Increased Risk of Drug-Induced Hyponatremia during High Temperatures
Abstract
:1. Introduction
2. Method
2.1. Statistical Analysis
2.2. Ethical Considerations
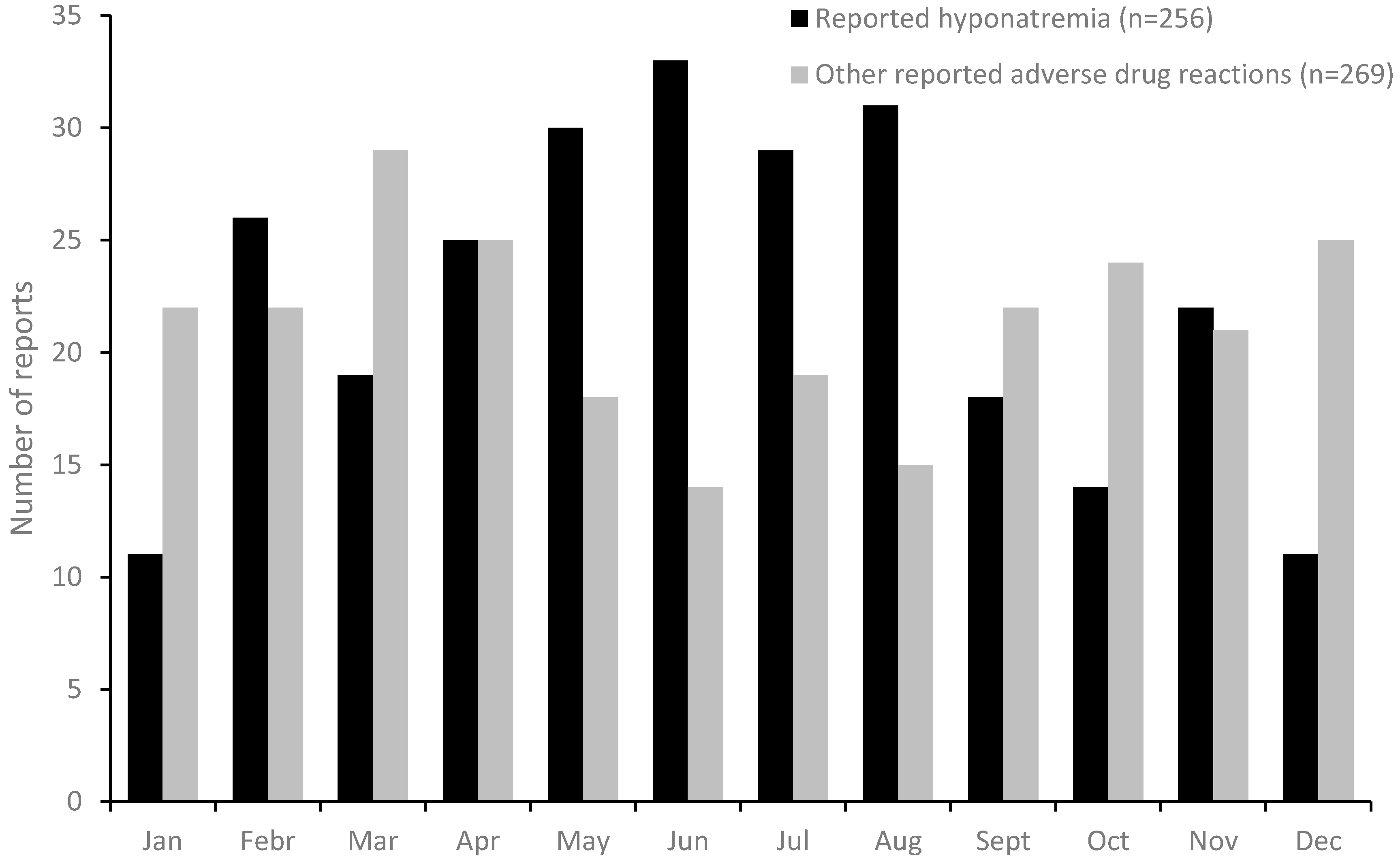
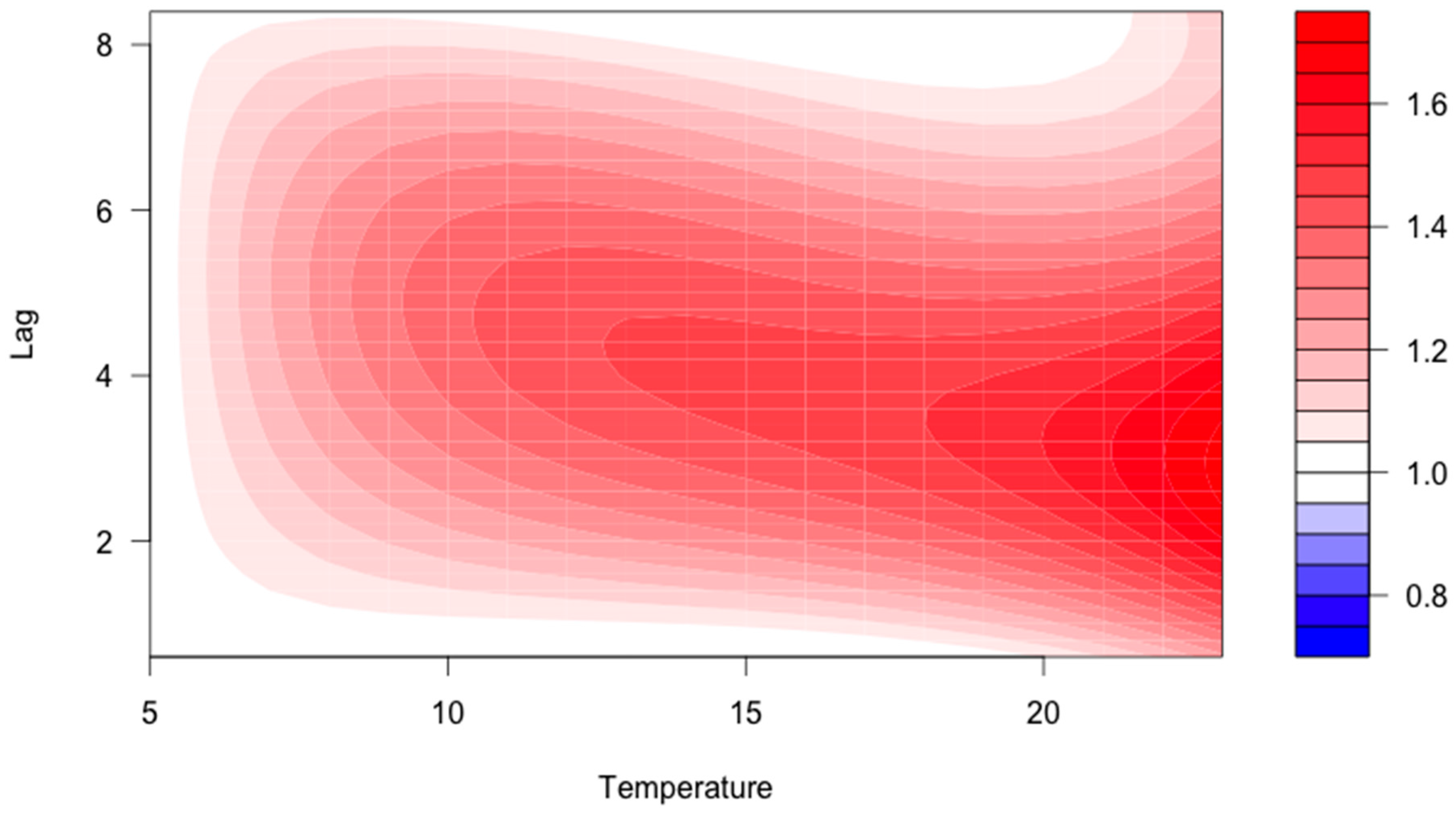
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Astrom, D.O.; Forsberg, B.; Rocklov, J. Heat wave impact on morbidity and mortality in the elderly population: A review of recent studies. Maturitas 2011, 69, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Hajat, S.; Kosatky, T. Heat-related mortality: A review and exploration of heterogeneity. J. Epidemiol. Community Health 2010, 64, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Josseran, L.; Fouillet, A.; Caillere, N.; Brun-Ney, D.; Ilef, D.; Brucker, G.; Medeiros, H.; Astagneau, P. Assessment of a syndromic surveillance system based on morbidity data: Results from the Oscour network during a heat wave. PLoS ONE 2010, 5, e11984. [Google Scholar] [CrossRef] [PubMed]
- Pfortmueller, C.A.; Funk, G.C.; Leichtle, A.B.; Fiedler, G.M.; Schwarz, C.; Exadaktylos, A.K.; Lindner, G. Electrolyte disorders and in-hospital mortality during prolonged heat periods: A cross-sectional analysis. PLoS ONE 2014, 9, e92150. [Google Scholar] [CrossRef] [PubMed]
- Mohan, S.; Gu, S.; Parikh, A.; Radhakrishnan, J. Prevalence of hyponatremia and association with mortality: Results from NHANES. Am. J. Med. 2013, 126, 1127–1137 e1. [Google Scholar] [CrossRef] [PubMed]
- Waikar, S.S.; Mount, D.B.; Curhan, G.C. Mortality after hospitalization with mild, moderate, and severe hyponatremia. Am. J. Med. 2009, 122, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Liamis, G.; Milionis, H.; Elisaf, M. A review of drug-induced hyponatremia. Am. J. Kidney Dis. 2008, 52, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Rodenburg, E.M.; Hoorn, E.J.; Ruiter, R.; Lous, J.J.; Hofman, A.; Uitterlinden, A.G.; Stricker, B.H.; Visser, L.E. Thiazide-associated