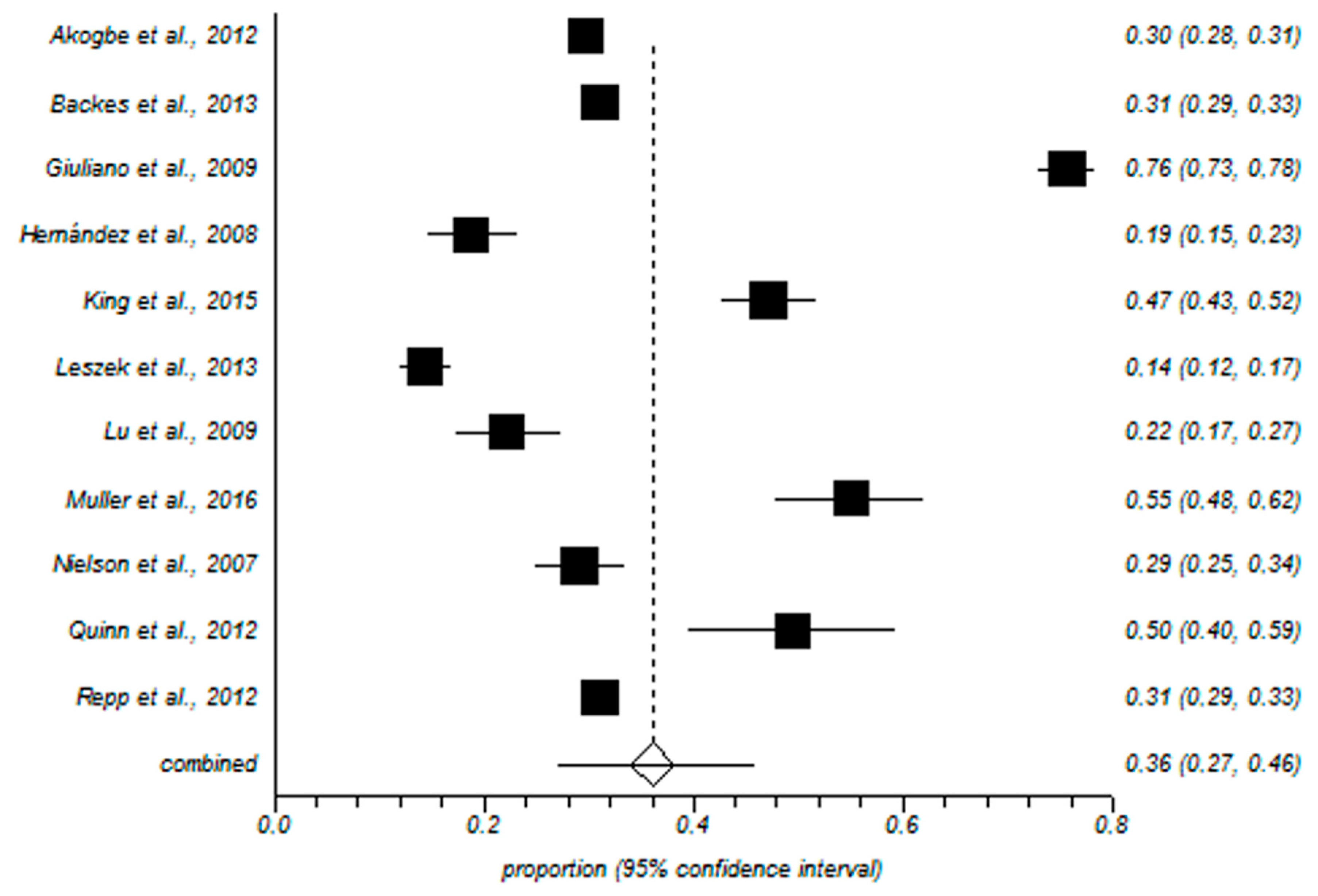
3. Results
3.1. Meta-Analysis
3.2. HPV and the Initiation of Sexual Relations or Promiscuity
3.3. Circumcision
3.4. Condom Use
3.5. Smoking
3.6. Vaccination
3.7. Miscellaneous
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflict of Interest
Authorship Declaration
References
- McQuillan, G.; Kruszon-Moran, D.; Markowitz, L.E.; Unger, E.R.; Paulose-Ram, R. Prevalence of HPV in adults aged 18–69: United States, 2011–2014. NCHS Data Brief. 2017, 280, 1–8. [Google Scholar]
- Schiffman, M.; Castle, P.E.; Jeronimo, J.; Rodriguez, A.C.; Wacholder, S. Human papillomavirus and cervical cancer. Lancet 2007, 370, 890–907. [Google Scholar] [CrossRef]
- Krashias, G.; Koptides, D.; Christodoulou, C. HPV prevalence and type distribution in Cypriot Women with cervical cytological abnormalities. BMC Infect. Dis. 2017, 17, 346. [Google Scholar] [CrossRef] [PubMed]
- Agsalda-Garcia, M.; Shieh, T.; Chuang, E.; Loi, N.; Milne, C.; Fang, R.; Lim, E.; Killeen, J.; Shiramizu, B. Human Papillomavirus-16 DNA Quantitation differentiates high-grade anal neoplasia. Int. J. Environ. Res. Public Health 2018, 15, 1690. [Google Scholar] [CrossRef] [PubMed]
- Nohata, N.; Abba, M.; Gutkind, J.S. Unraveling the oral cancer lncRNAome: Identification of novel lncRNAs associated with malignant progression and HPV infection. Oral Concol. 2016, 59, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Ghosn, M.; Kourie, H.R.; Abdayem, P.; Antoun, J.; Nasr, D. Anal cancer treatment: Current status and future perspective. World J. Gastroenterol. 2015, 21, 2294–2302. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.G.; Marquez-Domínguez, L.; Reyes-Leyva, J.; Vallejo-Ruiz, V. Aspectos generales de la estructura, la clasificación y la replicación del virus del papiloma humano. Rev. Med. Inst. Mex. Seguro Soc. 2015, 53, 166–171. [Google Scholar]
