Disease and Health Inequalities Attributable to Air Pollutant Exposure in Detroit, Michigan
Abstract
1. Introduction
1.1. Background
1.2. Objectives
2. Materials and Methods
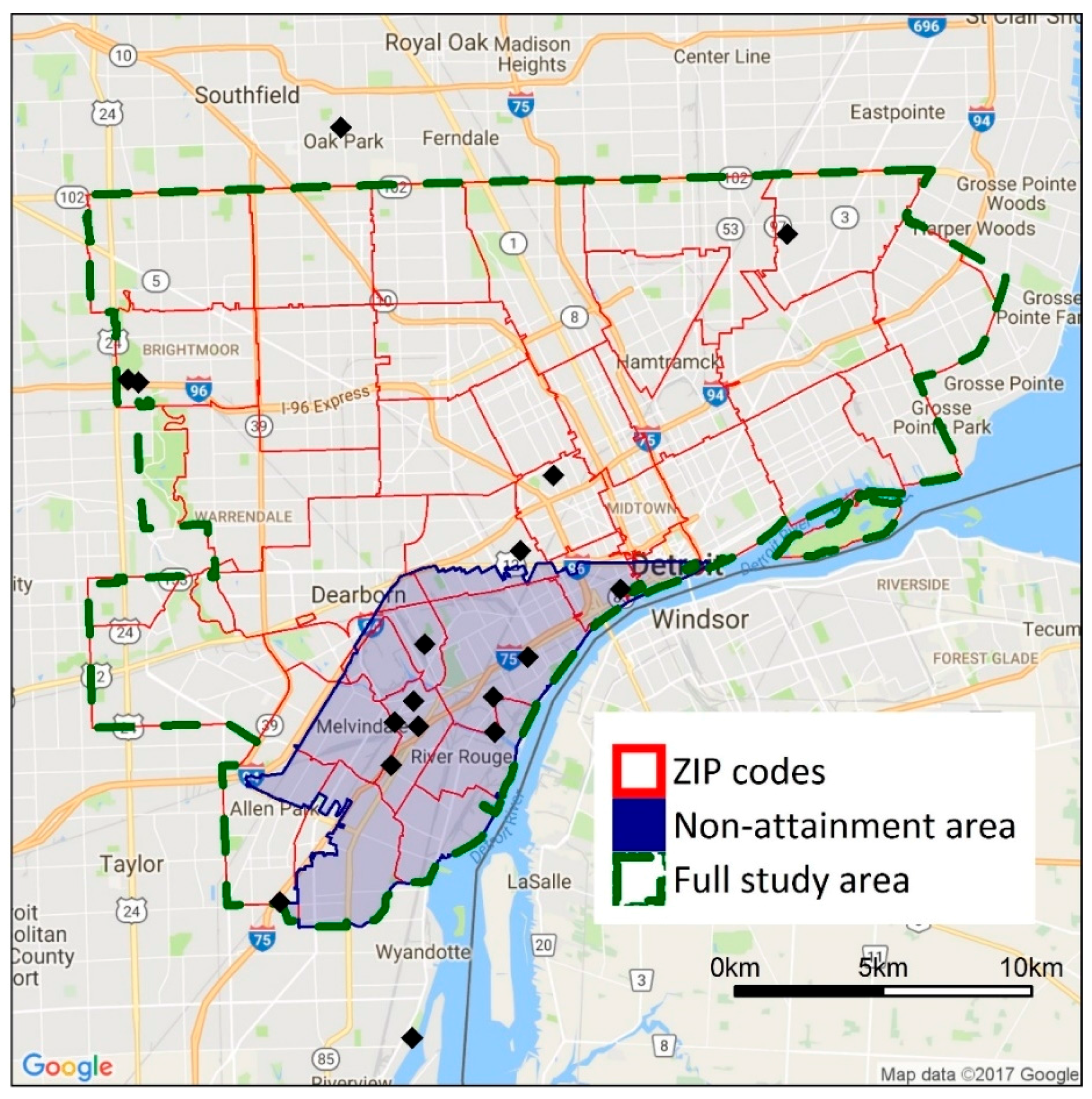
2.1. Study Area, Spatial Resolution, and Study Population
2.2. Health Impact Assessment
2.3. Exposure Assessment
2.4. Apportionment of Exposures to Source Categories
2.5. Inequality Metrics
3. Results
3.1. Daily Population Exposures at the Census Block Level
3.2. Burden of Disease
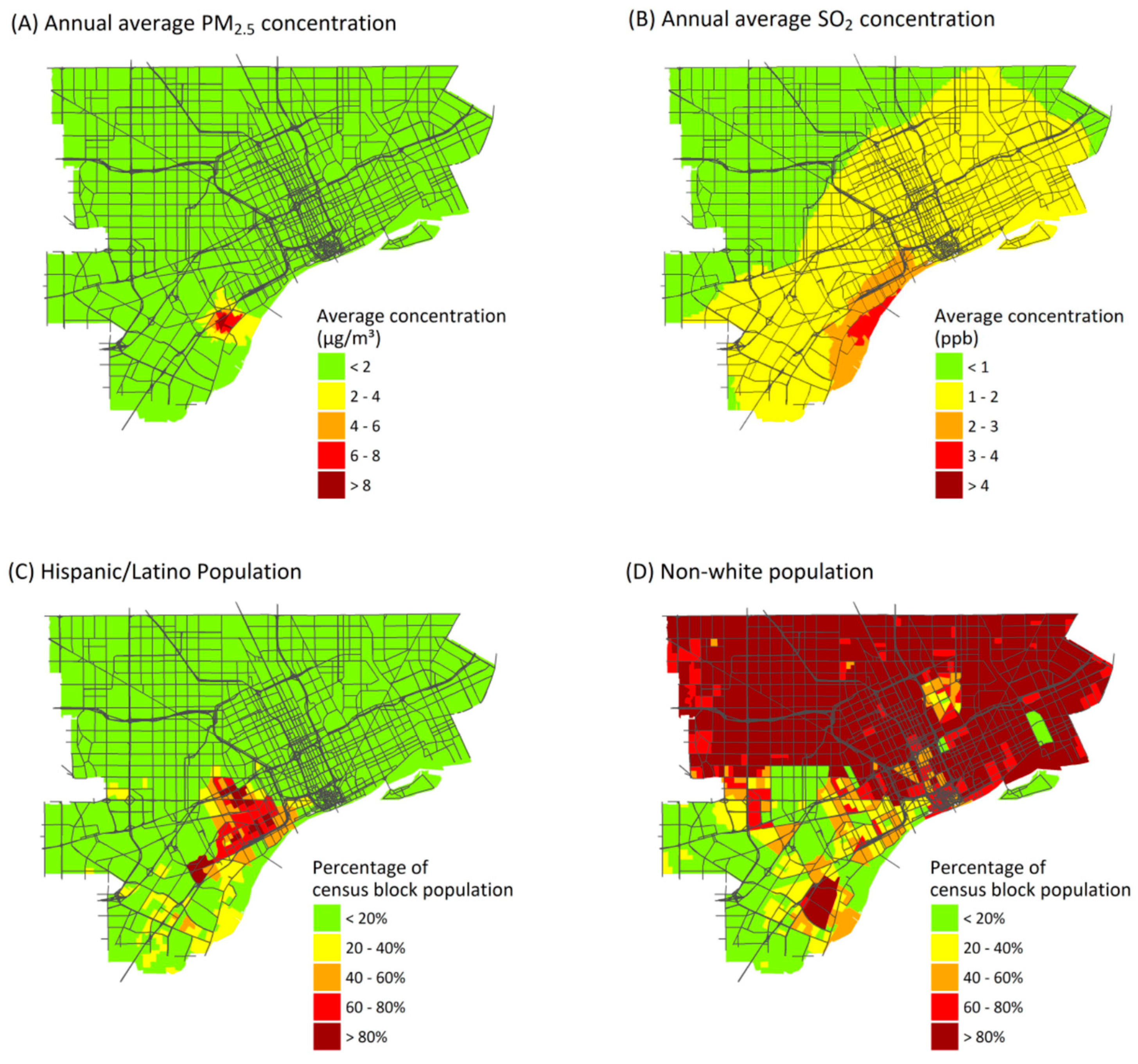
3.3. Spatial Distribution and Inequality of Exposures and Attributable Health Burden
3.4. Atkinson Index
3.5. Concentration Index
4. Discussion
4.1. Burden of Disease Attributable to Ambient Air Pollutant Exposures below the NAAQS
4.2. Intra-Urban Inequality in the Health Burden Attributable to Ambient Air Pollution
4.3. Exposures as a Poor Proxy for Health Risks in Urban-Scale Inequality Assessments
4.4. Using Urban-Scale HIAs Incorporating Inequality Metrics in AQM Decision Making
4.5. Uncertainty in the Quantitative Health Impact Assessments
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
References
- Solomon, G.M.; Morello-Frosch, R.; Zeise, L.; Faust, J.B. Cumulative environmental impacts: Science and policy to protect communities. Annu. Rev. Public Health 2016, 37, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Mohai, P.; Pellow, D.; Roberts, J.T. Environmental Justice. In Annual Review of Environment and Resources; Annual Reviews: Palo Alto, CA, USA, 2009; Volume 34, pp. 405–430. ISBN 978-0-8243-2334-9. [Google Scholar]
- O’Neill, M.S.; Breton, C.V.; Devlin, R.B.; Utell, M.J. Air pollution and health: Emerging information on susceptible populations. Air Qual. Atmos. Health 2012, 5, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Sacks, J.D.; Stanek, L.W.; Luben, T.J.; Johns, D.O.; Buckley, B.J.; Brown, J.S.; Ross, M. Particulate matter-induced health effects: Who is susceptible? Environ. Health Perspect. 