Association between Pesticide Profiles Used on Agricultural Fields near Maternal Residences during Pregnancy and IQ at Age 7 Years
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population: CHAMACOS Cohort
2.2. Characterization of Prenatal Pesticide Use near Maternal Residences
2.3. Assessment of Cognitive Development
2.4. Covariates
2.5. Statistical Analysis
2.5.1. Bayesian Profile Regression
2.5.2. Characterizing Pesticide-Use Profile Clusters
2.5.3. Pesticide Importance by Bayesian Kernel Machine Regression
2.6. Mapping Pesticide Use Profiles
3. Results
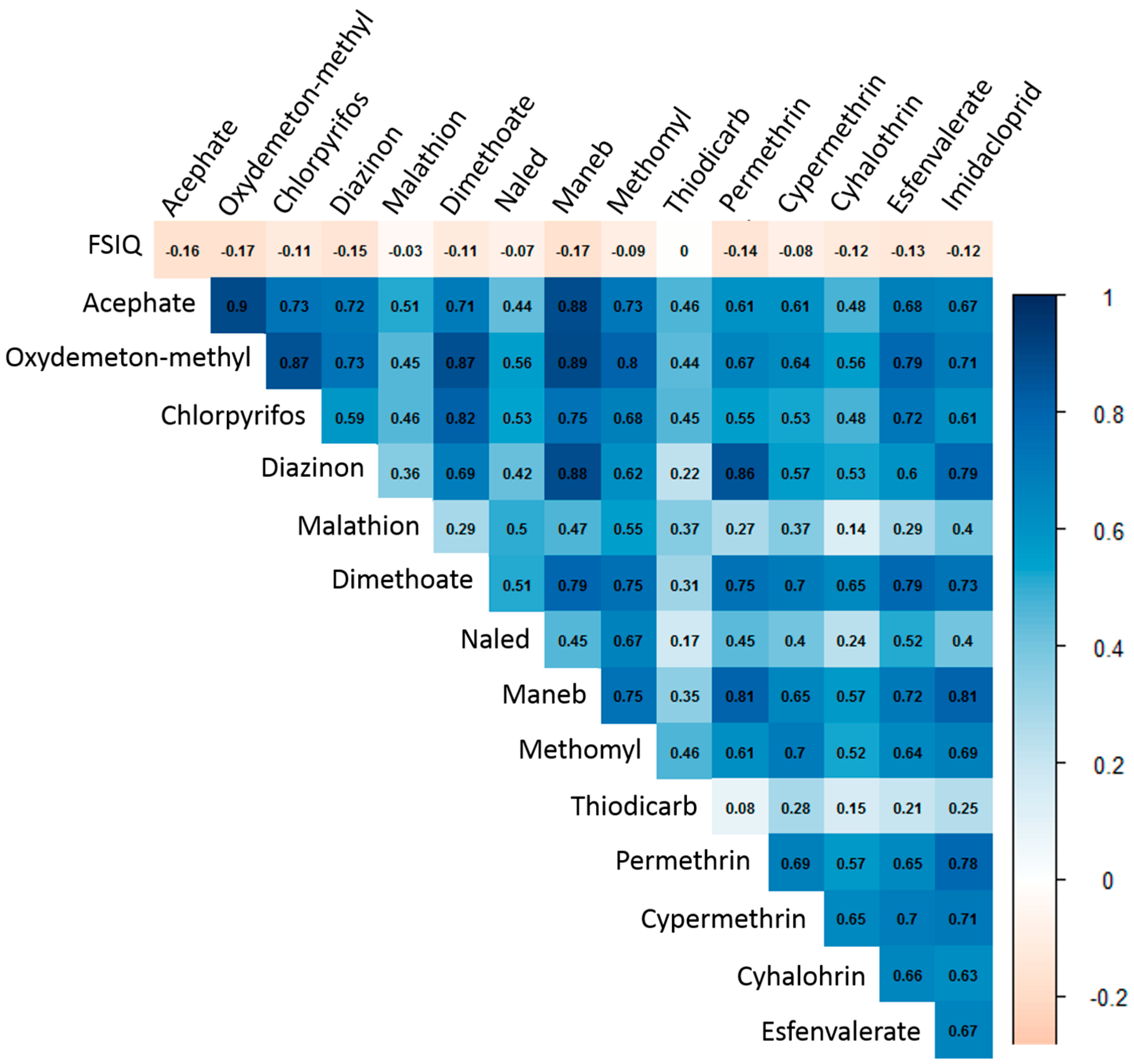
3.1. Pesticide Use Summaries
3.2. Pesticide Use Profile Clusters
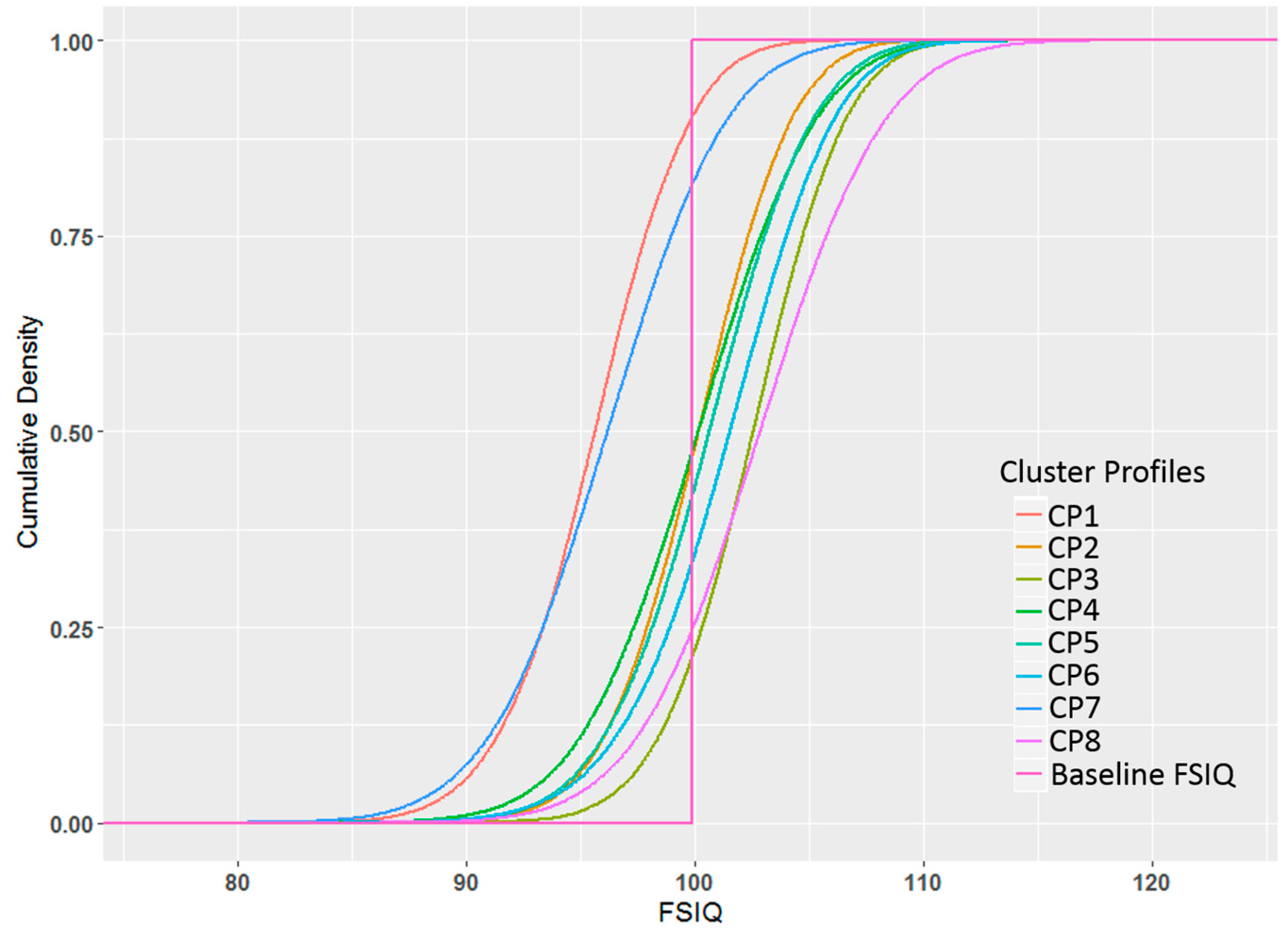
3.3. FSIQ Results
3.4. Comparison of BPR Clustering When Excluding the Outcome
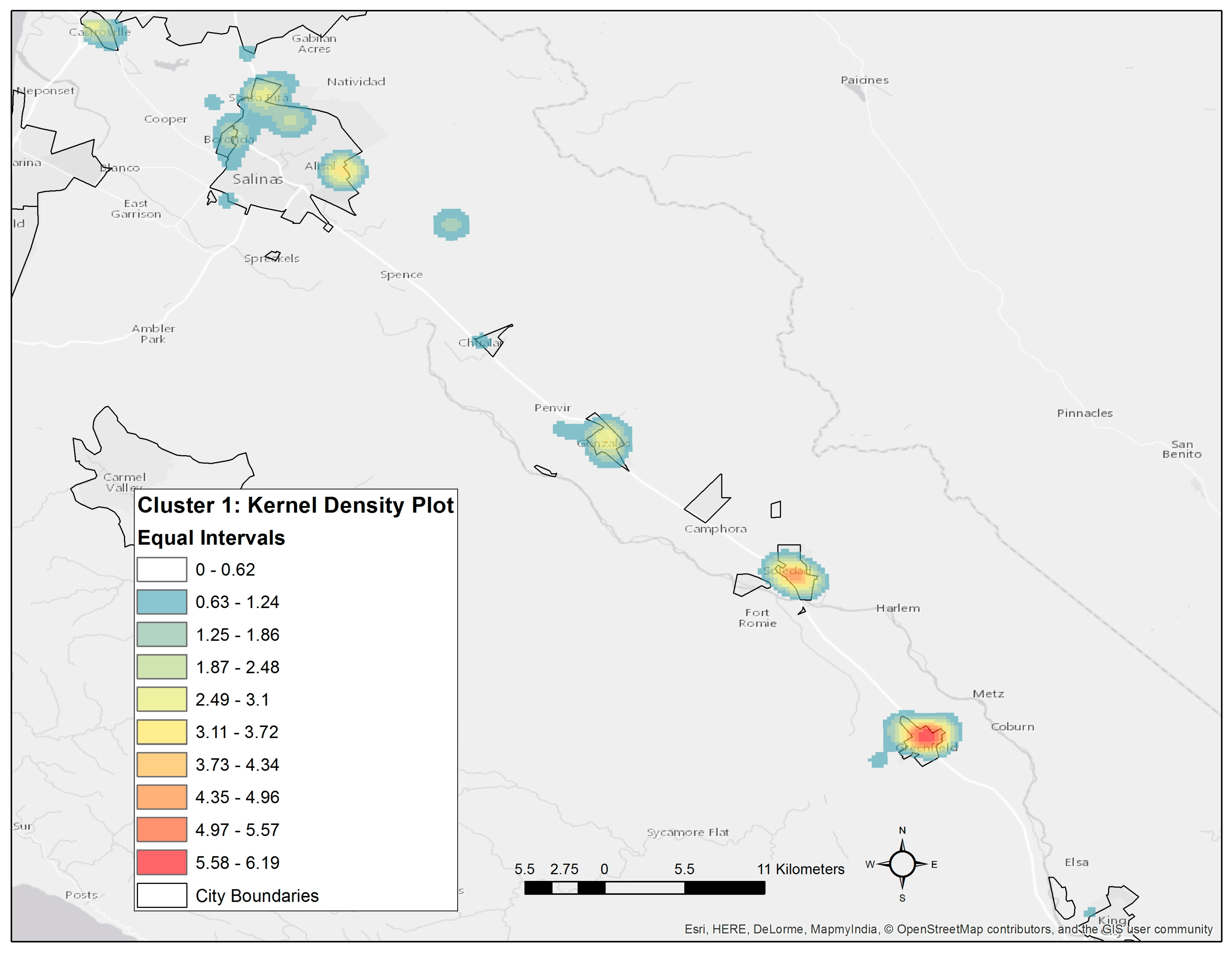
3.5. Spatial Patterns of Risk and Potential Exposure
3.6. Variable Selection from BKMR
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Engel, S.M.; Berkowitz, G.S.; Barr, D.B.; Teitelbaum, S.L.; Siskind, J.; Meisel, S.J.; Wetmur, J.G.; Wolff, M.S. Prenatal Organophosphate metabolite and organochlorine levels and performance on the brazelton neonatal behavioral assessment scale in a multiethnic pregnancy cohort. Am. J. Epidemiol. 2007, 165, 1397–1404. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Kogut, K.; Huen, K.; Harley, K.G.; Bouchard, M.; Bradman, A.; Boyd-Barr, D.; Johnson, C.; Holland, N. Organophosphate pesticide exposure, PON1, and neurodevelopment in school-age children from the CHAMACOS study. Environ. Res. 2014, 134, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, F.W.; Harley, K.G.; Kogut, K.; Chevrier, J.; Mora, A.M.; Sjödin, A.; Eskenazi, B. Prenatal DDT and DDE exposure and child IQ in the CHAMACOS cohort. Environ. Int. 2015, 85, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Horton, M.K.; Kahn, L.G.; Perera, F.; Barr, D.B.; Rauh, V. Does the home environment and the sex of the child modify the adverse effects of prenatal exposure to chlorpyrifos on child working memory? Neurotoxicol. Teratol. 