Asbestos-Related Disorders in Germany: Background, Politics, Incidence, Diagnostics and Compensation
Abstract
:1. Introduction
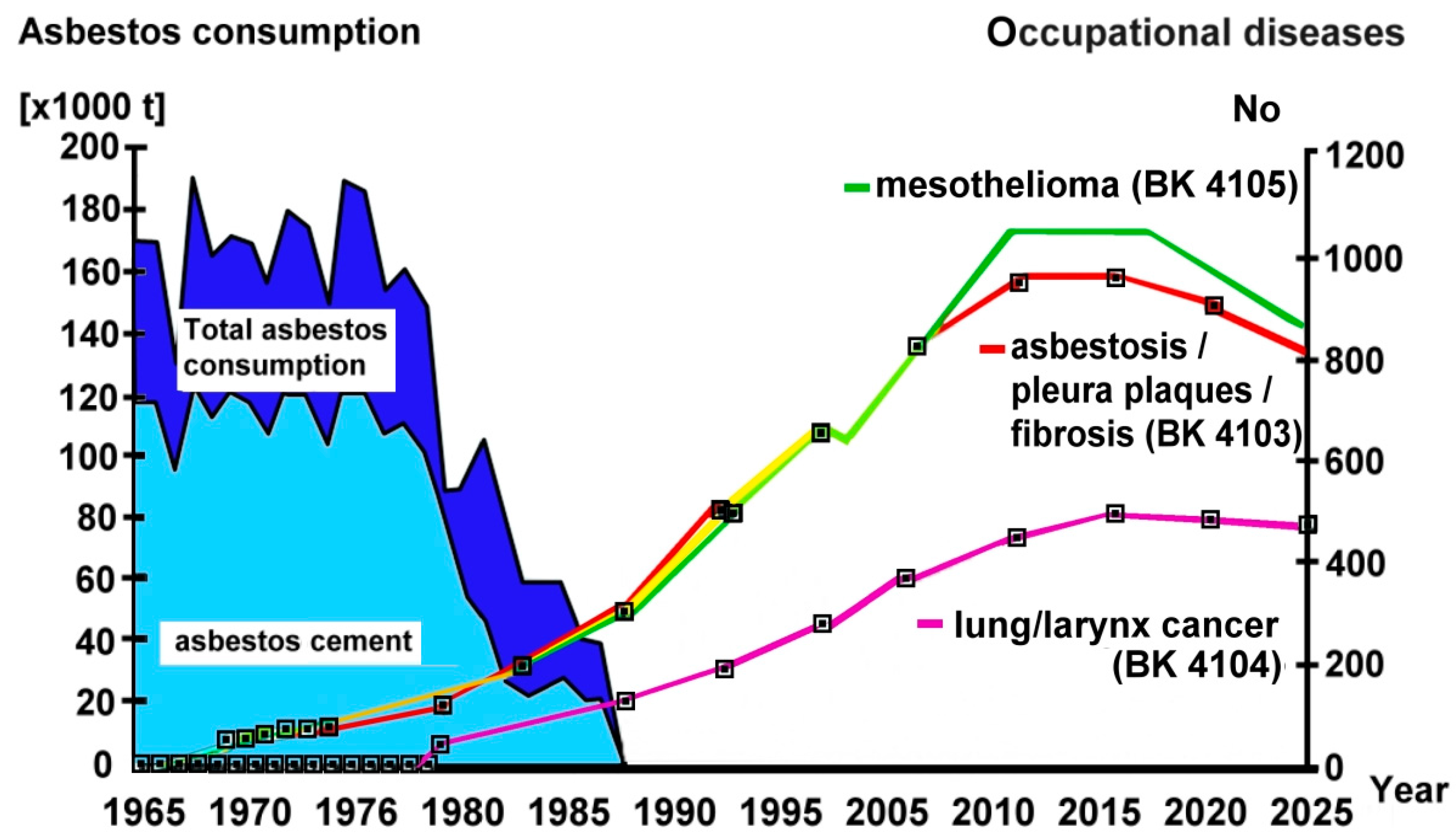
1.1. Asbestos Consumption and Occupational Exposure in Germany
1.2. Figures on Exposed Subjects and on Asbestos Exposure Levels
1.3. Asbestos Threshold Limit Value
1.4. Medical Surveillance and Screening
1.5. Pressure on Recognition and Compensation of Asbestos-Related Diseases
1.6. Schedule of Asbestos-Related Diseases
1.6.1. European Union
1.6.2. History of the List of Asbestos-Related Diseases in Germany
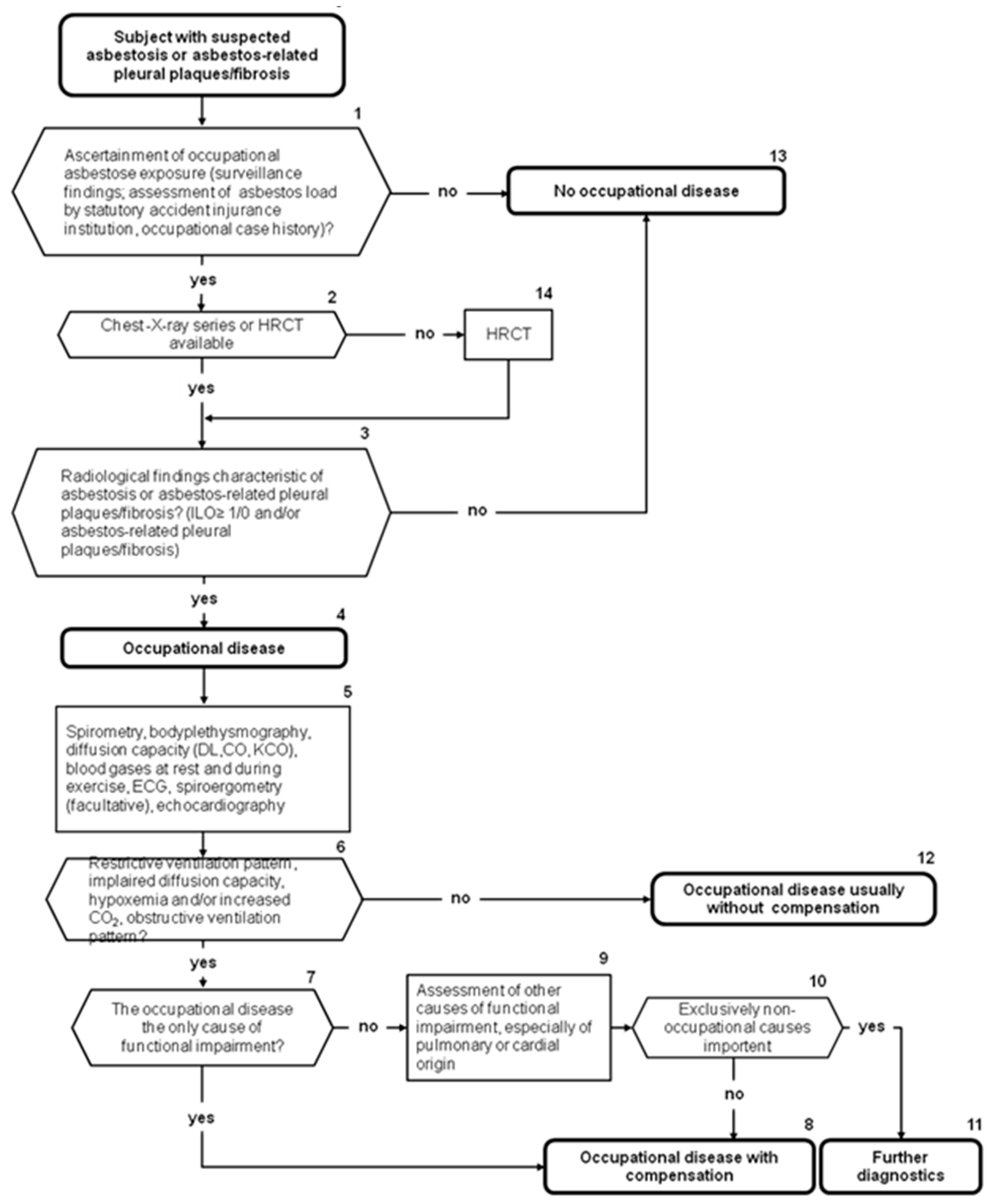
1.7. German Guideline on Diagnosis of and Compensation for Asbestos-Related Diseases
1.8. Asbestos-Related Diseases in Germany
1.9. Health and the Economic Burden on Society
1.10. Role of Stakeholders—Their Socio-Political Contributionsand Influence on Policy-Making Processes
1.11. Factors Initiating Improvement of Working Conditions and a Definitive Asbestos Ban
2. Discussion
Lessons Learnt from the Burden of Asbestos-Related Disorders and Deaths
Past Pandemic and Current Battlefields
3. What Still Has to Be Done
Key Notes
- Distribution of scientific knowledge on the harm of asbestos and the related enormous economic burden on society has increased the concern of exposed workers and the public.
- This has forced policymakers to initiate regulations on effective preventative measures and compensation for asbestos victims.
- Latency periods until appropriate regulations and an asbestos ban come into force are associated with many new asbestos-related diseases and, therefore, such latency periods should be minimized.
- Fair compensation for asbestos victims is needed.