- Cardona-Arias, J.; Puerta, S.J.; Flórez, D.J. Prevalencia del virus papiloma humano y sus factores de riesgo en hombres: Revisión sistemática. Infectio 2011, 15, 268–276. [Google Scholar] [CrossRef]
- Husein-El, A.H.; Cañadas-De la Fuente, G.A. Squamous-cell carcinoma of the penis with human papillomavirus. N. Engl. J. Med. 2016, 374, 164. [Google Scholar] [CrossRef] [PubMed]
- Feixue, W.; Kai, Y.; Xin, W.; Lan, J.; Huang, S.; Sheng, W.; Zhao, J.; Su, Y.; Wang, Y.; Li, Y.; et al. Human papillomavirus prevalence and associated factors in Women and Men in South China: A population-based study. Emerg. Microbes Infect. 2016, 5, e119. [Google Scholar] [CrossRef] [PubMed]
- Goldstone, S.E.; Jessen, H.; Palefsky, J.M.; Giuliano, A.R.; Moreira, E.D.; Vardas, E.; Aranda, C.; Hillman, R.J.; Ferris, D.G.; Coutlee, F.; et al. Quadrivalent HPV vaccine efficacy against disease related to vaccine and non-vaccine HPV types in Males. Vaccine 2013, 31, 3849–3855. [Google Scholar] [CrossRef] [PubMed]
- Meites, E.; Gorbach, P.M.; Gratzer, B.; Panicker, G.; Steinau, M.; Collins, T. Monitoring for human papillomavirus vaccine impact among gay, bisexual, and other men who have sex with Men—United States, 2012–2014. J. Infect. Dis. 2016, 214, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- OCEBM Levels of Evidence Working Group. The Oxford 2011 Levels of Evidence. Oxford Centre for Evidence-Based Medicine. Available online: http://www.cebm.net/wp-content/uploads/2014/06/CEBM-Levels-of-Evidence-2.1.pdf (accessed on 1 November 2017).
