In Vitro Investigations of Human Bioaccessibility from Reference Materials Using Simulated Lung Fluids
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reference Materials
2.2. In Vitro Inhalation Bioaccessibility Protocol
2.3. Statistical Analyses
3. Results and Discussion
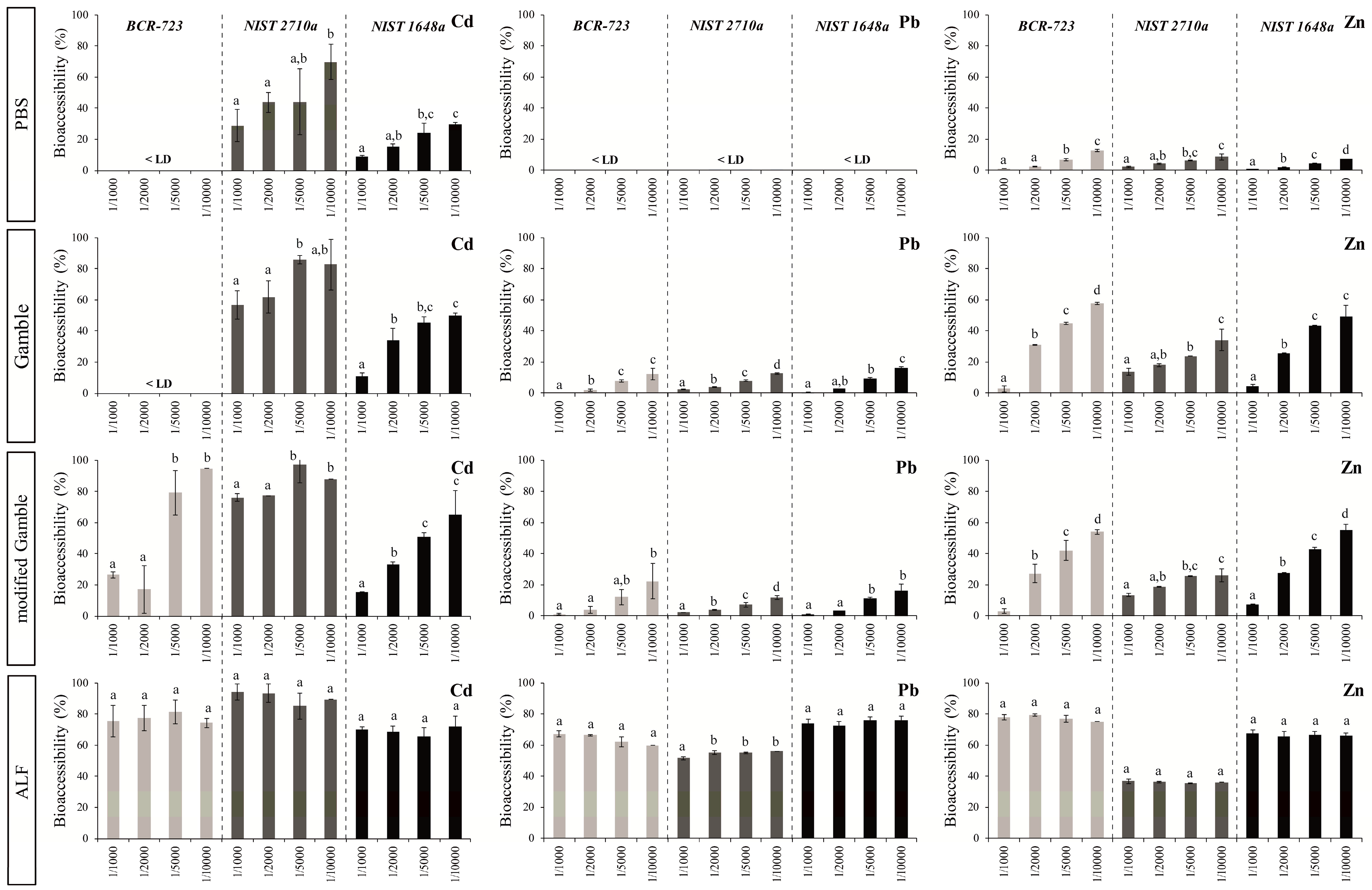
3.1. Influence of the S/L Ratio on Bioaccessibility
3.2. Comparison of in vitro Tests Using PBS, Gamble’s Solution and ALF
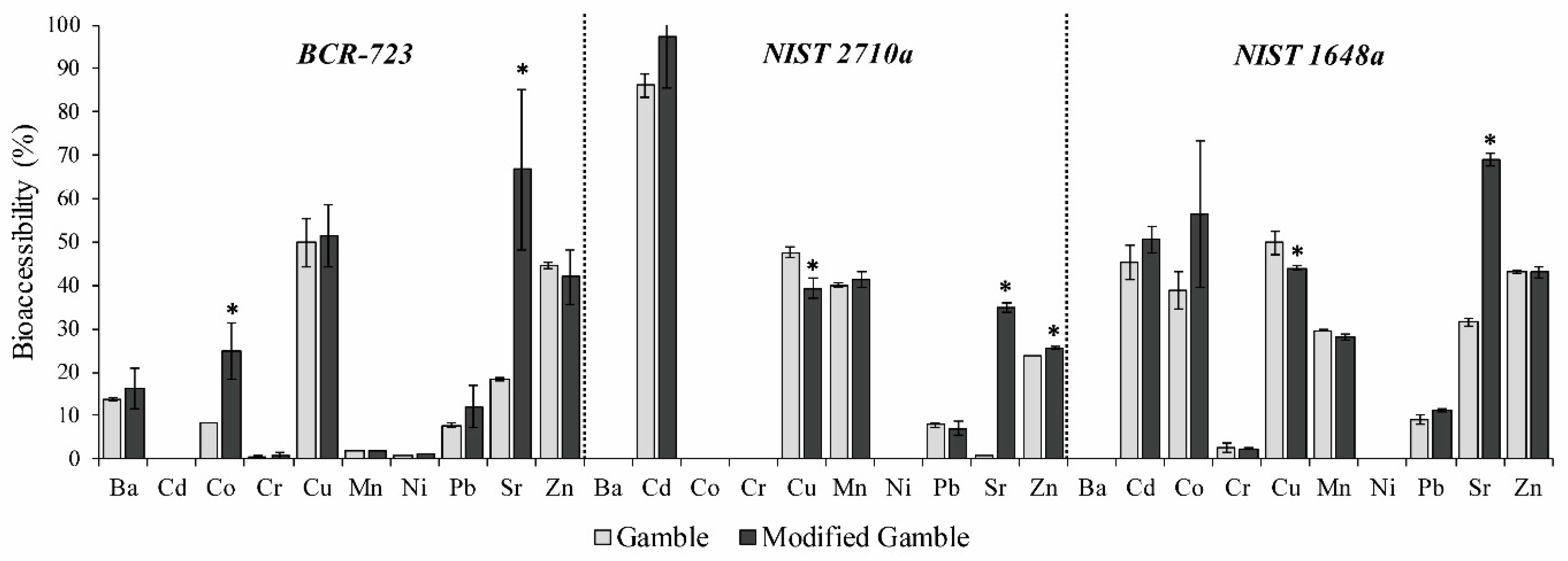
3.3. Influence of Addition of Surfactant in the Gamble’s Solution on Lung Bioaccessibility
4. Conclusions
- Bioaccessibility values are critically dependent on SLF pH;
- Bioaccessibility is closely associated with the lung solution used. Indeed, the chemical composition of lung fluid (with similar pH) had an impact on pulmonary bioaccessibility and can favor the mobilization or immobilization of elements from SRMs;
- Addition of surfactant (DPPC) resulted in no significant change in the bioaccessibility of most of the metallic elements studied;
- Bioaccessibility was speciation- and element-dependent, with percentages varying from 0.04% for Pb to 86.0% for Cd;
- For PBS and Gamble’s solutions, S/L ratios influenced dissolution, while for ALF, bioaccessibility is independent of S/L ratios over a large range from 1/1000 to 1/10,000.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Plumlee, G.S.; Ziegler, T.L. The medical geochemistry of dusts, soils and other earth materials. In Environmental Geochemistry, Treatise on Geochemistry; Lollar, B.S., Holland, H.D., Turekian, K.K., Eds.; Elsevier Ltd.: Oxford, UK, 2003; Volume 9, pp. 263–310. [Google Scholar]
- Taylor, D.M.; Williams, D.R. Bio-inorganic chemistry and its pharmaceutical applications. In Introduction to the Principles of Drug Design and Action; Smith, H.J., Ed.; Harwood Academic: Amsterdam, The Netherlands, 1998; pp. 509–538. [Google Scholar]
- Goyer, R.A.; Clarkson, T.W. Toxic effects of metals. In Casarett and Soull’s Toxicology: The Basic Science of Poisons, 6th ed.; Klassen, C.D., Ed.; McGraw-Hill: New York, NY, USA, 2001; pp. 811–868. [Google Scholar]
- Abrahams, P.W. Soils: Their implications to human health. Sci. Total Environ. 2002, 291, 1–32. [Google Scholar] [CrossRef]
- Bérubé, K.A.; Jones, T.P.; Williamson, B.J.; Winters, C.; Morgan, A.J.; Richards, R.J. Physicochemical characterisation of diesel exhaust particles: Factors for assessing biological activity. Atmos. Environ. 1999, 33, 1599–1614. [Google Scholar] [CrossRef]
- Nieboer, E.; Fletcher, G.G.; Thomassen, Y. Relevance of reactivity determinants of exposure assessment and biological monitoring of the elements. J. Environ. Monitor. 1999, 1, 1–14. [Google Scholar] [CrossRef]
- Jarup, L.; Pershagen, O.; Wall, S. Cumulative arsenic exposure and lung cancer in smelter workers—A case-control study. Am. J. Ind. Med. 1989, 15, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Antonini, J.M.; Murthy, G.G.K.; Rogers, R.A.; Albert, R.; Ulrich, G.D.; Brain, J.D. Pneumotoxicity and pulmonary clearance of different welding fumes after intratracheal instillation in the rat. Toxicol. Appl. Pharm. 1996, 140, 188–199. [Google Scholar] [CrossRef]