- Alternative harmless products that replace asbestos should be promoted.
- Expensive cleaning up of asbestos-containing materials, e.g., in buildings, has to be taken into account.
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Fahr, T. Kristallbildung in der Lunge. Deut. Med. Wochenschr. 1914, 30, 1548–1549. [Google Scholar]
- BAuA. National Asbestos Profile for Germany; Federal Institute for Occupational Safety and Health: Dortmund, Germany, 2014. [Google Scholar]
- Kameda, T.; Takahashi, K.; Kim, R.; Jiang, Y.; Movahed, M.; Park, E.K.; Rantanen, J. Asbestos: Use, bans and disease burden in Europe. Bull. World Health Organ. 2014, 92, 790–797. [Google Scholar] [CrossRef] [PubMed]
- Coenen, W.; Schenk, H. Ermittlung von Risikogruppen bei Asbestexponierten. Die BG 1990, 12, 718–725. [Google Scholar]
- Tossavainen, A. Consensus report. Asbestos, asbestosis, and cancer: The Helsinki criteria for diagnosis and attribution. Scand. J. Work Environ. Health 1997, 23, 311–316. [Google Scholar] [CrossRef]
- Lissner, L.; Kuhl, K.; Knaupinen, T.; Uuksulainen, S. Exposure to carcinogens and work-related cancer: A review of assessment methods. In European Risk Observatory Report; European Agency for Safety and Health at Work: Luxembourg, 2014; pp. 1–64. [Google Scholar]
- Enviromental Working Group. Asbestos: Think Again: America’s Asbestos Epidemic. Available online: https://www.ewg.org/research/asbestos-think-again/americas-asbestos-epidemic#.WlxeDTcxmSQ (accessed on 4 March 2004).
- Occupational Safety & Health Administration. Safety and Health Topics: Asbestos. Available online: http://www.osha.gov/SLTC/asbestos/index.html (accessed on 2 January 2011).
- Arendt, M.; Bauer, H.D.; Blome, H.; Blome, O.; Bonk, L.; Brock, T.; Chromy, W.; Gabriel, S.; Guldner, K.H.; Karsten, H.; et al. Faserjahre—Berufsgenossenschaftliche Hinweise zur Ermittlung der Kummulativen Asbestfaserstaub-Dosis am Arbeitsplatz (Faserjahre) und Bearbeitungshinweise zur Berufskrankheit Nr. 4104 “Lungenkrebs Oder Kehlkopfkrebs” Durch Asbest, 4th ed.; Hauptverband der Gewerblichen Berufsgenossenschaften (HVBG) (BK-Report): St. Augustin, Germany, 2007; Volume 1. [Google Scholar]
- Deutsche Gesetzliche Unfallversicherung. BK-Report 1/2013 Faserjahre; Deutsche Gesetzliche Unfallversicherung (DGUV): Rheinbreitbach, Germany, 2013. [Google Scholar]
- Enviromental Working Group. The Asbestos Epidemic in America. Available online: http://www.ewg.org/sites/asbestos/facts/fact1.php (accessed on 1 January 2011).
- Hagemeyer, O.; Otten, H.; Kraus, T. Asbestos consumption, asbestos exposure and asbestos-related occupational diseases in Germany. Int. Arch. Occup. Environ. Health 2006, 79, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Burdorf, A.; Swuste, P. An expert system for the evaluation of historical asbestos exposure as diagnostic criterion in asbestos-related diseases. Ann. Occup. Hyg. 1999, 43, 57–66. [Google Scholar] [CrossRef]
- Baur, X.; Czuppon, A. Regulation and compensation of asbestos diseases in Germany. In The Treatment and Prevention of Asbestos Diseases; Peters, G., Peters, B., Eds.; Lexis Law Publishing: Charlottesville, VA, USA, 1997; pp. 405–419. [Google Scholar]
- BAuA. TRGS 519 Asbest: Abbruch-, Sanierungs- oder Instandhaltungsarbeiten. Available online: https://www.baua.de/DE/Angebote/Rechtstexte-und-Technische-Regeln/Regelwerk/TRGS/pdf/TRGS-519.pdf?__blob=publicationFile (accessed on 14 January 2018).
