Comparison of Gastric versus Gastrointestinal PBET Extractions for Estimating Oral Bioaccessibility of Metals in House Dust
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples and Reference Materials
2.2. Physiologically Based Extraction Technique (PBET)
2.3. Instrumental Analysis and Calculation of Percent Bioaccessibility
3. Results
3.1. Comparison of G-Alone and GI Percent Bioaccessibility in NIST CRMs
3.2. Variability of Total and Bioaccessible Metals in House Dust Samples
3.3. Comparison of G-Alone and GI Percent Bioaccessibility in House Dust Samples
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Leech, J.A.; Nelson, W.C.; Burnett, R.T.; Aaron, S.; Raizenne, M.E. It’s about time: A comparison of Canadian and American time-activity patterns. J. Expo. Anal. Environ. Epidemiol. 2002, 12, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.; Jones-Otazo, H.; Petrovic, S.; Mitchell, I.; Bonvalot, Y.; Williams, D.; Richardson, G.M. Revisiting dust and soil ingestion rates based on hand-to-mouth transfer. Hum. Ecol. Risk Assess. 2013, 19, 158–188. [Google Scholar] [CrossRef]
- Turner, A. Oral bioaccessibility of trace metals in household dust: A review. Environ. Geochem. Health 2011, 33, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Bradham, K.D.; Laird, B.D.; Rasmussen, P.E.; Schoof, R.A.; Serda, S.M.; Siciliano, S.D.; Hughes, M.F. Assessing the bioavailability and risk from metal-contaminated soils and dusts. Hum. Ecol. Risk Assess. 2014, 20, 272–286. [Google Scholar] [CrossRef]
- Koch, I.; Reimer, K.J.; Bakker, M.I.; Basta, N.T.; Cave, M.R.; Denys, S.; Dodd, M.; Hale, B.A.; Irwin, R.; Lowney, Y.W.; et al. Variability of bioaccessibility results using seventeen different methods on a standard reference material, NIST 2710. J. Environ. Sci. Health A Toxic Hazard. Subst. Environ. Eng. 2013, 48, 641–655. [Google Scholar] [CrossRef] [PubMed]
- Ruby, M.V.; Davis, A.; Schoof, R.; Eberle, S.; Sellstone, C.M. Estimation of lead and arsenic bioavailability using a physiologically based extraction test. Environ. Sci. Technol. 1996, 30, 422–430. [Google Scholar] [CrossRef]
- Ruby, M.V.; Schoof, R.; Brattin, W.; Goldade, M.; Post, G.; Harnois, M.; Mosby, E.; Casteel, S.W.; Berti, W.; Carpenter, M.; et al. Advances in evaluating the oral bioavailability of inorganics in soil for use in human health risk assessment. Environ. Sci. Technol. 1999, 33, 3697–3705. [Google Scholar] [CrossRef]
- Rodriguez, R.R.; Basta, N.T.; Casteel, S.W.; Pace, L.W. An in vitro gastrointestinal method to estimate bioavailable arsenic in contaminated soils and solid media. Environ. Sci. Technol. 1999, 33, 642–649. [Google Scholar] [CrossRef]
- Oomen, A.G.; Hack, A.; Minekus, M.; Zeijdner, E.; Cornelis, C.; Schoeters, G.; Verstraete, W.; van de Wiele, T.; Wragg, J.; Rompelberg, C.J.M.; et al. Comparison of five in vitro digestion models to study the bioaccessibility of soil contaminants. Environ. Sci. Technol. 2002, 36, 3326–3334. [Google Scholar] [CrossRef] [PubMed]
- Drexler, J.W.; Brattin, W.J. An in vitro procedure for estimation of lead relative bioavailability: With validation. Hum. Ecol. Risk Assess. 2007, 13, 383–401. [Google Scholar] [CrossRef]
- Saikat, S.; Barnes, B.; Westwood, D. A review of laboratory results for bioaccessibility values of arsenic, lead and nickel in contaminated UK soils. J. Environ. Sci. Health A Toxic Hazard. Subst. Environ. Eng. 2007, 42, 1213–1221. [Google Scholar] [CrossRef] [PubMed]
- Wragg, J.; Cave, M.; Basta, N.; Brandon, E.; Casteel, S.; Denys, S.; Gron, C.; Oomen, A.; Reimer, K.; Tack, K.; et al. An inter-laboratory trial of the unified BARGE bioaccessibility method for arsenic, cadmium and lead in soil. Sci. Total Environ. 2011, 409, 4016–4030. [Google Scholar] [CrossRef] [PubMed]
- Juhasz, A.L.; Weber, J.; Smith, E.; Naidu, R.; Rees, M.; Rofe, A.; Kuchel, T.; Sansom, L. Assessment of four commonly employed in vitro arsenic bioaccessibility assays for predicting in vivo relative arsenic bioavailability in contaminated soils. Environ. Sci. Technol. 2009, 43, 9487–9494. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (USEPA). Estimation of Relative Bioavailability of Lead in Soil and Soil-Like Materials Using In Vivo and In Vitro Methods; EPA 9285.7-77; U.S. Environmental Protection Agency: Washington, DC, USA, 2007.
