Evaluation and Implementation of a Proactive Telephone Smoking Cessation Counseling for Parents: A Study Protocol of an Effectiveness Implementation Hybrid Design
Abstract
1. Introduction
2. Materials and Methods
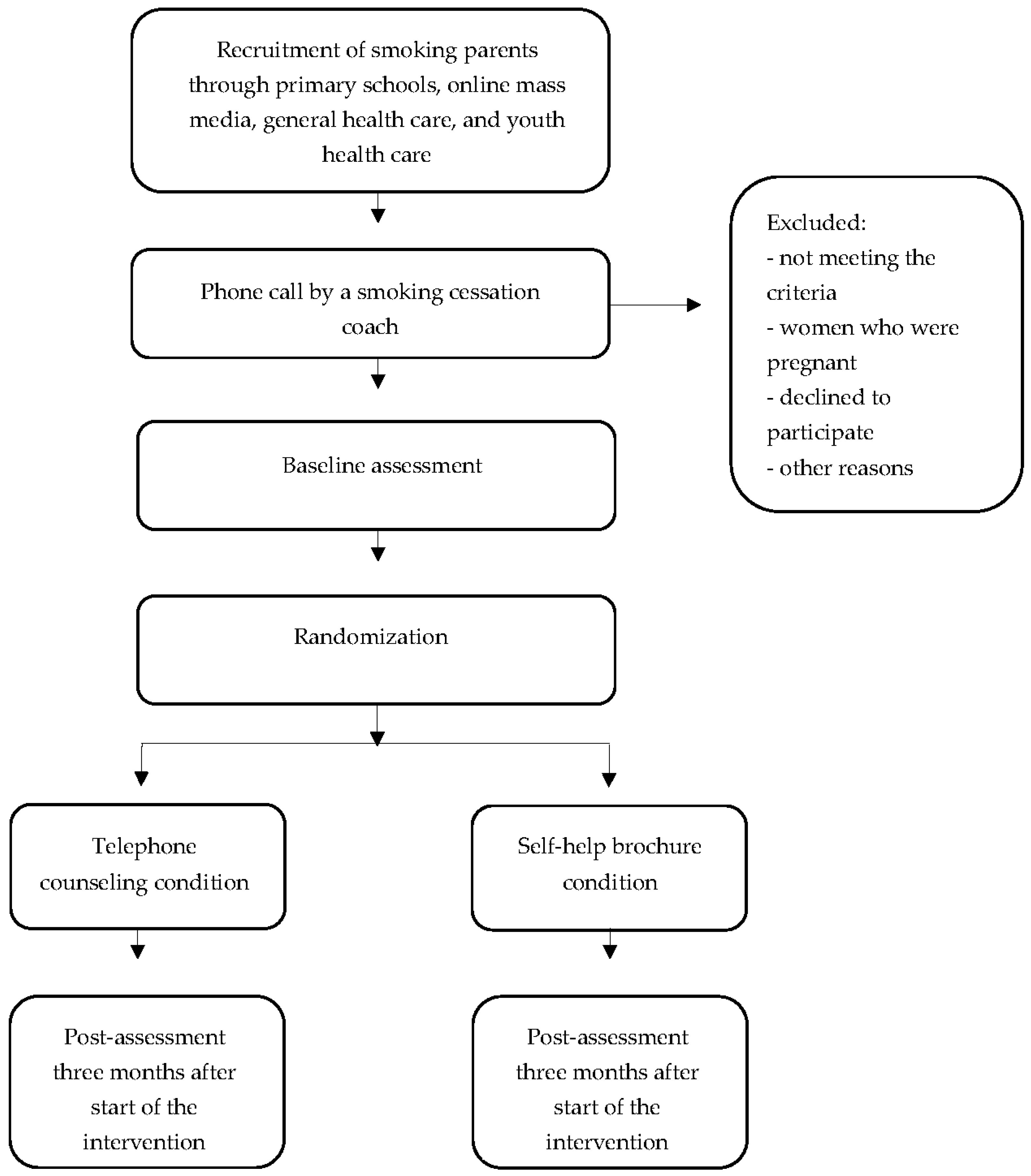
2.1. Study Design
2.2. Participants and Recruitment
2.2.1. Mass Media Approach
Primary Schools
Online Mass Media
2.2.2. Health Care Approach
2.3. Inclusion Criteria
2.4. Study Conditions
2.4.1. Telephone Counseling Condition
2.4.2. Control Condition
2.5. Data Collection
2.6. Randomization
2.7. Sample Size
2.8. Outcomes
2.9. Economic Evaluation
2.10. Statistical Analyses
2.11. Process Evaluation
3. Discussion
Strengths and Limitations
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. WHO Report on the Global Tobacco Epidemic, 2013: Enforcing Bans on Tobacco Advertising, Promotion and Sponsorship; World Health Organization: Geneva, Switzerland, 2013; ISBN 9789240691605. [Google Scholar]
- Van Laar, M.W.; van Ooyen-Houben, M.M.J.; Cruts, A.A.N.; Meijer, R.F.; Croes, E.A.; Ketelaars, A.P.M.; van der Pol, P.M. Nationale Drug Monitor: Jaarbericht 2016; Trimbos-Instituut: Utrecht, The Netherlands, 2016; ISBN 9789052537658. [Google Scholar]
- Ter Weijde, W.; Croes, E.; Verdurmen, J.; Monshouwer, K. Factsheet Meeroken; Trimbos-Instituut: Utrecht, The Netherlands, 2015. [Google Scholar]
- Difranza, J.R.; Aligne, C.A.; Weitzman, M. Prenatal and Postnatal Environmental Tobacco Smoke Exposure and Children’s Health. Pediatrics 2004, 113, 1007–1015. [Google Scholar] [CrossRef] [PubMed]
- Hofhuis, W.; De Jongste, J.C.; Merkus, M. Adverse Health Effects of Prenatal and Postnatal Tobacco Smoke Exposure on Children. Arch. Dis. Child. 2003, 88, 1086–1090. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.K.; Hermansson, G.; Ludvigsson, J. How Should Parents Protect Their Children from Environmental Tobacco-Smoke Exposure in the Home? Pediatrics 2004, 113, e291–e295. [Google Scholar] [CrossRef] [PubMed]
- Bricker, J.B.; Leroux, B.G.; Peterson, A.V.; Kealey, K.A.; Sarason, I.G.; Andersen, M.R.; Marek, P.M. Nine-year Prospective Relationship Between Parental Smoking Cessation and Children’s Daily Smoking. Addiction 2003, 98, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Otten, R.; Engels, R.C.M.E.; Van De Ven, M.O.M.; Bricker, J.B. Parental Smoking and Adolescent Smoking Stages: The Role of Parents’ Current and Former Smoking, and Family Structure. J. Behav. Med. 2007, 30, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Jha, P.; Ramasundarahettige, C.; Landsman, V.; Rostron, B.; Thun, M.; Anderson, R.N.; McAfee, T.; Peto, R. 21st-Century Hazards of Smoking and Benefits of Cessation in the United States. N. Engl. J. Med. 2013, 368, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Hymowitz, N.; Schwab, J.; Haddock, C.K.; Pyle, S.; Moore, G.; Meshberg, S. The Pediatric Resident Training on Tobacco Project: Baseline Findings from the Parent/Guardian Tobacco Survey. Prev. Med. 2005, 41, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Winickoff, J.P.; Tanski, S.E.; McMillen, R.C.; Hipple, B.J.; Friebely, J.; Healey, E.A. A National Survey of the Acceptability of Quitlines to Help Parents Quit Smoking. Pediatrics 2006, 117, e695–e700. [Google Scholar] [CrossRef] [PubMed]