- Borak, J.; Cohen, H.; Hethmon, T.A. Copper exposure and metal fume fever: Lack of evidence for a causal relationship. Am. Ind. Hyg. Assoc. J. 2000, 61, 832–836. [Google Scholar] [CrossRef]
- Herting, G.; Odnevall, W.I.; Leygraf, C. Factors that influence the release of metals from stainless steels exposed to physiological media. Corros. Sci. 2006, 48, 2120–2132. [Google Scholar] [CrossRef]
- Bastian, S.; Busch, W.; Kühnel, D.; Springer, A.; Meißner, T.; Holke, R.; Scholz, S.; Iwe, M.; Pompe, W.; Gelinsky, M.; et al. Toxicity of tungsten carbide and cobalt-doped tungsten carbide nanoparticles in mammalian cells in vitro. Environ. Health Persp. 2009, 117, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Goodman, J.E.; Prueitt, R.L.; Thakali, S.; Oller, A.R. The nickel ion bioavailability model of the carcinogenic potential of nickel-containing substances in the lung. Cr. Rev. Toxicol. 2011, 41, 142–174. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, V.; Breznan, D.; Goegan, P.; Nadeau, D.; Karthikeyan, S.; Brook, J.R.; Vincent, R. Effects of ambient air particles on nitric oxide production in macrophage cell lines. Cell Biol. Toxicol. 2004, 20, 221–239. [Google Scholar] [CrossRef] [PubMed]
- Dagher, Z.; Garçon, G.; Billet, S.; Gosset, P.; Ledoux, F.; Courcot, D.; Aboukais, A.; Shirali, P. Activation of different pathways of apoptosis by air pollution particulate matter (PM2.5) in human epithelial lung cells (L132) in culture. Toxicology 2006, 225, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Frelon, S.; Chazel, V.; Tourlonias, E.; Blanchardon, E.; Bouisset, P.; Pourcelot, L.; Paquet, F. Risk assessment after internal exposure to black sand from Camargue: Uptake and prospective dose calculation. Radiat. Prot. Dosim. 2007, 127, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Wragg, J.; Klinck, B. The bioaccessibility of lead from Welsh mine waste using a respiratory uptake test. J. Environ. Sci. Heal. 2007, 42, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.D.; Dang, Z.; Huang, W.L.; Yang, C. Chemical speciation of fine particle bound trace metals. Int. J. Environ. Sci. Technol. 2009, 6, 337–346. [Google Scholar] [CrossRef]
- Barrett, J.E.S.; Taylor, K.G.; Hudson-Edwards, K.A.; Charnock, J.M. Solid-phase speciation of Pb in urban road dust sediment: A XANES and EXAFS study. Environ. Sci. Technol. 2010, 44, 2940–2946. [Google Scholar] [CrossRef] [PubMed]
- Broadway, A.; Cave, M.R.; Wragg, J.; Fordyce, F.M.; Bewley, R.J.F.; Graham, M.C.; Ngwenya, B.T.; Farmer, J.G. Determination of the bioaccessibility of chromium in Glasgow soil and the implications for human health risk assessment. Sci. Total Environ. 2010, 409, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Dockery, D.W.; Pope, C.A. Acute respiratory effects of particulate air pollution. Annu. Rev. Publ. Health 1994, 15, 107–132. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.; Dockery, D.W.; Neas, L.M. Is daily mortality associated specifically with fine particulates? J. Air Waste Manage. Assoc. 1996, 46, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Pope, C.A.; Burnett, R.T.; Thun, M.J.; Cale, E.E.; Krewsi, D.; Ito, K.; Thurston, G.D. Lung cancer, cardiopulmonary mortality and long-term exposure to fine particulate air pollution. J. Am. Med. Assoc. 2002, 287, 1132–1141. [Google Scholar] [CrossRef]
