Adsorption Property and Mechanism of Oxytetracycline onto Willow Residues
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
2.2. Preparation of Willow Residues
2.3. Characterization of Willow Residues
2.4. Detection Method of OTC in Solution
2.5. Adsorption Procedure
2.5.1. Adsorption Kinetics
2.5.2. Adsorption Isotherm
2.5.3. Thermodynamics
3. Results and Discussion
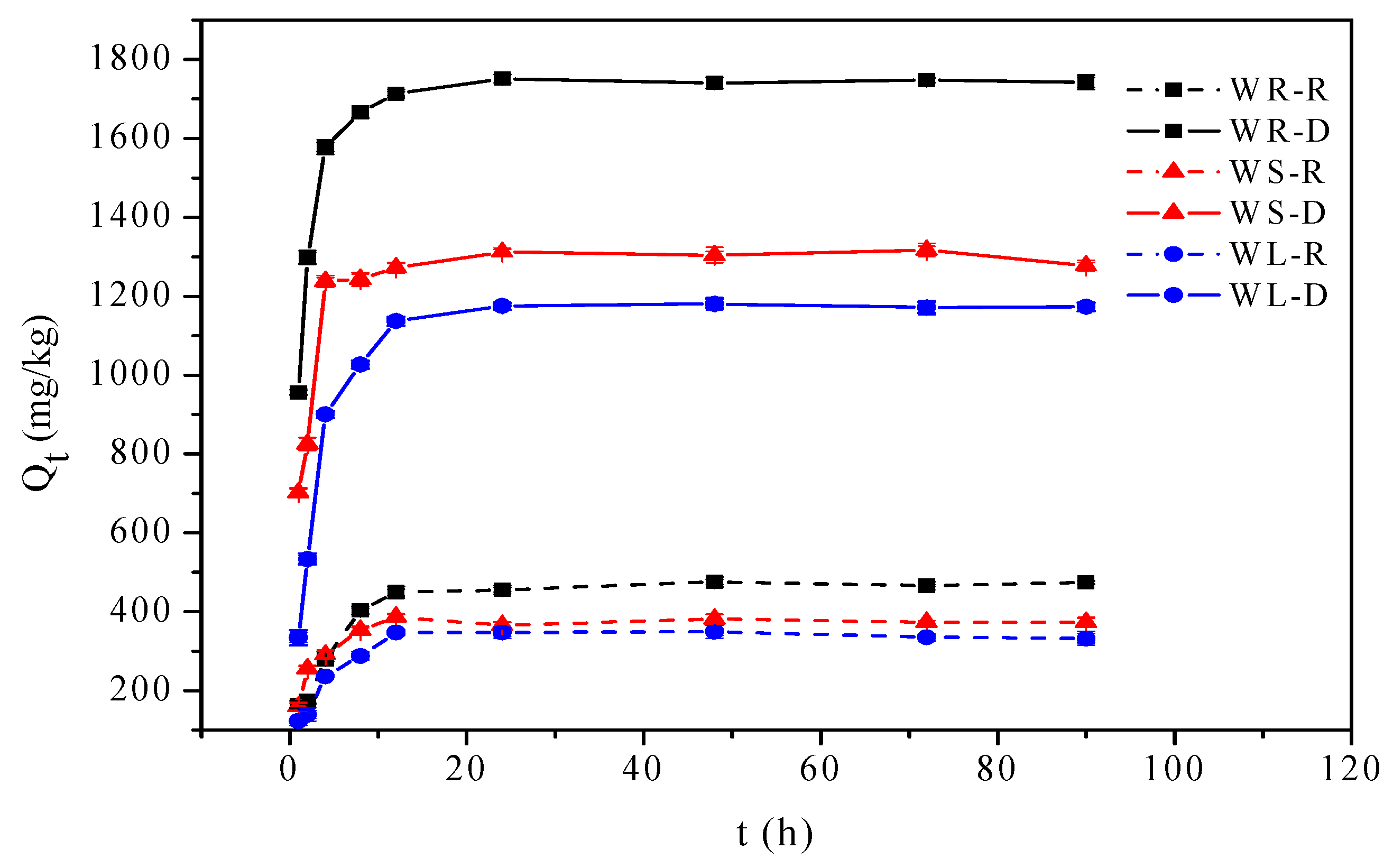
3.1. Adsorption Kinetics
3.2. Adsorption Isotherms
3.3. Thermodynamics
3.4. Analysis of the Adsorption Mechanism of OTC Adsorption onto Willow Residues
3.4.1. SEM Analysis
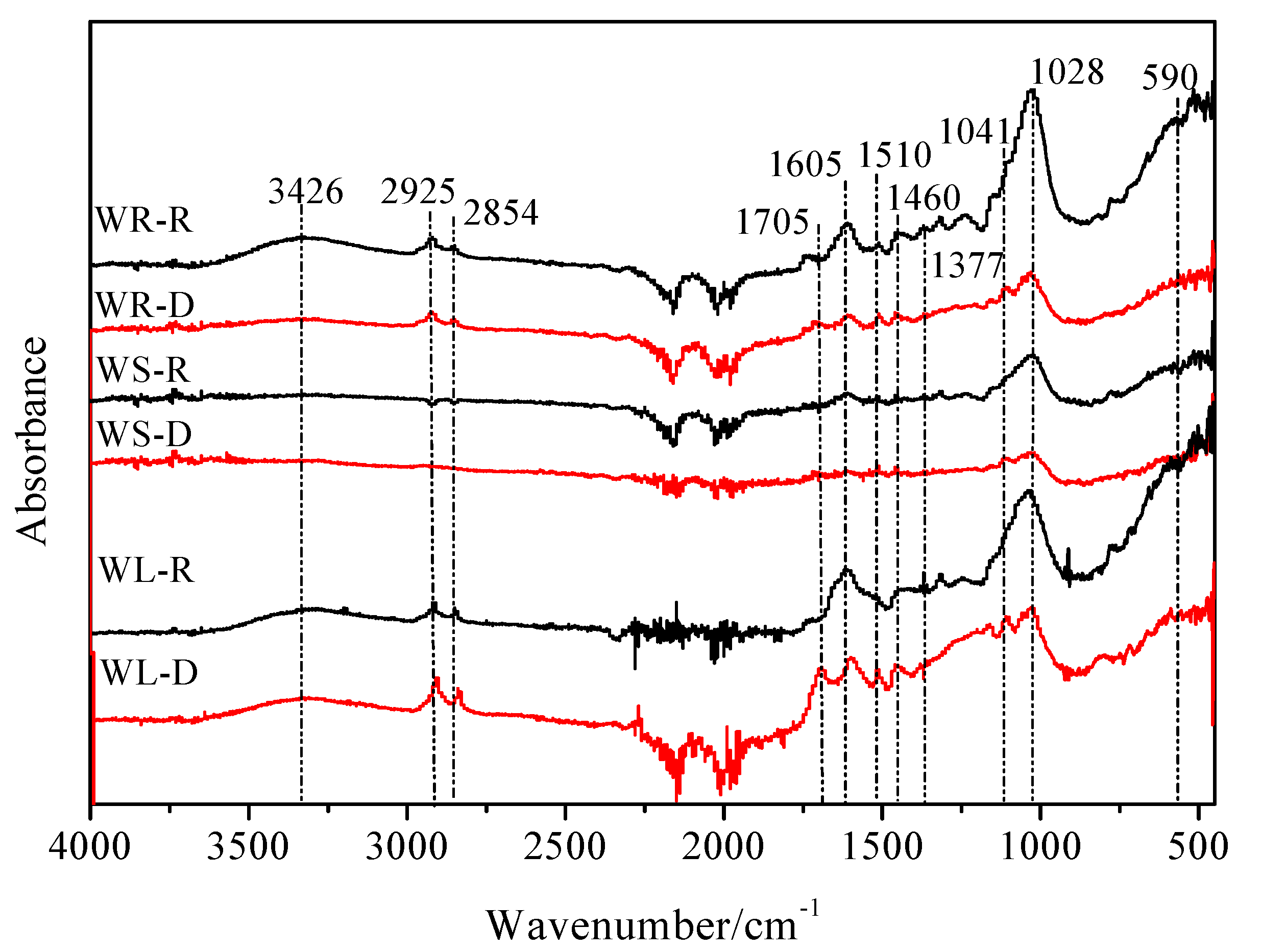
3.4.2. FTIR Spectra Analysis
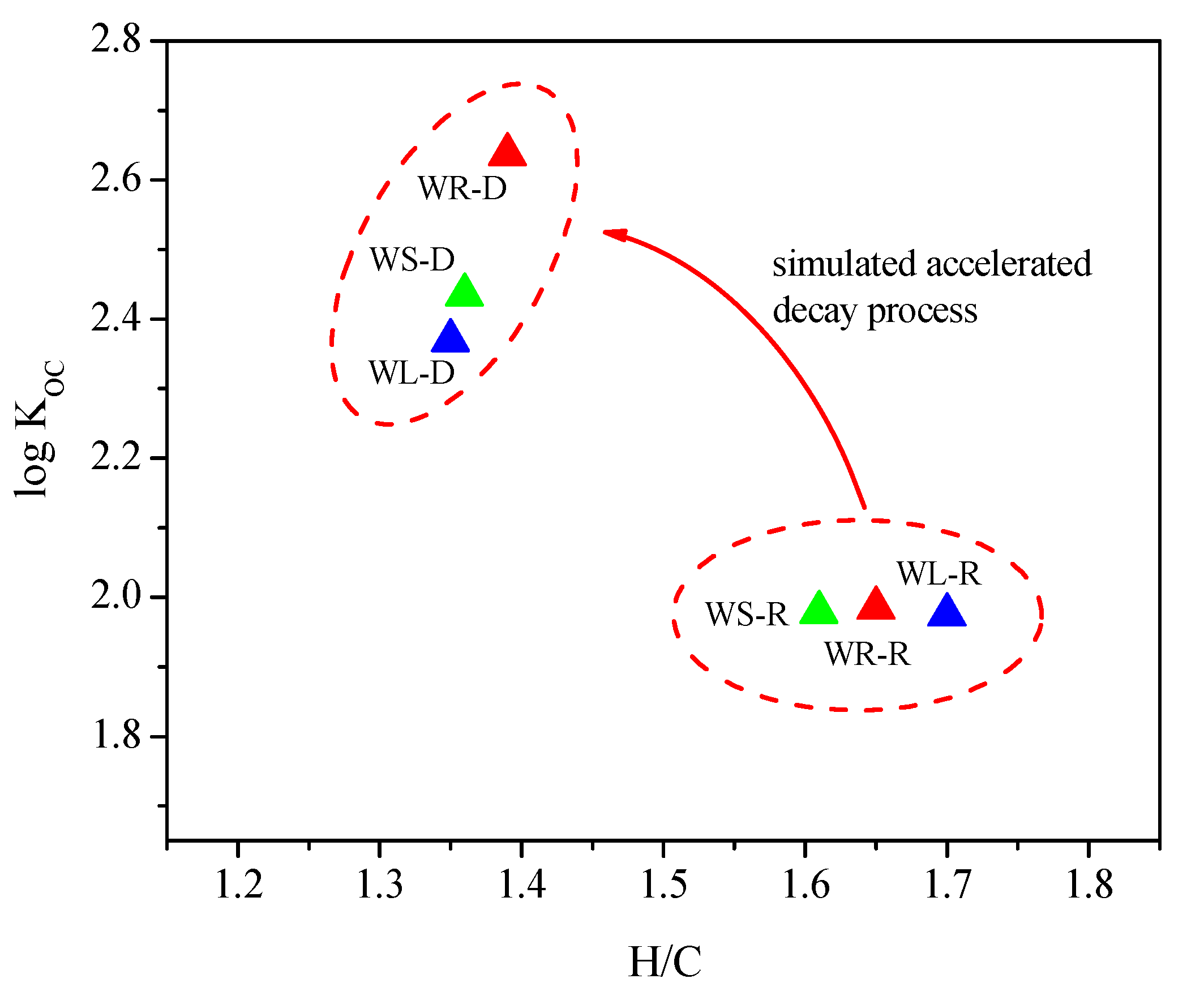
3.4.3. Elemental Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sun, Y.Y.; Yue, Q.Y.; Gao, B.Y.; Li, Q.; Huang, L.H.; Yao, F.J.; Xu, X. Preparation of activated carbon derived from cotton linter fibers by fused NaOH activation and its application for oxytetracycline (OTC) adsorption. J. Colloid Interface Sci. 2012, 368, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Kay, P.; Blackwell, P.A.; Boxall, A.B. Fate of veterinary antibiotics in a macroporous tile drained clay soil. Environ. Toxicol. Chem. 2004, 23, 1136–1144. [Google Scholar] [CrossRef] [PubMed]
- Karci, A.; Balcioglu, I.A. Investigation of the tetracycline, sulfonamide, and fluoroquinolone antimicrobial compounds in animal manure and agricultural soils in Turkey. Sci. Total Environ. 2009, 407, 4652–4664. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Liu, T.Z.; Wang, T. The fate of antibiotics in environment and its ecotoxicology. Acta Ecol. Sin. 2006, 26, 265–269. (In Chinese) [Google Scholar]