2012, 34, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Lovasi, G.S.; Quinn, J.W.; Rauh, V.A.; Perera, F.P.; Andrews, H.F.; Garfinkel, R.; Hoepner, L.; Whyatt, R.; Rundle, A. Chlorpyrifos exposure and urban residential environment characteristics as determinants of early childhood neurodevelopment. Am. J. Public Health 2011, 101, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Rauh, V.A.; Perera, F.P.; Horton, M.K.; Whyatt, R.M.; Bansal, R.; Hao, X.; Liu, J.; Barr, D.B.; Slotkin, T.A.; Peterson, B.S. Brain anomalies in children exposed prenatally to a common organophosphate pesticide. Proc. Natl. Acad. Sci. USA 2012, 109, 7871–7876. [Google Scholar] [CrossRef] [PubMed]
- Rauh, V.; Arunajadai, S.; Horton, M.; Perera, F.; Hoepner, L.; Barr, D.B.; Whyatt, R. Seven-year neurodevelopmental scores and prenatal exposure to chlorpyrifos, a common agricultural pesticide. Environ. Health Perspect. 2011, 119, 1196–1201. [Google Scholar] [CrossRef] [PubMed]
- Ribas-Fito, N.; Torrent, M.; Carrizo, D.; Munoz-Ortiz, L.; Julvez, J.; Grimalt, J.O.; Sunyer, J. In utero exposure to background concentrations of DDT and cognitive functioning among preschoolers. Am. J. Epidemiol. 2006, 164, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Viel, J.-F.; Warembourg, C.; Le Maner-Idrissi, G.; Lacroix, A.; Limon, G.; Rouget, F.; Monfort, C.; Durand, G.; Cordier, S.; Chevrier, C. Pyrethroid insecticide exposure and cognitive developmental disabilities in children: The PELAGIE mother–child cohort. Environ. Int. 2015, 82, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Young, J.G.; Eskenazi, B.; Gladstone, E.A.; Bradman, A.; Pedersen, L.; Johnson, C.; Barr, D.B.; Furlong, C.E.; Holland, N.T. Association between in utero organophosphate pesticide exposure and abnormal reflexes in neonates. NeuroToxicology 2005, 26, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, M.F.; Chevrier, J.; Harley, K.G.; Kogut, K.; Vedar, M.; Calderon, N.; Trujillo, C.; Johnson, C.; Bradman, A.; Barr, D.B.; et al. Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environ. Health Perspect. 2011, 119, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Gunier, R.B.; Bradman, A.; Harley, K.G.; Kogut, K.; Eskenazi, B. Prenatal residential proximity to agricultural pesticide use and IQ in 7-year-old children. Environ. Health Perspect. 2016. [Google Scholar] [CrossRef] [PubMed]
