Longitudinal Study-Based Dementia Prediction for Public Health
Abstract
:1. Introduction
2. Previous Work
3. Longitudinal Study-Based Dementia Prediction
3.1. Workflow
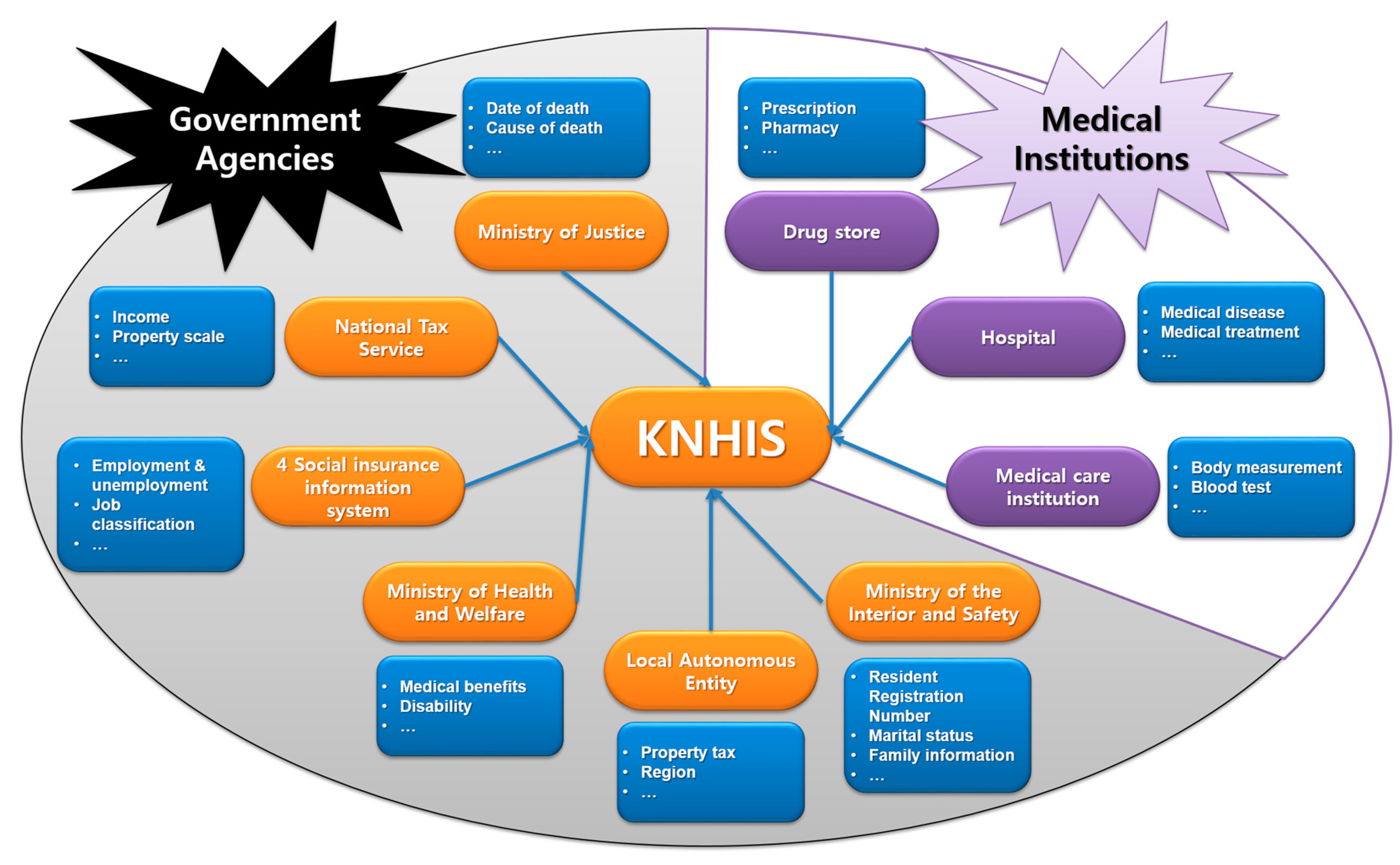
3.2. Introduction of KNHIS-SC DB
3.3. Feature Selection
3.3.1. Feature Analysis
3.3.2. Preprocessing
3.4. Dementia Prediction Using Longitudinal Public Health Data
4. Experimental Results
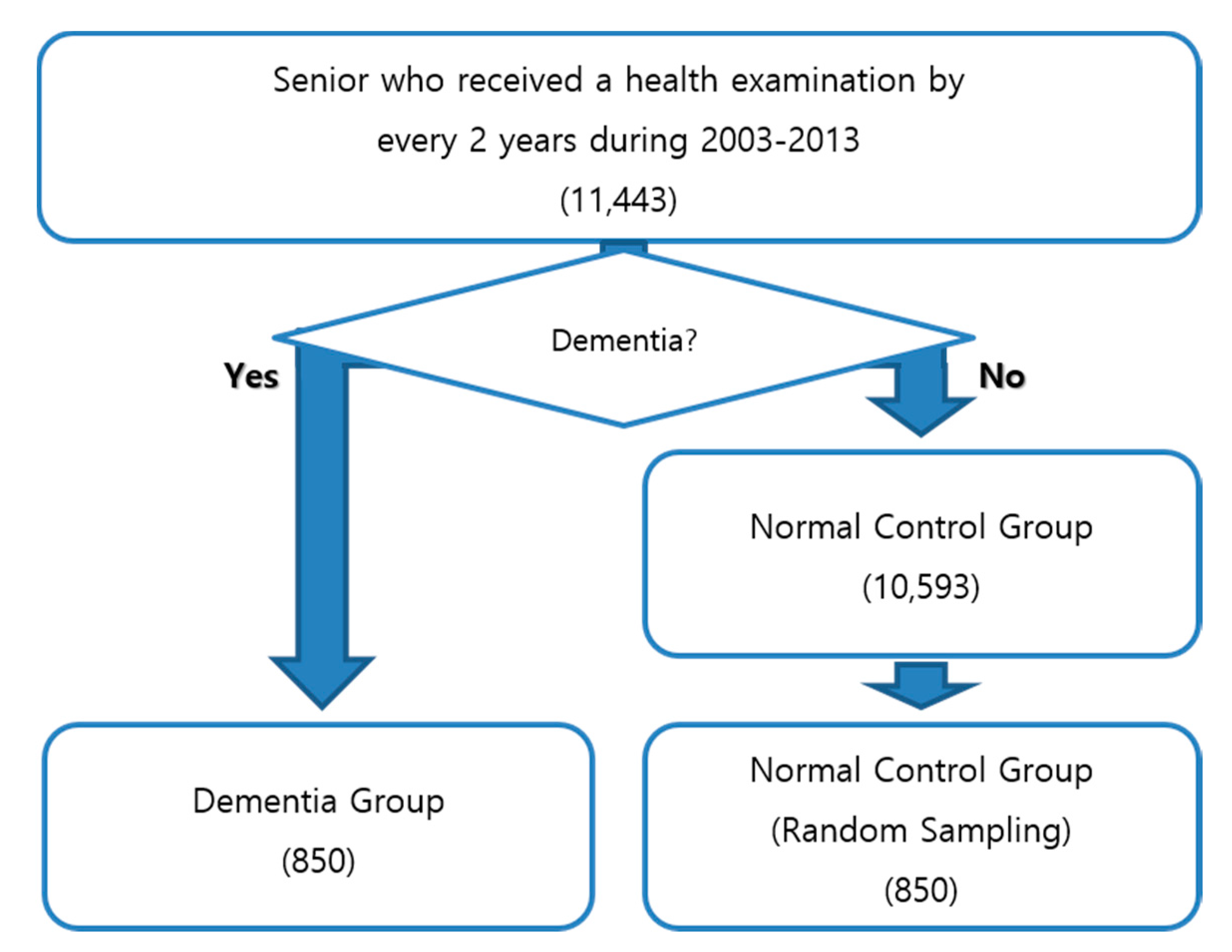
4.1. Sampling
4.2. Experimental Setting
4.3. Evaluation
4.4. Results
5. Discussion
- (1)
- KNHIS-SC DB is a database of records created by doctors’ medical service activities. The diagnosis rate of dementia in Korea is 73.6% (as of 2015) [43], which is higher than in some countries of in Europe (44–67%) [44]. As for the dementia diagnosis rate, the proposed approach does not appear to demonstrate a good performance. The proposed approach used a gold standard data in Korea.
- (2)
- The proposed approach considered the NC based on only a diagnostic history of dementia. However, the elderly may have symptoms or diseases related with dementia. Such cases have ambiguous attributes to classify DM and NC, and may have a negative effect on the performance of dementia prediction. Therefore, the proposed approach involved an extra experiment excluding NC who had dementia-related symptoms or diseases in their medical history. The experimental result showed 81.4% F-measure (89.3 precision and 74.8 recall). Compared with the previous experiments (including NC who have dementia-related symptoms or diseases), dementia prediction slightly increased performance (+0.5% F-measure). This was because the attributes of NC became clearly different from DM.
- (3)
- Since the current approach used the personal history of dementia, the results may be biased. However, the approach tried to follow the same method with the same data with human physicians. As for the reference, the experiment excluding dementia features demonstrated 73.3% accuracy, 77.2% precision, 66.1% recall, and 71.2% F-measure.
- (4)
- Since the data from the KNHIS-SC DB in the proposed approach are newly released, there has been no previous work to handle it. To compare with the proposed method and the previous methods, the baseline experiment used only data from a particular year, and other experiments used personal medical history to prove the effectiveness of personal medical history.
- (5)
- The proposed approach dealt with personal medical history according to year, i.e., if a person has a medical record for such a disease at least one time during the corresponding year, the proposed approach considered him or her as having that disease in one total year. Thus, the current method to deal with personal medical history is not sophisticated and it needs to handle personal medical history by the month, week, or day for a more specific dementia prediction.
- (6)
- In addition, the proposed approach did not consider disease phase, but considered whether a person had certain diseases or not. To analyze disease patterns more exquisitely, we will develop and apply an algorithm that can allocate the phase for any diseases based on drug dosage and dose schedule screening.
