Association between Exposure to Endocrine Disruptors in Drinking Water and Preterm Birth, Taking Neighborhood Deprivation into Account: A Historic Cohort Study
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Area
2.2. Individual Data
2.3. Area-Level Data
2.4. Exposure
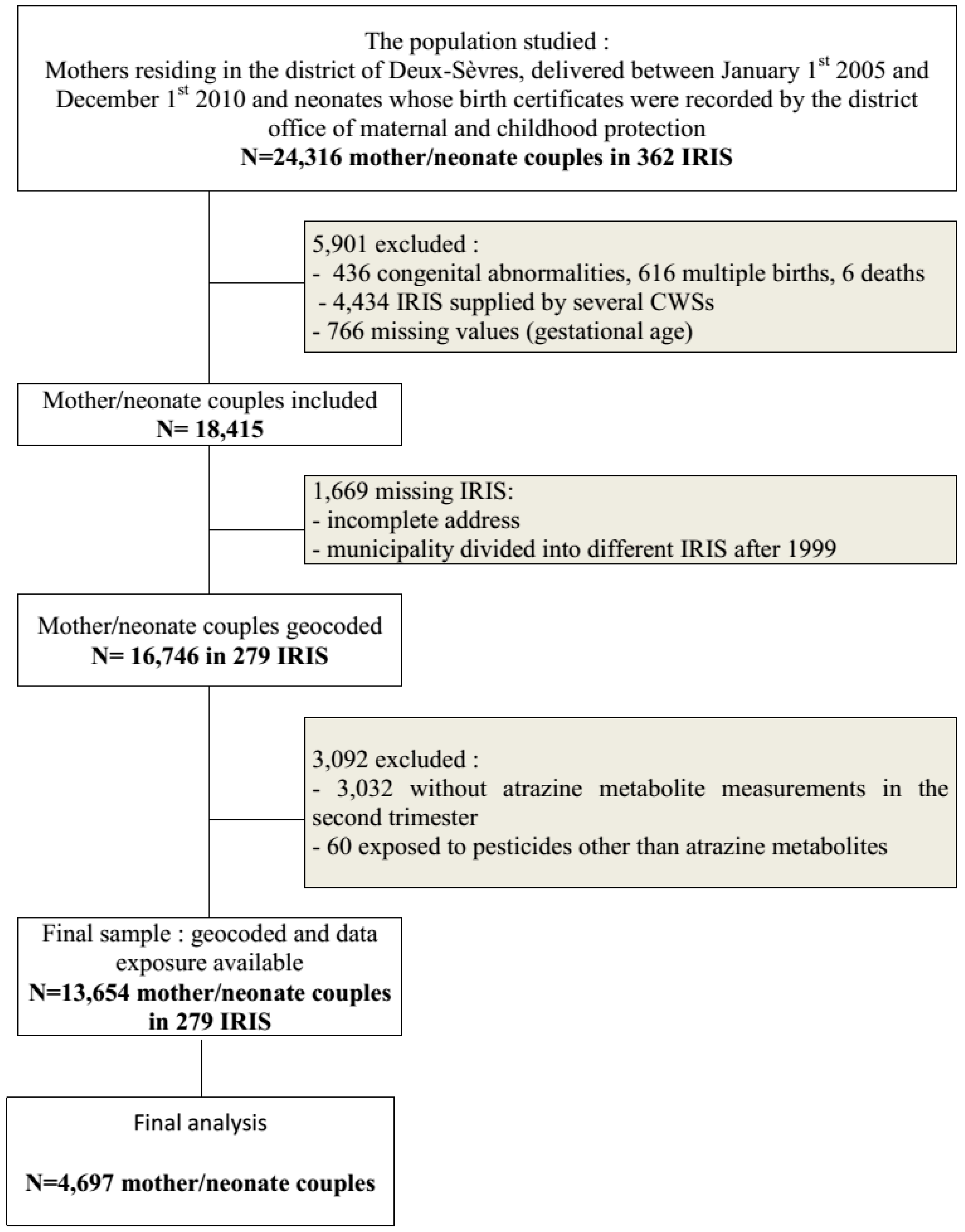
2.5. Analysis Dataset
2.6. Exclusions to the Dataset Included
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ancel, P.Y. Epidemiology of preterm births. Rev. Prat. 2012, 62, 362–365. [Google Scholar] [PubMed]
- Kramer, M.S.; Demissie, K.; Yang, H.; Platt, R.W.; Sauve, R.; Liston, R. The contribution of mild and moderate preterm birth to infant mortality. Fetal and Infant Health Study Group of the Canadian Perinatal Surveillance System. JAMA 2000, 284, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Grellier, J.; Bennett, J.; Patelarou, E.; Smith, R.B.; Toledano, M.B.; Rushton, L.; Briggs, D.J.; Nieuwenhuijsen, M.J. Exposure to disinfection by-products, fetal growth, and prematurity, a systematic review and meta-analysis. Epidemiology 2010, 21, 300–313. [Google Scholar] [CrossRef] [PubMed]
- Stieb, D.M.; Chen, L.; Eshoul, M.; Judek, S. Ambient air pollution, birth weight and preterm birth, a systematic review and meta-analysis. Environ. Res. 2012, 117, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Auger, N.; Park, A.L.; Gamache, P.; Pampalon, R.; Daniel, M. Weighing the contributions of material and social area deprivation to preterm birth. Soc. Sci. Med. 2012, 75, 1032–1037. [Google Scholar] [CrossRef] [PubMed]
- Calling, S.; Li, X.; Sundquist, J.; Sundquist, K. Socioeconomic inequalities and infant mortality of 46,470 preterm infants born in Sweden between 1992 and 2006. Paediatr. Perinat. Epidemiol. 2011, 25, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Jansen, P.W.; Tiemeier, H.; Jaddoe, V.W.; Hofman, A.; Steegers, E.A.; Verhulst, F.C.; Mackenbach, J.P.; Raat, H. Explaining educational inequalities in preterm birth, the generation r study. Arch. Dis. Child Fetal Neonatal. 2009, 94, F28–F34. [Google Scholar] [CrossRef] [PubMed]
- Messer, L.C.; Vinikoor, L.C.; Laraia, B.A.; Kaufman, J.S.; Eyster, J.; Holzman, C.; Culhane, J.; Elo, I.; Burke, J.G.; O’Campo, P. Socioeconomic domains and associations with preterm birth. Soc. Sci Med. 2008, 67, 1247–1257. [Google Scholar] [CrossRef] [PubMed]
- Niedhammer, I.; Murrin, C.; O’Mahony, D.; Daly, S.; Morrison, J.J.; Kelleher, C.C. Explanations for social inequalities in preterm delivery in the prospective Lifeways cohort in the Republic of Ireland. Eur. J. Public Health 2011, 22, 533–538. [Google Scholar] [CrossRef] [PubMed]