- Li, H.-B.; Cui, X.-Y.; Li, K.; Li, J.; Juhasz, A.L.; Ma, L.Q. Assessment of in vitro lead bioaccessibility in house dust and its relationship to in vivo lead relative bioavailability. Environ. Sci. Technol. 2014, 48, 8548–8555. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, P.E. Can metal concentrations in indoor dust be predicted from soil geochemistry? Can. J. Anal. Sci. Spectrosc. 2004, 49, 166–174. [Google Scholar]
- Rasmussen, P.E.; Beauchemin, S.; Nugent, M.; Dugandzic, R.; Lanouette, M.; Chénier, M. Influence of matrix composition on the bioaccessibility of copper, zinc, and nickel in urban residential dust and soil. Hum. Ecol. Risk Assess. 2008, 14, 351–371. [Google Scholar] [CrossRef]
- Dodd, M.; Rasmussen, P.E.; Chénier, M. Comparison of two in vitro extraction protocols for assessing metals’ bioaccessibility using dust and soil reference materials. Hum. Ecol. Risk Assess. 2013, 19, 1014–1027. [Google Scholar]
- Turner, A.; Ip, K.-H. Bioaccessibility of metals in dust from the indoor environment: Application of a physiologically based extraction test. Environ. Sci. Technol. 2007, 41, 7851–7856. [Google Scholar] [CrossRef] [PubMed]
- Poggio, L.; Vršcaj, B.; Schulin, R.; Hepperle, E.; Ajmone Marsan, F. Metals pollution and human bioaccessibility of topsoils in Grugliasco (Italy). Environ. Pollut. 2009, 157, 680–689. [Google Scholar] [CrossRef] [PubMed]
- Karadas, C.; Kara, D. Chemometric evaluation for the relation of BCR sequential extraction method and in vitro gastro-intestinal method for the assessment of metal bioavailability in contaminated soils in Turkey. Environ. Sci. Pollut. Res. 2012, 19, 1280–1295. [Google Scholar] [CrossRef] [PubMed]
- De Miguel, E.; Mingot, J.; Chacón, E.; Charlesworth, S. The relationship between soil geochemistry and the bioaccessibility of trace elements in playground soil. Environ. Geochem. Health 2012, 34, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Sialelli, J.; Urquhart, G.J.; Davidson, C.M.; Hursthouse, A.S. Use of a physiologically based extraction test to estimate the human bioaccessibility of potentially toxic elements in urban soils from the city of Glasgow, UK. Environ. Geochem. Health 2010, 32, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Levallois, P.; St-Laurent, J.; Gauvin, D.; Courteau, M.; Prévost, M.; Campagna, C.; Lemieux, F.; Nour, S.; D’Amour, M.; Rasmussen, P.E. The impact of drinking water, indoor dust and paint on blood lead levels of children aged 1–5 years in Montréal (Québec, Canada). J. Expo. Sci. Environ. Epidemiol. 2014, 24, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, P.E.; Beauchemin, S.; Chénier, M.; Levesque, C.; MacLean, L.C.W.; Marro, L.; Jones-Otazo, H.; Petrovic, S.; McDonald, L.T.; Gardner, H.D. Canadian House Dust Study: Lead bioaccessibility and speciation. Environ. Sci. Technol. 2011, 45, 4959–4965. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, P.E.; Levesque, C.; Chénier, M.; Gardner, H.D.; Jones-Otazo, H.; Petrovic, S. Canadian House Dust Study: Population-based concentrations, loads and loading rates of arsenic, cadmium, chromium, copper, nickel, lead, and zinc inside urban homes. Sci. Total Environ. 2013, 443, 520–529. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (USEPA). Standard Operating Procedure for an In Vitro Bioaccessibility Assay for Lead in Soil; EPA 9200.1-86; United States Environmental Protection Agency: Washington, DC, USA, 2008; pp. 1–10.