- Zhou, Q.X.; Luo, Y.; Wang, M.E. Environmental residues and ecotoxicity of antibiotics and their resistance gene pollution. Asian J. Ecotoxicol. 2007, 2, 243–251. (In Chinese) [Google Scholar]
- Gao, L.C.; Yang, S.K.; Wang, D.; Wang, G. Influence of humic acid colloid on adsorption of oxytetracycline in sediment. Asian J. Chem. 2014, 26, 8303–8308. [Google Scholar] [CrossRef]
- Hu, L.H.; Stemig, A.M.; Wammer, K.H.; Strathmann, T.J. Oxidation of antibiotics during water treatment with potassium permanganate: Reaction pathways and deactivation. Environ. Sci. Technol. 2011, 45, 3635–3642. [Google Scholar] [CrossRef] [PubMed]
- Sturini, M.; Speltini, A.; Maraschi, F.; Profumo, A.; Pretali, L.; Fasani, E.; Albini, A. Photochemical degradation of marbofloxacin and enrofloxacin in natural waters. Environ. Sci. Technol. 2010, 44, 4564–4569. [Google Scholar] [CrossRef] [PubMed]
- Dirany, A.; Sirés, I.; Oturan, N.; Özcan, A.; Oturan, M.A. Electrochemical treatment of the antibiotic sulfachloropyridazine: Kinetics, reaction pathways, and toxicity evolution. Environ. Sci. Technol. 2012, 46, 4074–4082. [Google Scholar] [CrossRef] [PubMed]
- Kovalova, L.; Siegrist, H.; Singer, H.; Wittmer, A.; McArdell, C.S. Hospital wastewater treatment by membrane bioreactor: Performance and efficiency for organic micropollutant elimination. Environ. Sci. Technol. 2012, 46, 1536–1545. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.H.; Sun, Y.Y.; Wang, W.L.; Yue, Q.Y.; Yang, T. Comparative study on characterization of activated carbons prepared by microwave and conventional heating methods and application in removal of oxytetracycline (OTC). Chem. Eng. J. 2011, 171, 1446–1453. [Google Scholar] [CrossRef]
- Wang, M.Q.; Liu, X.H.; Wu, L.Y.; Li, A.M. Adsorption behaviors of oxytetracycline on synthetic resins. Adv. Mater. Res. 2013, 726–731, 573–576. [Google Scholar] [CrossRef]