- Akogbe, G.O.; Ajidahun, A.; Sirak, B.; Anic, G.M.; Papenfuss, M.R.; Fulp, W.J. Race and prevalence of human papillomavirus infection among Men residing in Brazil, Mexico, and the United States. Int. J. Cancer 2012, 131, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Auvert, B.; Sobngwi-Tambekou, J.; Cutler, E.; Nieuwoudt, M.; Lissouba, P.; Puren, A.; Taljaard, D. Effect of male circumcision on the prevalence of high-risk human papillomavirus in young Men: Results of a randomized controlled trial conducted in Orange Farm, South Africa. J. Infect. Dis. 2009, 199, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Backes, D.M.; Bleeker, M.C.G.; Meijer, C.J.L.M.; Hudgens, M.G.; Agot, K.; Bailey, R.C.; Ndinya-Achola, J.O.; Hayombe, J.; Hogewoning, C.J.; Moses, S.; et al. Male circumcision is associated with a lower prevalence of human papillomavirus-associated penile lesions among Kenyan Men. Int. J. Cancer 2012, 130, 1888–1897. [Google Scholar] [CrossRef] [PubMed]
- Davidson, C.L.; Richter, K.L.; Van der Linde, M.; Coetsee, J.; Boy, S.C. Prevalence of oral and oropharyngeal human papillomavirus in a sample of South African Men: A pilot study. S. Afr. Med. J. 2014, 104, 358–361. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.R.; Lazcano, E.; Villa, L.L.; Flores, R.; Salmeron, J.; Lee, J.H.; Papenfuss, M.; Abrahamsen, M.; Baggio, M.L.; Silva, R.; et al. Circumcision and sexual behavior: Factors independently associated with human papillomavirus (HPV) detection among Men in the HIM Study. Int. J. Cancer 2009, 124, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Hernández, B.Y.; Wilkens, L.R.; Zhu, X.; McDuffie, K.; Thompson, P.; Shvetsov, Y.B.; Ning, L.; Goodman, M.T. Circumcision and human papillomavirus infection in Men: A site-specific comparison. J. Infect. Dis. 2008, 197, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Ingles, D.J.; Pierce Campbell, C.M.; Messina, J.A.; Stoler, M.H.; Lin, H.Y.; Fulp, W.J.; Abrahamsen, M.; Sirak, B.A.; O´Keefe, M.T.; Papenfuss, M.; et al. Human Papillomavirus Virus (HPV) genotype and age-specific analyses of external genital lesions among Men in the HPV Infection in Men (HIM) Study. J. Infect. Dis. 2015, 211, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- King, E.M.; Gilson, R.; Beddows, S.; Soldan, K.; Panwar, K.; Young, C. Human papillomavirus DNA in men who have sex with Men: Type-specific prevalence, risk factors and implications for vaccination strategies. Br. J. Cancer 2015, 112, 1585–1593. [Google Scholar] [CrossRef] [PubMed]
- Leszek, W.; Slawomir, D.; Andrzej, M. Incidence and prevalence of multiple types of genital human papillomavirus (HPV) infection in Men: A study in Poland. Ginekol. Pol. 2013, 84, 112–115. [Google Scholar]
- Lu, W.Y.; Nielson, C.M.; Flores, R.; Abrahamsen, M.; Papenfuss, M.; Harris, R.B.; Giuliano, A.R. Factors associated with acquisition and clearance of human papillomavirus infection in a cohort of US Men: A prospective study. J. Infect. Dis. 2009, 199, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Müller, E.E.; Rebe, K.; Chirwa, T.F.; Struthers, H.; McIntyre, J.; Lewis, D.A. The prevalence of human papillomavirus infections and associated risk factors in men-who-have-sex-with-men in Cape Town, South Africa. Infect. Dis. 2016, 16, 440. [Google Scholar] [CrossRef] [PubMed]