- Stead, L.F.; Perera, R.; Lancaster, T. Telephone Counselling for Smoking Cessation. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Hollis, J.F.; McAfee, T.A.; Fellows, J.L.; Zbikowski, S.M.; Stark, M.; Riedlinger, K. The Effectiveness and Cost Effectiveness of Telephone Counselling and the Nicotine Patch in a State Tobacco Quitline. Tob. Control 2007, 16, i53–i59. [Google Scholar] [CrossRef] [PubMed]
- Schuck, K.; Bricker, J.B.; Otten, R.; Kleinjan, M.; Brandon, T.H.; Engels, R.C.M.E. Effectiveness of Proactive Quitline Counselling for Smoking Parents Recruited through Primary Schools: Results of a Randomized Controlled Trial. Addiction 2014, 109, 830–841. [Google Scholar] [CrossRef] [PubMed]
- Flay, B.R. Efficacy and Effectiveness Trials (and other Phases of Research) in the Development of Health Promotion Programs. Prev. Med. 1986, 15, 451–474. [Google Scholar] [CrossRef]
- McClure, J.B.; Greene, S.M.; Wiese, C.; Johnson, K.E.; Alexander, G.; Strecher, V. Interest in an Online Smoking Cessation Program and Effective Recruitment Strategies: Results from Project Quit. J. Med. Internet Res. 2006, 8, e14. [Google Scholar] [CrossRef] [PubMed]
- McDonald, P.W. Population-based Recruitment for Quit-smoking Programs: An Analytic Review of Communication Variables. Prev. Med. 1999, 28, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Prochaska, J.O.; Velicer, W.F.; Fava, J.L.; Rossi, J.S.; Tsoh, J.Y. Evaluating a Population-based Recruitment Approach and a Stage-based Expert System Intervention for Smoking Cessation. Addict. Behav. 2001, 26, 583–602. [Google Scholar] [CrossRef]
- Velicer, W.F.; Prochaska, J.O.; Fava, J.L.; Laforge, R.G.; Rossi, J.S. Interactive versus Noninteractive Interventions and Dose–Response Relationships for Stage-Matched Smoking Cessation Programs in a Managed Care Setting. Health Psychol. 1999, 18, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.R.; Marcy, T.W.; Naud, S. Interest in Treatments to Stop Smoking. J. Subst. Abuse Treat. 2009, 36, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Marcano Belisario, J.S.; Bruggeling, M.N.; Gunn, L.H.; Brusamento, S.; Car, J. Interventions for Recruiting Smokers into Cessation Programmes. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef] [PubMed]