- BAuA. Hazardous Substances Ordinance (GefStoffV); Bundesministerium für Justiz und Verbraucherschutz: Berlin, Germany, 2013; pp. 1–61. [Google Scholar]
- BAuA. Gefahrstoffverordnung (GefStoffV); BMAS: Berlin, Germany, 2017; pp. 1–67. [Google Scholar]
- McCulloch, J.; Tweedale, G. Defending the Indefensible: The Global Asbestos Industry and Its Fight for Survival; Oxford University Press: New York, NY, USA, 2008. [Google Scholar]
- Lilienfeld, D.E. The silence: The asbestos industry and early occupational cancer research—A case study. Am. J. Public Health 1991, 81, 791–800. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, J. Saving the asbestos industry, 1960 to 2006. Public Health Rep. 2006, 121, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Wagner, J.C.; Sleggs, C.A.; Marchand, P. Diffuse pleural mesothelioma and asbestos exposure in the North Western Cape Province. Br. J. Ind. Med. 1960, 17, 260–271. [Google Scholar] [CrossRef] [PubMed]
- Selikoff, I.J. Asbestos disease-1990–2020: The risks of asbestos risk assessment. Toxicol. Ind. Health 1991, 7, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Selikoff, I.J.; Churg, J.; Hammond, E.C. Asbestos Exposure and Neoplasia. JAMA 1964, 188, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Selikoff, J.; Churg, J. Biological Effects of Asbestos; New York Academy of Sciences: New York, NY, USA, 1965. [Google Scholar]
- Peto, J.; Hodgson, J.T.; Matthews, F.E.; Jones, J.R. Continuing increase in mesothelioma mortality in Britain. Lancet 1995, 345, 535–539. [Google Scholar] [CrossRef]
- Peto, J.; Decarli, A.; La Vecchia, C.; Levi, F.; Negri, E. The European mesothelioma epidemic. Br. J. Cancer 1999, 79, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Pelucchi, C.; Malvezzi, M.; La Vecchia, C.; Levi, F.; Decarli, A.; Negri, E. The Mesothelioma epidemic in Western Europe: An update. Br. J. Cancer 2004, 90, 1022–1024. [Google Scholar] [CrossRef] [PubMed]
- Banaei, A.; Auvert, B.; Goldberg, M.; Gueguen, A.; Luce, D.; Goldberg, S. Future trends in mortality of French men from mesothelioma. Occup. Environ. Med. 2000, 57, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, J.T.; McElvenny, D.M.; Darnton, A.J.; Price, M.J.; Peto, J. The expected burden of mesothelioma mortality in Great Britain from 2002 to 2050. Br. J. Cancer 2005, 92, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Marinaccio, A.; Montanaro, F.; Mastrantonio, M.; Uccelli, R.; Altavista, P.; Nesti, M.; Costantini, A.S.; Gorini, G. Predictions of mortality from pleural mesothelioma in Italy: A model based on asbestos consumption figures supports results from age-period-cohort models. Int. J. Cancer 2005, 115, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Murayama, T.; Takahashi, K.; Natori, Y.; Kurumatani, N. Estimation of future mortality from pleural malignant mesothelioma in Japan based on an age-cohort model. Am. J. Ind. Med. 2006, 49, 1–7. [Google Scholar] [CrossRef] [PubMed]
- IARC. Asbestos (Chrysotile, Amosite, Crocidolite, Tremolite, Actinolite and Anthophyllite). In IARC Monographs Volume 100C; IARC/WHO: Lyon, France, 2012. [Google Scholar]
- National Academies. Asbestos selected cancers. In Asbestos: Selected Cancer; The National Academies Collection: Washington, DC, USA, 2006. [Google Scholar]
- Rice, J. The global reorganization and revitalization of the asbestos industry, 1970–2007. Int. J. Health Serv. 2011, 41, 239–254. [Google Scholar] [CrossRef] [PubMed]
- Kazan-Allen, L. The asbestos war. Int. J. Occup. Environ. Health 2003, 9, 173–193. [Google Scholar] [CrossRef] [PubMed]
- Collegium Ramazzini. The 18th Collegium Ramazzini statement: The global health dimensions of asbestos and asbestos-related diseases. Scand. J. Work Environ. Health 2016, 42, 86–90. [Google Scholar]
- IJPC. 2012. Available online: http://www.ijpc-se.org/documents/01.JPC-SE-Position_Statement_on_Asbestos-June_4_2012-Summary_and_Appendix_A_English.pdf (accessed on 14 January 2018).
- European Commission. Information Notices on Occupational Diseases: A Guide to Diagnosis. Available online: https://publications.europa.eu/en/publication-detail/-/publication/5d549879-7f75-4e89-b737-e19f530d1236 (accessed on 14 October 2009).