hyponatremia: A population-based study. Am. J. Kidney Dis. 2013, 62, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Ambrosi, P.; Villani, P.; Bouvenot, G. Hyponatremia in elderly patients treated with thiazide diuretics and incited to drink abundantly during the heat wave. La Presse Med. 2004, 33, 535–536. [Google Scholar] [CrossRef]
- Martin-Latry, K.; Goumy, M.P.; Latry, P.; Gabinski, C.; Begaud, B.; Faure, I.; Verdoux, H. Psychotropic drugs use and risk of heat-related hospitalisation. Eur. Psychiatry 2007, 22, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Chow, K.M.; Szeto, C.C.; Kwan, B.C.; Li, P.K. Influence of climate on the incidence of thiazide-induced hyponatraemia. Int. J. Clin. Pract. 2007, 61, 449–452. [Google Scholar] [CrossRef] [PubMed]
- World health organization. International Drug Monitoring: the Role of National Centres. Report of a WHO meeting. World Health Organ. Tech. Rep. Ser. 1972, 498, 1–25. [Google Scholar]
- World Health Organisation. International Drug Monitoring: The Role of the Hospital; Technical Report Series; WHO: Geneva, Switzerland, 1972. [Google Scholar]
- Gasparrini, A. Distributed lag linear and non-linear models in R: The package dlnm. J. Stat. Softw. 2011, 43, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Hajat, S.; O’Connor, M.; Kosatsky, T. Health effects of hot weather: From awareness of risk factors to effective health protection. Lancet 2010, 375, 856–863. [Google Scholar] [CrossRef]
- Yong, S.A.; Reid, D.A.; Tobin, A.E. Heatwave hyponatraemia: A case series at a single Victorian tertiary centre during January 2014. Intern Med. J. 2015, 45, 211–214. [Google Scholar] [CrossRef] [PubMed]
- Rocklöv, J.; Forsberq, B.; Ebi, K.; Bellander, T. Susceptibility to mortality related to temperature and heat and cold wave duration in the population of Stockholm County, Sweden. Glob. Health Action 2014. [Google Scholar] [CrossRef] [PubMed]