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Ethics
References
- Anand, P.; Singh, B. A review on cholinesterase inhibitors for Alzheimer’s disease. Arch. Pharmacol. Res. 2013, 36, 375–399. [Google Scholar] [CrossRef] [PubMed]
- Prince, M.; Wimo, A.; Guerchet, M.; Ali, G.C.; Wu, Y.T.; Prina, M. World Alzheimer Report 2015. The Global Impact of Dementia. An Analysis of Prevalence, Incidence, Cost & Trends; Alzheimer’s Disease International: London, UK, 2015. [Google Scholar]
- Shin, S.A.; Choo, D.S.; Han, T.H.; Park, J.H.; Kim, J.H.; Bang, J.H. NHIS Big Data and Health Services-Consolidated Ageing Well Strategy in Korea. In Proceedings of the 1st International Conference on Information and Communication Technologies for Ageing Well and e-Health (ICT4AgeingWell 2015), Lisbon, Portugal, 20–22 May 2015; pp. 143–148. [Google Scholar]
- Lloyd-Sherlock, P.; McKee, M.; Ebrahim, S.; Gorman, M.; Greengross, S.; Prince, M.; Ferrucci, L. Population ageing and health. Lancet 2012, 379, 1295–1296. [Google Scholar] [CrossRef]
- Ministry of Health & Welfare. Nationwide Study on the Prevalence of Dementia in Korean Elders 2012; Ministry of Health & Welfare: Sejong, Korea, 2013.
- Kim, S. Analysis on Management Policies for the Dementia; National Assembly Budget Office: Seoul, Korea, 2014.
- Prince, M.; Bryce, R.; Ferri, C. World Alzheimer Report 2011. The Benefits of Early Diagnosis and Intervention; Alzheimer’s Disease International: London, UK, 2011. [Google Scholar]
- Byeon, H.; Lee, Y.; Lee, S.Y.; Lee, K.S.; Moon, S.Y.; Kim, H.; Choi, S.H. Association of alcohol drinking with verbal and visuospatial memory impairment in older adults: Clinical Research Center for Dementia of South Korea (CREDOS) study. Int. Psychogeriatr. 2015, 27, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Herrett, E.; Gallagher, A.M.; Bhaskaran, K.; Forbes, H.; Mathur, R.; van Staa, T.; Smeeth, L. Data resource profile: Clinical practice research datalink (CPRD). Int. J. Epidemiol. 2015, 44, 827–836. [Google Scholar] [CrossRef] [PubMed]
- Jesano, R. Free drug information sources on the Web: Government sites. J. Hosp. Librariansh. 2010, 10, 145–151. [Google Scholar] [CrossRef]
- Broniatowski, D.A.; Paul, M.J.; Dredze, M. National and local influenza surveillance through Twitter: An analysis of the 2012–2013 influenza epidemic. PLoS ONE 2013, 8, e83672. [Google Scholar] [CrossRef] [PubMed]
- Feldman, S.; Hanover, J.; Burghard, C.; Schubmehl., D. Unlocking the power of unstructured data. IDC Health Insights 2012, HI235064, 1–10. [Google Scholar]
- Hwang, D.; Kim, S.; Choi, H.; Oh, I.H.; Kim, B.S.; Choi, H.R.; Won, C.W. Calcium-Channel Blockers and Dementia Risk in Older Adults-National Health Insurance Service-Senior Cohort (2002–2013). Circ. J. 2016, 80, 2336–2342. [Google Scholar] [CrossRef] [PubMed]
- National Health Insurance Sharing Service. Available online: https://nhiss.nhis.or.kr (accessed on 15 May 2017).