- Ellickson, K.M.; Meeker, R.J.; Gallo, M.A.; Buckley, B.T.; Lioy, P.J. Oral bioavailability of lead and arsenic from a NIST standard reference soil material. Arch. Environ. Contam. Toxicol. 2001, 40, 128–135. [Google Scholar] [PubMed]
- Le Bot, B.; Gilles, E.; Durand, S.; Glorennec, P. Bioaccessible and quasi-total metals in soil and indoor dust. Eur. J. Mineral. 2010, 22, 651–657. [Google Scholar] [CrossRef]
- Wragg, J.; Cave, M. Assessment of a geochemical extraction procedure to determine the solid phase fractionation and bioaccessibility of potentially harmful elements in soils: A case study using the NIST 2710 reference soil. Anal. Chim. Acta 2012, 722, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, P.E.; Beauchemin, S.; Maclean, L.C.W.; Chénier, M.; Levesque, C.; Gardner, H.D. Impact of humidity on speciation and bioaccessibility of Pb, Zn, Co and Se in house dust. J. Anal. At. Spectrom. 2014, 29, 1206–1217. [Google Scholar] [CrossRef]
- Jiang, M.; Nakamatsu, Y.; Jensen, K.A.; Utsunomiya, S. Multi-scale analysis of the occurrence of Pb, Cr and Mn in the NIST standards: Urban Dust (SRM 1649a) and Indoor Dust (SRM 2584). Atmos. Environ. 2014, 82, 364–374. [Google Scholar] [CrossRef]
- Ibanez, Y.; Bot, B.L.E.; Glorennec, P. House-dust metal content and bioaccessibility: A review. Eur. J. Mineral. 2010, 22, 629–637. [Google Scholar] [CrossRef]
- Juhasz, A.L.; Weber, J.; Naidu, R.; Gancarz, D.; Rofe, A.; Todor, D.; Smith, E. Determination of cadmium relative bioavailability in contaminated soils and its prediction using in vitro methodologies. Environ. Sci. Technol. 2010, 44, 5240–5247. [Google Scholar] [CrossRef] [PubMed]