- Roberts, E.M.; English, P.B.; Grether, J.K.; Windham, G.C.; Somberg, L.; Wolff, C. Maternal residence near agricultural pesticide applications and autism spectrum disorders among children in the California Central Valley. Environ. Health Perspect. 2007, 115, 1482–1489. [Google Scholar] [CrossRef] [PubMed]
- Zaunbrecher, V.; Hattis, D.; Melnick, R.; Kegley, S.; Malloy, T.; Froines, J. Exposure and Interaction: The Potential Health Impacts of Using Multiple Pesticides; UCLA Sustainable Technology & Policy Progra: Los Angeles, CA, USA, 2016. [Google Scholar]
- Moser, V.C.; Simmons, J.E.; Gennings, C. Neurotoxicological interactions of a five-pesticide mixture in preweanling rats. Toxicol. Sci. Off. J. Soc. Toxicol. 2006, 92, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Rizzati, V.; Briand, O.; Guillou, H.; Gamet-Payrastre, L. Effects of pesticide mixtures in human and animal models: An update of the recent literature. Chem. Biol. Interact. 2016, 254, 231–246. [Google Scholar] [CrossRef] [PubMed]
- Hernández, A.F.; Parrón, T.; Tsatsakis, A.M.; Requena, M.; Alarcón, R.; López-Guarnido, O. Toxic effects of pesticide mixtures at a molecular level: Their relevance to human health. Toxicology 2013, 307, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Moser, V.C.; Padilla, S.; Simmons, J.E.; Haber, L.T.; Hertzberg, R.C. Impact of chemical proportions on the acute neurotoxicity of a mixture of seven carbamates in preweanling and adult rats. Toxicol. Sci. 2012, 129, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Bobb, J.F.; Valeri, L.; Claus Henn, B.; Christiani, D.C.; Wright, R.O.; Mazumdar, M.; Godleski, J.J.; Coull, B.A. Bayesian kernel machine regression for estimating the health effects of multi-pollutant mixtures. Biostatistics 2015, 16, 493–508. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.M.; Gennings, C.; Hauser, R.; Webster, T.F. What Can Epidemiological studies tell us about the impact of chemical mixtures on human health? Environ. Health Perspect. 2016, 124, A6–A9. [Google Scholar] [CrossRef] [PubMed]