- Lee, S.; Kawachi, I.; Berkman, L.F.; Grodstein, F. Education, other socioeconomic indicators, and cognitive function. Am. J. Epidemiol. 2003, 157, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Brust, J. Ethanol and cognition: Indirect effects, neurotoxicity and neuroprotection: A review. Int. J. Environ. Res. Public Health 2010, 7, 1540–1557. [Google Scholar] [CrossRef] [PubMed]
- Gillum, R.F.; Kwagyan, J.; Obisesan, T.O. Smoking, cognitive function and mortality in a US national cohort study. Int. J. Environ. Res. Public Health 2011, 8, 3628–3636. [Google Scholar] [CrossRef] [PubMed]
- Kisby, G.E.; Spencer, P.S. Is neurodegenerative disease a long-latency response to early-life genotoxin exposure? Int. J. Environ. Res. Public Health 2011, 8, 3889–3921. [Google Scholar] [CrossRef] [PubMed]
- Ngandu, T.; Lehtisalo, J.; Levälahti, E.; Laatikainen, T.; Lindström, J.; Peltonen, M.; Solomon, A.; Ahtiluoto, S.; Antikainen, R.; Hänninen, T.; et al. Recruitment and baseline characteristics of participants in the finnish geriatric intervention study to prevent cognitive impairment and disability (FINGER)—A randomized controlled lifestyle trial. Int. J. Environ. Res. Public Health 2014, 11, 9345–9360. [Google Scholar] [CrossRef] [PubMed]
- Ojo, O.; Brooke, J. Evaluating the association between diabetes, cognitive decline and dementia. Int. J. Environ. Res. Public Health 2015, 12, 8281–8294. [Google Scholar] [CrossRef] [PubMed]
- Shim, Y.S.; Yang, D.W.; Yoon, B.; Lee, Y.; Hong, C.H.; Seo, S.W.; Kim, S.Y. Clinical predictors for mild cognitive impairment progression in a Korean cohort. Dement. Neurocogn. Disord. 2016, 15, 68–74. [Google Scholar] [CrossRef]
- Walters, K.; Hardoon, S.; Petersen, I.; Iliffe, S.; Omar, R.Z.; Nazareth, I.; Rait, G. Predicting dementia risk in primary care: Development and validation of the Dementia Risk Score using routinely collected data. BMC Med. 2016, 14, 6. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Factors predicting perioperative delirium and acute exacerbation of behavioral and psychological symptoms of dementia based on admission data in elderly patients with proximal femoral fracture: A retrospective study. Geriatr. Gerontol. Int. 2016, 16, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Allen, G.I.; Amoroso, N.; Anghel, C.; Balagurusamy, V.; Bare, C.J.; Beaton, D.; Bellotti, R.; Bennett, D.A.; Boehme, K.L.; Boutros, P.C.; et al. Crowdsourced estimation of cognitive decline and resilience in Alzheimer’s disease. Alzheimer Dement. 2016, 12, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Panwar, B.; Dodge, H.H.; Li, H.; Hampstead, B.M.; Albin, R.L.; Paulson, H.L.; Guan, Y. COMPASS: A computational model to predict changes in MMSE scores 24-months after initial assessment of Alzheimer’s disease. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Stephan, B.C.; Tzourio, C.; Auriacombe, S.; Amieva, H.; Dufouil, C.; Alpérovitch, A.; Kurth, T. Usefulness of data from magnetic resonance imaging to improve prediction of dementia: Population based cohort study. BMJ 2015, 350, h2863. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, F.; Epelbaum, S.; Coley, N.; Cantet, C.; Ousset, P.J.; Hampel, H.; Bakardjian, H.; Lista, S.; Vellas, B.; Dubois, B.; et al.; et al. Prediction of Alzheimer’s Disease Dementia: Data from the GuidAge Prevention Trial. J. Alzheimer’s Dis. 2015, 48, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Nowrangi, M.A.; Rosenberg, P.B.; Leoutsakos, J.M.S. Subtle changes in daily functioning predict conversion from normal to mild cognitive impairment or dementia: An analysis of the NACC database. Int. Psychogeriatr. 2016, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ellendt, S. Predicting Stability of Mild Cognitive Impairment (MCI): Findings of a Community Based Sample. Curr. Alzheimer Res. 2016, 14, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Seong, S.C.; Kim, Y.Y.; Khang, Y.H.; Park, J.H.; Kang, H.J.; Lee, H.; Do, C.H.; Song, J.S.; Hyon Bang, J.; Ha, S.; et al. Data Resource Profile: The National Health Information Database of the National Health Insurance Service in South Korea. Int. J. Epidemiol. 2016, 46, 799–800. [Google Scholar]
- Statistics Korea. Population Statistics Based on Resident Registration. Available online: http://kosis.kr/eng/statisticsList/statisticsList_01List.jsp?vwcd=MT_ETITLE&parentId=A (accessed on 17 August 2017).
- Kim, S.Y.; Kim, S.; Choi, S.E.; Kim, B.S.; Choi, H.R.; Hwang, D.; Won, C.W. Number of daily antihypertensive drugs and the risk of osteoporotic fractures in older hypertensive adults: National health insurance service–Senior cohort. J. Cardiol. 2017, 70, 80–85. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems; World Health Organization: Geneva, Switzerland, 2004; Volume 1. [Google Scholar]
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems; World Health Organization: Geneva, Switzerland, 2004; Volume 2. [Google Scholar]
- Ministry of Health and Welfare. Available online: http://www.mohw.go.kr (accessed on 15 May 2017).
- Korea Institute for Health and Social Affairs. The Social Burden and Care Management for People with Dementia; Goreo C&P: Seoul, Korea, 2016. [Google Scholar]
- Kang, I.O.; Lee, S.Y.; Kim, S.Y.; Park, C.Y. Economic cost of dementia patients according to the limitation of the activities of daily living in Korea. Int. J. Geriatr. Psychiatry 2007, 22, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Korean Dementia Association. Dementia a Clinical, 3rd ed.; Freeacademy: Seoul, Korea, 2012. [Google Scholar]
- Fatima, M.; Pasha, M. Survey of Machine Learning Algorithms for Disease Diagnostic. J. Intell. Learn. Syst. Appl. 2017, 9, 1–16. [Google Scholar] [CrossRef]
- Noble, W.S. What is a support vector machine? Nat. Biotechnol. 2006, 24, 1565–1567. [Google Scholar] [CrossRef] [PubMed]
- Evered, L.; Scott, D.A.; Silbert, B. Cognitive decline associated with anesthesia and surgery in the elderly does this contribute to dementia prevalence. Curr. Opin. Psychiatry 2017, 30, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Wolters, F.J.; Zonneveld, H.I.; Hofman, A.; van der Lugt, A.; Koudstaal, P.J.; Vernooij, M.W.; Ikram, M.A. Cerebral Perfusion and the Risk of Dementia A Population-Based Study. Circulation 2017. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Dementia. Seongnam, Korea. Available online: https://www.nid.or.kr /info/today_list.aspx (accessed on 17 August 2017).