- Ljung, K.; Oomen, A.; Duits, M.; Selinus, O.; Berglund, M. Bioaccessibility of metals in urban playground soils. J. Environ. Sci. Health A Toxic Hazard. Subst. Environ. Eng. 2007, 42, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.; Cox, S.F.; McKinley, J.M.; Ofterdinger, U. Soil-geochemical factors controlling the distribution and oral bioaccessibility of nickel, vanadium and chromium in soil. Appl. Geochem. 2014, 51, 255–267. [Google Scholar] [CrossRef]
- Walker, S.R.; Jamieson, H.E.; Rasmussen, P.E. Application of synchrotron microprobe methods to solid-phase speciation of metals and metalloids in house dust. Environ. Sci. Technol. 2011, 45, 8233–8240. [Google Scholar] [CrossRef] [PubMed]
- Beauchemin, S.; MacLean, L.C.W.; Rasmussen, P.E. Lead speciation in indoor dust: A case study to assess old paint contribution in a Canadian urban house. Environ. Geochem. Health 2011, 33, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Beauchemin, S.; Rasmussen, P.E.; Mackinnon, T.; Chénier, M.; Boros, K. Zinc in house dust: Speciation, bioaccessibility, and impact of humidity. Environ. Sci. Technol. 2014, 48, 9022–9029. [Google Scholar] [CrossRef] [PubMed]
- MacLean, L.C.W.; Beauchemin, S.; Rasmussen, P.E. Lead speciation in house dust from Canadian urban homes using EXAFS, Micro-XRF, and Micro-XRD. Environ. Sci. Technol. 2011, 45, 5491–5497. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Demisie, W.; Zhang, M.-K. The function of digestive enzymes on Cu, Zn, and Pb release from soil in in vitro digestion tests. Environ. Sci. Pollut. Res. 2013, 20, 4993–5002. [Google Scholar] [CrossRef] [PubMed]