- Schuck, K.; Otten, R.; Kleinjan, M.; Bricker, J.B.; Engels, R.C. School-Based Promotion of Cessation Support: Reach of Proactive Mailings and Acceptability of Treatment in Smoking Parents Recruited into Cessation Support through Primary Schools. BMC Public Health 2013, 13, 381. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.S.M.; Mak, Y.W.; Loke, A.Y.; Lam, T.-H. Smoking Cessation Intervention in Parents of Young Children: A Randomised Controlled Trial. Addiction 2005, 100, 1731–1740. [Google Scholar] [CrossRef] [PubMed]
- Lando, H.A.; Hellerstedt, W.L.; Pirie, P.L.; McGovern, P.G. Brief Supportive Telephone Outreach as a Recruitment and Intervention Strategy for Smoking Cessation. Am. J. Public Health 1992, 82, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Tzelepis, F.; Paul, C.L.; Walsh, R.A.; Wiggers, J.; Knight, J.; Lecathelinais, C.; Daly, J.; Neil, A.; Girgis, A. Telephone Recruitment into a Randomized Controlled Trial of Quitline Support. Am. J. Prev. Med. 2009, 37, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Van der Putten, N. Publieksinformatie: Jaarbericht 2016. Available online: https://assets.trimbos.nl/docs/c8228127-2349-4bc9-b349-521bb3f912f1.pdf (accessed on 28 September 2017).
- Graham, A.L.; Milner, P.; Saul, J.E.; Pfaff, L. Correction: Online Advertising as a Public Health and Recruitment Tool: Comparison of Different Media Campaigns to Increase Demand for Smoking Cessation Interventions. J. Med. Internet Res. 2009, 11, e2. [Google Scholar] [CrossRef]
- Heffner, J.L.; Wyszynski, C.M.; Comstock, B.; Mercer, L.D.; Bricker, J. Overcoming Recruitment Challenges of Web-Based Interventions for Tobacco Use: The Case of Web-Based Acceptance and Commitment Therapy for Smoking Cessation. Addict. Behav. 2013, 38, 2473–2476. [Google Scholar] [CrossRef] [PubMed]
- Smit, E.S.; Hoving, C.; Cox, V.C.M.; De Vries, H. Influence of Recruitment Strategy on the Reach and Effect of a Web-Based Multiple Tailored Smoking Cessation Intervention among Dutch Adult Smokers. Health Educ. Res. 2012, 27, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Hipple, B.; Nabi-Burza, E.; Hall, N.; Regan, S.; Winickoff, J.P. Distance-Based Training in Two Community Health Centers to Address Tobacco Smoke Exposure of Children. BMC Pediatr. 2013, 13, 56. [Google Scholar] [CrossRef] [PubMed]
- Solberg, L.I.; Boyle, R.G.; Davidson, G.; Magnan, S.J.; Carlson, C.L. Patient Satisfaction and Discussion of Smoking Cessation during Clinical Visits. Mayo Clin. Proc. 2001, 76, 138–143. [Google Scholar] [CrossRef]
- Winickoff, J.P.; Nabi-Burza, E.; Chang, Y.; Regan, S.; Drehmer, J.; Finch, S.; Wasserman, R.; Ossip, D.; Hipple, B.; Woo, H.; et al. Sustainability of a Parental Tobacco Control Intervention in Pediatric Practice. Pediatrics 2014, 134, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Winickoff, J.P.; Hibberd, P.L.; Case, B.; Sinha, P.; Rigotti, N.A. Child Hospitalization An Opportunity for Parental Smoking Intervention. Am. J. Prev. Med. 2001, 21, 218–220. [Google Scholar] [CrossRef]