- Organisation for Economic Co-Operation and Development. Addressing Dementia the OECD Response; OECD Publishing: Paris, France, 2015; Available online: http://www.oecd.org/health/addressing-dementia-9789264231726-en.htm (accessed on 17 August 2017).

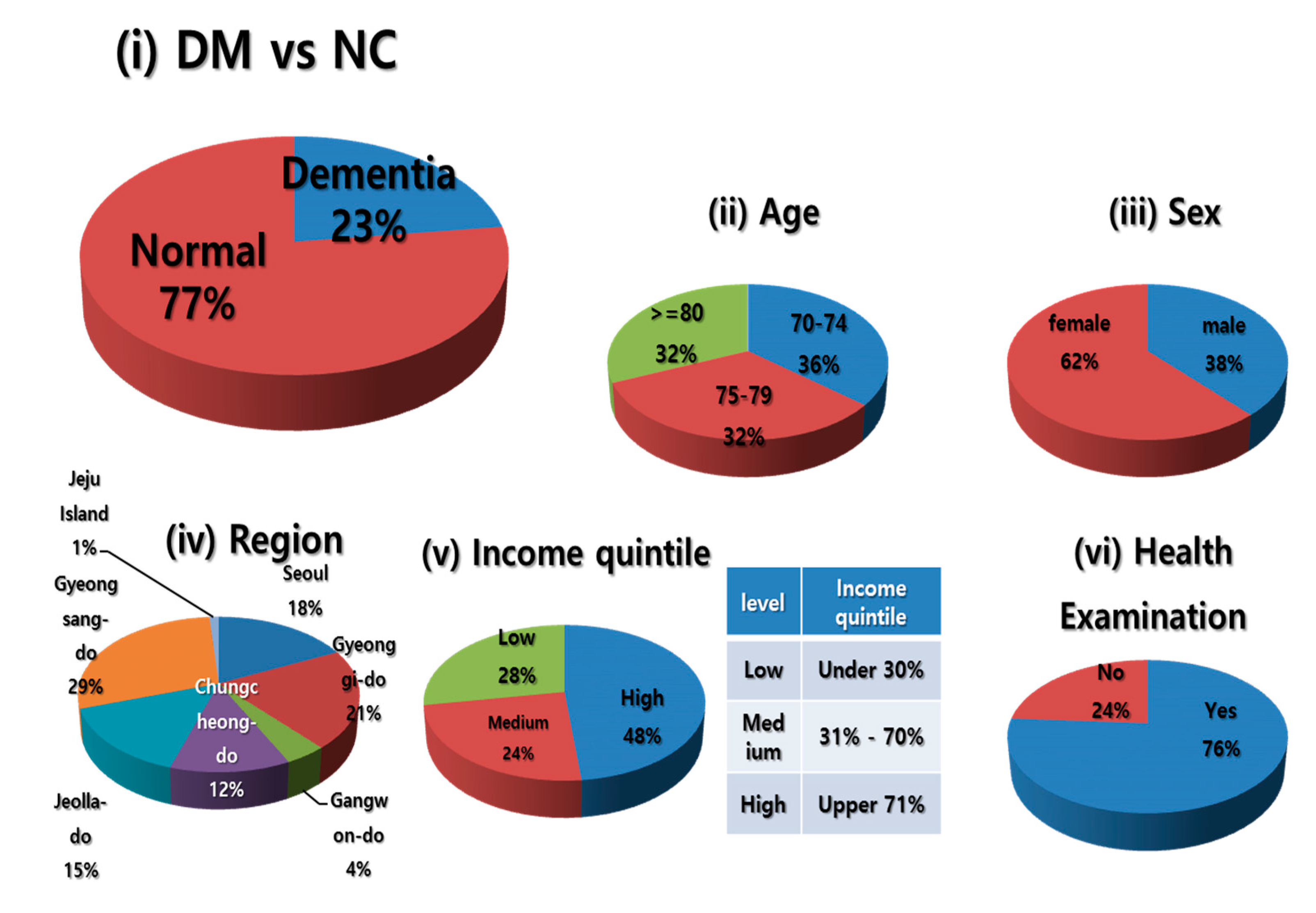
| Item | Classification | KNHIS-SC DB (%) | Population Statistics Based on Resident Registration (%) |
|---|---|---|---|
| Sex | Male | 38 | 39 |
| Female | 62 | 61 | |
| Sum | 100 | 100 | |
| Age | 70–74 | 36 | 42 |
| 75–79 | 32 | 30 | |
| 80+ | 32 | 28 | |
| Sum | 100 | 100 | |
| Region | Seoul | 18 | 18 |
| Gyeonggi-do | 21 | 23 | |
| Gyeongsang-do | 29 | 28 | |
| Jeolla-do | 15 | 14 | |
| Gangwon-do | 4 | 4 | |
| Chungcheong-do | 12 | 12 | |
| Jeju Island | 1 | 1 | |
| Sum | 100 | 100 |
| Year | Total | Age, Years | |||||
|---|---|---|---|---|---|---|---|
| 60–64 | 65–69 | 70–74 | 75–79 | 80–85 | 85+ | ||
| 2002 | 558,147 | 196,116 | 147,361 | 97,657 | 61,217 | 35,215 | 20,581 |
| 2003 | 557,195 | 161,261 | 157,155 | 105,339 | 66,978 | 41,351 | 25,111 |
| 2004 | 539,278 | 122,144 | 164,629 | 111,768 | 71,595 | 42,769 | 26,373 |
| 2005 | 521,967 | 85,776 | 167,293 | 120,415 | 76,092 | 44,336 | 28,055 |
| 2006 | 504,417 | 46,113 | 173,449 | 128,909 | 80,206 | 45,795 | 29,945 |
| 2007 | 487,460 | 186,057 | 135,631 | 84,926 | 47,983 | 32,863 | |
| 2008 | 470,005 | 151,331 | 142,687 | 89,766 | 50,578 | 35,643 | |
| 2009 | 452,631 | 114,559 | 149,864 | 96,030 | 54,763 | 37,415 | |
| 2010 | 436,395 | 80,560 | 153,026 | 104,029 | 58,905 | 39,875 | |
| 2011 | 422,171 | 43,571 | 160,221 | 112,412 | 63,101 | 42,866 | |
| 2012 | 405,614 | 172,606 | 118,931 | 67,529 | 46,548 | ||
| 2013 | 388,493 | 140,503 | 125,795 | 71,993 | 50,202 | ||
| PIE-DB | Demographic information (sex, age, area of residence) |
| Death related information (date of death, cause of death) | |
| Types of health insurance (health insurance subscribers/medical benefits) | |
| Socio-economic level and other information (income quintile, disability registration information) | |
| MT-DB | Medical institution information |
| Medical care benefit costs | |
| Information on medical subjects and medical diseases | |
| Details of medical examination, treatment, surgery and other acts, treatment materials, etc. | |
| Detailed diseases history | |
| In-house/outpatient prescription drug prescription | |
| GHE-DB | Major test results such as body measurement, blood test |
| Results of interview about history, lifestyle | |
| Balance, bone density test, depression, cognitive function test result | |
| MCI-DB | Medical utilization, medical institution type and establishment division, medical institution, local information |
| Information on the number of beds, doctors, equipment, etc. | |
| LCI-DB | Long-term care application and judgment result |
| Doctor’s note | |
| Billing statement | |
| Basic information on long-term care facilities |
| No. | Feature | Class | |
|---|---|---|---|
| Normal | Abnormal | ||
| 1 | Body mass index (kg/m2) | 0–29 | 30–300 |
| 2 | Waist circumference (cm) | Male: 50–90, Female: 50–85 | Male: 90–130, Female: 85–130 |
| 3 | Blood pressure highest (mmHg) | 60–139 | 140–400 |
| 4 | Blood pressure lowest (mmHg) | 40–89 | 90–250 |
| 5 | Blood glucose before meals (g/dL) | 25–125 | 126–999 |