- Al Saadi, M.M. Air pollution and respiratory health: An evolving epidemic. Pak. J. Med. Sci. 2010, 26, 239–243. [Google Scholar]
- Boevski, I.; Daskalova, N.; Havezov, I. Determination of barium, chromium, cadmium, manganese, lead and zinc in atmospheric particulate matter by inductively coupled plasma atomic emission spectrometry (ICP-AES). Spectrochim. Acta 2000, 55, 1643–1657. [Google Scholar] [CrossRef]
- Costa, D.L.; Dreher, K.L. Bioavailable transition metals in particulate matter mediate cardiopulmonary injury in healthy and compromised animal models. Environ. Health Persp. 1997, 105, 1053–1060. [Google Scholar] [CrossRef]
- Adamson, I.Y.R.; Prieditis, H.; Vincent, R. Pulmonary toxicity of an atmospheric particulate sample is due to the soluble fraction. Toxicol. Appl. Pharm. 1999, 157, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Adamson, I.Y.R.; Prieditis, H.; Hedgecock, C.; Vincent, R. Zinc is the toxic factor in the lung response to an atmospheric particulate sample. Toxicol. Appl. Pharm. 2000, 166, 111–119. [Google Scholar] [CrossRef] [PubMed]
- McNeilly, J.D.; Heal, M.R.; Beverland, I.J.; Howe, A.; Gibson, M.D.; Hibbs, L.R.; MacNee, W.; Donaldson, K. Soluble transition metals cause the proinflammatory effects of welding fumes in vitro. Toxicol. Appl. Pharm. 2004, 196, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Molina, R.M.; Schaider, L.A.; Donaghey, T.C.; Shine, J.P.; Brain, J.D. Mineralogy affects geoavailability, bioaccessibility and bioavailability of zinc. Environ. Poll. 2013, 182, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Colin, J.L.; Jaffrezo, J.L.; Gros, J.M. Solubility of major species in precipitation: Factors of variation. Atmos. Environ. 1990, 24, 537–544. [Google Scholar] [CrossRef]
- Desboeufs, K.V.; Losno, R.; Colin, J.L. Factors influencing aerosol solubility during cloud processes. Atmos. Environ. 2001, 35, 3529–3537. [Google Scholar] [CrossRef]
- Gamble, J.L. Chemical Anatomy, Physiology, and Pathology of Extracellular Fluid: A Lecture Syllabus, 6th ed.; Harvard University Press: Cambridge, UK, 1942; p. 164. [Google Scholar]
- Kanapilly, G.M. Alveolar microenvironment and its relationship to the retention and transport into the blood of aerosols deposited in the alveoli. Health Phys. 1977, 32, 89–100. [Google Scholar] [CrossRef] [PubMed]
- De Meringo, A.; Morscheidt, C.; Thélohan, S.; Tiesler, H. In vitro assessment of biodurability: Acellular systems. Environ. Health Persp. 1994, 102, 47–53. [Google Scholar] [CrossRef]
- Brain, J.D. Mechanisms, measurement, and significance of lung macrophage function. Environ. Health Persp. 1992, 97, 5–10. [Google Scholar] [CrossRef]
