Community-Based Management of Child Malnutrition in Zambia: HIV/AIDS Infection and Other Risk Factors on Child Survival
Abstract
:1. Introduction
2. Materials and Methods
2.1. Setting
2.2. Malnutrition Classification, Management and Integration of CMAM Components
2.3. SFP Activities and Data Collection
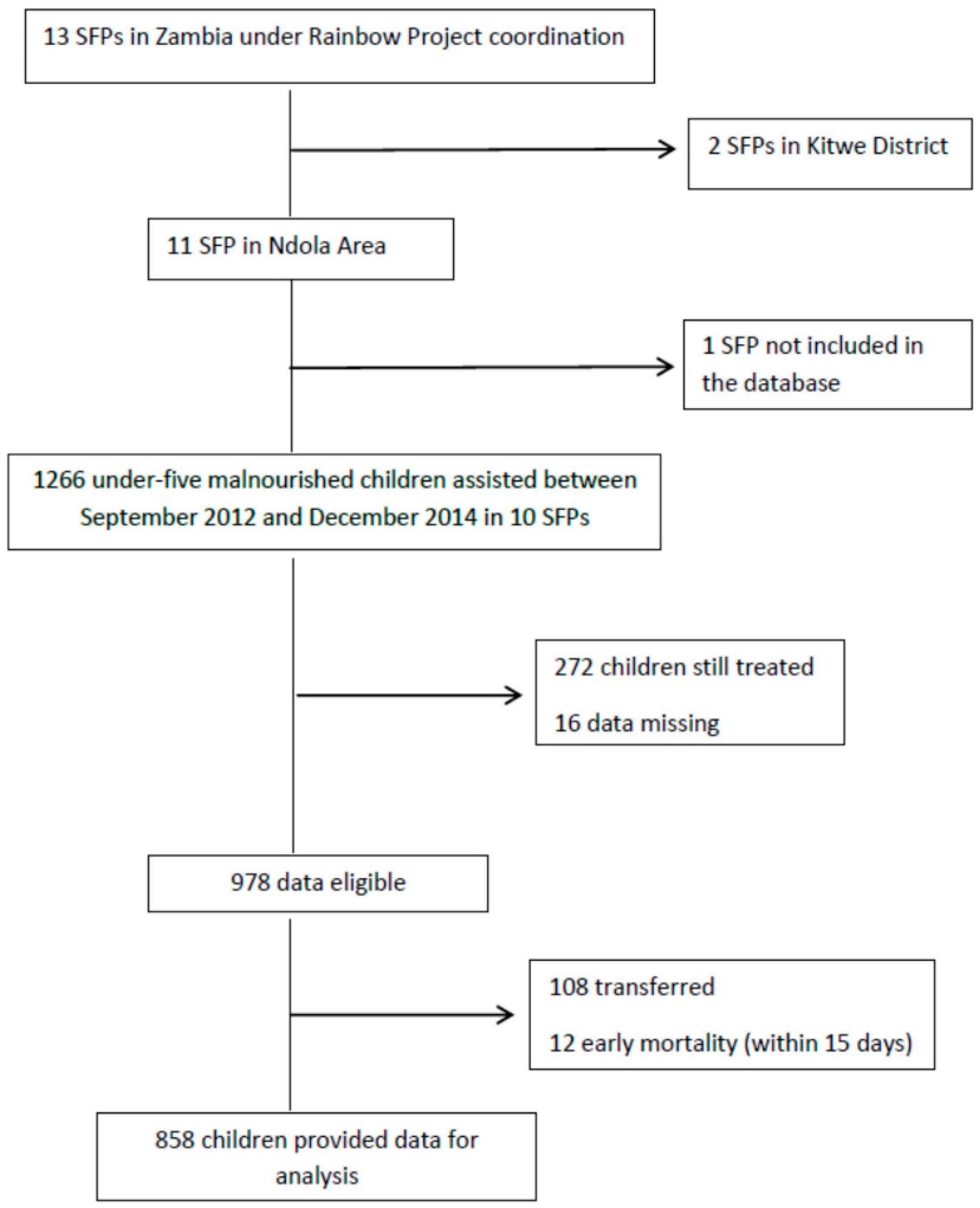
2.4. Study Population, Statistical Analysis, and Ethical Considerations
2.5. Program Outcomes and Performance Indicators
3. Results
3.1. Performance of the Rainbow Project Based on International Standards