| 6 | Total cholesterol (mg/dL) | 40–229 | 230–999 |
| 7 | Hemoglobin (g/dL) | Male: 12–16.5, Female: 10–15.5 | Male: 0–12, Female: 0–10 |
| 8 | Urine protein | Negative | Positive |
| 9 | Serum GOT (U/L) | 0–50 | 51–999 |
| 10 | Serum GPT (U/L) | 0–45 | 46–999 |
| 11 | Gamma GTP (U/L) | Male: 11–77, Female: 8–45 | Male: 78–999, Female: 46–999 |
| Disease Classification | Disease |
|---|---|
| Degenerative diseases | (1) Parkinson’s disease, (2) Huntington’s disease, (3) Pick’s disease, (4) Progressive palsy, (5) Multiple system atrophy, (6) Genetic disorder, (7) Motor neuron disease, (8) Multiple sclerosis |
| Deficiency disease | (1) Thiamine (B1): Wernicke’s encephalopathy, (2) Vitamin B12: Pernicious anemia, (3) Nicotinic acid: Pellagra |
| Endocrine and organ dysfunction diseases | (1) Hypothyroidism, (2) Lack of adrenal function and Cushing’s syndrome, (3) Hypothyroidism and hypertrophy, (4) Loss of renal function, (5) Liver failure, (6) Loss of lung function |
| Tumor disease | (1) Primary brain tumor, (2) Paraneoplastic limbic encephalitis, (3) Metastatic brain tumor |
| Chronic infection | (1) Human immunodeficiency virus, (2) Neurosyphilis, (3) Parvovirus, (4) Prion disease, (5) Tuberculosis, (6) Fungi, (7) Protozoa, (8) Sarcoidosis, (9) Whipple’s disease |
| Head trauma and extensive brain injury | (1) Chronic subdural hematoma, (2) Anoxic syndrome, (3) Encephalitis, (4) Normal pressure hydrocephalus |
| Toxic disease | (1) Drug and drug addiction, (2) Alcoholism, (3) Heavy metal poisoning, (4) Organic toxins |
| Mental illness | (1) Depression, (2) Schizophrenia, (3) Conversion reaction |
| Other | (1) Vasculitis, (2) CADASIL, (3) Acute intermittent porphyria, (4) Repeated seizures |
| Year | DB | Features |
|---|---|---|
| 2013 | PIE-DB | (1) Sex, (2) Age, (3) Income quintile |
| GHE-DB | (1) Height, (2) Weight, (3) Body mass index, (4) Waist, (5) Blood pressure highest, (6) Blood pressure lowest, (7) Blood sugar before meals, (8) Total cholesterol, (9) Hemoglobin, (10) Urine protein, (11) Serum GOT, (12) Serum GPT, (13) Gamma GTP, (14) History of personal illness: Stroke, Heart disease, High blood pressure, Diabetes, Hyperlipidemia, Phthisis, Cancer, (15) History of family illness: Stroke, Heart disease, High blood pressure, Diabetes, cancer | |
| MT-DB | Personal disease history diagnosis by every year |
| DB | Features | |
|---|---|---|
| PIE-DB | Increasing/decreasing compared to 2013 [Income quintile] | |
| Class changing compared to 2013 [Income quintile] | ||
| GHE-DB | Features of baseline | Increasing/decreasing compared to 2013 [(1) Height, (2) Weight, (3) Body mass index, (4) Waist, (5) Blood pressure highest, (6) Blood pressure lowest, (7) Blood sugar before meals, (8) Total cholesterol, (9) Hemoglobin, (10) Urine protein, (11) Serum GOT, (12) Serum GPT, (13) Gamma GTP, (14) History of personal illness: Stroke, Heart disease, High blood pressure, Diabetes, Hyperlipidemia, Phthisis, cancer, (15) History of family illness: Stroke, Heart disease, High blood pressure, Diabetes, Cancer] |