- O’Campo, P.; Burke, J.G.; Culhane, J.; Elo, I.T.; Eyster, J.; Holzman, C.; Messer, L.C.; Kaufman, J.S.; Laraia, B.A. Neighborhood deprivation and preterm birth among non-Hispanic Black and White women in eight geographic areas in the United States. Am. J. Epidemiol. 2008, 167, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, S.; Bonsel, G.J.; Steegers-Theunissen, R.P.; Mackenbach, J.P.; Steyerberg, E.W.; Raat, H.; Verbrugh, H.A.; Tiemeier, H.W.; Hofman, A.; Birnie, E.; et al. Individual accumulation of heterogeneous risks explains perinatal inequalities within deprived neighbourhoods. Eur. J. Epidemiol. 2011, 26, 165–180. [Google Scholar] [CrossRef] [PubMed]
- Zeitlin, J.; Combier, E.; Levaillant, M.; Lasbeur, L.; Pilkington, H.; Charreire, H.; Rivera, L. Neighbourhood socio-economic characteristics and the risk of preterm birth for migrant and non-migrant women, a study in a French district. Paediatr. Perinat. Epidemiol. 2011, 25, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Bourguignon, J.P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-disrupting chemicals, an Endocrine Society scientific statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, K.K.; O’Neill, M.S.; Meeker, J.D. Environmental contaminant exposures and preterm birth, a comprehensive review. J. Toxicol. Environ. Health 2013, 16, 69–113. [Google Scholar] [CrossRef] [PubMed]
- Goodman, M.; Mandel, J.S.; DeSesso, J.M.; Scialli, A.R. Atrazine and pregnancy outcomes: A systematic review of epidemiologic evidence. Birth Defects Res. B Dev. Reprod. Toxicol. 2014, 101, 215–236. [Google Scholar] [CrossRef] [PubMed]
- Rinsky, J.L.; Hopenhayn, C.; Golla, V.; Browning, S.; Bush, H.M. Atrazine exposure in public drinking water and preterm birth. Public Health Rep. 2012, 127, 72–80. [Google Scholar] [PubMed]
- Ochoa-Acuna, H.; Frankenberger, J.; Hahn, L.; Carbajo, C. Drinking-water herbicide exposure in Indiana and prevalence of small-for-gestational-age and preterm delivery. Environ. Health Perspect. 2009, 117, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, C.M.; Durand, G.; Coutte, M.B.; Chevrier, C.; Cordier, S. Atrazine in municipal drinking water and risk of low birth weight, preterm delivery, and small-for-gestational-age status. Occup. Environ. Med. 2005, 62, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Charafeddine, R.; Boden, L. Does income inequality modify the association between air pollution and health? Environ. Res. 2008, 106, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Genereux, M.; Auger, N.; Goneau, M.; Daniel, M. Neighbourhood socioeconomic status, maternal education and adverse birth outcomes among mothers living near highways. J. Epidemiol. Community Health 2008, 62, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Yi, O.; Kim, H.; Ha, E. Does area level socioeconomic status modify the effects of PM(10) on preterm delivery? Environ. Res. 2010, 110, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Bukowski, J.; Somers, G.; Bryanton, J. Agricultural contamination of groundwater as a possible risk factor for growth restriction or prematurity. J. Occup. Environ. Med. 2001, 43, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Guillette, L.J., Jr.; Edwards, T.M. Is nitrate an ecologically relevant endocrine disruptor in vertebrates? Integr. Comp. Biol. 2005, 45, 19–27. [Google Scholar] [CrossRef] [PubMed]