- World Health Organization (WHO). Elimination of asbestos related diseases. In WHO Fact Sheet N 343; World Health Organization (WHO): Geneva, Switzerland, 2010. [Google Scholar]
- Proctor, R.N. The Nazi War on Cancer; Princeton University Press: Princeton, NJ, USA, 2000. [Google Scholar]
- Edelman, D.A. Laryngeal cancer and occupational exposure to asbestos. Int. Arch. Occup. Environ. Health 1989, 61, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Konetzke, G. Das Larynxkarzinom aus arbeitsmedizinischer und onkologischer Sicht unter Berücksichtigung der in der ehemaligen DDR zur Frage des Ursachenzusammenhanges gewonnenen Erkenntnisse. In BK-Report 2/94; HVGB: Sankt Augustin, Germany, 1994. [Google Scholar]
- Maier, H.; Sennewald, E.; Dietz, A.; Fischer, G.; Gewelke, U.; Heller, W.; Kura, N.; Zöller, J. Risikofaktoren für Plattenepithelkarzinome im Kopf-Hals-Bereich. Ergebnisse der Heidelberger Fallkontrollstudien; HVGB: Sankt Augustin, Germany, 1994. [Google Scholar]
- Bundesregierung. Berufskrankheiten-Verordnung vom 31.10.1997 (BGBl I S. 2623), zuletzt geändert durch Verordnung vom 11.6.2009. BGBl I 2009, 30, 1273–1276. [Google Scholar]
- Camargo, M.C.; Stayner, L.T.; Straif, K.; Reina, M.; Al-Alem, U.; Demers, P.A.; Landrigan, P.J. Occupational exposure to asbestos and ovarian cancer: A meta-analysis. Environ. Health Perspect. 2011, 119, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.K.; Burnett, C.A.; Freund, E.; Walker, J.; Lalich, N.; Sestito, J. Gastrointestinal cancer mortality of workers in occupations with high asbestos exposures. Am. J. Ind. Med. 1997, 31, 713–718. [Google Scholar] [CrossRef]
- Baur, X.; Clasen, M.; Fisseler-Eckhoff, A.; Heger, M.; Hering, K.G.; Hofmann-Preiss, K.; Kohler, D.; Kranig, A.; Kraus, T.; Letzel, S.; et al. Diagnostics and expert opinion of asbestos-induced occupational diseases. Pneumologie 2011, 65, e1–e47. [Google Scholar] [CrossRef] [PubMed]
- Hering, K.G.; Kraus, T. Coding CT-classification in occupational and environmental respiratory disease (OERD). In International Classification of HRCT for Occupational and Environmental Respiratory Diseases; Kusaka, Y., Hering, K.G., Parker, J.E., Eds.; Springer: Tokyo, Japan, 2005; pp. 15–23. [Google Scholar]
- BAuA. Safety and Health at Work 2015—Report on Accident Prevention at Work; BAuA: Dortmund, Germany, 2016. [Google Scholar]
- DGUV. Entschädigungsleistungen; DGUV: St. Augustin, Germany, 2017. [Google Scholar]
- Baur, X.; Woitowitz, H.J. Lung Cancer as an Occupational Disease. Pneumologie 2016, 70, 510–513. [Google Scholar] [PubMed]
- Takala, Y. Eliminating Occupational Cancer in Europe and Globally; ETUI Aisbl: Brussels, Belgium, 2015. [Google Scholar]
- World Health Organization (WHO). International Programm on Chemical Safty—Asbestos; World Health Organization (WHO): Geneva, Switzerland, 2015. [Google Scholar]
- International Labour Organization (ILO). Asbestos: The Iron Grip of Latency. Available online: http://www.ilo.org/global/about-the-ilo/press-and-media-centre/news/WCMS_076282/lang--en/index.htm (accessed on 10 January 2006).
- LaDou, J. The asbestos cancer epidemic. Environ. Health Perspect. 2004, 112, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.T.; Takahashi, K.; Karjalainen, A.; Hoshuyama, T.; Wilson, D.; Kameda, T.; Chan, C.C.; Wen, C.P.; Furuya, S.; Higashi, T.; et al. Ecological association between asbestos-related diseases and historical asbestos consumption: An international analysis. Lancet 2007, 369, 844–849. [Google Scholar] [CrossRef]
- HSE. The Burden of Occupational Cancer in Great Britain; Overview Report; Health and Safety Executive: London, UK, 2012.
- Tompa, E.; Kalcevich, C.; McLeod, C.; Lebeau, M.; Song, C.; McLeod, K.; Kim, J.; Demers, P.A. The economic burden of lung cancer and mesothelioma due to occupational and para-occupational asbestos exposure. Occup. Environ. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). The Human and Financial Burden of Asbestos in the WHO European Region: Meeting Report, Bonn, Germany; WHO Regional Office for Europe: Copenhagen, Denmark, 2012. [Google Scholar]
- ICA. Safe Use of Chrysotile. Available online: http://www.chrysotileassociation.com/en/sfuse/default.php (accessed on 14 January 2018).
- Ruff, K. Exposé of the International Chrysotile Association. Available online: https://www.rightoncanada.ca/?p=1862 (accessed on 18 February 2013).