- Coker, E.; Liverani, S.; Ghosh, J.K.; Jerrett, M.; Beckerman, B.; Li, A.; Ritz, B.; Molitor, J. Multi-pollutant exposure profiles associated with term low birth weight in Los Angeles County. Environ. Int. 2016, 91, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Coull, B.A.; Bobb, J.F.; Wellenius, G.A.; Kioumourtzoglou, M.-A.; Mittleman, M.A.; Koutrakis, P.; Godleski, J.J. Part 1. Statistical learning methods for the effects of multiple air pollution constituents. Res. Rep. Health Eff. Inst. 2015, 183, 5–50. [Google Scholar]
- Molitor, J.; Papathomas, M.; Jerrett, M.; Richardson, S. Bayesian profile regression with an application to the national survey of children’s health. Biostatistics 2010, 11, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Oakes, M.; Baxter, L.; Long, T.C. Evaluating the application of multipollutant exposure metrics in air pollution health studies. Environ. Int. 2014, 69, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Park, E.S.; Symanski, E.; Han, D.; Spiegelman, C. Part 2. Development of enhanced statistical methods for assessing health effects associated with an unknown number of major sources of multiple air pollutants. Res. Rep. Health Eff. Inst. 2015, 183, 51–113. [Google Scholar]
- Pirani, M.; Best, N.; Blangiardo, M.; Liverani, S.; Atkinson, R.W.; Fuller, G.W. Analysing the health effects of simultaneous exposure to physical and chemical properties of airborne particles. Environ. Int. 2015, 79, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Zanobetti, A.; Austin, E.; Coull, B.A.; Schwartz, J.; Koutrakis, P. Health effects of multi-pollutant profiles. Environ. Int. 2014, 71, 13–19. [Google Scholar] [CrossRef] [PubMed]
- CDPR. Summary of Pesticide Use Report Data 2014 Indexed by Chemical; California Environmental Protection Agency Department of Pesticide Regulation: Sacramento, CA, USA, 2016.