- Winickoff, J.P.; Hillist, V.J.; Palfrey, J.S.; Perrin, J.M.; Rigotti, N.A. A Smoking Cessation Intervention for Parents of Children Who Are Hospitalized for Respiratory Illness: The Stop Tobacco Outreach Program. Pediatrics 2003, 111, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Nederlands Centrum Jeugdgezondheid (NCJ). Landelijk Professioneel Kader: Uitvoering Basispakket Jeugdgezondheidszorg (JGZ). Available online: https://assets.ncj.nl/docs/9c8aba38-2e8d-4fef-a346-ab7dab7f8bc3.pdf (accessed on 28 July 2017).
- Nederlands Centrum Jeugdgezondheid (NCJ). Leeftijdspecifieke Preventie. Available online: www.ncj.nl (accessed on 28 July 2017).
- Cluss, P.A.; Moss, D. Parent Attitudes about Pediatricians Addressing Parental Smoking. Ambul. Pediatr. 2002, 2, 485–488. [Google Scholar] [CrossRef]
- Frankowski, B.L.; Weaver, S.O.; Secker-Walker, R.H. Advising Parents to Stop Smoking: Pediatricians’ and Parents’ Attitudes. Pediatrics 1993, 91, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, S.G.; Kuijlaars, J.S.; Mesters, I.; Muris, J.W.M.; van Schayck, C.P.; Dompeling, E.; Feron, F.J.M. Addressing Passive Smoking in Children. PLoS ONE 2014, 9, e93220. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Stable, E.J.; Juarez-Reyes, M.; Kaplan, C.P.; Fuentes-Afflick, E.; Gildengorin, V.; Millstein, S.G. Counseling Smoking Parents of Young Children. Arch. Pediatr. Adolesc. Med. 2001, 155, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Bernet, A.C.; Willens, D.E.; Bauer, M.S. Effectiveness-Implementation Hybrid Designs: Implications for Quality Improvement Science. Implement. Sci. 2013, 8, S2. [Google Scholar] [CrossRef]
- Curran, G.M.; Bauer, M.; Mittman, B.; Pyne, J.M.; Stetler, C. Effectiveness-Implementation Hybrid Design: Combining Elements of Clinical Effectiveness and Implementation Research to Enhance Public Health. Med. Care 2012, 50, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Rollnick, S.; Miller, W.R. What is Motivational Interviewing? Behav. Cogn. Psychother. 1995, 23, 325–344. [Google Scholar] [CrossRef]
- Rollnick, S.; Butler, C.C.; Kinnersley, P.; Gregory, J.; Mash, B. Motivational Interviewing. BMJ 2010, 340, 1242–1245. [Google Scholar] [CrossRef] [PubMed]
- Heckman, C.J.; Egleston, B.L.; Hofmann, M.T. Efficacy of Motivational Interviewing for Smoking Cessation. Tob. Control 2011, 19, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Hettema, J.E.; Hendricks, P.S. Motivational Interviewing for Smoking Cessation: A Meta-Analytic Review. J. Consult. Clin. Psychol. 2010, 78, 868–884. [Google Scholar] [CrossRef] [PubMed]
- West, R. Assessing Smoking Cessation Performance in NHS Stop Smoking Services: The Russell Standard (Clinical). Available online: http://www.ncsct.co.uk/usr/pub/assessing-smoking-cessation-performance-in-nhs-stop-smoking-services-the-russell-standard-clinical.pdf (accessed on 1 July 2017).