2011, 119, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Morello-Frosch, R.; Zuk, M.; Jerrett, M.; Shamasunder, B.; Kyle, A.D. Understanding the cumulative impacts of inequalities in environmental health: Implications for policy. Health Aff. 2011, 30, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Su, J.G.; Jerrett, M.; Morello-Frosch, R.; Jesdale, B.M.; Kyle, A.D. Inequalities in cumulative environmental burdens among three urbanized counties in California. Environ. Int. 2012, 40, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Su, J.G.; Morello-Frosch, R.; Jesdale, B.M.; Kyle, A.D.; Shamasunder, B.; Jerrett, M. An index for assessing demographic inequalities in cumulative environmental hazards with application to Los Angeles, California. Environ. Sci. Technol. 2009, 43, 7626–7634. [Google Scholar] [CrossRef] [PubMed]
- Pratt, G.C.; Vadali, M.L.; Kvale, D.L.; Ellickson, K.M. Traffic, air pollution, minority and socio-economic status: Addressing inequities in exposure and risk. Int. J. Environ. Res. Public Health 2015, 12, 5355–5372. [Google Scholar] [CrossRef] [PubMed]
- August, L.M.; Faust, J.B.; Cushing, L.; Zeise, L.; Alexeeff, G.V. Methodological considerations in screening for cumulative environmental health impacts: Lessons learned from a pilot study in California. Int. J. Environ. Res. Public Health 2012, 9, 3069–3084. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.I.; Hanna, S.R. Spatial and temporal variability in urban fine particulate matter concentrations. Environ. Pollut. 2011, 159, 2009–2015. [Google Scholar] [CrossRef] [PubMed]
- Matte, T.D.; Ross, Z.; Kheirbek, I.; Eisl, H.; Johnson, S.; Gorczynski, J.E.; Kass, D.; Markowitz, S.; Pezeshki, G.; Clougherty, J.E. Monitoring intraurban spatial patterns of multiple combustion air pollutants in New York City: Design and implementation. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Sadd, J.L.; Pastor, M.; Morello-Frosch, R.; Scoggins, J.; Jesdale, B. Playing it safe: Assessing cumulative impact and social vulnerability through an environmental justice screening method in the South Coast Air Basin, California. Int. J. Environ. Res. Public Health 2011, 8, 1441–1459. [Google Scholar] [CrossRef] [PubMed]
- US Environmental Protection Agency. EJ 2020 Action Agenda: Environmental Justice Strategic Plan 2016–2020; US EPA: Washington, DC, USA, 2016.