| Class changing compared to 2013 [(1) Body mass index, (2) Waist, (3) Blood pressure highest, (4) Blood pressure lowest, (5) Blood sugar before meals, (6) Total cholesterol, (7) Hemoglobin, (8) Urine protein, (9) Serum GOT, (10) Serum GPT, (11) Gamma GTP, (12) History of personal illness: Stroke, Heart disease, High blood pressure, Diabetes, Hyperlipidemia, Phthisis, Cancer, (13) History of family illness: Stroke, Heart disease, High blood pressure, Diabetes, Cancer] | ||
| MT-DB | Personal disease history diagnosis by every year | |
| Baseline | Longitudinal Model 1 | |||||
|---|---|---|---|---|---|---|
| Year | 2013 | 2003–2013 | 2005–2013 | 2007–2013 | 2009–2013 | 2011–2013 |
| Number of features | 55 | 366 | 314 | 262 | 210 | 158 |
| True positive | 614 | 638 | 613 | 630 | 648 | 648 |
| False positive | 317 | 285 | 280 | 282 | 274 | 292 |
| True negative | 533 | 565 | 570 | 568 | 576 | 558 |
| False negative | 236 | 212 | 237 | 220 | 202 | 202 |
| Accuracy (%) | 67.5 | 70.8 | 69.6 | 70.5 | 72.0 | 70.9 |
| Precision (%) | 66.0 | 69.1 | 68.6 | 69.1 | 70.3 | 68.9 |
| Recall (%) | 72.2 | 75.1 | 72.1 | 74.1 | 76.2 | 76.2 |
| F-measure (%) | 69.0 | 72.0 | 70.3 | 71.5 | 73.1 | 72.4 |
| Baseline | Longitudinal Model 2 | |||||
|---|---|---|---|---|---|---|
| Year | 2013 | 2003–2013 | 2005–2013 | 2007–2013 | 2009–2013 | 2011–2013 |
| Number of features | 55 | 709 | 559 | 409 | 259 | 113 |
| True positive | 614 | 623 | 625 | 633 | 619 | 611 |
| False positive | 317 | 78 | 79 | 82 | 69 | 67 |
| True negative | 533 | 772 | 771 | 768 | 781 | 783 |
| False negative | 236 | 227 | 225 | 217 | 231 | 239 |
| Accuracy (%) | 67.5 | 82.1 | 82.1 | 82.4 | 82.4 | 82.0 |
| Precision (%) | 66.0 | 88.9 | 88.8 | 88.5 | 90.0 | 90.1 |
| Recall (%) | 72.2 | 73.3 | 73.5 | 74.5 | 72.8 | 71.9 |
| F-measure (%) | 69.0 | 80.3 | 80.4 | 80.9 | 80.5 | 80.0 |
| DB | Primary Disease Group | Type | Features |
|---|---|---|---|
| MT-DB | E; Endocrine, nutritional and metabolic diseases (8) | Known | (1) Other disorders of pancreatic internal secretion, (2) Vitamin D deficiency, (3) Other disorders of thyroid, (4) Malnutrition-related diabetes mellitus |
| Newly detected | (1) Hyperfunction of pituitary gland, (2) Hypofunction and other disorders of pituitary gland, (3) Other disorders of adrenal gland, (4) Unspecified protein-energy malnutrition | ||
| F; Mental and behavioural disorders (13) | Known | (1) Dementia in Alzheimer’s disease, (2) Vascular dementia, (3) Mental and behavioural disorders due to use of alcohol, (4) Acute and transient psychotic disorders, (5) Unspecified nonorganic psychosis, (6) Unspecified dementia, (7) Bipolar affective disorder, (8) Depressive episode, (9) Delirium, not induced by alcohol and other psychoactive substances, (10) Eating disorders, (11) Psychological and behavioural factors associated with disorders or diseases classified elsewhere, (12) Other mental disorders due to brain damage and dysfunction and to physical disease, (13) Schizophrenia | |
| G; Diseases of the nervous system (17) | Known | (1) Parkinson’s disease, (2) Secondary parkinsonism, (3) Parkinsonism in diseases classified elsewhere, (4) Alzheimer’s disease, (5) Other degenerative diseases of nervous system NEC, (6) Epilepsy, (7) Status epilepticus, (8) Transient cerebral ischaemic attacks and related syndromes, (9) Vascular syndromes of brain in cerebro- vascular diseases, (10) Disorders of other cranial nerves, (11) Hemiplegia, (12) Paraplegia and tetraplegia, (13) Other paralytic syndromes, (14) Hydrocephalus, (15) Other disorders of brain, (16) Other disorders of nervous system, NEC, (17) Other disorders of nervous system in diseases classified elsewhere | |