- Albracht, G. Trade unions and the Federal Environment Agency—Instigators of an asbestos ban in Germany. In The Long and Winding Road to an Asbestos Free Workplace; Cremers, J., Gehring, R., Eds.; European Institute for Construction Labour Research, International Books: Brussels, Belgium, 2013; pp. 77–88. [Google Scholar]
- Michaels, D. Doubt Is Their Product How Industrys Assault on Science Threatens Your Health; Oxford University Press: New York, NY, USA, 2016. [Google Scholar]
- Axelson, O.; Balbus, J.M.; Cohen, G.; Davis, D.; Donnay, A.; Doolittle, R.; Duran, B.M.; Egilman, D.; Epstein, S.S.; Goldman, L.; et al. Re: Regulatory Toxicology and Pharmacology. Int. J. Occup. Environ. Health 2003, 9, 386–389. [Google Scholar] [PubMed]
- Boden, L.I.; Ozonoff, D. Litigation-generated science: Why should we care? Environ. Health Perspect. 2008, 116, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Diitales Heimatmuseum Bremen, Kampf Gegen Asbest auf der Vulkan-Werft. Available online: http://digitales-heimatmuseum.de/kampf-gegen-asbest-auf-der-vulkan-werft/ (accessed on 4 November 2014).
- Craighead, J.E.; Abraham, J.L.; Churg, A.; Green, F.H.; Kleinerman, J.; Pratt, P.C.; Seemayer, T.A.; Vallyathan, V.; Weill, H. The pathology of asbestos-associated diseases of the lungs and pleural cavities: Diagnostic criteria and proposed grading schema. Report of the Pneumoconiosis Committee of the College of American Pathologists and the National Institute for Occupational Safety and Health. Arch. Pathol. Lab. Med. 1982, 106, 544–596. [Google Scholar] [PubMed]
- Baur, X. Asbestos: Socio-legal and Scientific Controversies and Unsound Science in the Context of the Worldwide Asbestos Tragedy—Lessons to be Learned. Pneumologie 2016, 70, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Hammar, S.P.; Abraham, J.L. Commentary on pathologic diagnosis of asbestosis and critique of the 2010 Asbestosis Committee of the College of American Pathologists (CAP) and Pulmonary Pathology Society’s (PPS) update on the diagnostic criteria for pathologic asbestosis. Am. J. Ind. Med. 2015, 58, 1034–1039. [Google Scholar] [CrossRef] [PubMed]
- Baur, X.; Woitowitz, H.J.; Budnik, L.T.; Egilman, D.; Oliver, C.; Frank, A.; Soskolne, C.L.; Landrigan, P.J.; Lemen, R.A. Asbestos, asbestosis, and cancer: The Helsinki criteria for diagnosis and attribution. Critical need for revision of the 2014 update. Am. J. Ind. Med. 2017, 60, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Baur, X. Response to the letter of R. Merget, I. Feder and A. Tannapfel. Pneumologie 2017, 71, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Woitowitz, H.J. The theory of asbestos bodies is dead—German Mesothelioma register—What next? Zbl. Arbeitsmed. 2016, 66, 232–238. [Google Scholar] [CrossRef]
- Takaro, T.K.; Davis, D.; Van Rensburg, S.J.; Arroyo Aguilar, R.S.; Algranti, E.; Bailar, J.C., 3rd; Belpoggi, F.; Berlin, M.; Bhattacharya, S.; Bonnier Viger, Y.V.; et al. Scientists appeal to Quebec Premier Charest to stop exporting asbestos to the developing world. Int. J. Occup. Environ. Health 2010, 16, 241–248. [Google Scholar] [PubMed]
- Midori, N.; Courtice, P.A.; Demers, T.K.; Takaro, S.V.; Ahktar Ahmad, S.K.; Davies, H.W.; Siddique, Z. Asbestos-related disease in Bangladeshi ship breakers: A pilot study. Int. J. Occup. Environ. Health 2011, 17, 144–153. [Google Scholar]
- Motley, R.L.; Patrick, C.W., Jr.; McGinness Kearse, A. Medicolegal aspects of asbestos-related diseases: A plaintiff’s attorney’s perspective. In Pathology of Asbestos-Associated Diseases; Roggli, V.L., Oury, T.D., Sporn, T.A., Eds.; Springer: New York, NY, USA; Berlin/Heidelberg, Germany, 2004; pp. 355–376. [Google Scholar]
- Bernstein, D.M. The health risk of chrysotile asbestos. Curr. Opin. Pulm. Med. 2014, 20, 366–370. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. A Review of Human Carcinogens: Arsenic, Metals, Fibres, and Dusts; International Agency for Research on Cancer (IARC): Lyon, France, 2012. [Google Scholar]
- Straif, K.; Benbrahim-Tallaa, L.; Baan, R.; Grosse, Y.; Secretan, B.; El Ghissassi, F.; Bouvard, V.; Guha, N.; Freeman, C.; Galichet, L.; et al. A review of human carcinogens—Part C: Metals, arsenic, dusts, and fibres. Lancet Oncol. 2009, 10, 453–454. [Google Scholar] [CrossRef]
- Council of the European Union. Council Directive 83/477/EEC of 19 September 1983 on the Protection of Workers from the Risks Related to Exposure to Asbestos at Work (Second Individual Directive within the Meaning of Article 8 of Directive 80/1107/EEC) (83/477/EEC) (OJ L 263, 24.9.1983, p. 25), as Last Amended by Directive 2007/30/EC of the European Parliament and of the Council of 20 June 2007 (OJ L 165 21 27.6.2007). 2007. Available online: https://publications.europa.eu/en/publication-detail/-/publication/6276d1bc-b8e4-47e4-a9a7-8e64e3cb8041/language-en (accessed on 14 January 2018).