- INSEE. Poverty in Poitou-Charentes. 2004. Available online: http://www.insee.fr/fr/themes/document.asp?reg_id=29&ref_id=23981 (accessed on 6 June 2016).
- Collet, M.; Vilain, A. French birth certificates of 8th day. Validity of 2006 and 2007. Available online: http://fulltext.bdsp.ehesp.fr/Ministere/Drees/SerieSourcesMethodes/2012/28/seriesource_method28.pdf (accessed on 6 June 2016).
- Liberatos, P.; Link, B.G.; Kelsey, J.L. The measurement of social class in epidemiology. Epidemiol. Rev. 1988, 10, 87–121. [Google Scholar] [PubMed]
- Pornet, C.; Delpierre, C.; Dejardin, O.; Grosclaude, P.; Launay, L.; Guittet, L.; Lang, T.; Launoy, G. Construction of an adaptable European transnational ecological deprivation index, the French version. J. Epidemiol. Community Health 2012, 66, 982–989. [Google Scholar] [CrossRef] [PubMed]
- CMH. Maurice Halbwachs Center. Available online: http://www.cmh.ens.fr (accessed on 6 June 2016).
- Migeot, V.; Albouy-Llaty, M.; Carles, C.; Limousi, F.; Strezlec, S.; Dupuis, A.; Rabouan, S. Drinking-water exposure to a mixture of nitrate and low-dose atrazine metabolites and small-for-gestational age (SGA) babies, a historic cohort study. Environ. Res. 2013, 122, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Hein, M.J.; Deddens, J.A.; Hines, C.J. Analysis of lognormally distributed exposure data with repeated measures and values below the limit of detection using SAS. Ann. Occup. Hyg. 2011, 55, 97–112. [Google Scholar] [CrossRef] [PubMed]
- Voltolini, C.; Torricelli, M.; Conti, N.; Vellucci, F.L.; Severi, F.M.; Petraglia, F. Understanding spontaneous preterm birth, from underlying mechanisms to predictive and preventive interventions. Reprod. Sci. 2013, 20, 1274–1292. [Google Scholar] [CrossRef] [PubMed]
- Behrman, R.E.; Butler, A.S. Preterm Birth, Causes, Consequences, and Prevention, 1st ed.; The National Academy Press: Washington, DC, USA, 2007; p. 229. [Google Scholar]
- Chaix, B.; Chauvin, P. The contribution of multilevel models in contextual analysis in the field of social epidemiology, a review of literature. Rev. Epidemiol. Sante. Publ. 2002, 50, 489–499. [Google Scholar]
- Chevrier, C.; Limon, G.; Monfort, C.; Rouget, F.; Garlantezec, R.; Petit, C.; Durand, G.; Cordier, S. Urinary biomarkers of prenatal atrazine exposure and adverse birth outcomes in the PELAGIE birth cohort. Environ. Health Perspect. 2011, 119, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
- UE. Directive 98/83/CE du Conseil Européen du 3 Novembre 1998 Relative à la Qualité des Eaux Destinées à la Consommation Humaine. 1998. Available online: https://www.legifrance.gouv.fr/affichTexte.do?cidTexte=JORFTEXT000000521549 (accessed on 6 June 2016).