- Mukhtar, A.; Limbeck, A. Recent developments in assessment of bio-accessible trace metal fractions in airborne particulate matter: A review. Anal. Chim. Acta 2013, 774, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, C.L.S. Analytical methods for assessing metal bioaccessibility in airborne particulate matter: A scoping review. Anal. Chim. Acta 2015, 877, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Paustenbach, D.J.; Meyer, D.M.; Sheehan, P.J.; Lau, V. An assessment and quantitative uncertainty analysis of the health risks to workers exposed to chromium contaminated soils. Toxicol. Ind. Health 1991, 7, 159–196. [Google Scholar] [CrossRef] [PubMed]
- Roesems, G.; Hoet, P.H.M.; Demedts, M.; Nemery, B. In vitro toxicity of cobalt and hard metal dust in rat and human type II pneumocytes. Pharmacol. Toxicol. 1997, 81, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Soukup, J.M.; Ghio, A.J.; Becker, S. Soluble components of Utah Valley particulate pollution alter alveolar macrophage function in vivo and in vitro. Inhal. Toxicol. 2000, 12, 401–414. [Google Scholar] [PubMed]
- Heal, M.R.; Hibbs, L.R.; Agius, R.M.; Beverland, I.J. Total and water-soluble trace metal content of urban background PM10, PM2.5 and black smoke in Edinburgh, UK. Atmos. Environ. 2005, 39, 1417–1430. [Google Scholar] [CrossRef]
- Mwinyihja, M.; Strachan, N.J.C.; Rotariu, O.; Standing, D.; Meharg, A.; Killham, K. Ecotoxicological screening of Kenyan tannery dust using a luminescent-based bacterial biosensor. Int. J. Environ. Heal. Res. 2006, 16, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, M.; Gómez, D.; Dawidowski, L.; Gautier, E.; Smichowski, P. Determination of water-soluble and insoluble compounds in size classified airborne particulate matter. Microchem. J. 2009, 91, 133–139. [Google Scholar] [CrossRef]
- Fernández-Espinoza, A.J.; Rodríguez, M.T.; Barragán de la Rosa, F.J.; Jiménez Sánchez, J.C. A chemical speciation of trace metals for fine urban particles. Atmos. Environ. 2002, 36, 773–780. [Google Scholar] [CrossRef]
- Gómez, D.; Dos Santos, M.; Fujiwara, F.; Polla, G.; Marrero, J.; Dawidowski, L.; Smichowski, P. Fractionation of metals and metalloids by chemical bonding from particles accumulated by electrostatic precipitation in an Argentine thermal power plant. Microchem. J. 2007, 85, 276–284. [Google Scholar] [CrossRef]
- Zanetti, G.; Fubini, B. Surface interaction between metallic cobalt and tungsten carbide particles as a primary cause of hard metal lung disease. J. Mater. Chem. 1997, 7, 1647–1654. [Google Scholar] [CrossRef]
- Sadakane, K.; Ichinose, T.; Takano, H.; Yanagisawa, R.; Sagai, M.; Yoshikawa, T.; Shibamoto, T. Murine strain differences in airway inflammation induced by diesel exhaust particles and house dust mite allergen. Int. Arch. Allergy Imm. 2002, 128, 220–228. [Google Scholar] [CrossRef]
- Midlander, K.; Pan, J.; Leygraf, C. Elaboration of a test method for the study of metal release from stainless steel particles in artificial biological media. Corros. Sci. 2006, 48, 2855–2866. [Google Scholar] [CrossRef]
- Midlander, K.; Wallinder, I.O.; Leygraf, C. In vitro studies of copper release from powder particles in synthetic biological media. Environ. Poll. 2007, 145, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Arora, D.; Shah, K.A.; Halquist, M.S.; Sakagami, M. In vitro aqueous fluid-capacity-limited dissolution testing of respirable aerosol drug particles generated from inhaler products. Pharm. Res. 2010, 27, 786–795. [Google Scholar] [CrossRef] [PubMed]