- Nielson, C.M.; Harris, R.B.; Dunne, E.F.; Abrahamsen, M.; Papenfuss, M.R.; Flores, R. Risk factors for anogenital human papillomavirus infection in Men. J. Infect. Dis. 2007, 196, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Quinn, R.; Salvatierra, J.; Solari, V.; Calderon, M.; Ton, T.G.N.; Zunt, J.R. Human papillomavirus infection in men who have sex with Men in Lima, Peru. Aids 2012, 28, 1734–1738. [Google Scholar] [CrossRef] [PubMed]
- Repp, K.K.; Nielson, C.M.; Fu, R.; Schafer, S.; Lazcano-Ponce, E.; Salmerón, J.; Quiterio, M.; Villa, L.L.; Giuliano, A.R. Male human papillomavirus prevalence and association with condom use in Brazil, Mexico, and the United States. J. Infect. Dis. 2012, 205, 1287–1293. [Google Scholar] [CrossRef] [PubMed]
- Tarnaud, C.; Lissouba, P.; Cutler, E.; Puren, A.; Taljaard, D.; Auvert, B. Association of Low-Risk Human papillomavirus infection with Male circumcision in young Men: Results from a longitudinal study conducted in Orange Farm (South Africa). Infect. Dis. Obstet. Gynecol. 2011, 2011, 567408. [Google Scholar] [CrossRef] [PubMed]
- Vardas, E.; Giuliano, A.R.; Goldstone, S.; Palefsky, J.M.; Moreira, E.D.; Penny, M.E.; Aranda, C.; Jessen, H.; Moi, H.; Ferris, D.G.; et al. External genital human papillomavirus prevalence and associated factors among heterosexual Men on 5 continents. J. Infect. Dis. 2011, 203, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Dunne, E.F.; Nielson, C.M.; Stone, K.M.; Markowitz, L.E.; Giuliano, A.R. Prevalence of HPV infection among Men: A systematic review of the literature. J. Infect. Dis. 2006, 194, 1044–1057. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.P.H.; Wong, J.Y.H.; Lau, A.Y.Y.; Fong, D.Y. Gender and sexual orientation differences in human papillomavirus (HPV) vaccine uptake among Chinese young adults. Int. J. Environ. Res. Public Health 2018, 15, 1099. [Google Scholar] [CrossRef] [PubMed]
- Nielson, C.M.; Harris, R.B.; Dunne, E.F.; Abrahamsen, M.; Papenfuss, M.R.; Dune, E.F.; Markowitz, L.E.; Giuliano, A.R. Multiple-Type human papillomavirus infection in male anogenital sites: Prevalence and associated factors. Cancer Epidemiol. Biomark. Prev. 2009, 18, 1077–1083. [Google Scholar] [CrossRef] [PubMed]
- Aghakhani, A.; Mamishi, S.; Sabeti, S.; Bidari-Zerehpoosh, F.; Banifazl, M.; Bavand, A.; Ramezani, A. Gender and age-specific seroprevalence of human papillomavirus 16 and 18 in general population in Tehran, Iran. Med. Microbiol. Immunol. 2017, 206, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Mercado Gutiérrez, M.R.; Cuns, C.A.; Gómez Dorronsoro, M.L.; Paniello Alastruey, I.; Mallor Giménez, F.; Lozano Escario, F.; Santamaría, M. Influence of age in the prevalence of high risk human papiloma virus in Women with pre-neoplasic cervical lesions in Navarra, Spain. Rev. Esp. Salud. Pública 2017, 91, e1–e8. [Google Scholar]
- Rositch, A.F.; Hudgens, M.G.; Backes, D.M.; Moses, S.; Agot, K.; Nyagaya, E.; Snijders, P.J.; Meijer, C.J.; Bailey, R.C.; Smith, J.S. Vaccine-relevant human papillomavirus (HPV) infections and future acquisition of high-risk HPV types in Men. J. Infect. Dis. 2012, 206, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Olesen, T.B.; Mwaiselage, J.; Iftner, T.; Kahesa, C.; Rasch, V.; Frederiksen, K.; Munk, C.; Kjaer, S.K. Risk factors for genital human papillomavirus among Men in Tanzania. J. Med. Virol. 2017, 89, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Gray, R.H.; Serwadda, D.; Kong, X.; Makumbi, F.; Kigozi, G.; Gravitt, P.E.; Watya, S.; Nalugoda, F.; Ssempijja, V.; Tobian, A.A.R.; et al. Male circumcision decreases acquisition and increases clearance of high risk human papillomavirus in HIV-negative Men: A randomized trial in Rakai, Uganda. J. Infect. Dis. 2010, 201, 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.; Monsour, E.; Klausner, J.D.; Galea, J.T. Sociodemographic and behavioral correlates of anogenital warts and human papilloma virus-related knowledge among Men who have sex with Men and trans Women in Lima, Peru. Sex. Transm. Dis. 2015, 42, 198–201. [Google Scholar] [CrossRef] [PubMed]