- Jones, R.M.; Stayner, L.T.; Demirtas, H. Multiple imputation for assessment of exposures to drinking water contaminants: Evaluation with the Atrazine Monitoring Program. Environ. Res. 2014, 134, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Albouy-Llaty, M.; Dupuis, A.; Grignon, C.; Strezlec, S.; Pierre, F.; Rabouan, S.; Migeot, V. Estimating drinking-water ingestion and dermal contact with water in a French population of pregnant women: the EDDS cohort study. J. Expo. Sci. Environ. Epidemiol. 2015, 25, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Doria, M.F. Bottled water versus tap water, understanding consumers’ preferences. J. Water Health 2006, 4, 271–276. [Google Scholar] [PubMed]
- Nieuwenhuijsen, M.J. Exposure Assessment in Occupational and Environmental Eidemiology, 1st ed.; Oxford University Press: New York, NY, USA, 2003; p. 416. [Google Scholar]
- Pantazis, C.; Townsend, P.; Gordon, D. The Necessities of life in Britain. Available online: http://www.bris.ac.uk/poverty/pse/work_pap.htm (accessed on 6 June 2016).
- Kramer, M.S.; Wilkins, R.; Goulet, L.; Seguin, L.; Lydon, J.; Kahn, S.R.; McNamara, H.; Dassa, C.; Dahhou, M.; Masse, A.; et al. Investigating socio-economic disparities in preterm birth, evidence for selective study participation and selection bias. Paediatr. Perinat. Epidemiol. 2009, 23, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Health Regional Observatory. Mother and Child Health in Poitou-Charentes; Health Regional Observatory: Poitiers, France, 2013. [Google Scholar]
- Ribet, C.; Melchior, M.; Lang, T.; Zins, M.; Goldberg, M.; Leclerc, A. Characterisation and measurement of social position in epidemiologic studies. Rev. Epidemiol. Sante. Publ. 2007, 55, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Blondel, B.; Lelong, N.; Kermarrec, M.; Goffinet, F. Trends in perinatal health in France from 1995 to 2010. Results from the French National Perinatal Surveys. J. Gynecol. Obstet. Biol. Reprod. (Paris) 2010, 41, e1–e15. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.R.; Dunlop, A.L.; Hogue, C.J. Measuring Women’s cumulative neighborhood deprivation exposure using longitudinally linked vital records, a method for life course MCH research. Matern. Child Health J. 2014, 18, 478–487. [Google Scholar] [CrossRef] [PubMed]
| Pairs | Missing Values | Preterm Birth | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | (%) | n | (%) | n | % | ORa | 95% CI | p | |||||
| Individual data | |||||||||||||
| Maternal age | 0.375 | ||||||||||||
| <20 years | 331 | (2%) | 26 | (0.1%) | 16 | (4.8%) | 1.165 | [ | 0.699 | ; | 1.941 | ] | |
| 20–35 years | 11,198 | (82%) | 468 | (4.2%) | 1 | ||||||||
| >35 years | 2099 | (15%) | 101 | (4.8%) | 1.159 | [ | 0.930 | ; | 1.445 | ] | |||
| Mother’s occupation a | 0.561 | ||||||||||||
| Disadvantaged | 4218 | (31%) | 140 | (1%) | 179 | (4.2%) | 1 | ||||||
| Moderately advantaged | 7575 | (56%) | 330 | (4.4%) | 1.129 | [ | 0.845 | ; | 1.508 | ] | |||
| Advantaged | 1721 | (13%) | 65 | (3.8%) | 1.160 | [ | 0.885 | ; | 1.522 | ] | |||