- Liu, J.G.; Li, J.X.; Chen, Y.C.; Liu, B.; Jiang, X.L.; Yang, J. Adsorption of oxytetracycline wastewater by modified corn straw. Environ. Ecol. Three Gorges 2009, 2, 28–30. (In Chinese) [Google Scholar]
- Boving, T.B.; Zhang, W. Removal of aqueous-phase polynuclear aromatic hydrocarbons using aspen wood fibers. Chemosphere 2004, 54, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.L.; Yuan, M.X.; Liu, H. Removal of polycyclic aromatic hydrocarbons from aqueous solution using plant residue materials as a biosorbent. J. Hazard. Mater. 2011, 188, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.M.; Chen, B.L. Removal of polycyclic aromatic hydrocarbons from aqueous solution by raw and modified plant residue materials as biosorbents. J. Environ. Sci. 2014, 26, 737–748. [Google Scholar] [CrossRef]
- Lin, D.H.; Pan, B.; Zhu, L.Z.; Xing, B.S. Characterization and phenanthrene sorption of tea leaf powders. J. Agric. Food Chem. 2007, 55, 5718–5724. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.L.; Li, Y.G.; Guo, Y.T.; Zhu, L.Z.; Schnoor, J.L. Role of the extractable lipids and polymeric lipids in sorption of organic contaminants onto plant Cuticles. Environ. Sci. Technol. 2008, 42, 1517–1523. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.W.; Zhan, Y.H.; Zhu, Z.L.; Xing, Y.Q. Adsorption of tannic acid from aqueous solution onto surfactant-modified zeolite. J. Hazard. Mater. 2011, 193, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.H.; Yang, S.K.; Zhao, Y.; Chen, J. Adsorption behaviors of oxytetracycline onto sediment in the Weihe River, Shaanxi, China. J. Chem. 2013, 2013, 10. [Google Scholar] [CrossRef]