- Parliament and Council EU. Directive 2003/18/EC of the European Parliament and of the Council of 27 March 2003 Amending Council Directive 83/477/EEC on the Protection of Workers from the Risks Related to Exposure to Asbestos at Work OJ L 97/48 15.4.2003. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2003:097:0048:0052:EN:PDF (accessed on 14 January 2018).
- European Commission. REACH Restrictions—Asbestos Exemptions by Member States—Report. Available online: http://ec.europa.eu/DocsRoom/documents/13166 (accessed on 16 October 2015).
- ICA. Rotterdam Convention—COP8 Meeting 2017. Available online: http://www.chrysotileassociation.com/data/ICA_Rotterdam-Convention_2017-COP8_v4F.pdf (accessed on14 January 2018).
- LaDou, J.; Castleman, B.; Frank, A.; Gochfeld, M.; Greenberg, M.; Huff, J.; Joshi, T.K.; Landrigan, P.J.; Lemen, R.; Myers, J.; et al. The case for a global ban on asbestos. Environ. Health Perspect. 2010, 118, 897–901. [Google Scholar] [CrossRef] [PubMed]
- Castleman, B. “Controlled use” of asbestos. Int. J. Occup. Environ. Health 2003, 9, 294–298. [Google Scholar] [PubMed]
- Egilman, D.; Roberts, M. Re: Controlled use of asbestos. Int. J. Occup. Environ. Health 2004, 10, 99–103. [Google Scholar] [PubMed]
| Implementation (Year) | Fibre Type | TLV (mg/m3), fibres/m3 |
|---|---|---|
| 1973 | Chrysotile * | (0.15) |
| 1976 | Chrysotile * | 2 × 106; (0.10) |
| 1979 | asbestos (chrysotile, crocidolite, amosite) | 106; (0.05) |
| 1985 | crocidolite | 5 × 105; (0.025) |
| 1990 | chrysotile, crocidolite, amosite | 2.5 × 105 |
| 1996 | asbestos (chrysotile, crocidolite, amosite) | no TLV 15 × 105 fibres/m3 initiate preventative measures for sanitation workers (personal safety medical surveillance programme) |
| 2014 | asbestos (chrysotile, crocidolite, amosite) | no TLV 10 × 105 fibres/m3 initiate preventative measures for sanitation workers (personal safety medical surveillance programme) |
| L 238/30 | Official Journal of the European Union.9/25/2003 ANNEX I |
|---|---|
| 3 | Diseases caused by the inhalation of substances and agents not included under other headings |
| 301 | Diseases of the respiratory system and cancers |
| 301.21 | Asbestosis |
| 301.22 | Mesothelioma following the inhalation of asbestos dust |
| 301.31 | Pneumoconioses caused by dusts of silicates |
| 302 | Complication of asbestos in the form of bronchial cancer |
| (a) | |
| Occupational Disease | Year |
| Asbestosis | 1936 |
| Lung cancer in connection with asbestosis | 1942 |
| Mesothelioma of pleura and/or peritoneum | 1977 |
| Asbestosis or asbestos-related pleural plaques or fibrosis | 1988 |
| Lung cancer combined with asbestosis or asbestos-related pleural plaques or fibrosis | 1988 |
| Lung cancer combined with asbestosis, asbestos-related pleural plaques or fibrosis or evidence of cumulative exposure to asbestos dust in the workplace of at least 25 asbestos fiber-years | 1992 |
| Larynx cancer combined with asbestosis, asbestos-related pleural plaques or fibrosis or evidence of cumulative exposure to asbestos dust in the workplace of at least 25 asbestos fiber-years | 1997 |
| (b) | |
| Occupational Disease No. | |
| 4103 | Asbestosis or diseases of the pleura (plaques, fibrosis) caused by asbestos dust |