- Pierce Campbell, C.M.; Lin, H.Y.; Fulp, W.; Papenfuss, M.R.; Salmerón, J.J.; Quiterio, M.M.; Lazcano-Ponce, E.; Villa, L.L.; Giuliano, A.R. Consistent condom use reduces the genital human papillomavirus burden among high risk Men: The HPV Infection in Men Study. J. Infect. Dis. 2013, 208, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Rocha Rodríguez, R.; Juárez Juárez, M.E.; Ruiz Jiménez, M.M.; Ramírez Banda, X.G.; Gaytán Sanchéz, M.R.; Contreras Valero, P. Identification of risk factors to acquire the human papilloma virus in sexual-server. Rev. Cuba Obstet. Ginecol. 2012, 38, 244–255. [Google Scholar]
- Oyediran, K.A.; Feyisetan, O.I.; Akpan, T. Predictors of condom-use among young never married males in Nigeria. J. Health Popul. Nutr. 2011, 29, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Williams, W.W.; Li, J.; Dorell, C.; Yankey, D.; Kepka, D.; Dunne, E.F. Human papillomavirus (HPV) vaccine initiation and HPV and vaccine awareness among men 18–26 years in the United States, 2010. Am. J. Prev. Med. 2013, 44, 330–388. [Google Scholar] [CrossRef] [PubMed]
| Author, Year (Country of Study) [Reference] | Sample | Design | HPV Risk Factors | EL | RG |
|---|---|---|---|---|---|
| Akogbe et al., 2013 (Brazil, Mexico and USA) [15]. | n = 3909 | Cohort study | Asian/Pacific Islanders had the lowest prevalence of all types of HPV (42.3%) compared to black (66.2%), Mexican (62.3), other (67.3%) and white races (71.5%). Asian/Pacific Islanders had the lowest prevalence of oncogenic HPV (18.9%), compared to black (32.2%), whites (31.4%), Mexican (27.5%) and other (27.1%). | 2b | B |
| Auvert et al., 2009 (South Africa) [16]. | n = 1264 | Clinical trial | Male circumcision protects against high risk-HPV (23.2% prevalence in uncircumcised men against 14% in circumcised men). | 2b | B |
| Backes DM et al., 2012 (Kenya) [17]. | n = 2509 | Clinical trial | Male circumcision was strongly associated with low probability of flat penile lesions (OR = 0.02) and with higher probability of popular lesions (OR = 3.0). Men with flat penile lesions have higher prevalence of the five most common HPV types comparing to men without flat penile lesions. | 1a | A |
| Davidson et al., 2014 (South Africa) [18]. | n = 128 | Cross-sectional study | More sexual partners increase prevalence of oral and oropharyngeal HPV prevalence. No statistically significant differences in HPV prevalence were found regarding age, smoking, alcohol consumption, oral sex, HIV and clinical lesions. | 2c | B |