- Rhodus, J.; Fulk, F.; Autrey, B.; O’Shea, S.; Roth, A. A Review of Health Impact Assessments in the U.S.: Current State-of-Science, Best Practices and Area for Improvement; U.S. Environmental Protection Agency: Washington, DC, USA, 2013.
- US Environmental Protection Agency. Regulatory Impact Analysis of the Proposed Revisions to the National Ambient Air Quality Standards for Ground-Level Ozone; United States Environmental Protection Agency: Research Triangle Park, NC, USA, 2014.
- US Environmental Protection Agency. Regulatory Impact Analysis for the Final Revisions to the National Ambient Air Quality Standards for Particulate Matter; Office of Air Quality Planning and Standards: Research Triangle Park, NC, USA, 2012.
- US Environmental Protection Agency. Final Regulatory Impact Analysis (RIA) for the SO2 National Ambient Air Quality Standards (NAAQS); Office of Air Quality Planning and Standards: Research Triangle Park, NC, USA, 2010.
- US Environmental Protection Agency. Final Regulatory Impact Analysis (RIA) for the NO2 National Ambient Air Quality Standards (NAAQS); Office of Air Quality Planning and Standards: Washington, DC, USA, 2010.
- Fann, N.; Lamson, A.D.; Anenberg, S.C.; Wesson, K.; Risley, D.; Hubbell, B.J. Estimating the National Public Health Burden Associated with exposure to ambient PM2.5 and ozone. Risk Anal. 2012, 32, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Fann, N.; Roman, H.A.; Fulcher, C.M.; Gentile, M.A.; Hubbell, B.J.; Wesson, K.; Levy, J.I. Maximizing health benefits and minimizing inequality: Incorporating local-scale data in the design and evaluation of air quality policies. Risk Anal. 2011, 31, 908–922. [Google Scholar] [CrossRef] [PubMed]
- Kheirbek, I.; Wheeler, K.; Walters, S.; Kass, D.; Matte, T. PM2.5 and ozone health impacts and disparities in New York City: Sensitivity to spatial and temporal resolution. Air Qual. Atmos. Health 2013, 6, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Michigan Department of Environmental Quality. Sulfur Dioxide One-Hour National Ambient Air Quality Standard Nonattainment State Implementation Plan for Wayne County (Partial); MDEQ: Lansing, MI, USA, 2016.
- Michigan Department of Environmental Quality. Recommendations for Area Designations for the Ozone National Ambient Air Quality Standard 2016; MDEQ: Lansing, MI, USA, 2016.
- Michigan Department of Health and Human Services. Hospitalizations by Selected Diagnosis. Available online: http://www.mdch.state.mi.us/pha/osr/CHI/hospdx/frame.html (accessed on 8 February 2016).
- Schulz, A.J.; Mentz, G.B.; Sampson, N.; Ward, M.; Anderson, R.; de Majo, R.; Israel, B.A.; Lewis, T.C.; Wilkins, D. Race and the distribution of social and physical environmental risk: A case example from the Detroit metropolitan area. Bois Rev. Soc. Sci. Res. Race 2016, 13, 285–304. [Google Scholar] [CrossRef] [PubMed]
- US Census Bureau 2010–2014 American Community Survey (ACS) 5-Year Estimates. Available online: https://www.census.gov/programs-surveys/acs/ (accessed on 6 October 2016).
- US Census Bureau Detroit QuickFacts. Available online: http://quickfacts.census.gov/qfd/states/26/2622000.html (accessed on 9 June 2015).
- Batterman, S.; Chambliss, S.; Isakov, V. Spatial resolution requirements for traffic-related air pollutant exposure evaluations. Atmos. Environ. 2014, 94, 518–528. [Google Scholar] [CrossRef] [PubMed]
- US Census Bureau TIGER/Line® with Selected Demographic and Economic Data. Available online: http://www.census.gov/geo/maps-data/data/tiger-data.html (accessed on 2 July 2015).
- Martenies, S.E.; Wilkins, D.; Batterman, S.A. Health impact metrics for air pollution management strategies. Environ. Int. 2015, 85, 84–95. [Google Scholar] [CrossRef] [PubMed]
- US Environmental Protection Agency. Integrated Science Assessment for Oxides of Nitrogen—Health Criteria; US Environmental Protection Agency: Washington, DC, USA, 2016.
- US Environmental Protection Agency. Integrated Science Assessment (ISA) for Sulfur Oxides—Health Criteria (Second External Review Draft); US Environmental Protection Agency: Washington, DC, USA, 2016.
- US Environmental Protection Agency. Integrated Science Assessment for Ozone and Related Photochemical Oxidants; US Environmental Protection Agency: Washington, DC, USA, 2013.
- US Environmental Protection Agency. Integrated Science Assessment for Particulate Matter; US EPA: Research Triangle Park, NC, USA, 2009.
- US Environmental Protection Agency. Integrated Science Assessment (ISA) for Sulfur Dioxide (Health Criteria); National Center for Environmental Assessment: Washington, DC, USA, 2008.
- Bell, M.L.; Peng, R.D.; Dominici, F. The exposure-response curve for ozone and risk of mortality and the adequacy of current ozone regulations. Environ. Health Perspect. 2006, 114, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Daniels, M.J.; Dominici, F.; Samet, J.M.; Scott, L. Zeger estimating particulate matter-mortality dose-response curves and threshold levels: An analysis of daily time-series for the 20 largest USA cities. Am. J. Epidemiol. 2000, 152, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.; Coull, B.; Laden, F.; Ryan, L. The effect of dose and timing of dose on the association between airborne particles and survival. Environ. Health Perspect. 2008, 116, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Propper, R.; Wong, P.; Bui, S.; Austin, J.; Vance, W.; Alvarado, A.; Croes, B.; Luo, D. Ambient and emission trends of toxic air contaminants in California. Environ. Sci. Technol. 2015, 49, 11329–11339. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J. Quantifying the burden of disease: The technical basis for disability-adjusted life years. Bull. World Health Organ. 1994, 72, 429–445. [Google Scholar] [PubMed]
- Ontario Ministry of Environment and Climate Change. Air Quality Ontario. Available online: http://www.airqualityontario.com/history/index.php (accessed on 30 July 2016).