| 4104 | Lung or larynx cancer * - combined with asbestosis - combined with diseases of the pleura caused by asbestos dust or - if there is evidence of a cumulative exposure to asbestos dust in the workplace of at least 25 fiber-years {25 × 106 [(fiber/m3) × years]} |
| 4105 | Mesothelioma of the pleura, the peritoneum or the pericardium caused by asbestos |
| 4111 | Lung cancer caused by the interaction of asbestos dust and polycyclic aromatic hydrocarbons, by evidence of exposure to a cumulative dose which equates a probability of causation of at least 50 percent |
| Asbestos-Induced Occupational Diseases (OD) in Germany in 2016, 2015, 2014, 2013 | |||||
|---|---|---|---|---|---|
| Occupational Diseases No. | Reported OD | Acknowledged OD | Newly Compensated OD | Deaths | Year |
| 4103 Asbestosis, Pleural fibrosis/plaques | 3607 | 2183 | 578 | 166 | 2016 |
| 3712 | 2002 | 541 | 165 | 2015 | |
| 3602 | 1997 | 603 | 153 | 2014 | |
| 3636 | 1926 | 582 | 159 | 2013 | |
| 4104 Lung or larynx cancer | 4368 | 912 | 814 | 618 | 2016 |
| 4482 | 773 | 715 | 593 | 2015 | |
| 4343 | 834 | 766 | 595 | 2014 | |
| 4079 | 794 | 711 | 559 | 2013 | |
| 4105 Mesothelioma | 1304 | 1031 | 944 | 857 | 2016 |
| 1417 | 958 | 881 | 812 | 2015 | |
| 1380 | 1048 | 976 | 817 | 2014 | |
| 1425 | 978 | 904 | 734 | 2013 | |
| 4114 Lung cancers due to asbestos + PAH | 125 | 22 | 19 | 15 | 2016 |
| 138 | 33 | 28 | 11 | 2015 | |
| 132 | 23 | 20 | 18 | 2014 | |
| 142 | 24 | 24 | 17 | 2013 | |
| Total (2016) | 9404 | 4148 | 2353 | 1656 | |
| Country | Number of Mesothelioma Cases | Costs 2009 a (€) | Number of Lung Cancer Cases | Costs 2012 b (€) |
|---|---|---|---|---|
| Austria | 80 | 10,000,000 | 160 | 487,001,280 |
| Belgium | 156 | 19,500,000 | 2512 | 7,645,920,096 |
| Denmark | 71 | 8,875,000 | 142 | 432,213,636 |
| Finland | 75 | 9,375,000 | 150 | 456,563,700 |
| France | 826 | 103,250,000 | 1652 | 5,028,288,216 |
| Germany | 1063 | 132,875,000 | 2126 | 6,471,029,508 |
| Italy | 1235 | 15,437,500 | 2470 | 7,518,082,260 |
| The Netherlands | 395 | 49,375,000 | 790 | 2,404,568,820 |
| Norway | 54 | 6,750,000 | 108 | 328,725,864 |
| Poland | 96 | 12,000,000 | 192 | 584,401,536 |
| Portugal | 19 | 2,375,000 | 38 | 115,662,804 |
| Romania | 58 | 7,250,000 | 116 | 353,075,928 |
| Span | 263 | 32,875,000 | 526 | 1,601,016,708 |
| Sweden | 123 | 15,375,000 | 246 | 748,764,468 |
| United Kingdom | 1891 | 236,375,000 | 3782 | 11,511,492,756 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baur, X. Asbestos-Related Disorders in Germany: Background, Politics, Incidence, Diagnostics and Compensation. Int. J. Environ. Res. Public Health 2018, 15, 143. https://doi.org/10.3390/ijerph15010143
Baur X. Asbestos-Related Disorders in Germany: Background, Politics, Incidence, Diagnostics and Compensation. International Journal of Environmental Research and Public Health. 2018; 15(1):143. https://doi.org/10.3390/ijerph15010143
Chicago/Turabian StyleBaur, Xaver. 2018. "Asbestos-Related Disorders in Germany: Background, Politics, Incidence, Diagnostics and Compensation" International Journal of Environmental Research and Public Health 15, no. 1: 143. https://doi.org/10.3390/ijerph15010143
APA StyleBaur, X. (2018). Asbestos-Related Disorders in Germany: Background, Politics, Incidence, Diagnostics and Compensation. International Journal of Environmental Research and Public Health, 15(1), 143. https://doi.org/10.3390/ijerph15010143