| Giuliano et al., 2009 (Brazil, México and USA) [19]. | n = 998 | Cohort study | Statistically significant associations (p < 0.05): Between 30–34 years, the probability of suffering oncogenic HPV (OR = 1.99) and any type of HPV (OR = 1.81) is higher. Reduced risk of any type of HPV infection in Asians/Pacific Islands (OR = 0.18) and in mixed races (OR = 0.74). Reduced risk of oncogenic HPV in Asians/Pacific Islands (OR = 0.32). Increased risk of oncogenic HPV (OR = 2.18) and of any type of HPV (OR = 2.12) in Brazil. Higher risk of oncogenic HPV (OR = 2.27) and of any type of HPV (OR = 2.63) in divorced/separated. Lower risk of oncogenic HPV (OR = 0.70) in non-smokers. Increased risk of oncogenic HPV (OR = 9.01) and of any type of HPV (OR = 8.15) is related to a greater number of couples. Lower risk of oncogenic HPV (OR = 0.30) and of any type of HPV (OR = 0.44) if they have not had sex in the last 3 months and Increased risk of oncogenic HPV (OR = 3.43) and of any type of HPV (OR = 3.05) is related to a greater number of couples in the last 3 months. Greater risk of oncogenic HPV (OR = 1.45) and of any type of HPV (OR = 1.40) if they have had anal sex. Lower risk of oncogenic HPV (OR = 0.47) and of any type of HPV (OR = 0.52) if they have never been diagnosed with sexually transmitted disease. Increased risk of any type of HPV (OR = 1.57) if they have genital herpes. Increased risk of oncogenic HPV (OR = 2.27) and for any type of HPV (OR = 2.19) if they have had partners with genital warts. | 2b | B |
| Hernández et al., 2008 (USA) [20]. | n = 351 | Cohort study | Smoking, sex with men, lifetime number of female sex partners, history of genital warts and circumcision are associated with HPV. History of sex with men was inversely associated with HPV infection of the shaft (OR = 0.44). Lifetime number of female sex partners was positively associated with HPV infection of the shaft and scrotum (OR = 6.93). Uncircumcised men have higher prevalence rate of HPV of the glans (OR = 1.96) and oncogenic infection (OR = 2.51). | 2b | B |
| Ingles et al., 2015 (USA, Brazil, México) [21]. | n = 2754 | Cohort study | High-risk type HPV is more prevalent in penile intraepithelial neoplasia (85.7%) and other genital external lesions (21%) than in condiloma (8.2%). | 2b | B |
| King et al., 2015 (England) [22]. | n = 511 | Cross-sectional study | Higher HPV prevalence rate (92.6%) in HIV-positive men who have sex with men compare with HIV-negative men who have sex with men (71.1%). There is a 4.7% increase in the odd of HPV infection per year when modeling age as a continuous variable (18–40 years). When treating age as a categorical variable there are no significant differences in HPV infection. | 2c | B |
| Leszek et al., 2013 (Poland) [23]. | n = 820 | Cross-sectional study | Higher risk of HVP infection with more than three sexual partners in the last 12 months (OR = 1.44). HPV infection is greater in men younger than 34 years (OR = 1.08). Higher HPV infection in men who do not use condoms (OR = 1.86). | 2c | B |
| Lu et al., 2009 (USA) [24]. | n = 377 | Cohort study | There is a reduced risk of HPV infection with having a college education or higher (Hazard rate = 0.3) and with older age at first sexual intercourse (Hazard rate = 0.9). There is an increased risk of HPV infection in smokers (Hazard rate = 2.4). | 2b | B |
| Müller et al., 2016 (South Africa) [25]. | n = 200 | Cross-sectional study | Men with HIV have higher prevalence rate of anal and oropharyngeal HPV. Men who have sex with men are more likely to have anal HPV than men who have sex with woman (85% vs. 26.8%). HPV infection is significantly associated with receiving an income, having sex with men only, engaging in group sex in their lifetime, being HIV positive and practicing receptive anal sex. | 2c | B |