- Sommet, A.; Durrieu, G.; Lapeyre-Mestre, M.; Montastruc, J.L.; Association of French PharmacoVigilance Centres. A comparative study of adverse drug reactions during two heat waves that occurred in France in 2003 and 2006. Pharmacoepidemiol. Drug Saf. 2012, 21, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Verbalis, J.G. Disorders of body water homeostasis. Best Pract. Res. Clin. Endocrinol. Metab. 2003, 17, 471–503. [Google Scholar] [CrossRef]
- Upadhyay, A.; Jaber, B.L.; Madias, N.E. Incidence and prevalence of hyponatremia. Am. J. Med. 2006, 119, S30–S35. [Google Scholar] [CrossRef] [PubMed]
- Hazell, L.; Shakir, S.A. Under-reporting of adverse drug reactions : A systematic review. Drug Saf. 2006, 29, 385–396. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jönsson, A.K.; Lövborg, H.; Lohr, W.; Ekman, B.; Rocklöv, J. Increased Risk of Drug-Induced Hyponatremia during High Temperatures. Int. J. Environ. Res. Public Health 2017, 14, 827. https://doi.org/10.3390/ijerph14070827
Jönsson AK, Lövborg H, Lohr W, Ekman B, Rocklöv J. Increased Risk of Drug-Induced Hyponatremia during High Temperatures. International Journal of Environmental Research and Public Health. 2017; 14(7):827. https://doi.org/10.3390/ijerph14070827
Chicago/Turabian StyleJönsson, Anna K, Henrik Lövborg, Wolfgang Lohr, Bertil Ekman, and Joacim Rocklöv. 2017. "Increased Risk of Drug-Induced Hyponatremia during High Temperatures" International Journal of Environmental Research and Public Health 14, no. 7: 827. https://doi.org/10.3390/ijerph14070827
APA StyleJönsson, A. K., Lövborg, H., Lohr, W., Ekman, B., & Rocklöv, J. (2017). Increased Risk of Drug-Induced Hyponatremia during High Temperatures. International Journal of Environmental Research and Public Health, 14(7), 827. https://doi.org/10.3390/ijerph14070827