- Copley, M.; Son, Y.J.; McConville, J. Dissolution testing for inhaled drugs. Pharm. Tech. Europe 2010, 22, 1–5. [Google Scholar]
- Son, Y.J.; Horng, M.; Copley, M.; McConville, J.T. Optimization of an in vitro dissolution test method for inhalation formulations. Dissolut. Technol. 2010, 17, 6–13. [Google Scholar] [CrossRef]
- Ansoborlo, E.; Chalabreysse, J.; Escallon, S.; Hengé-Napoli, M.H. In vitro solubility of uranium tetrafluoride with oxidizing medium compared with in vivo solubility in rats. Int. J. Radiat. Biol. 1990, 58, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Brock, T.; Stopford, W. Bioaccessibility of metals in human health risk assessment: Evaluating risk from exposure to cobalt compounds. J. Environ. Monitor. 2003, 5, 71N–76N. [Google Scholar]
- Colombo, C.; Monhemius, A.J.; Plant, J.A. Platinum, palladium and rhodium release from vehicle exhaust catalysts and road dust exposed to simulated lung fluids. Ecotox. Environ. Safe. 2008, 71, 722–730. [Google Scholar] [CrossRef] [PubMed]
- Stopford, W.; Turner, J.; Cappellini, D.; Brock, T. Bioaccessibility testing of cobalt compounds. J. Environ. Monitor. 2003, 5, 675–680. [Google Scholar] [CrossRef]
- Ansoborlo, E.; Chalabreysse, J.; Hengé-Napoli, H.; Pujol, E. In vitro chemical and cellular tests applied to uranium trioxide with different hydration states. Environ. Health Persp. 1992, 97, 139–143. [Google Scholar] [CrossRef]
- Lison, D.; Lauwerys, R. Cobalt bioavailability from hard metal particles—Further evidence that cobalt alone is not responsible for the toxicity of hard metal particles. Arch. Toxicol. 1994, 68, 528–531. [Google Scholar] [CrossRef] [PubMed]
- Saint-Georges, F.; Garcon, G.; Escande, F.; Abbas, I.; Verdin, A.; Gosset, P.; Mulliez, P.; Shirali, P. Loss of heterozygosity and/or microsatellite instability in multiple critical regions of 3p and 9p chromosomes in human epithelial lung cells (L132) exposed to air pollution particulate matter (PM2.5). In Genotoxicity: Evaluation, Testing and Prediction; Nova Science Publishers: New York, NY, USA, 2009. [Google Scholar]
- Pritchard, R.J.; Ghio, A.J.; Lehmann, J.R.; Winsett, D.W.; Tepper, J.S.; Park, P.; Gilmour, M.I.; Dreher, K.L.; Costa, D.L. Oxidant generation and lung injury after particulate air pollutant exposure increase with the concentrations of associated metals. Inhal. Toxicol. 1996, 8, 457–477. [Google Scholar] [CrossRef]
- Vallyathan, V.; Pack, D.; Leonard, S.; Lawson, R.; Schenker, M.; Castranova, V. Comparative in vitro toxicity of grape- and citrus-farm dusts. J. Tox. Env. Health Part 2007, 70, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Caboche, J.; Esperenza, P.; Bruno, M.; Alleman, L.Y. Developement of an in vitro method to estimate lung bioaccessibility of metals from atmospheric particles. J. Environ. Monitor. 2011, 13, 621–630. [Google Scholar]
- Boisa, N.; Elom, N.; Dean, J.R.; Deary, M.E.; Bird, G.; Entwistle, J.A. Development and application of an inhalation bioaccessibility method (IBM) for lead in the PM10 size fraction of soil. Environ. Int. 2014, 70, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, C.L.; Zereini, F. Characterizing metal (loid) solubility in airborne PM10, PM2.5 and PM1 in Frankfurt, Germany using simulated lung fluids. Atmos. Environ. 2014, 89, 282–289. [Google Scholar] [CrossRef]