- Husereau, D.; Drummond, M.; Petrou, S.; Carswell, C.; Moher, D.; Greenberg, D.; Augustovski, F.; Briggs, A.H.; Mauskopf, J.; Loder, E. Consolidated Health Economic Evaluation Reporting Standards (CHEERS) Statement. Eur. J. Health Econ. 2013, 14, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Hakkaart-van Roijen, L.; van der Linden, N.; Bouwmans, C.; Kanters, T.; Swan Tan, S. Kostenhandleiding: Methodologie van Kostenonderzoek en Referentieprijzen voor Economische Evaluaties in de Gezondheidszorg; Erasmus University Rotterdam: Rotterdam, The Netherlands, 2016. [Google Scholar]
- Bouwmans, C.; De Jong, K.; Timman, R.; Zijlstra-Vlasveld, M.; Van der Feltz-Cornelis, C.; Tan, S.S.; Hakkaart-van Roijen, L. Feasibility, Reliability and Validity of a Questionnaire on Healthcare Consumption and Productivity Loss in Patients with a Psychiatric Disorder (TiC-P). BMC Health Serv. Res. 2013, 13, 217. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, G.R.A.; Becker, E.S.; Smit, F.; Rinck, M.; Spijker, J. Investigating the (Cost-) Effectiveness of Attention Bias Modification (ABM) for Outpatients with Major Depressive Disorder (MDD): A Randomized Controlled Trial Protocol. BMC Psychiatry 2016, 16, 370. [Google Scholar] [CrossRef] [PubMed]
- Hoch, J.S.; Rockx, M.A.; Krahn, A.D. Using the Net Benefit Regression Framework to Construct Cost-Effectiveness Acceptability Curves: An Example Using Data from a Trial of External Loop Recorders versus Holter Monitoring for Ambulatory Monitoring of “Community Acquired” Syncope. BMC Health Serv. Res. 2006, 6, 68. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Mohler, D. CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomised Trials. Trials 2010, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Steckler, A.B.; Linnan, L. (Eds.) Process Evaluation for Public Health Interventions and Research: An Overview. In Process Evaluation for Public Health Interventions and Research; Jossey-Bass: San Francisco, CA, USA, 2002; pp. 1–23. ISBN 9780787959760. [Google Scholar]
- Johnson, R.B.; Onwuegbuzie, A. Mixed Methods Research: A Research Paradigm Whose Time Has Come. Educ. Res. 2004, 33, 14–26. [Google Scholar] [CrossRef]
- Fendrich, M.; MacKesy-Amiti, M.E.; Johnson, T.P.; Hubbell, A.; Wislar, J.S. Tobacco-Reporting Validity in an Epidemiological Drug-Use Survey. Addict. Behav. 2005, 30, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Hayes, K.; Jackson, C.; Dickinson, D.; Miller, A.L. Providing Antismoking Socialization to Children after Quitting Smoking: Does It Help Parents Stay Quit? Am. J. Health Promot. 2017. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scheffers-van Schayck, T.; Otten, R.; Engels, R.; Kleinjan, M. Evaluation and Implementation of a Proactive Telephone Smoking Cessation Counseling for Parents: A Study Protocol of an Effectiveness Implementation Hybrid Design. Int. J. Environ. Res. Public Health 2018, 15, 97. https://doi.org/10.3390/ijerph15010097
Scheffers-van Schayck T, Otten R, Engels R, Kleinjan M. Evaluation and Implementation of a Proactive Telephone Smoking Cessation Counseling for Parents: A Study Protocol of an Effectiveness Implementation Hybrid Design. International Journal of Environmental Research and Public Health. 2018; 15(1):97. https://doi.org/10.3390/ijerph15010097
Chicago/Turabian StyleScheffers-van Schayck, Tessa, Roy Otten, Rutger Engels, and Marloes Kleinjan. 2018. "Evaluation and Implementation of a Proactive Telephone Smoking Cessation Counseling for Parents: A Study Protocol of an Effectiveness Implementation Hybrid Design" International Journal of Environmental Research and Public Health 15, no. 1: 97. https://doi.org/10.3390/ijerph15010097
APA StyleScheffers-van Schayck, T., Otten, R., Engels, R., & Kleinjan, M. (2018). Evaluation and Implementation of a Proactive Telephone Smoking Cessation Counseling for Parents: A Study Protocol of an Effectiveness Implementation Hybrid Design. International Journal of Environmental Research and Public Health, 15(1), 97. https://doi.org/10.3390/ijerph15010097