- Wahab, M.A.; Jellali, S.; Jedidi, N. Ammonium biosorption onto sawdust: FTIR analysis, kinetics and adsorption isotherms modeling. Bioresour. Technol. 2010, 101, 5070–5075. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.S.; Zheng, T.; Wang, P.; Jiang, J.P.; Li, N. Adsorption isotherm, kinetic and mechanism studies of some substituted phenols on activated carbon fibers. Chem. Eng. J. 2010, 157, 348–356. [Google Scholar] [CrossRef]
- Sharma, P.; Kaur, H. Sugarcane bagasse for the removal of erythrosin B and methylene blue from aqueous waste. Appl. Water Sci. 2011, 1, 135–145. [Google Scholar] [CrossRef]
- Yu, Y.; Zhuang, Y.Y.; Wang, Z.H.; Qiu, M.Q. Adsorption of water-soluble dyes onto modified resin. Chemosphere 2004, 54, 425–430. [Google Scholar] [CrossRef]
- Liu, Y. Is the free energy change of adsorption correctly calculated? Chem. Eng. J. 2009, 54, 1981–1985. [Google Scholar] [CrossRef]
- Li, Y.H.; Di, Z.C.; Ding, J.; Wu, D.H.; Luan, Z.K.; Zhu, Y.Q. Adsorption thermodynamic, kinetic and desorption studies of Pb2+ on carbon nanotubes. Water Res. 2005, 39, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.L.; Johnson, E.J.; Chefetz, B.; Zhu, L.Z.; Xing, B.S. Sorption of polar and nonpolar aromatic organic contaminants by plant cuticular materials: Role of polarity and accessibility. Environ. Sci. Technol. 2005, 39, 6138–6146. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.G.; Chen, B.L.; Zhu, L.Z. Single-solute and bi-solute sorption of phenanthrene and pyrene onto pine needle cuticular fractions. Environ. Pollut. 2010, 158, 2478–2484. [Google Scholar] [CrossRef] [PubMed]

| Samples | Qe,exp | Pseudo-First-Order Model | Pseudo-Second-Order Model | ||||
|---|---|---|---|---|---|---|---|
| R2 | k1 | Qe,cal | R2 | k2 | Qe,cal | ||
| (mg/kg) | (1/h) | (mg/kg) | (kg/mg·h) | (mg/kg) | |||