- Zischka, M.; Schramel, P.; Muntau, H.; Rehnert, A.; Gomez, M.; Stojanik, B.; Wannemaker, G.; Dams, R.; Quevauviller, P.; Maier, E.A. A new certified reference material for the quality control of palladium, platinum and rhodium in road dust, BCR-723. Trend. Anal. Chem. 2002, 21, 851–868. [Google Scholar] [CrossRef]
- Gray, J.E.; Plumlee, G.S.; Morman, S.A.; Higueras, P.L.; Crock, J.G.; Lowers, H.A.; Witten, M.L. In vitro studies evaluating leaching of mercury from mine waste calcine using simulated human body fluids. Environ. Sci. Technol. 2010, 44, 4782–4788. [Google Scholar] [CrossRef] [PubMed]
- Drysdale, M.; Ljung Bjorjlund, K.; Jamieson, H.E.; Weinstein, P.; Cook, A.; Watkins, R.T. Evaluating the respiratory bioaccessibility of nickel in soil through the use of a simulated lung fluid. Environ. Geochem. Health 2012, 34, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Oller, A.R.; Cappellini, D.; Henderson, R.G.; Bates, H.K. Comparison of nickel release in solutions used for the identification of water-soluble nickel exposures and in synthetic lung fluids. J. Environ. Monitor. 2009, 11, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Rennard, S.I.; Basset, G.; Lecossier, D.; O’Donnell, K.M.; Pinkston, P.; Martin, P.G.; Crystal, R.G. Estimation of volume of epithelial lining fluid recovered by lavage using urea as marker of dilution. J. Appl. Physiol. 1986, 60, 532–538. [Google Scholar] [PubMed]
- Anderson, S.D. Asthma provoked by exercise, hyperventilation, and the inhalation of non-isotonic aerosols. In Asthma Basic Mechanisms and Clinical Management, 2nd ed.; Academic Press: New York, NY, USA, 1992; pp. 473–490. [Google Scholar]
- Weibel, E.R. Morphological basis of alveolar-capillary gas exchange. Physiol. Rev. 1973, 53, 419–495. [Google Scholar] [PubMed]
- Macklin, C.C. Pulmonary sumps, dust accumulations, alveolar fluid and lymph vessels. Acta Anat. 1955, 23, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Potgieter-Vermaak, S.; Rotondo, G.; Novakovic, V.; Rollins, S. Component-specific toxic concerns of the inhalable fraction of urban road dust. Environ. Geochem. Health 2012, 34, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Zereini, F.; Wiseman, C.L.; Püttmann, W. In vitro investigations of platinum, palladium, and rhodium mobility in urban airborne particulate matter (PM10, PM2.5, and PM1) using simulated lung fluids. Environ. Sci. Technol. 2012, 46, 10326–10333. [Google Scholar] [CrossRef] [PubMed]
- Schaider, L.A.; Senn, D.B.; Brabander, D.J.; McCarthy, K.D.; Shine, J.P. Characterization of zinc, lead, and cadmium in mine waste: Implications for transport, exposure, and bioavailability. Environ. Sci. Technol. 2007, 41, 4164–4171. [Google Scholar] [CrossRef] [PubMed]
- Kida, A.; Yukio, N.; Imada, T. Chemical speciation and leaching properties of elements in municipal incinerator ashes. Waste Manage. 1996, 16, 527–536. [Google Scholar] [CrossRef]