| Smoking during pregnancy | 7404 | (54%) | 0.110 | ||||||||||
| no | 5012 | (80%) | 200 | (4.0%) | 1 | ||||||||
| yes | 1238 | (20%) | 62 | (5.0%) | 1.269 | [ | 0.947 | ; | 1.699 | ] | |||
| Single-parent family | 0 | (0%) | 0.330 | ||||||||||
| no | 12,957 | (95%) | 551 | (4.3%) | 1 | ||||||||
| yes | 697 | (5%) | 35 | (5.0%) | 1.190 | [ | 0.839 | ; | 1.690 | ] | |||
| History of preterm birth | 4547 | (33%) | <10−3 | ||||||||||
| no | 8675 | (95%) | 374 | (4.3%) | 1 | ||||||||
| yes | 432 | (5%) | 59 | (13.7%) | 3.511 | [ | 2.618 | ; | 4.708 | ] | |||
| Primiparity | 654 | (5%) | <10−3 | ||||||||||
| no | 7439 | (57%) | 267 | (3.6%) | 1 | ||||||||
| yes | 5561 | (43%) | 284 | (5.1%) | 1.446 | [ | 1.219 | ; | 1.715 | ] | |||
| Quality of follow up b | 2995 | (22%) | <10−3 | ||||||||||
| high | 10,246 | (96%) | 48 | (3.6%) | 1 | ||||||||
| low | 413 | (4%) | 369 | (11.6%) | 3.520 | [ | 2.560 | ; | 4.840 | ] | |||
| European deprivation index c | |||||||||||||
| Tertile 1 (least deprived) | 5756 | (42%) | 0 | (0%) | 234 | (4.1%) | 1 | 0.05 | |||||
| Tertile 2 | 5202 | (38%) | 213 | (4.1%) | 1.008 | [ | 0.384 | ; | 1.218 | ] | |||
| Tertile 3 (most deprived) | 2696 | (20%) | 139 | (5.2%) | 1.283 | [ | 1.035 | ; | 1.591 | ] | |||
| Exposure to atrazine metabolites during second trimester | |||||||||||||
| 2-hydroxy-atrazine | |||||||||||||
| 0.013 µg/L | 7145 | (52%) | 0 | (0%) | 299 | (4.2%) | 1 | 0.642 | |||||
| 0.013–0.02 µg/L | 585 | (4%) | 29 | (5.0%) | 1.194 | [ | 0.808 | ; | 1.765 | ] | |||
| >0.02 µg/L | 5924 | (43%) | 258 | (4.4%) | 1.043 | [ | 0.879 | ; | 1.236 | ] | |||
| Exposure to nitrate during second trimester | |||||||||||||
| Nitrates | |||||||||||||
| <16.1 mg/L | 4493 | (33%) | 0 | (0%) | 186 | (4.0%) | 1 | 0.433 | |||||
| 16.1–27.2 mg/L | 4508 | (33%) | 209 | (5.0%) | 1.126 | [ | 0.920 | ; | 1.378 | ] | |||
| >27.2 mg/L | 4480 | (33%) | 187 | (4.0%) | 1.009 | [ | 0.820 | ; | 1.241 | ] | |||
| 2-hydroxyatrazine Exposure during Second Trimester | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.013 µg/L | 0.013–0.020 µg/L | >0.020 µg/L | |||||||
| Pairs n (%) | n | % | n | % | n | % | p | ||
| European deprivation index | 0.004 | ||||||||
| Tertile 1 (least deprived) | 5756 | (42%) | 3079 | (53.5%) | 221 | (3.8%) | 2456 | (42.7%) | |
| Tertile 2 | 5202 | (38%) | 2664 | (51.2%) | 262 | (5.0%) | 2276 | (43.8%) | |
| Tertile 3 (most deprived) | 2696 | (20%) | 1402 | (52.0%) | 102 | (3.8%) | 1192 | (44.2%) | |
| Live in rural area | <10−3 | ||||||||
| no | 9505 | (70%) | 5265 | (55.4%) | 282 | (3.0%) | 3958 | (41.6%) | |
| yes | 4149 | (30%) | 1880 | (45.3%) | 303 | (7.3%) | 1966 | (47.4%) | |
| Season during second trimester | <10−3 | ||||||||
| summer | 3377 | (25%) | 1703 | (50.4%) | 191 | (5.6%) | 1483 | (43.9%) | |
| autumn | 3696 | (27%) | 1550 | (41.9%) | 19 | (0.5%) | 2127 | (57.5%) | |
| winter | 2890 | (21%) | 1751 | (61.6%) | 97 | (3.4%) | 1042 | (36.1%) | |
| spring | 3691 | (27%) | 2141 | (58.0%) | 278 | (7.5%) | 1272 | (34.5%) | |
| Police 9 | Preterm Birth Risk | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Model 1 (n = 13,654) | Model 2 (n = 4697) | Model 3 (n = 4697) | |||||||||||||||||||