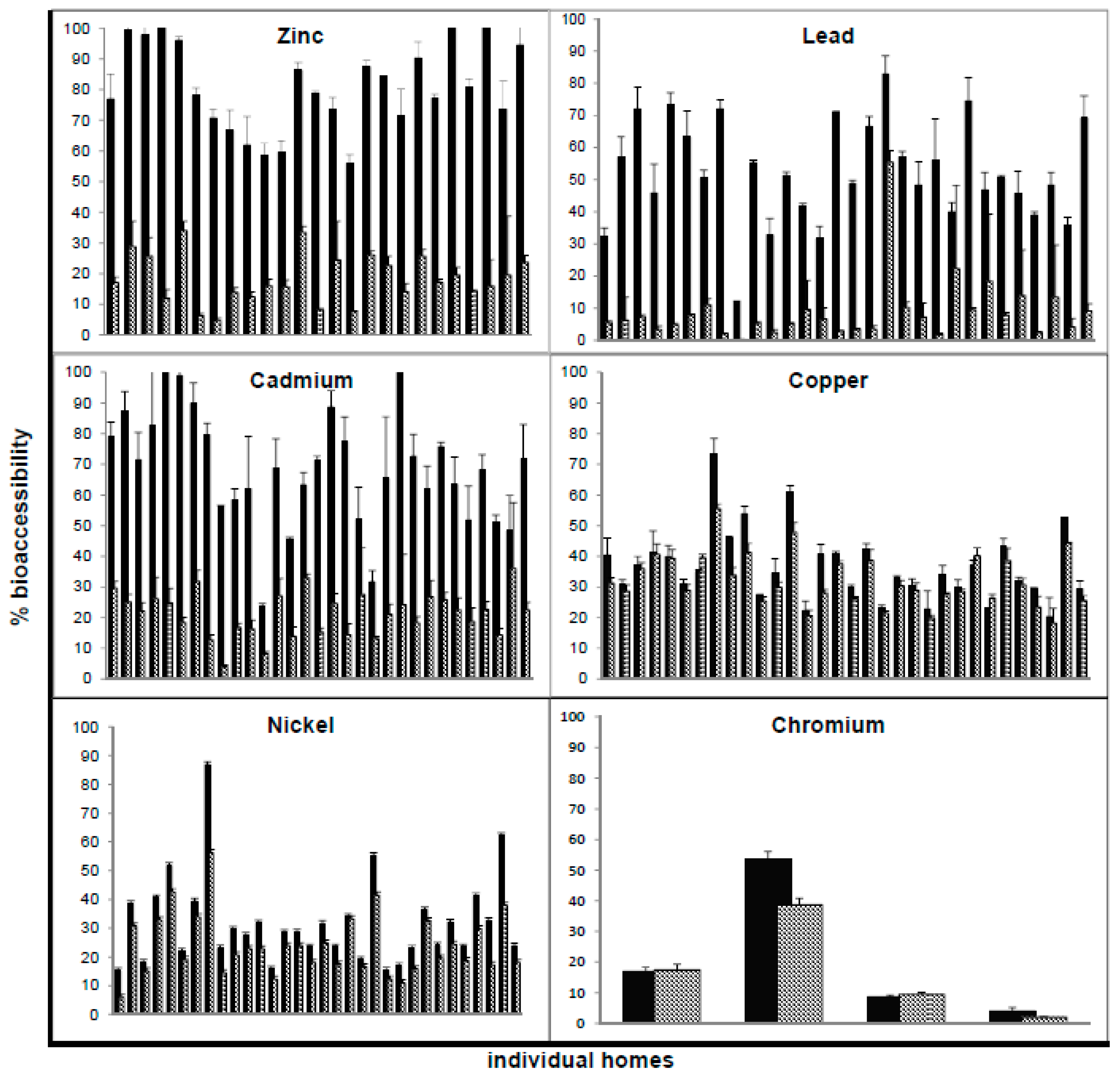
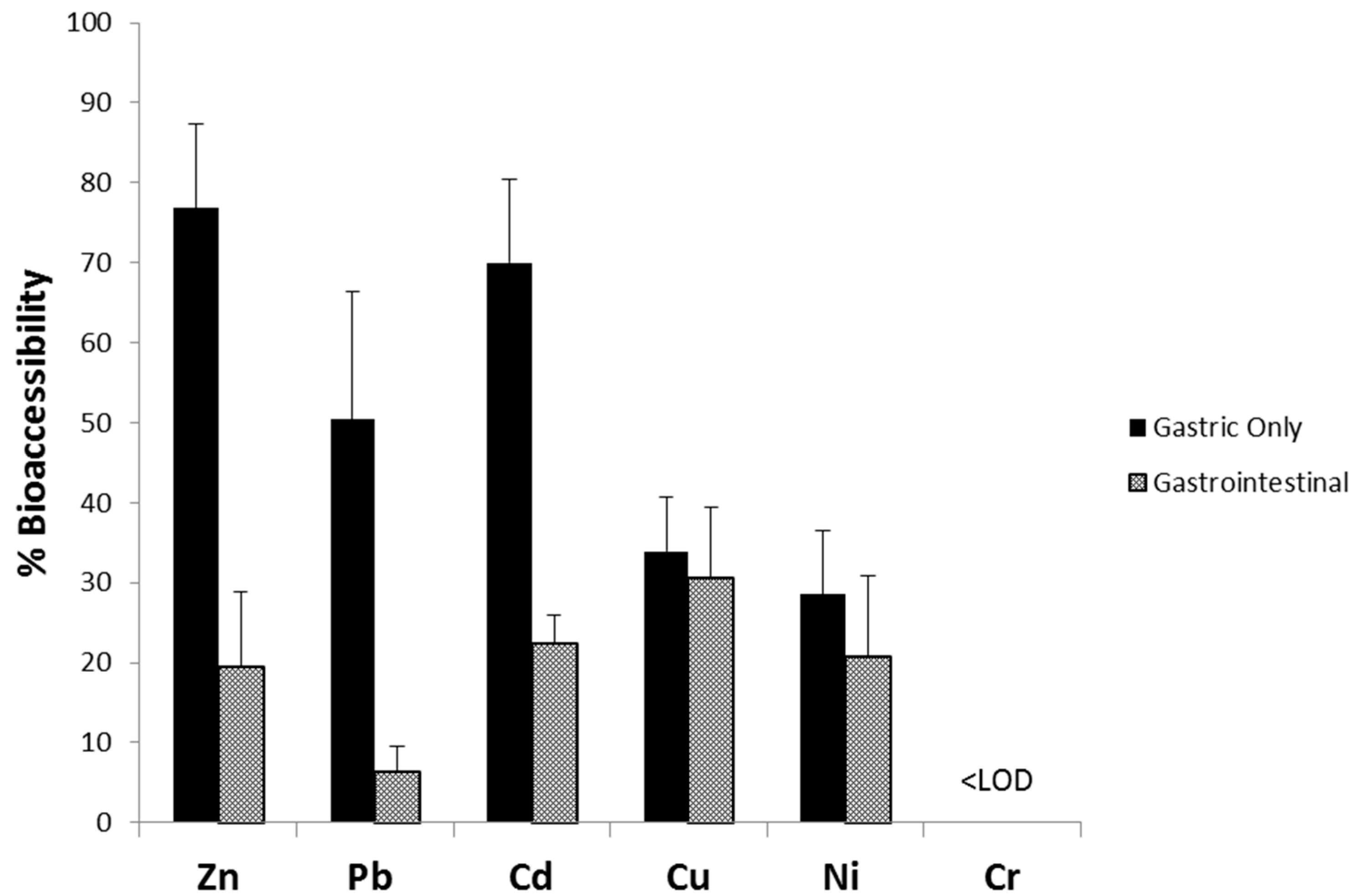
| Metal | Complete Set (n = 225) | Subset Used for PBET (n = 33) | ||||
|---|---|---|---|---|---|---|
| Mean (SD) | Range | Median | Mean (SD) | Range | Median | |
| Zn | 979 (610) | 27.6–5190 | 879 | 1177 (1007) | 27.6–5190 | 982 |
| Pb | 415 (838) | 6.2–8000 | 176 | 962 (1787) | 6.2–8000 | 195 |
| Cd | 4.4 (4.4) | <LOD–38.6 | 3.2 | 6.0 (7.8) | <LOD–38.6 | 2.9 |
| Cu | 278 (413) | 4.7–5810 | 205 | 266 (204) | 4.7–1020 | 213 |
| Ni | 80.7 (145) | 2.4–2120 | 61.2 | 78.6 (67.0) | 2.4–313 | 60.0 |
| Cr | 82.6 (73.6) | 1.4–868 | 67.1 | 117 (152) | 1.4–868 | 86.4 |
| Sample | Extraction | Zn | Pb | Cd | Cu | Ni | Cr |
|---|---|---|---|---|---|---|---|
| NIST 2710 (n = 3) | Total (mg/kg) | 6952 ± 91 | 5532 ± 80 | 21.8 ± 0.2 | 2950 ± 130 | 14.3 ± 1 | 39 a |
| G-alone (%) | 28.8 ± 1.3 | 64.9 ± 1.7 | 74.4 ± 2.1 | 63.3 ± 0.8 | 14.0 ± 0.4 | <LOD | |
| GI (%) | 11.4 ± 1.0 | 15.7 ± 0.6 | 39.3 ± 2.5 | 44.6 ± 2.0 | 9.6 ± 1.2 | <LOD | |
| NIST 2710a (n = 6) | Total (mg/kg) | 4180 ± 150 | 5520 ± 30 | 12.3 ± 0.3 | 3420 ± 50 | 8 ± 1 | 23 ± 6 |
| G-alone (%) | 42.3 ± 1.8 | 45.7 ± 1.4 | 45.6 ± 1.5 | 53.0 ± 1.8 | 12.8 ± 0.7 | <LOD | |
| GI (%) | 12.1 ± 0.6 | 4.9 ± 0.2 | 22.7 ± 0.7 | 37.2 ± 0.6 | 8.2 ± 0.5 | <LOD | |
| NIST 2583 (n = 5) | Total (mg/kg) | 896 ± 56.7 b | 85.9 ± 7.2 | 7.3 ± 3.7 | 233 ± 19.4 b | 93.9 ± 8.4 b | 80 ± 22 |
| G-alone (%) | 95.3 ± 2.6 | 31.1 ± 1.1 | 69.7 ± 5.2 | 33.0 ± 1.0 | 26.4 ± 2.4 | <LOD | |