- US Environmental Protection Agency. AQS Data Mart. Available online: https://aqs.epa.gov/api (accessed on 30 July 2016).
- Van Buuren, S.; Groothuis-Oudshoorn, K. MICE: Multivariate Imputation by Chained Equations in R. J. Stat. Softw. 2011, 25. [Google Scholar] [CrossRef]
- Michigan Department of Environmental Quality. MDEQ-Michigan Air Emissions Reporting System (MAERS). Annual Pollutant Totals Query. Available online: http://www.deq.state.mi.us/maers/emissions_query.asp (accessed on 25 March 2016).
- US Environmental Protection Agency. National Emissions Inventory. Available online: http://www.epa.gov/ttn/chief/net/2005inventory.html#inventorydata (accessed on 10 March 2015).
- Milando, C.W.; Martenies, S.E.; Batterman, S.A. Assessing concentrations and health impacts of air quality management strategies: Framework for Rapid Emissions Scenario and Health impact ESTimation (FRESH-EST). Environ. Int. 2016, 94, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Cimorelli, A.J.; Perry, S.G.; Venkatram, A.; Weil, J.C.; Paine, R.J.; Wilson, R.B.; Lee, R.F.; Peters, W.D.; Brode, R.W. AERMOD: A dispersion model for industrial source applications. Part I: General model formulation and boundary layer characterization. J. Appl. Meteorol. 2005, 44, 682–693. [Google Scholar] [CrossRef]
- US Environmental Protection Agency. 2014 National Emissions Inventory (NEI) Data. Available online: https://www.epa.gov/air-emissions-inventories/2014-national-emissions-inventory-nei-data (accessed on 13 January 2017).
- Snyder, M.G.; Venkatram, A.; Heist, D.K.; Perry, S.G.; Petersen, W.B.; Isakov, V. RLINE: A line source dispersion model for near-surface releases. Atmos. Environ. 2013, 77, 748–756. [Google Scholar] [CrossRef]
- US Environmental Protection Agency. MOVES (Motor Vehicle Emission Simulator). Available online: http://www.epa.gov/oms/models/moves/ (accessed on 6 July 2015).
- Milando, C.; Huang, L.; Batterman, S. Trends in PM2.5 emissions, concentrations and apportionments in Detroit and Chicago. Atmos. Environ. 2016, 129, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Harper, S.; Ruder, E.; Roman, H.A.; Geggel, A.; Nweke, O.; Payne-Sturges, D.; Levy, J.I. Using inequality measures to incorporate environmental justice into regulatory analyses. Int. J. Environ. Res. Public Health 2013, 10, 4039–4059. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.I.; Greco, S.L.; Melly, S.J.; Mukhi, N. Evaluating efficiency-equality tradeoffs for mobile source control strategies in an urban area. Risk Anal. 2009, 29, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.I.; Wilson, A.M.; Zwack, L.M. Quantifying the efficiency and equity implications of power plant air pollution control strategies in the United States. Environ. Health Perspect. 2007, 115, 743–750. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, O.; van Doorslaer, E.; Wagstaff, A.; Lindelow, M. Analyzing Health Equity Using Household Survey Data: A Guide to Techniques and Their Implementation; The World Bank: Washington, DC, USA, 2008. [Google Scholar]
- Cushing, L.; Faust, J.; August, L.M.; Cendak, R.; Wieland, W.; Alexeeff, G. Racial/ethnic disparities in cumulative environmental health impacts in California: Evidence from a statewide environmental justice screening tool (CalEnviroScreen 1.1). Am. J. Public Health 2015, 105, 2341–2348. [Google Scholar] [CrossRef] [PubMed]
- Boehmer, T.K.; Foster, S.L.; Henry, J.R.; Woghiren-Akinnifesi, E.L.; Yip, F.Y. Centers for Disease Control and Prevention (CDC). Residential proximity to major highways—United States, 2010. Morb. Mortal. Wkly. Rep. 2013, 62 (Suppl. 3), 46–50. [Google Scholar]
- Tian, N.; Xue, J.; Barzyk, T.M. Evaluating socioeconomic and racial differences in traffic-related metrics in the United States using a GIS approach. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.; Bind, M.-A.; Koutrakis, P. Estimating causal effects of local air pollution on daily deaths: Effect of low levels. Environ. Health Perspect. 2017, 125, 23. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Zanobetti, A.; Kloog, I.; Coull, B.A.; Koutrakis, P.; Melly, S.J.; Schwartz, J.D. Low-concentration PM2.5 and mortality: Estimating acute and chronic effects in a population-based study. Environ. Health Perspect. 2016, 124, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Goodkind, A.L.; Coggins, J.S.; Marshall, J.D. A spatial model of air pollution: The impact of the concentration-response function. J. Assoc. Environ. Resour. Econ. 2014, 1, 451–479. [Google Scholar] [CrossRef]
- Pope, C.A.; Cropper, M.; Coggins, J.; Cohen, A. Health benefits of air pollution abatement policy: Role of the shape of the concentration-response function. J. Air Waste Manag. Assoc. 2015, 65, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Dabdub, D.; Seinfeld, J.H. Chemical coupling between atmospheric ozone and particulate matter. Science 1997, 277, 116–119. [Google Scholar] [CrossRef]
- Sacks, J.D.; Fann, N.; Owens, E.O.; Costa, D.L. Using science to shape policy. In Air Pollution and Health Effects; Nadadur, S.S., Hollingsworth, J.W., Eds.; Molecular and Integrative Toxicology; Springer: London, UK, 2015; pp. 403–436. ISBN 978-1-4471-6668-9. [Google Scholar]
- Batterman, S.; Ganguly, R.; Harbin, P. High resolution spatial and temporal mapping of traffic-related air pollutants. Int. J. Environ. Res. Public Health 2015, 12, 3646–3666. [Google Scholar] [CrossRef] [PubMed]
- Padró-Martínez, L.T.; Patton, A.P.; Trull, J.B.; Zamore, W.; Brugge, D.; Durant, J.L. Mobile monitoring of particle number concentration and other traffic-related air pollutants in a near-highway neighborhood over the course of a year. Atmos. Environ. 2012, 61, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Patton, A.P.; Perkins, J.; Zamore, W.; Levy, J.I.; Brugge, D.; Durant, J.L. Spatial and temporal differences in traffic-related air pollution in three urban neighborhoods near an interstate highway. Atmos. Environ. 2014, 99, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Schulz, A.J.; Williams, D.R.; Israel, B.A.; Lempert, L.B. Racial and spatial relations as fundamental determinants of health in Detroit. Milbank Q. 2002, 80, 677–707. [Google Scholar] [CrossRef] [PubMed]
- De la Cruz, P.; Brittingham, A. The Arab Population: 2000; Census 2000 Brief; Census Bureau: Suitland, MD, USA, 2003.