- CDPR Pesticide Use Reporting. Available online: http://www.cdpr.ca.gov/docs/pur/purmain.htm (accessed on 8 July 2016).
- Gunier, R.B.; Ward, M.H.; Airola, M.; Bell, E.M.; Colt, J.; Nishioka, M.; Buffler, P.A.; Reynolds, P.; Rull, R.P.; Hertz, A.; et al. Determinants of agricultural pesticide concentrations in carpet dust. Environ. Health Perspect. 2011, 119, 970–976. [Google Scholar] [CrossRef] [PubMed]
- Harnly, M.; McLaughlin, R.; Bradman, A.; Anderson, M.; Gunier, R. Correlating agricultural use of organophosphates with outdoor air concentrations: A particular concern for children. Environ. Health Perspect. 2005, 113, 1184–1189. [Google Scholar] [CrossRef] [PubMed]
- Harnly, M.E.; Bradman, A.; Nishioka, M.; McKone, T.E.; Smith, D.; McLaughlin, R.; Kavanagh-Baird, G.; Castorina, R.; Eskenazi, B. Pesticides in dust from homes in an agricultural area. Environ. Sci. Technol. 2009, 43, 8767–8774. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Johnson, B.; Segawa, R. Empirical relationship between use, area, and ambient air concentration of methyl bromide. J. Environ. Qual. 2005, 34, 420–428. [Google Scholar] [PubMed]
- Bradman, A.; Eskenazi, B.; Barr, D.B.; Bravo, R.; Castorina, R.; Chevrier, J.; Kogut, K.; Harnly, M.E.; McKone, T.E. Organophosphate urinary metabolite levels during pregnancy and after delivery in women living in an agricultural community. Environ. Health Perspect. 2005, 113, 1802–1807. [Google Scholar] [CrossRef] [PubMed]
- Bradman, A.; Castorina, R.; Boyd Barr, D.; Chevrier, J.; Harnly, M.E.; Eisen, E.A.; McKone, T.E.; Harley, K.; Holland, N.; Eskenazi, B. Determinants of organophosphorus pesticide urinary metabolite levels in young children living in an agricultural community. Int. J. Environ. Res. Public Health 2011, 8, 1061–1083. [Google Scholar] [CrossRef] [PubMed]
- Bigelow, J.L.; Dunson, D.B. Bayesian semiparametric joint models for functional predictors. J. Am. Stat. Assoc. 2009, 104, 26–36. [Google Scholar] [CrossRef]
- Chung, Y.; Dunson, D.B. Nonparametric bayes conditional distribution modeling with variable selection. J. Am. Stat. Assoc. 2009, 104, 1646–1660. [Google Scholar] [CrossRef] [PubMed]
- Dunson, D.B. Nonparametric Bayes local partition models for random effects. Biometrika 2009, 96, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Dunson, D.B.; Herring, A.H.; Siega-Riz, A.M. Bayesian inference on changes in response densities over predictor clusters. J. Am. Stat. Assoc. 2008, 103, 1508–1517. [Google Scholar] [CrossRef]
- Eskenazi, B.; Bradman, A.; Castorina, R. Exposures of children to organophosphate pesticides and their potential adverse health effects. Environ. Health Perspect. 1999, 107, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Rowe, C.; Gunier, R.; Bradman, A.; Harley, K.G.; Kogut, K.; Parra, K.; Eskenazi, B. Residential proximity to organophosphate and carbamate pesticide use during pregnancy, poverty during childhood, and cognitive functioning in 10-year-old children. Environ. Res. 2016, 150, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Lonare, M.; Kumar, M.; Raut, S.; Badgujar, P.; Doltade, S.; Telang, A. Evaluation of imidacloprid-induced neurotoxicity in male rats: A protective effect of curcumin. Neurochem. Int. 2014, 78, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Gill, K.D.; Flora, G.; Pachauri, V.; Flora, S.J.S. Neurotoxicity of organophosphates and carbamates. In Anticholinesterase Pesticides; Satoh, T., Gupta, R.C., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 237–265. [Google Scholar]
- Costello, S.; Cockburn, M.; Bronstein, J.; Zhang, X.; Ritz, B. Parkinson’s disease and residential exposure to maneb and paraquat from agricultural applications in the central valley of California. Am. J. Epidemiol. 2009, 169, 919–926. [Google Scholar] [CrossRef] [PubMed]