| ORa | 95% CI | p | ORa | 95% CI | p | ORa | 95% CI | p | |||||||||||||
| Exposure data during second trimester | |||||||||||||||||||||
| 2-hydroxyatrazine | |||||||||||||||||||||
| 0.013–0.020 µg/L vs. 0.013 µg/L | 1.168 | [ | 0.786 | ; | 1.736 | ] | 0.706 | 0.938 | [ | 0.421 | ; | 2.090 | ] | 0.944 | 0.929 | [ | 0.666 | ; | 1.345 | ] | 0.944 |
| >0.020 µg/L vs. 0.013 µg/L | 1.042 | [ | 0.876 | ; | 1.239 | ] | 0.945 | [ | 0.665 | ; | 1.343 | ] | 0.946 | [ | 0.705 | ; | 1.412 | ] | |||
| Live in rural area: yes vs. no | 1.058 | [ | 0.884 | ; | 1.266 | ] | 0.540 | 1.301 | [ | 0.948 | ; | 1.784 | ] | 0.103 | 1.316 | [ | 0.949 | ; | 1.827 | ] | 0.100 |
| Season: autumn vs. summer | 0.854 | [ | 0.678 | ; | 1.074 | ] | 0.462 | 0.662 | [ | 0.433 | ; | 1.012 | ] | 0.217 | 0.662 | [ | 0.433 | ; | 1.012 | ] | 0.217 |
| Season: winter vs. summer | 0.923 | [ | 0.726 | ; | 1.173 | ] | 1.007 | [ | 0.675 | ; | 1.502 | ] | 1.006 | [ | 0.674 | ; | 1.501 | ] | |||
| Season: spring vs. summer | 0.852 | [ | 0.678 | ; | 1.071 | ] | 0.891 | [ | 0.611 | ; | 1.301 | ] | 0.883 | [ | 0.605 | ; | 1.290 | ] | |||
| Individual data | |||||||||||||||||||||
| Maternal age | |||||||||||||||||||||
| <20 vs. 20 to 35 years | 1.022 | [ | 0.448 | ; | 2.331 | ] | 0.176 | 0.983 | [ | 0.430 | ; | 2.247 | ] | 0.1611 | |||||||
| >35 vs. 20 to 35 years | 1.453 | [ | 0.981 | ; | 2.150 | ] | 1.465 | [ | 0.990 | ; | 2.170 | ] | |||||||||
| Mother’s occupation a | |||||||||||||||||||||
| Disadvantaged vs. Advantaged | 1.280 | [ | 0.697 | ; | 2.351 | ] | 0.726 | 1.194 | [ | 0.646 | ; | 2.207 | ] | 0.839 | |||||||
| Moderately advantaged vs. Advantaged | 1.202 | [ | 0.677 | ; | 2.136 | ] | 1.183 | [ | 0.665 | ; | 2.101 | ] | |||||||||
| Smoking during pregnancy: yes vs. no | 1.076 | [ | 0.746 | ; | 1.551 | ] | 0.696 | 1.075 | [ | 0.745 | ; | 1.550 | ] | 0.701 | |||||||
| Single-parent family: yes vs. no | 0.827 | [ | 0.433 | ; | 1.579 | ] | 0.565 | 0.783 | [ | 0.409 | ; | 1.500 | ] | 0.461 | |||||||
| History of preterm birth: yes vs. no | 5.946 | [ | 3.723 | ; | 9.497 | ] | <10−3 | 5.890 | [ | 3.686 | ; | 9.414 | ] | <10−3 | |||||||
| Primiparity: yes vs. no | 2.099 | [ | 1.473 | ; | 2.992 | ] | <10−3 | 2.089 | [ | 1.465 | ; | 2.979 | ] | <10−3 | |||||||
| Quality of follow-up: low vs. high | 4.606 | [ | 2.566 | ; | 8.268 | ] | <10−3 | 4.487 | [ | 2.494 | ; | 8.074 | ] | <10−3 | |||||||
| European deprivation index (EDI) | |||||||||||||||||||||
| Tertile 2 vs. Tertile 1 (least deprived) | 0.998 | [ | 0.705 | ; | 1.412 | ] | 0.204 | ||||||||||||||
| Tertile 3 (most deprived) vs. Tertile 1 | 1.363 | [ | 0.929 | ; | 2.000 | ] | |||||||||||||||
| Police 9 | Preterm Birth Risk | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Model 1 (n = 8735) | Model 2 (n = 3806) | |||||||||||||
| ORa | 95% CI | p | ORa | 95% CI | p | |||||||||
| Exposure data during second trimester | ||||||||||||||
| 2-hydroxyatrazine | ||||||||||||||
| 0.013–0.020 µg/L vs. 0.013 µg/L | 1.162 | [ | 0.779 | ; | 1.733 | ] | 0.504 | 0.968 | [ | 0.432 | ; | 2.169 | ] | 0.171 |