- Baker, E.N.; Baker, H.M.; Anderson, B.F.; Reeves, R.D. Chelation of nickel (II) by citrate, the crystal structure of a nickel-citrate complex, K2[Ni(C6H5O7)(H2O2)2]2·4H2O. Inorg. Chim. Acta 1983, 78, 281–285. [Google Scholar] [CrossRef]
- Bernaudat, F.; Bulow, L. Rapid evaluation of nickel binding properties of His-tagged lactate dehydrogenases using surface plasmon resonance. J. Chromatogr. 2005, 1066, 219–224. [Google Scholar] [CrossRef]
- Harris, W.R.; Silberman, D. Time-dependent leaching of coal fly ash by chelating agents. Environ. Sci. Technol. 1983, 17, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Wang, W.; Chan, C.Y.; Cheung, K.C.; Man, Y.B.; Wang, X.; Wong, M.H. Contamination and risk assessment (based on bioaccessibility via ingestion and inhalation) of metal (loid)s in outdoor and indoor particles from urban centers of Guangzhou, China. Sci. Total Environ. 2014, 479–480, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Vousta, D.; Samara, C. Labile and bioaccessible fractions of heavy metals in the airborne particulate matter from urban and industrial areas. Atmos. Environ. 2002, 36, 3583–3590. [Google Scholar]
- Niu, J.; Rasmussen, P.E.; Hassan, N.H.; Vincent, R. Concentration distribution and bioaccessibility of trace elements in nano and fine urban airborne particulate matter: Influence of particle size. Water Air Soil Poll. 2010, 213, 211–225. [Google Scholar] [CrossRef]
- Stebounova, L.V.; Adamcakova-Dodd, A.; Kim, J.S.; Park, H.; O’Shaughnessy, P.T.; Grassian, V.H.; Thorne, P.S. Nanosilver induces minimal lung toxicity or inflammation in a subacute murine inhalation model. Particle Fibre Toxicology 2011, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Morton, N.S. Pulmonary surfactant: Physiology, pharmacology and clinical uses. Brit. J. Hosp. Med. 1989, 42, 52–58. [Google Scholar]
- Davies, N.M.; Feddah, M.R. A novel method for assessing dissolution of aerosol inhaler products. Int. J. Pharm. 2003, 255, 175–187. [Google Scholar] [CrossRef]
- Son, Y.J.; McConville, J.T. Development of a standardized dissolution test method for inhaled pharmaceutical formulations. Int. J. Pharm. 2009, 382, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Riley, T.; Christopher, D.; Arp, J.; Casazza, A.; Colombani, A.; Cooper, A.; Dey, M.; Maas, J.; Mitchell, J.; Reiners, M.; et al. Challenges with developing in vitro dissolution tests for orally inhaled products (OIPs). Am. Assoc. Pharm. Sci. 2012, 13, 978–989. [Google Scholar] [CrossRef] [PubMed]
| Elements | BCR-723 | NIST 2710a | NIST 1648a |
|---|---|---|---|
| Ba | 460 ± 33 | 792 ± 36 | 737 2 |
| Cd | 2.5 ± 0.4 | 12.3 ± 0.3 | 73.7 ± 2.3 |
| Co | 29.8 ± 1.6 | 5.99 ± 0.14 | 17.97 ± 0.68 |
| Cr | 440 ± 18 | 23 ± 6 | 402 ± 13 |
| Cu | 226 ± 3 1 | 3420 ± 50 | 610 ± 70 |
| Mn | 1280 ± 40 | 2140 ± 60 | 790 ± 44 |
| Ni | 171 ± 3 | 8 ± 1 | 81.1 ± 6.8 |
| Pb | 866 ± 16 | 5520 ± 30 | 6550 ± 330 |
| Sr | 254 ± 19 | 255 ± 7 | 215 ± 17 |
| Zn | 1660 ± 100 | 4180 ± 150 | 4800 ± 270 |
| Composition (g·L−1) | PBS 1 | Gamble 2 | Modified Gamble | ALF 3 |
|---|---|---|---|---|
| NaCl | 8.77 | 6.779 | 6.779 | 3.21 |
| Na2HPO4 | 1.28 | 0.071 | ||
| NaHCO3 | 2.268 | 2.268 | ||
| Trisodium citrate dihydrate | 0.055 | 0.055 | 0.077 | |