- DeMicco, A.; Cooper, K.R.; Richardson, J.R.; White, L.A. Developmental neurotoxicity of pyrethroid insecticides in zebrafish embryos. Toxicol. Sci. 2010, 113, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, A.F.M.; Strickland, J.D.; Crofton, K.M.; Gennings, C.; Shafer, T.J. Effects of an environmentally-relevant mixture of pyrethroid insecticides on spontaneous activity in primary cortical networks on microelectrode arrays. NeuroToxicology 2016. [Google Scholar] [CrossRef] [PubMed]
- Wechsler, D. Wechsler Intelligence Scale for Children; Hardcount Assessment: San Antonio, TX, USA, 2003. [Google Scholar]
- Woodcock, R.; Johnson, M. Woodcock-Johnson Psycho-Educational Battery-Revised; Riverside: Itasca, IL, USA, 1990. [Google Scholar]
- Sugland, B.W.; Zaslow, M.; Smith, J.R.; Brooks-Gunn, J.; Coates, D.; Blumenthal, C.; Moore, K.A.; Griffin, T.; Bradley, R. The Early Childhood HOME inventory and HOME-short form in differing racial/ethnic groups: Are there differences in underlying structure, internal consistency of subscales, and patterns of prediction? J. Fam. Issues 1995, 16, 632–663. [Google Scholar] [CrossRef]
- Dunn, M.; Dunn, L.M. Peabody Picture Vocabulary Test—Revised; AGS: Circle Pines, MN, USA, 1981. [Google Scholar]
- Gilks, W.R.; Richardson, S.; Spiegelhalter, D.J. Markov Chain Monte Carlo in Practice; Gilks, W.R., Richardson, S., Spiegelhalter, D.J., Eds.; Chapman & Hall: Boca Raton, FL, USA, 1998. [Google Scholar]
- Jain, S.; Neal, R.M. Splitting and merging components of a nonconjugate Dirichlet process mixture model. Bayesian Anal. 2007, 2, 445–472. [Google Scholar] [CrossRef]
- Liverani, S.; Hastie, D.I.; Azizi, L.; Papathomas, M.; Richardson, S. PReMiuM: An R package for profile regression mixture models using dirichlet processes. J. Stat. Softw. 2015, 64, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Hastie, D.I.; Liverani, S.; Azizi, L.; Richardson, S.; Stücker, I. A semi-parametric approach to estimate risk functions associated with multi-dimensional exposure profiles: Application to smoking and lung cancer. BMC Med. Res. Methodol. 2013, 13, 129. [Google Scholar] [CrossRef] [PubMed]
- Papathomas, M.; Molitor, J.; Richardson, S.; Riboli, E.; Vineis, P. Examining the joint effect of multiple risk factors using exposure risk profiles: Lung cancer in nonsmokers. Environ. Health Perspect. 2010, 119, 84–91. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Environmental Epidemiology; National Academy Press: Washington, DC, USA, 1997; Volume 2. [Google Scholar]
- Papathomas, M.; Molitor, J.; Hoggart, C.; Hastie, D.; Richardson, S. Exploring data from genetic association studies using bayesian variable selection and the dirichlet process: Application to searching for gene × Gene patterns: Searching for gene × gene patterns. Genet. Epidemiol. 2012, 36, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Greenland, S. Methods for epidemiologic analyses of multiple exposures: A review and comparative study of maximum-likelihood, preliminary-testing, and empirical-bayes regression. Stat. Med. 1993, 12, 717–736. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.C.; Witte, J.S.; Greenland, S. Dissecting effects of complex mixtures: Who? Afraid of informative priors? Epidemiology 2007, 18, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Liverani, S.; Lavigne, A.; Blangiardo, M. Modelling collinear and spatially correlated data. Spat. Spatio-Temporal Epidemiol. 2016, 18, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Rull, R.P.; Ritz, B.; Shaw, G.M. Neural tube defects and maternal residential proximity to agricultural pesticide applications. Am. J. Epidemiol. 