| >0.020 µg/L vs. 0.013 µg/L | 1.164 | [ | 0.863 | ; | 1.570 | ] | 1.625 | [ | 0.975 | ; | 2.710 | ] | ||
| Live in rural area: yes vs. no | 1.076 | [ | 0.859 | ; | 1.347 | ] | 0.526 | 1.228 | [ | 0.860 | ; | 1.754 | ] | 0.258 |
| Season: autumn vs. summer | 0.750 | [ | 0.564 | ; | 0.998 | ] | 0.051 | 0.683 | [ | 0.437 | ; | 1.067 | ] | 0.390 |
| Season: winter vs. summer | 0.794 | [ | 0.592 | ; | 1.064 | ] | 0.913 | [ | 0.588 | ; | 1.418 | ] | ||
| Season: spring vs. summer | 0.693 | [ | 0.524 | ; | 0.916 | ] | 0.824 | [ | 0.545 | ; | 1.245 | ] | ||
| Individual data | ||||||||||||||
| Maternal age | ||||||||||||||
| <20 vs. 20 to 35 years | 1.167 | [ | 0.502 | ; | 2.713 | ] | 0.255 | |||||||
| >35 vs. 20 to 35 years | 1.426 | [ | 0.931 | ; | 2.184 | ] | ||||||||
| Mother’s occupation a | ||||||||||||||
| Disadvantaged vs. Advantaged | 1.069 | [ | 0.541 | ; | 2.111 | ] | 0.820 | |||||||
| Moderately advantaged vs. Advantaged | 0.951 | [ | 0.497 | ; | 1.819 | ] | ||||||||
| Smoking during pregnancy: yes vs. no | 1.046 | [ | 0.702 | ; | 1.557 | ] | 0.826 | |||||||
| Single-parent family: yes vs. no | 0.746 | [ | 0.378 | ; | 1.473 | ] | 0.399 | |||||||
| History of preterm birth: yes vs. no | 6.185 | [ | 3.677 | ; | 10.405 | ] | <10−3 | |||||||
| Primiparity: yes vs. no | 1.875 | [ | 1.285 | ; | 2.736 | ] | 0.001 | |||||||
| Quality of follow-up: low vs. high | 4.807 | [ | 2.544 | ; | 9.082 | ] | <10−3 | |||||||
| Preterm Birth Risk | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Model 1 (n = 13,481) | Model 2 (n = 4625) | Model 3 (n = 4625) | |||||||||||||||||||
| ORa | 95% CI | p | ORa | 95% CI | p | ORa | 95% CI | p | |||||||||||||
| Exposure data during second trimester | |||||||||||||||||||||
| Exposure to nitrates and atrazine metabolites a | |||||||||||||||||||||
| P0N2 vs. P0N1 | 1.289 | [ | 0.996 | ; | 1.668 | ] | 0.454 | 0.890 | [ | 0.552 | ; | 1.433 | ] | 0.380 | 0.861 | [ | 0.529 | ; | 1.404 | ] | 0.383 |
| P0N3 vs. P0N1 | 1.093 | [ | 0.836 | ; | 1.428 | ] | 0.752 | [ | 0.461 | ; | 1.226 | ] | 0.755 | [ | 0.461 | ; | 1.235 | ] | |||
| P1N1 vs. P0N1 | 1.211 | [ | 0.899 | ; | 1.630 | ] | 1.305 | [ | 0.747 | ; | 2.281 | ] | 1.297 | [ | 0.740 | ; | 2.274 | ] | |||
| P1N2 vs. P0N1 | 1.079 | [ | 0.657 | ; | 1.771 | ] | 1.103 | [ | 0.559 | ; | 2.179 | ] | 1.077 | [ | 0.542 | ; | 2.138 | ] | |||
| P1N3 vs. P0N1 | 1.196 | [ | 0.822 | ; | 1.739 | ] | 1.044 | [ | 0.577 | ; | 1.892 | ] | 1.062 | [ | 0.586 | ; | 1.925 | ] | |||
| Live in rural area: yes vs. no | 1.077 | [ | 0.895 | ; | 1.297 | ] | 0.430 | 1.196 | [ | 0.858 | ; | 1.669 | ] | 0.291 | 1.198 | [ | 0.842 | ; | 1.703 | ] | 0.315 |
| Season: autumn vs. summer | 0.841 | [ | 0.669 | ; | 1.058 | ] | 0.399 | 0.654 | [ | 0.427 | ; | 1.003 | ] | 0.143 | 0.653 | [ | 0.426 | ; | 1.002 | ] | 0.140 |
| Season: winter vs. summer | 0.907 | [ | 0.710 | ; | 1.158 | ] | 1.084 | [ | 0.721 | ; | 1.629 | ] | 1.086 | [ | 0.723 | ; | 1.632 | ] | |||
| Season: spring vs. summer | 0.842 | [ | 0.668 | ; | 1.061 | ] | 0.935 | [ | 0.634 | ; | 1.377 | ] | 0.931 | [ | 0.631 | ; | 1.373 | ] | |||