| Raw willow roots (WR-R) | 464.32 | 0.9740 | 0.2409 | 467.42 | 0.9700 | 0.0008 | 486.91 |
| Desugared willow roots (WR-D) | 1745.84 | 0.9853 | 0.7649 | 1727.43 | 0.9819 | 0.0007 | 1793.56 |
| Raw willow stems (WS-R) | 376.58 | 0.9564 | 0.5317 | 374.62 | 0.9098 | 0.0024 | 388.55 |
| Desugared willow stems (WS-D) | 1302.04 | 0.9591 | 0.7166 | 1289.20 | 0.9483 | 0.0009 | 1337.26 |
| Raw willow leaves (WL-R) | 342.69 | 0.9686 | 0.2909 | 344.65 | 0.9362 | 0.0012 | 370.24 |
| Desugared willow leaves (WL-D) | 1175.11 | 0.9881 | 0.3368 | 1163.17 | 0.9436 | 0.0004 | 1283.42 |
| Samples | Langmuir Model | Freundlich Model | |||||
|---|---|---|---|---|---|---|---|
| Qm | KL | R2 | RL | KF | n | R2 | |
| (mg/kg) | (L/mg) | (mg/kg) (L/mg)1/n | |||||
| WR-R | 20,223.57 | 0.0027 | 0.9912 | (0.93, 0.99) | 56.43 | 1.03 | 0.9906 |
| WR-D | 21,932.29 | 0.0116 | 0.9844 | (0.74, 0.95) | 301.81 | 1.15 | 0.9796 |
| WS-R | 5936.72 | 0.0087 | 0.9624 | (0.79, 0.96) | 21.04 | 1.10 | 0.9544 |
| WS-D | 12,188.92 | 0.0146 | 0.9967 | (0.70, 0.93) | 233.00 | 1.23 | 0.9965 |
| WL-R | 8216.63 | 0.0055 | 0.9687 | (0.86, 0.97) | 16.17 | 1.06 | 0.9657 |
| WL-D | 9780.79 | 0.0186 | 0.9961 | (0.64, 0.91) | 196.98 | 1.15 | 0.9689 |
| Samples | T/K | ΔG/kJ·mol−1 | ΔH/kJ·mol−1 | ΔS/J·mol−1·K−1 |
|---|---|---|---|---|
| WR-R | 298 | −10.28 | 37.49 | 160.29 |
| 308 | −11.88 | |||
| 318 | −13.48 | |||
| WR-D | 298 | −14.35 | 39.18 | 179.63 |
| 308 | −16.15 | |||
| 318 | −17.94 | |||
| WS-R | 298 | −10.21 | 33.05 | 145.18 |
| 308 | −11.67 | |||
| 318 | −13.12 | |||
| WS-D | 298 | −11.76 | 31.68 | 145.76 |
| 308 | −13.21 | |||
| 318 | −14.67 | |||
| WL-R | 298 | −9.51 | 38.36 | 160.63 |
| 308 | −10.91 | |||
| 318 | −12.72 | |||
| WL-D | 298 | −11.95 | 20.47 | 108.79 |
| 308 | −13.04 | |||