| I; Diseases of the circulatory system (7) | Known | (1) Hypertensive renal disease, (2) Subsequent myocardial infarction, (3) Cerebral infarction, (4) Cerebrovascular disorders in diseases classified elsewhere, (5) Sequelae of cerebrovascular disease, (6) Aortic aneurysm and dissection, (7) Stroke, not specified as haemorrhage or infarction | |
| N; Diseases of the genitourinary system (8) | Known | (1) Acute nephritic syndrome, (2) Chronic kidney disease, (3) Glomerular disorders in diseases classified elsewhere | |
| Newly detected | (1) Calculus of lower urinary tract, (2) Urethral stricture, (3) Other disorders of male genital organs, (4) Inflammatory disease of uterus, except cervix, (5) Polyp of female genital tract | ||
| M; Diseases of the musculoskeletal system and connective tissue (3) | Newly detected | (1) Kyphosis and lordosis, (2) Spinal osteochondrosis (3) Psoriatic and enteropathic arthropathies | |
| R; Symptoms, signs and abnormal clinical and laboratory findings, NEC (13) | Known | (1) Faecal incontinence, (2) Abnormalities of gait and mobility, (3) Unspecified urinary incontinence, (4) Somnolence, stupor and coma, (5) Other symptoms and signs involving cognitive functions and awareness, (6) Other symptoms and signs involving general sensations and perceptions, (7) Symptoms and signs involving appearance and behavior, | |
| Newly detected | (1) Ascites, (2) Retention of urine, (3) Voice disturbances, (4) Malaise and fatigue, (5) Enlarged lymph nodes, (6) Systemic Inflammatory Response Syndrome | ||
| S; Injury, poisoning and certain other consequences of external causes (6) | Known | (1) Fracture of skull and facial bones, (2) Open wound of thorax, (3) Injury of other and unspecified intrathoracic organs, (4) Open wound of forearm, (5) Fracture at wrist and hand level, (6) Injury of muscle and tendon at hip and thigh level | |
| GHE-DB | - | - | (1) Total cholesterol, (2) Hemoglobin, (3) Serum GOT, (4) Serum GPT, (5) Gamma GTP |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Chun, H.-W.; Kim, S.; Coh, B.-Y.; Kwon, O.-J.; Moon, Y.-H. Longitudinal Study-Based Dementia Prediction for Public Health. Int. J. Environ. Res. Public Health 2017, 14, 983. https://doi.org/10.3390/ijerph14090983
Kim H, Chun H-W, Kim S, Coh B-Y, Kwon O-J, Moon Y-H. Longitudinal Study-Based Dementia Prediction for Public Health. International Journal of Environmental Research and Public Health. 2017; 14(9):983. https://doi.org/10.3390/ijerph14090983
Chicago/Turabian StyleKim, HeeChel, Hong-Woo Chun, Seonho Kim, Byoung-Youl Coh, Oh-Jin Kwon, and Yeong-Ho Moon. 2017. "Longitudinal Study-Based Dementia Prediction for Public Health" International Journal of Environmental Research and Public Health 14, no. 9: 983. https://doi.org/10.3390/ijerph14090983
APA StyleKim, H., Chun, H.-W., Kim, S., Coh, B.-Y., Kwon, O.-J., & Moon, Y.-H. (2017). Longitudinal Study-Based Dementia Prediction for Public Health. International Journal of Environmental Research and Public Health, 14(9), 983. https://doi.org/10.3390/ijerph14090983