| Individual data | |||||||||||||||||||||
| Maternal age | |||||||||||||||||||||
| <20 vs. 20 to 34 years | 1.001 | [ | 0.476 | ; | 2.527 | ] | 0.135 | 0.967 | [ | 0.422 | ; | 2.216 | ] | 0.123 | |||||||
| >34 vs. 20 to 34 years | 1.494 | [ | 0.888 | ; | 1.993 | ] | 1.507 | [ | 1.017 | ; | 2.234 | ] | |||||||||
| Mother’s occupation b | |||||||||||||||||||||
| Disadvantaged vs. Advantaged | 1.269 | [ | 0.692 | ; | 2.329 | ] | 0.743 | 1.187 | [ | 0.643 | ; | 2.191 | ] | 0.834 | |||||||
| Moderately advantaged vs. Advantaged | 1.206 | [ | 0.680 | ; | 2.138 | ] | 1.185 | [ | 0.668 | ; | 2.101 | ] | |||||||||
| Smoking during pregnancy: yes vs. no | 1.069 | [ | 0.739 | ; | 1.545 | ] | 0.723 | 1.070 | [ | 0.740 | ; | 1.548 | ] | 0.717 | |||||||
| Single-parent family: yes vs. no | 0.809 | [ | 0.424 | ; | 1.545 | ] | 0.521 | 0.770 | [ | 0.402 | ; | 1.474 | ] | 0.430 | |||||||
| History of preterm birth: yes vs. no | 6.055 | [ | 3.778 | ; | 9.704 | ] | <10−3 | 6.014 | [ | 3.750 | ; | 9.644 | ] | <10−3 | |||||||
| Primiparity : yes vs. no | 2.157 | [ | 1.509 | ; | 3.084 | ] | <10−3 | 2.148 | [ | 1.502 | ; | 3.071 | ] | <10−3 | |||||||
| Quality of follow up c : high vs. low | 4.709 | [ | 2.616 | ; | 8.475 | ] | <10−3 | 4.590 | [ | 2.543 | ; | 8.283 | ] | <10−3 | |||||||
| European deprivation index | |||||||||||||||||||||
| Tertile 2 vs. Tertile 1 (least deprived) | 1.006 | [ | 0.703 | ; | 1.438 | ] | 0.232 | ||||||||||||||
| Tertile 3 vs. Tertile 1 (least deprived) | 1.354 | [ | 0.914 | ; | 2.006 | ] | |||||||||||||||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albouy-Llaty, M.; Limousi, F.; Carles, C.; Dupuis, A.; Rabouan, S.; Migeot, V. Association between Exposure to Endocrine Disruptors in Drinking Water and Preterm Birth, Taking Neighborhood Deprivation into Account: A Historic Cohort Study. Int. J. Environ. Res. Public Health 2016, 13, 796. https://doi.org/10.3390/ijerph13080796
Albouy-Llaty M, Limousi F, Carles C, Dupuis A, Rabouan S, Migeot V. Association between Exposure to Endocrine Disruptors in Drinking Water and Preterm Birth, Taking Neighborhood Deprivation into Account: A Historic Cohort Study. International Journal of Environmental Research and Public Health. 2016; 13(8):796. https://doi.org/10.3390/ijerph13080796
Chicago/Turabian StyleAlbouy-Llaty, Marion, Frédérike Limousi, Camille Carles, Antoine Dupuis, Sylvie Rabouan, and Virginie Migeot. 2016. "Association between Exposure to Endocrine Disruptors in Drinking Water and Preterm Birth, Taking Neighborhood Deprivation into Account: A Historic Cohort Study" International Journal of Environmental Research and Public Health 13, no. 8: 796. https://doi.org/10.3390/ijerph13080796
APA StyleAlbouy-Llaty, M., Limousi, F., Carles, C., Dupuis, A., Rabouan, S., & Migeot, V. (2016). Association between Exposure to Endocrine Disruptors in Drinking Water and Preterm Birth, Taking Neighborhood Deprivation into Account: A Historic Cohort Study. International Journal of Environmental Research and Public Health, 13(8), 796. https://doi.org/10.3390/ijerph13080796