| GI (%) | 42.7 ± 1.8 | 13.4 ± 0.8 | 39.6 ± 3.2 | 31.8 ± 0.8 | 21.4 ± 1.1 | <LOD | |
| NIST 2584 (n = 5) | Total (mg/kg) | 2580 ± 150 | 9761 ± 67 | 10 ± 1.1 | 288 ± 29.4 b | 84.6 ± 10.1 b | 135 ± 9.1 |
| G-alone (%) | 84.7 ± 1.6 | 56.1 ± 1.7 | 80.1 ± 3.8 | 43.4 ± 0.27 | 23.9 ± 0.5 | <LOD | |
| GI (%) | 21.1 ± 0.6 | 7.6 ± 0.2 | 28.9 ± 1.2 | 42.3 ± 1.24 | 18.7 ± 0.8 | <LOD |
| Metal | Gastric Phase | Gastrointestinal Phase | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LOD | Mean (SD) | Median | 95th Percentile | # Samples >LOD * | LOD | Mean (SD) | Median | 95th Percentile | # Samples >LOD * | |
| Zn | ||||||||||
| Conc. (mg/kg) | 191 ++ | 955 (872) | 750 | 2325 | 27 | 71.1 | 214 (216) | 162 | 485 | 28 |
| % Bioaccessibility | 76.1 (16.4) | 76.9 | 100 | 28.3 (29.7) | 19.5 | 109 | ||||
| Pb | ||||||||||
| Conc. (mg/kg) | 3.2 | 578 (1192) | 96.1 | 3006 | 31 | 0.4 | 55.6 (111) | 15.2 | 238 | 32 |
| % Bioaccessibility | 51.6 (17.6) | 50.4 | 77.8 | 8.4 (9.7) | 6.2 | 20.0 | ||||
| Cd | ||||||||||
| Conc. (mg/kg) | 0.1 | 4.3 (5.6) | 2.1 | 17.1 | 33 | 0.1 | 1.3 (1.6) | 0.7 | 4.8 | 32 |
| % Bioaccessibility | 70.7 (22.0) | 70.0 | 105 | 20.7 (7.8) | 22.4 | 32.3 | ||||
| Cu | ||||||||||
| Conc. (mg/kg) | 2.6 | 91.2 (72.2) | 71.3 | 253 | 32 | 1.3 | 80.5 (57.0) | 72.9 | 211 | 33 |
| % Bioaccessibility | 36.1 (11.5) | 33.9 | 56.7 | 34.0 (12.9) | 30.5 | 50.9 | ||||
| Ni | ||||||||||
| Conc. (mg/kg) | 0.8 | 21.6 (19.1) | 16.5 | 52.9 | 32 | 0.9 | 15.6 (12.2) | 12.4 | 42.4 | 32 |
| % Bioaccessibility | 31.3 (15.2) | 28.5 | 58.0 | 23.9 (10.6) | 20.7 | 42.0 | ||||