- Padela, A.I.; Heisler, M. The association of perceived abuse and discrimination after 11 September 2001, with psychological distress, level of happiness, and health status among Arab Americans. Am. J. Public Health 2010, 100, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Samari, G. Islamophobia and public health in the United States. Am. J. Public Health 2016, 106, 1920–1925. [Google Scholar] [CrossRef] [PubMed]
- Robert Wood Johnson Foundation Michigan: County Health Rankings & Roadmaps. Available online: http://www.countyhealthrankings.org/app/michigan/2014/rankings/outcomes/overall (accessed on 12 April 2017).
- Gray, S.C.; Edwards, S.E.; Miranda, M.L. Race, socioeconomic status, and air pollution exposure in North Carolina. Environ. Res. 2013, 126, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.R.; Diez-Roux, A.V.; Hajat, A.; Kershaw, K.N.; O’Neill, M.S.; Guallar, E.; Post, W.S.; Kaufman, J.D.; Navas-Acien, A. Race/ethnicity, residential segregation, and exposure to ambient air pollution: The multi-ethnic study of atherosclerosis (MESA). Am. J. Public Health 2014, 104, 2130–2137. [Google Scholar] [CrossRef] [PubMed]
- Pope, R.; Wu, J.; Boone, C. Spatial patterns of air pollutants and social groups: A distributive environmental justice study in the phoenix metropolitan region of USA. Environ. Manag. 2016, 58, 753–766. [Google Scholar] [CrossRef] [PubMed]
- Prochaska, J.D.; Nolen, A.B.; Kelley, H.; Sexton, K.; Linder, S.H.; Sullivan, J. Social determinants of health in environmental justice communities: Examining cumulative risk in terms of environmental exposures and social determinants of health. Hum. Ecol. Risk Assess. 2014, 20, 980–994. [Google Scholar] [CrossRef] [PubMed]
- Batterman, S.; Burke, J.; Isakov, V.; Lewis, T.; Mukherjee, B.; Robins, T. A comparison of exposure metrics for traffic-related air pollutants: Application to epidemiology studies in Detroit, Michigan. Int. J. Environ. Res. Public Health 2014, 11, 9553–9577. [Google Scholar] [CrossRef] [PubMed]
- Brender, J.D.; Maantay, J.A.; Chakraborty, J. Residential proximity to environmental hazards and adverse health outcomes. Am. J. Public Health 2011, 101, S37–S52. [Google Scholar] [CrossRef] [PubMed]
- US Environmental Protection Agency. Technical Support Document: EPA’s 2011 National-Scale Air Toxics Assessment; Office of Air Quality Planning and Standards: Research Triangle Park, NC, USA, 2015.
- Fann, N.; Fulcher, C.M.; Hubbell, B.J. The influence of location, source, and emission type in estimates of the human health benefits of reducing a ton of air pollution. Air Qual. Atmos. Health 2009, 2, 169–176. [Google Scholar] [CrossRef] [PubMed]
- De Hollander, A.E.; Melse, J.M.; Lebret, E.; Kramers, P.G. An aggregate public health indicator to represent the impact of multiple environmental exposures. Epidemiology 1999, 10, 606–617. [Google Scholar] [CrossRef] [PubMed]
- Haagsma, J.A.; Polinder, S.; Cassini, A.; Colzani, E.; Havelaar, A.H. Review of disability weight studies: Comparison of methodological choices and values. Popul. Health Metr. 2014, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, E.; Hurley, F. A review of the strengths and weaknesses of quantitative methods used in health impact assessment. Public Health 2009, 123, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Brody, S.D.; Peck, B.M.; Highfield, W.E. Examining localized patterns of air quality perception in Texas: A spatial and statistical analysis. Risk Anal. 2004, 24, 1561–1574. [Google Scholar] [CrossRef] [PubMed]
- Downey, L.; Willigen, M.V. Environmental stressors: The mental health impacts of living near industrial activity. J. Health Soc. Behav. 2005, 46, 289–305. [Google Scholar] [CrossRef] [PubMed]
- Basner, M.; Babisch, W.; Davis, A.; Brink, M.; Clark, C.; Janssen, S.; Stansfeld, S. Auditory and non-auditory effects of noise on health. Lancet 2014, 383, 1325–1332. [Google Scholar] [CrossRef]
- Bhatia, R.; Seto, E. Quantitative estimation in health impact assessment: Opportunities and challenges. Environ. Impact Assess. Rev. 2011, 31, 301–309. [Google Scholar] [CrossRef]
- Fehr, R.; Hurley, F.; Mekel, O.C.; Mackenbach, J.P. Quantitative health impact assessment: Taking stock and moving forward. J. Epidemiol. Community Health 2012, 66, 1088–1091. [Google Scholar] [CrossRef] [PubMed]