2006, 163, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Shelton, J.F.; Geraghty, E.M.; Tancredi, D.J.; Delwiche, L.D.; Schmidt, R.J.; Ritz, B.; Hansen, R.L.; Hertz-Picciotto, I. Neurodevelopmental disorders and prenatal residential proximity to agricultural pesticides: The CHARGE study. Environ. Health Perspect. 2014, 122, 1103. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Carmichael, S.L.; Roberts, E.M.; Kegley, S.E.; Padula, A.M.; English, P.B.; Shaw, G.M. Residential agricultural pesticide exposures and risk of neural tube defects and orofacial clefts among offspring in the San Joaquin Valley of California. Am. J. Epidemiol. 2014, 179, 740–748. [Google Scholar] [CrossRef] [PubMed]
- Cedergreen, N. Quantifying synergy: A systematic review of mixture toxicity studies within environmental toxicology. PLoS ONE 2014, 9, e96580. [Google Scholar] [CrossRef] [PubMed]
- Crépet, A.; Héraud, F.; Béchaux, C.; Gouze, M.E.; Pierlot, S.; Fastier, A.; Leblanc, J.C.; Le Hégarat, L.; Takakura, N.; Fessard, V.; et al. The PERICLES research program: An integrated approach to characterize the combined effects of mixtures of pesticide residues to which the French population is exposed. Toxicology 2013, 313, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Crépet, A.; Tressou, J.; Graillot, V.; Béchaux, C.; Pierlot, S.; Héraud, F.; Leblanc, J.C. Identification of the main pesticide residue mixtures to which the French population is exposed. Environ. Res. 2013, 126, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Carlin, D.J.; Rider, C.V.; Woychik, R.; Birnbaum, L.S. Unraveling the health effects of environmental mixtures: An NIEHS priority. Environ. Health Perspect. 2013, 121, a6–a8. [Google Scholar] [CrossRef] [PubMed]
- Colovic, M.B.; Krstic, D.Z.; Lazarevic-Pasti, T.D.; Bondzic, A.M.; Vasic, V.M. Acetylcholinesterase inhibitors: Pharmacology and toxicology. Curr. Neuropharmacol. 2013, 11, 315–335. [Google Scholar] [CrossRef] [PubMed]
- Armon, R.H.; Hänninen, O. Environmental Indicators; Armon, R.H., Hänninen, O., Eds.; Springer: Dordrecht, The Netherlands, 2015. [Google Scholar]
- Vermeire, T.; McPhail, R.; Waters, M. D. Organophosphorous Pesticides in the Environment; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Davidson, C. California Amphibian Declines and Historic Pesticide Use; US Fish and Wildlife Service: Sacramento, CA, USA, 2003; pp. 1–50. [Google Scholar]
- Zhan, Y.; Zhang, M. Spatial and temporal patterns of pesticide use on California almonds and associated risks to the surrounding environment. Sci. Total Environ. 2014, 472, 517–529. [Google Scholar] [CrossRef] [PubMed]
- Curl, C.L.; Beresford, S.A.A.; Fenske, R.A.; Fitzpatrick, A.L.; Lu, C.; Nettleton, J.A.; Kaufman, J.D. Estimating pesticide exposure from dietary intake and organic food choices: The Multi-Ethnic Study of Atherosclerosis (MESA). Environ. Health Perspect. 2015, 123, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Bradman, A.; Kogut, K.; Eisen, E.A.; Jewell, N.P.; Quirós-Alcalá, L.; Castorina, R.; Chevrier, J.; Holland, N.T.; Barr, D.B.; Kavanagh-Baird, G.; et al. Variability of organophosphorous pesticide metabolite levels in spot and 24-h urine samples collected from young children during 1 week. Environ. Health Perspect. 2013, 121, 118–124. [Google Scholar] [PubMed]
- Zhang, X.; Driver, J.H.; Li, Y.; Ross, J.H.; Krieger, R.I. Dialkylphosphates (DAPs) in fruits and vegetables may confound biomonitoring in organophosphorus insecticide exposure and risk assessment. J. Agric. Food Chem. 2008, 56, 10638–10645. [Google Scholar] [CrossRef] [PubMed]

| Pesticide (Type) | Median (IQR) | Mean (SD) |
|---|---|---|
| Oxydemeton-methyl (OP) | 10.59 (1.95, 24.18) | 20.50 (34.89) |
| Acephate (OP) | 9.16 (2.54, 28.90) | 23.59 (41.99) |
| Chlorpyrifos (OP) | 9.83 (1.23, 29.89) | 24.10 (41.16) |
| Diazinon (OP) | 22.22 (11.86, 50.59) | 46.02 (70.47) |
| Malathion (OP) | 1.57 (0, 12.71) | 20.41 (49.58) |