| Cr | ||||||||||
| Conc. (mg/kg) | 20 | <LOD | <LOD | 40.6 | 4 | 18 | <LOD | <LOD | 36.3 | 4 |
| % Bioaccessibility | 20.9 (22.2) | 12.8 | 47.9 | 17.4 (15.9) | 14.5 | 35.8 | ||||
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boros, K.; Fortin, D.; Jayawardene, I.; Chénier, M.; Levesque, C.; Rasmussen, P.E. Comparison of Gastric versus Gastrointestinal PBET Extractions for Estimating Oral Bioaccessibility of Metals in House Dust. Int. J. Environ. Res. Public Health 2017, 14, 92. https://doi.org/10.3390/ijerph14010092
Boros K, Fortin D, Jayawardene I, Chénier M, Levesque C, Rasmussen PE. Comparison of Gastric versus Gastrointestinal PBET Extractions for Estimating Oral Bioaccessibility of Metals in House Dust. International Journal of Environmental Research and Public Health. 2017; 14(1):92. https://doi.org/10.3390/ijerph14010092
Chicago/Turabian StyleBoros, Kristina, Danielle Fortin, Innocent Jayawardene, Marc Chénier, Christine Levesque, and Pat E. Rasmussen. 2017. "Comparison of Gastric versus Gastrointestinal PBET Extractions for Estimating Oral Bioaccessibility of Metals in House Dust" International Journal of Environmental Research and Public Health 14, no. 1: 92. https://doi.org/10.3390/ijerph14010092
APA StyleBoros, K., Fortin, D., Jayawardene, I., Chénier, M., Levesque, C., & Rasmussen, P. E. (2017). Comparison of Gastric versus Gastrointestinal PBET Extractions for Estimating Oral Bioaccessibility of Metals in House Dust. International Journal of Environmental Research and Public Health, 14(1), 92. https://doi.org/10.3390/ijerph14010092