- Wolch, J.R.; Byrne, J.; Newell, J.P. Urban green space, public health, and environmental justice: The challenge of making cities “just green enough”. Landsc. Urban Plan. 2014, 125, 234–244. [Google Scholar] [CrossRef]
- Bertazzon, S.; Johnson, M.; Eccles, K.; Kaplan, G.G. Accounting for spatial effects in land use regression for urban air pollution modeling. Spat. Spat.-Temp. Epidemiol. 2015, 14–15, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Hoek, G.; Beelen, R.; de Hoogh, K.; Vienneau, D.; Gulliver, J.; Fischer, P.; Briggs, D. A review of land-use regression models to assess spatial variation of outdoor air pollution. Atmos. Environ. 2008, 42, 7561–7578. [Google Scholar] [CrossRef]
- Jerrett, M.; Burnett, R.T.; Ma, R.; Pope, C.A.; Krewski, D.; Newbold, K.B.; Thurston, G.; Shi, Y.; Finkelstein, N.; Calle, E.E.; et al. Spatial analysis of air pollution and mortality in Los Angeles. Epidemiology 2005, 16, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Baccini, M.; Grisotto, L.; Catelan, D.; Consonni, D.; Bertazzi, P.A.; Biggeri, A. Commuting-adjusted short-term health impact assessment of airborne fine particles with uncertainty quantification via Monte Carlo simulation. Environ. Health Perspect. 2015, 123, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Tchepel, O.; Dias, D. Quantification of health benefits related with reduction of atmospheric PM10 levels: Implementation of population mobility approach. Int. J. Environ. Health Res. 2011, 21, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Chart-Asa, C.; Gibson, J.M. Health impact assessment of traffic-related air pollution at the urban project scale: Influence of variability and uncertainty. Sci. Total Environ. 2015, 506–507, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Briggs, D.J.; Sabel, C.E.; Lee, K. Uncertainty in epidemiology and health risk and impact assessment. Environ. Geochem. Health 2009, 31, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, M. Statistical issues in health impact assessment at the state and local levels. Air Qual. Atmos. Health 2009, 2, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.I. Issues and uncertainties in estimating the health benefits of air pollution control. J. Toxicol. Environ. Health A 2003, 66, 1865–1871. [Google Scholar] [CrossRef] [PubMed]
- Mesa-Frias, M.; Chalabi, Z.; Vanni, T.; Foss, A.M. Uncertainty in environmental health impact assessment: Quantitative methods and perspectives. Int. J. Environ. Health Res. 2013, 23, 16–30. [Google Scholar] [CrossRef] [PubMed]
- US Environmental Protection Agency. Integrated Science Assessment for Oxides of Nitrogen—Health Criteria; US EPA: Washington, DC, USA, 2009.
- DeGuire, P.; Cao, B.; Wisnieski, L.; Strane, D.; Wahl, R.; Lyon-Callo, S.; Garcia, E. Detroit: The Current Status of the Asthma Burden; Michigan Department of Health and Human Services: Hart, MI, USA, 2016.
- Michigan Department of Health and Human Services. Michigan Asthma Surveillance, Data and Reports. Available online: http://www.michigan.gov/mdhhs/0,5885,7-339-71550_5104_5279-213824--,00.html (accessed on 8 February 2016).
- Batterman, S.A.; Lewis, T.; Robins, T.; Mentz, G.; Milando, C.W.; Mukherjee, B. Effects of SO2 exposures below the national ambient air quality standards in a cohor of children with asthma in Detroit, Michigan. Unpublished work. 2017. [Google Scholar]
- US Environmental Protection Agency. BenMAP User’s Manual; US EPA: Research Triangle Park, NC, USA, 2015.
- Hubbell, B.J.; Fann, N.; Levy, J.I. Methodological considerations in developing local-scale health impact assessments: Balancing national, regional, and local data. Air Qual. Atmos. Health 2009, 2, 99–110. [Google Scholar] [CrossRef]
- US Environmental Protection Agency. Guidelines for Preparing Economic Analyses 2010; US EPA: Washington, DC, USA, 2010.

| Pollutant | Source | Mean (SD) | Min | 25th | Median | 75th | 95th | Max |
|---|---|---|---|---|---|---|---|---|
| PM2.5 (μg/m3) | Regional | 8.3 (4.5) | 1.5 | 5.2 | 6.8 | 11.3 | 14.5 | 29.5 |
| Point | 0.5 (0.9) | 0.0 | 0.1 | 0.3 | 0.6 | 1.4 | 75.7 | |
| Mobile | 0.6 (0.5) | 0.0 | 0.3 | 0.4 | 0.7 | 1.6 | 12.7 | |