| Nielson et al., 2007 (USA) [26]. | n = 463 | Cross-sectional study | Sociodemographic factors are not related to HPV infection. Smoking is associated with HPV (OR = 1.8). Smoking 10 or more cigarettes per day is associated with HPV (OR = 3.0). Using condoms at least half time reduce HPV (OR = 0.5). Sexual factors such as high number of lifetime female sex partners, female partners on the past 3 months, and an increased frequency of intercourse in the past month and 3 months are significantly related to HPV. Current presence of genital warts (OR = 4.5) and having a female parte with abnormal pap smear (OR = 2.2) are related with HPV. | 2c | B |
| Quinn et al., 2012 (Peru) [27]. | n = 105 | Cross-sectional study | Being primary receptive as sex role is related with a higher prevalence of HPV, 56%, vs. 4% in insertive. Having exclusively men as sexual partners is related with higher HPV prevalence (92%) vs. 17.2% when having men and women. High risk HPV is related to not having a stable relationship, have sex work as primary income and having a greater number of life partners. | 2c | B |
| Repp et al., 2012 (Brazil, Mexico, USA) [28]. | n = 2621 | Cross-sectional study | There are statistically different prevalence rates of any HPV for men that always use condom (65.6%) and men do not always use it (71.9%). There are statistically different prevalence rates of any oncogenic HPV for men that always use condom (29.6%) and men do not always use it (35.4%). | 2c | B |
| Tarnaud et al., 2011 (South Africa) [29]. | n = 3274 | Cohort study | Low risk HPV genotypes significantly increase with the number of lifetime sexual partners. HIV and Herpes Virus are associated with an increase in low risk HPV genotypes. Number of low risk HPV genotypes decrease in participants with primary education and those that use condom. | 2b | B |
| Vardas et al., 2011 (5 continents) [30]. | n = 3463 | Cross-sectional study | Less condom use is associated with an increased risk of HPV (OR = 1.7). More than 3 lifetime female sexual partners increase prevalence HPV DNA detection (OR = 4.5). Africa has the highest risk of HPV (OR = 3.7). | 1a | A |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Álvarez, M.I.; Gómez-Urquiza, J.L.; Husein-El Ahmed, H.; Albendín-García, L.; Gómez-Salgado, J.; Cañadas-De la Fuente, G.A. Prevalence and Risk Factors of Human Papillomavirus in Male Patients: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2018, 15, 2210. https://doi.org/10.3390/ijerph15102210
Rodríguez-Álvarez MI, Gómez-Urquiza JL, Husein-El Ahmed H, Albendín-García L, Gómez-Salgado J, Cañadas-De la Fuente GA. Prevalence and Risk Factors of Human Papillomavirus in Male Patients: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2018; 15(10):2210. https://doi.org/10.3390/ijerph15102210
Chicago/Turabian StyleRodríguez-Álvarez, María Inmaculada, Jose L. Gómez-Urquiza, Husein Husein-El Ahmed, Luis Albendín-García, Juan Gómez-Salgado, and Guillermo A. Cañadas-De la Fuente. 2018. "Prevalence and Risk Factors of Human Papillomavirus in Male Patients: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 15, no. 10: 2210. https://doi.org/10.3390/ijerph15102210
APA StyleRodríguez-Álvarez, M. I., Gómez-Urquiza, J. L., Husein-El Ahmed, H., Albendín-García, L., Gómez-Salgado, J., & Cañadas-De la Fuente, G. A. (2018). Prevalence and Risk Factors of Human Papillomavirus in Male Patients: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 15(10), 2210. https://doi.org/10.3390/ijerph15102210