| 318 | −14.13 |
| Samples | Sugar (%) | N (%) | C (%) | H (%) | O (%) | H/C | (N + O)/C | O/C | Kd (L/kg) | Koc |
|---|---|---|---|---|---|---|---|---|---|---|
| WR-R | 40.45 | 0.43 | 43.47 | 5.97 | 46.80 | 1.65 | 0.82 | 0.81 | (49.42, 55.01) | 119.46 |
| WR-D | - | 0.16 | 50.53 | 5.86 | 43.83 | 1.39 | 0.65 | 0.65 | (196.42, 234.10) | 432.91 |
| WS-R | 48.67 | 0.64 | 45.76 | 6.14 | 44.32 | 1.61 | 0.74 | 0.73 | (38.88, 49.38) | 95.15 |
| WS-D | - | 0.33 | 55.61 | 6.31 | 38.25 | 1.36 | 0.52 | 0.52 | (129.36, 176.84) | 272.09 |
| WL-R | 61.11 | 1.68 | 42.28 | 5.99 | 41.36 | 1.70 | 0.77 | 0.73 | (37.66, 46.20) | 94.39 |
| WL-D | - | 1.05 | 58.61 | 6.60 | 33.38 | 1.35 | 0.44 | 0.43 | (124.34, 153.59) | 234.04 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, D.; Xu, H.; Yang, S.; Wang, W.; Wang, Y. Adsorption Property and Mechanism of Oxytetracycline onto Willow Residues. Int. J. Environ. Res. Public Health 2018, 15, 8. https://doi.org/10.3390/ijerph15010008
Wang D, Xu H, Yang S, Wang W, Wang Y. Adsorption Property and Mechanism of Oxytetracycline onto Willow Residues. International Journal of Environmental Research and Public Health. 2018; 15(1):8. https://doi.org/10.3390/ijerph15010008
Chicago/Turabian StyleWang, Di, Haiyang Xu, Shengke Yang, Wenke Wang, and Yanhua Wang. 2018. "Adsorption Property and Mechanism of Oxytetracycline onto Willow Residues" International Journal of Environmental Research and Public Health 15, no. 1: 8. https://doi.org/10.3390/ijerph15010008
APA StyleWang, D., Xu, H., Yang, S., Wang, W., & Wang, Y. (2018). Adsorption Property and Mechanism of Oxytetracycline onto Willow Residues. International Journal of Environmental Research and Public Health, 15(1), 8. https://doi.org/10.3390/ijerph15010008