| Area | 1.8 (2.8) | 0.0 | 0.2 | 1.0 | 2.2 | 6.3 | 29.4 | |
| Total | 10.7 (5.4) | 2.0 | 6.5 | 9.9 | 13.5 | 19.7 | 82.4 | |
| DPM (μg/m3) | Mobile | 0.5 (0.6) | 0.0 | 0.2 | 0.4 | 0.6 | 1.5 | 12.3 |
| O3 (ppb) | Regional | 38.3 (13.7) | 6.8 | 28.2 | 36.4 | 46.9 | 63.4 | 103.8 |
| SO2 (ppb) | Point | 1.1 (1.4) | 0.0 | 0.1 | 0.5 | 1.6 | 4.0 | 19.4 |
| NO2 (ppb) | Regional | 10.9 (5.1) | 2.6 | 7.7 | 9.7 | 12.9 | 23.0 | 30.2 |
| Point | 1.4 (1.1) | 0.0 | 0.5 | 1.1 | 1.9 | 3.5 | 17.0 | |
| Mobile | 10.2 (9.0) | 0.0 | 4.3 | 7.6 | 13.0 | 27.1 | 191.9 | |
| Area | 1.7 (3.0) | 0.0 | 0.0 | 0.0 | 2.6 | 8.8 | 17.2 | |
| Total | 23.5 (10.5) | 5.8 | 17.3 | 21.9 | 26.0 | 43.1 | 214.2 |
| Attributable Impacts | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Exposure Source | % of Attributable Burden Due to Each Pollutant | |||||||||
| Outcome (age group) | Estimated annual incidence 1 | Total (% 2) | Regional | Point | Mobile | Area | PM2.5 | O3 | SO2 | NO2 |
| Mortality (cases) | ||||||||||
| All-cause (>29) | 9400 | 520 (5.5) | 420 | 24 | 27 | 84 | 100 | 0 | 0 | 0 |
| Non-accidental (>29) | 8800 | 140 (1.5) | 140 | 0 | 0 | 0 | 0 | 100 | 0 | 0 |
| Infant (<1) | 200 | 6 (4.0) | 5 | 0 | 0 | 1 | 100 | 0 | 0 | 0 |
| Hospitalizations (cases) | ||||||||||
| Asthma (<65) | 3200 | 210 (6.7) | 140 | 17 | 46 | 16 | 51 | 0 | 3 | 46 |
| COPD (>65) | 1900 | 419 (22.4) | 330 | 48 | 40 | 12 | 5 | 62 | 10 | 23 |
| CVD (>65) | 9800 | 160 (1.6) | 130 | 7 | 8 | 8 | 100 | 0 | 0 | 0 |
| Pneumonia (>65) | 1500 | 250 (17.3) | 240 | 3 | 3 | 3 | 23 | 77 | 0 | 0 |
| Non-fatal MI (>17) | 2600 | 60 (2.3) | 48 | 3 | 3 | 3 | 100 | 0 | 0 | 0 |
| Asthma outcomes (cases) | ||||||||||
| Asthma ED visit (<18) | 9000 | 3300 (36.7) | 2600 | 160 | 450 | 120 | 15 | 51 | 2 | 31 |
| Day w/cough (6–14) | 1,700,000 | 210,000 (12.5) | 170,000 | 10,000 | 11,000 | 9500 | 100 | 0 | 0 | 0 |
| Day w/wheeze (6–14) | 1,100,000 | 17,000 (1.6) | 13,000 | 780 | 820 | 740 | 100 | 0 | 0 | 0 |
| Day w/SoB (6–14) | 1,000,000 | 21,000 (2.1) | 17,000 | 1000 | 1000 | 940 | 100 | 0 | 0 | 0 |
| 2+ symptoms (6–14) | 2,000,000 | 180,000 (8.6) | 110,000 | 12,000 | 45,000 | 9600 | 0 | 34 | 3 | 64 |
| Restricted days | ||||||||||
| MRAD (18–64) | 4,600,000 | 760,000 (16.7) | 700,000 | 16,000 | 18,000 | 18,000 | 44 | 56 | 0 | 0 |
| WLD (18–64) | 1,300,000 | 59,000 (4.7) | 47,000 | 2800 | 3000 | 3100 | 100 | 0 | 0 | 0 |
| MSD (6–14) | 2,700,000 | 570,000 (21.3) | 570,000 | 0 | 0 | 0 | 0 | 100 | 0 | 0 |
| Total DALYs (years) | 10,000 | 8100 | 470 | 560 | 1600 | 97 | 1 | 0.06 | 1.3 | |
| Monetized impact ($ million) | 6600 | 5500 | 240 | 280 | 830 | 78 | 21 | 0.03 | 0.5 | |
| Annual Average Exposures 2 | Annual Health Impact Risk | ||||||
|---|---|---|---|---|---|---|---|
| Pollutant | Source | All Blocks | ZIP Codes | NA Area 3 | All Blocks | ZIP Codes | NA Area 3 |
| PM2.5 | Regional 4 | - | - | - | 0.041 | 0.022 (46) | 0.038 (7) |
| Point | 0.101 | 0.139 (−37) | 0.107 (−5) | 0.126 | 0.154 (−22) | 0.157 (−25) | |
| Mobile | 0.079 | 0.057 (29) | 0.128 (−61) | 0.126 | 0.084 (34) | 0.153 (−21) | |
| Area | 0.070 | 0.019 (73) | 0.082 (−18) | 0.113 | 0.045 (60) | 0.111 (1) | |
| Total | 0.003 | 0.001 (62) | 0.003 (−13) | 0.045 | 0.023 (49) | 0.041 (8) | |
| O3 | Regional 4 | - | - | - | 0.040 | 0.023 (43) | 0.038 (4) |
| SO2 | Point | 0.064 | 0.055 (13) | 0.043 (33) | 0.155 | 0.075 (51) | 0.116 (25) |
| NO2 | Regional 4 | - | - | - | 0.133 | 0.038 (72) | 0.096 (28) |
| Point | 0.034 | 0.027 (23) | 0.042 (−21) | 0.159 | 0.057 (64) | 0.140 (12) | |
| Mobile | 0.084 | 0.055 (34) | 0.126 (−50) | 0.191 | 0.072 (62) | 0.203 (−7) | |
| Area | 0.130 | 0.101 (22) | 0.163 (−26) | 0.245 | 0.141 (43) | 0.225 (8) | |
| Total | 0.009 | 0.011 (−18) | 0.012 (−25) | 0.137 | 0.045 (67) | 0.104 (24) | |
| Concentration Index (×100) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Pollutant | Source | Percent Non-White | Percent Latino | Percent Less than HS | Median Income | Percent HH in Poverty | Percent POC | % FB |
| All census blocks | ||||||||
| PM2.5 | Regional | −6.7 | 3.0 | −1.0 | −4.1 | −1.2 | −6.4 | 6.5 |
| Point | 5.4 | −11.7 | −8.2 | −3.1 | −0.2 | 3.8 | −5.7 | |
| Mobile | −6.6 | 0.8 | −4.6 | −8.7 | −5.5 | −6.8 | 6.1 | |
| Area | −7.6 | 4.0 | 0.4 | −4.5 | −1.8 | −7.0 | 7.9 | |