| NH4Cl | 0.535 | 0.535 | ||
| Glycine | 0.375 | 0.375 | 0.059 | |
| NaH2PO4 | 1.872 | 1.872 | ||
| L-cysteine | 0.121 | 0.121 | ||
| NaOH | 6.0 | |||
| Citric acid | 20.8 | |||
| CaCl2·2H2O | 0.026 | 0.026 | 0.128 | |
| Na2SO4 | 0.039 | |||
| MgCl2·6H2O | 0.05 | |||
| Disodium tartrate | 0.09 | |||
| Sodium lactate | 0.085 | |||
| Sodium pyruvate | 0.172 | |||
| KH2PO4 | 1.36 | |||
| DPPC 4 (surfactant) | 0.01% | |||
| Properties | ||||
| pH | 7.3 ± 0.1 | 7.3 ± 0.1 | 7.3 ± 0.1 | 4.5 ± 0.1 |
| Ionic strength (mol·L−1) | 0.19 | 0.17 | 0.17 | 0.34 |
| Lung Fluids | Elements (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ba | Cd | Co | Cr | Cu | Mn | Ni | Pb | Sr | Zn | |
| BCR-723 | ||||||||||
| PBS | 0.5 ± 0.3 a | <LD | 14.0 ± 6.0 a | 0.8 ± 0.5 a | 4.1 ± 1.5 a | 0.9 ± 0.0 a | <LD | <LD | 22.1 ± 1.2 a | 6.8 ± 0.8 a |
| Gamble | 13.6 ± 0.4 b | <LD | 8.2 ± 0.0 a | 0.5 ± 0.3 a | 49.9 ± 5.6 b | 1.7 ± 0.0 b | 0.8 ± 0.0 a | 7.8 ± 0.6 a | 18.5 ± 0.4 a | 44.6 ± 0.8 b |
| ALF | 35.7 ± 0.5 b | 81.4 ± 7.6 a | 39.8 ± 15.3 a | 8.7 ± 0.0 b | 65.2± 3.7 c | 5.5 ± 0.1 c | 24.1 ± 3.7 b | 62.0 ± 3.2 b | 56.2 ± 3.2 b | 76.8 ± 2.2 c |
| NIST 2710a | ||||||||||
| PBS | <LD | 44.2 ± 21.2 a | 95.1 ± 52.7 a | 7.8 ± 0.0 | 8.3 ± 0.2 a | 28.7 ± 0.4 a | <LD | 0.04 ± 0.00 a | 2.8 ± 0.0 a | 6.2 ± 0.1 a |
| Gamble | <LD | 86.0 ± 2.8 a | <LD | <LD | 47.6 ± 1.4 b | 40.1 ± 0.7 b | <LD | 7.9 ± 0.4 b | 0.8 ± 0.2 a | 23.7 ± 0.1 b |
| ALF | 25.3 ± 0.4 a | 85.3 ± 8.4 a | 35.1 ± 0.0 a | <LD | 59.7 ± 1.4 c | 44.3 ± 0.2 c | <LD | 55.0 ± 0.5 c | 2.3 ± 0.5 a | 35.3 ± 0.1 c |
| NIST 1648a | ||||||||||
| PBS | <LD | 24.1 ± 6.2 a | 3.3 ± 0.0 a | 1.3 ± 0.4 a | 7.3 ± 1.8 a | 16.4 ± 1.4 a | <LD | <LD | 29.0 ± 23.8 a | 4.3 ± 0.2 a |
| Gamble | <LD | 45.2 ± 4.0 b | 38.8 ± 4.3 b | 2.7 ± 1.0 a | 49.9 ± 2.7 b | 29.6 ± 0.2 b | 3.3 ± 1.2 a | 9.1 ± 0.9 a | 31.5 ± 0.9 a | 43.2 ± 0.2 b |
| ALF | 52.8 ± 1.9 a | 65.6 ± 5.5 c | 35.0 ± 16.5 b | 8.7 ± 0.9 b | 55.0 ± 1.1 c | 46.8 ± 2.6 c | 12.2 ± 4.1 a | 75.9 ± 2.2 b | 50.9 ± 3.2 a | 66.2 ± 2.3 c |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pelfrêne, A.; Cave, M.R.; Wragg, J.; Douay, F. In Vitro Investigations of Human Bioaccessibility from Reference Materials Using Simulated Lung Fluids. Int. J. Environ. Res. Public Health 2017, 14, 112. https://doi.org/10.3390/ijerph14020112
Pelfrêne A, Cave MR, Wragg J, Douay F. In Vitro Investigations of Human Bioaccessibility from Reference Materials Using Simulated Lung Fluids. International Journal of Environmental Research and Public Health. 2017; 14(2):112. https://doi.org/10.3390/ijerph14020112
Chicago/Turabian StylePelfrêne, Aurélie, Mark R. Cave, Joanna Wragg, and Francis Douay. 2017. "In Vitro Investigations of Human Bioaccessibility from Reference Materials Using Simulated Lung Fluids" International Journal of Environmental Research and Public Health 14, no. 2: 112. https://doi.org/10.3390/ijerph14020112
APA StylePelfrêne, A., Cave, M. R., Wragg, J., & Douay, F. (2017). In Vitro Investigations of Human Bioaccessibility from Reference Materials Using Simulated Lung Fluids. International Journal of Environmental Research and Public Health, 14(2), 112. https://doi.org/10.3390/ijerph14020112