| Dimethoate (OP) | 3.47 (0.84, 16.12) | 11.67 (17.04) |
| Naled (OP) | 0.07 (0, 6.24) | 6.24 (14.62) |
| Maneb (Mn) | 55.20 (24.77, 123.60) | 108.90 (149.56) |
| Methomyl (Carb) | 10.00 (2.50, 28.07) | 25.09 (40.13) |
| Thiodicarb (Carb) | 0.45 (0, 1.67) | 1.67 (3.14) |
| Permethrin (PR) | 2.64 (1.03, 9.73) | 7.34 (10.47) |
| Cypermethrin (PR) | 0.007 (0, 0.47) | 0.48 (1.05) |
| Cyhalothrin (PR) | 0.13 (0.05, 0.42) | 0.40 (0.80) |
| Esfenvalerate (PR) | 0.15 (0.0003, 1.44) | 1.43 (5.17) |
| Imidacloprid (N) | 2.53 (1.37, 3.06) | 5.14 (6.29) |
| Cumulative (Summed) | 164.20 (67.53, 356.20) | 303.00 (395.75) |
| Overall | n | Mean (95% CI) a |
|---|---|---|
| 255 | 103.8 (102.1, 105.6) | |
| Cluster | ||
| CP1 | 59 | 98.6 (95.1, 102.2) |
| CP2 | 52 | 105.0 (101.2, 108.9) |
| CP3 | 50 | 106.7 (102.2, 111.1) |
| CP4 | 17 | 104.4 (97.0, 111.8) |
| CP5 | 43 | 104.8 (101.0, 108.7) |
| CP6 | 3 | 108.3 (84.7, 131.9) |
| CP7 | 18 | 101.4 (94.4, 108.4) |
| CP8 | 13 | 110.0 (102.6, 117.4) |
| Clusters | Adjusted Posterior Mean FSIQ a,b (95% Credible Intervals) | Mean FSIQ_c from FSIQ_CP3 a,c (95% Credible Intervals) | Probability FSIQ_c < FSIQ_CP3 d |
|---|---|---|---|
| CP1 | 95.5 (88.6, 102.0) | −6.9 (−11.7, −2.1) | 0.998 |
| CP2 | 100.1 (93.4, 106.3) | −2.4 (−7.2, 2.5) | 0.83 |
| CP3 | 102.5 (95.8, 108.7) | Ref | Ref |
| CP4 | 100.1 (91.8, 107.9) | −2.4 (−9.0, 4.3) | 0.76 |
| CP5 | 100.6 (93.1, 107.6) | −1.9 (−7.1, 3.3) | 0.77 |
| CP6 | 101.3 (93.2, 108.4) | −1.1 (−7.9, 3.4) | 0.47 |
| CP7 | 96.1 (87.7, 104.2) | −6.4 (−13.1, 0.5) | 0.97 |
| CP8 | 102.8 (94.1, 111.3) | 0.3 (−7.0, 7.8) | 0.47 |
| Clusters Excluding FSIQ Outcome in BPR | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Clusters Including FSIQ Outcome in BPR | CP1 | CP2 | CP3 | CP4 | CP5 | CP6 | CP7 | CP8 | CP9 | Row Total |
| CP1 | 55 | 0 | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 59 |
| CP2 | 0 | 8 | 22 | 0 | 0 | 0 | 4 | 0 | 18 | 52 |
| CP3 | 0 | 0 | 0 | 50 | 0 | 0 | 0 | 0 | 0 | 50 |
| CP4 | 0 | 1 | 0 | 0 | 15 | 0 | 0 | 1 | 0 | 17 |
| CP5 | 0 | 2 | 0 | 0 | 0 | 40 | 0 | 1 | 0 | 43 |
| CP6 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 3 |
| CP7 | 0 | 0 | 0 | 0 | 0 | 0 | 16 | 2 | 0 | 18 |
| CP8 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 10 | 0 | 13 |
| Column Total | 55 | 11 | 29 | 50 | 18 | 40 | 20 | 14 | 18 | 255 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coker, E.; Gunier, R.; Bradman, A.; Harley, K.; Kogut, K.; Molitor, J.; Eskenazi, B. Association between Pesticide Profiles Used on Agricultural Fields near Maternal Residences during Pregnancy and IQ at Age 7 Years. Int. J. Environ. Res. Public Health 2017, 14, 506. https://doi.org/10.3390/ijerph14050506
Coker E, Gunier R, Bradman A, Harley K, Kogut K, Molitor J, Eskenazi B. Association between Pesticide Profiles Used on Agricultural Fields near Maternal Residences during Pregnancy and IQ at Age 7 Years. International Journal of Environmental Research and Public Health. 2017; 14(5):506. https://doi.org/10.3390/ijerph14050506
Chicago/Turabian StyleCoker, Eric, Robert Gunier, Asa Bradman, Kim Harley, Katherine Kogut, John Molitor, and Brenda Eskenazi. 2017. "Association between Pesticide Profiles Used on Agricultural Fields near Maternal Residences during Pregnancy and IQ at Age 7 Years" International Journal of Environmental Research and Public Health 14, no. 5: 506. https://doi.org/10.3390/ijerph14050506
APA StyleCoker, E., Gunier, R., Bradman, A., Harley, K., Kogut, K., Molitor, J., & Eskenazi, B. (2017). Association between Pesticide Profiles Used on Agricultural Fields near Maternal Residences during Pregnancy and IQ at Age 7 Years. International Journal of Environmental Research and Public Health, 14(5), 506. https://doi.org/10.3390/ijerph14050506