| Total | −6.3 | 2.4 | −1.3 | −4.4 | −1.5 | −6.1 | 6.1 | |
| O3 | Regional | −6.2 | 3.0 | −0.6 | −3.4 | −0.5 | −5.9 | 6.1 |
| SO2 | Point | 8.1 | −13.3 | −11.1 | −3.3 | −6.9 | 6.0 | −12.4 |
| NO2 | Regional | 1.3 | −2.1 | −3.6 | −0.8 | −5.0 | 0.7 | −4.3 |
| Point | 5.8 | −9.3 | −8.9 | −2.8 | −7.0 | 4.1 | −10.1 | |
| Mobile | −1.0 | −3.4 | −7.2 | −5.0 | −8.4 | −2.3 | −2.6 | |
| Area | 3.6 | −0.6 | 2.8 | 4.4 | 0.2 | 4.1 | −4.7 | |
| Total | 0.8 | −3.0 | −4.9 | −2.3 | −6.1 | −0.1 | −3.9 | |
| ZIP codes | ||||||||
| PM2.5 | Regional | −8 (−19) | 4.1 (−35) | −0.5 (49) | −8.2 (−101) | −4.3 (−269) | −8.2 (−27) | 7.8 (−21) |
| Point | 8.5 (−58) | −23.7 (−102) | −12.9 (−57) | −6.1 (−95) | −6.1 (−2973) | 2.2 (41) | −8.2 (−44) | |
| Mobile | −4.4 (33) | −0.8 (193) | −1.4 (70) | −18.6 (−114) | −13.5 (−143) | −6.1 (11) | 1.1 (81) | |
| Area | −9.2 (−22) | 8.7 (−121) | 1 (−139) | −8.1 (−81) | −4.8 (−165) | −8.6 (−23) | 10.1 (−28) | |
| Total | −7.7 (−21) | 3 (−25) | −0.6 (57) | −8.1 (−85) | −3.9 (−166) | −8 (−32) | 7.4 (−21) | |
| O3 | Regional | −8.2 (−32) | 4.6 (−52) | −0.8 (−35) | −8.4 (−149) | −4.2 (−823) | −8.1 (−39) | 7.5 (−23) |
| SO2 | Point | 11.1 (−37) | −15.2 (−15) | −18.7 (−68) | −3.2 (4) | −7.2 (−4) | 8.3 (−40) | −12.1 (3) |
| NO2 | Regional | 1.5 (−16) | −0.9 (58) | −6.5 (−84) | 1.6 (296) | −2.2 (56) | 1.1 (−59) | −3 (30) |
| Point | 8.5 (−46) | −11 (−19) | −15.1 (−69) | −1.1 (59) | −5.7 (18) | 6.3 (−53) | −10.1 (0) | |
| Mobile | 1.9 (295) | −2.7 (20) | −9.1 (−27) | −7.8 (−55) | −10.3 (−24) | 0.2 (107) | −6.5 (−154) | |
| Area | −3.9 (207) | 8.4 (1421) | 4.8 (−72) | 7.5 (−69) | 4.8 (−2110) | −2.4 (158) | 9.4 (301) | |
| Total | 3.3 (−316) | −4.4 (−47) | −10.1 (−106) | 0.1 (105) | −4.2 (32) | 2.1 (1867) | −6.3 (−60) | |
| Census blocks in the SO2 non-attainment area | ||||||||
| PM2.5 | Regional | −3.8 (43) | 6.1 (−102) | 5.8 (702) | 0.6 (115) | 3 (362) | −1.3 (79) | 7.1 (−10) |
| Point | −11.3 (310) | 2.7 (123) | −3.2 (61) | −8.9 (−184) | −5.5 (−2663) | −11.6 (406) | 6.2 (208) | |
| Mobile | −9.2 (−40) | −0.9 (205) | −0.1 (98) | −6 (31) | −4.1 (26) | −9.3 (−37) | 1.8 (71) | |
| Area | −0.4 (95) | 7.8 (−98) | 10.9 (−2598) | 5.7 (227) | 6 (430) | 3.3 (147) | 7.7 (2) | |
| Total | −4.4 (31) | 5.4 (−125) | 5 (486) | −0.1 (97) | 2.1 (241) | −2.2 (64) | 6.6 (−8) | |
| O3 | Regional | −3.3 (46) | 6.5 (−117) | 6.5 (1168) | 1.5 (143) | 3.9 (965) | −0.8 (87) | 7.1 (−18) |
| SO2 | Point | −6.1 (175) | −11.2 (16) | −12.6 (−13) | −8.7 (−160) | −10.9 (−58) | −9.2 (254) | −10.1 (19) |
| NO2 | Regional | −0.6 (148) | −8.6 (−319) | −8.4 (−137) | −3.9 (−394) | −6.1 (−24) | −3.5 (609) | −9 (−109) |
| Point | −5.9 (202) | −12 (−29) | −13.4 (−49) | −9.7 (−252) | −11.9 (−71) | −9.7 (334) | −11.2 (−11) | |
| Mobile | −5.7 (−469) | −16.8 (−392) | −15.3 (−115) | −10.2 (−103) | −12.5 (−50) | −11.1 (−375) | −15 (−488) | |
| Area | 6.5 (−82) | 7.2 (1225) | 8.1 (−189) | 7.6 (−73) | 6.4 (−2835) | 9.2 (−125) | 7 (250) | |
| Total | −2.6 (429) | −11.3 (−279) | −10.6 (−117) | −6.1 (−170) | −8.4 (−37) | −6.2 (−5256) | −10.6 (−171) | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martenies, S.E.; Milando, C.W.; Williams, G.O.; Batterman, S.A. Disease and Health Inequalities Attributable to Air Pollutant Exposure in Detroit, Michigan. Int. J. Environ. Res. Public Health 2017, 14, 1243. https://doi.org/10.3390/ijerph14101243
Martenies SE, Milando CW, Williams GO, Batterman SA. Disease and Health Inequalities Attributable to Air Pollutant Exposure in Detroit, Michigan. International Journal of Environmental Research and Public Health. 2017; 14(10):1243. https://doi.org/10.3390/ijerph14101243
Chicago/Turabian StyleMartenies, Sheena E., Chad W. Milando, Guy O. Williams, and Stuart A. Batterman. 2017. "Disease and Health Inequalities Attributable to Air Pollutant Exposure in Detroit, Michigan" International Journal of Environmental Research and Public Health 14, no. 10: 1243. https://doi.org/10.3390/ijerph14101243
APA StyleMartenies, S. E., Milando, C. W., Williams, G. O., & Batterman, S. A. (2017). Disease and Health Inequalities Attributable to Air Pollutant Exposure in Detroit, Michigan. International Journal of Environmental Research and Public Health, 14(10), 1243. https://doi.org/10.3390/ijerph14101243