3.2. Anthropometric Analysis of Recovered Children
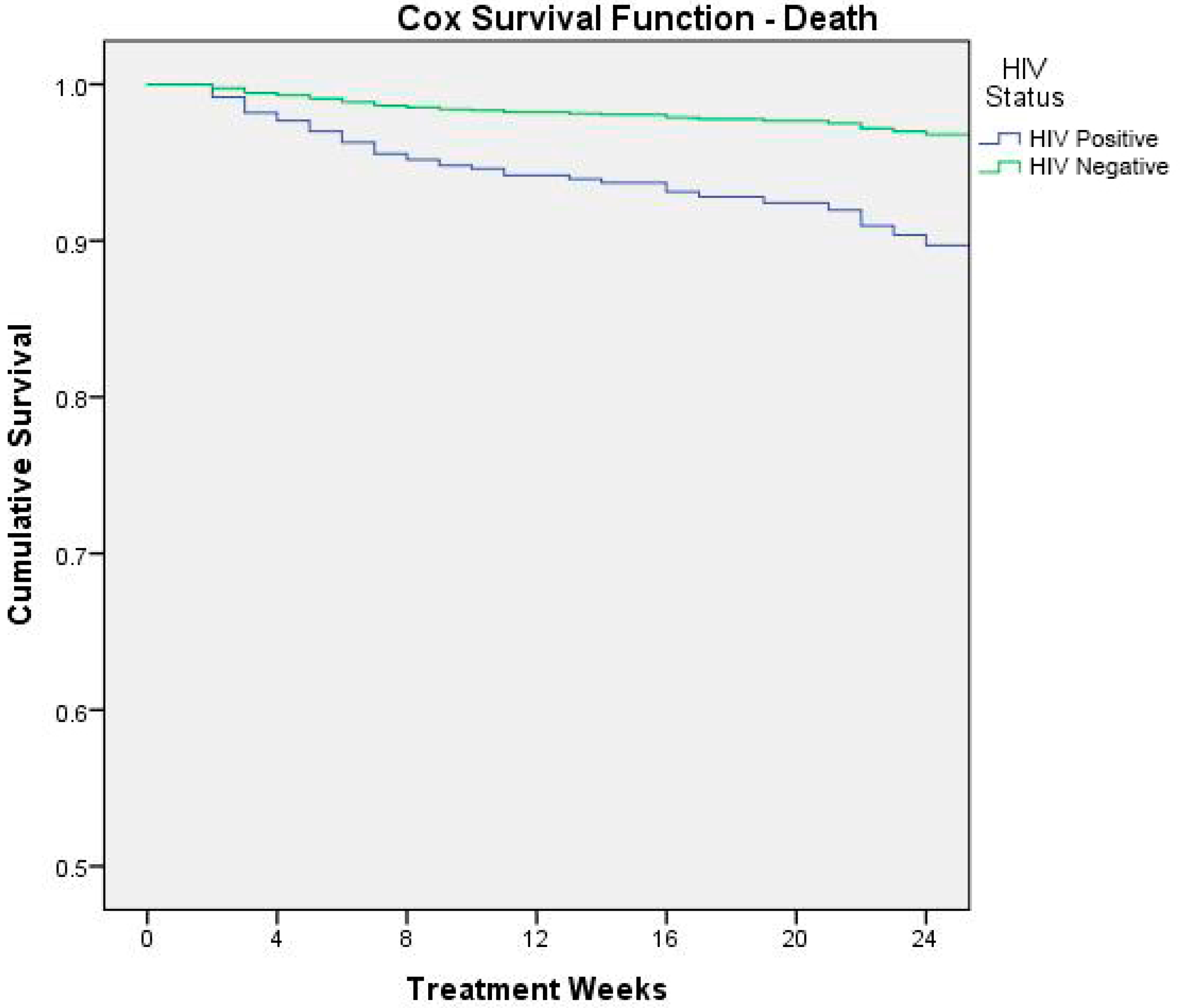
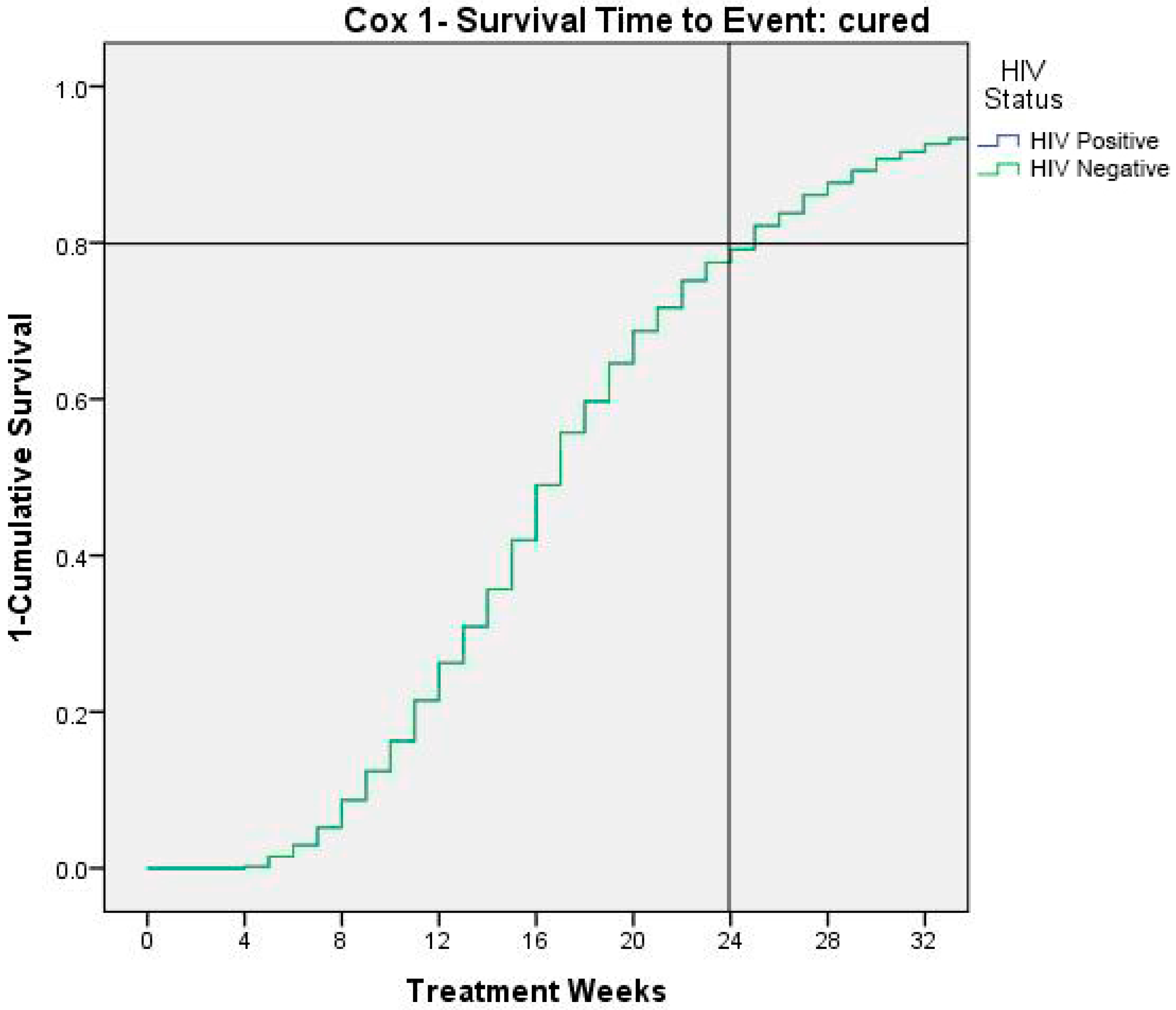
3.3. Predictors of Mortality and Cured
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| SFPs | Supplementary Feeding Programs |
| MAM | Moderate Acute Malnutrition |
| SAM | Severe Acute Malnutrition |
| OR | Odds Ratio |
| CI | Confidence Interval |
| HR | Hazard Ratio |
| SD | Standards Deviation |
| MUAC | Mid-Upper Arm Circumference |
| WAZ | Weight-for-Age Z-score |
| HIV | Human Immunodeficiency Virus |
| AIDS | Acquired Immune Deficiency Syndrome |
| CTC | Community-based Therapeutic Care |
| IC | Inpatient Care |
| WHO | World Health Organization |
| OTP | Outpatient Therapeutic Care |
| CMAM | Community-Management of Acute Malnutrition |
| NGOs | Non-Governmental Organizations |
| CBO | community-based organizations |
| DHMTs | District Health Management Teams |
| IYCF | Infant and Young Child Feeding |
| PMTCT | Prevention Mother-to-Child Transmission |
| UNICEF | United Nations International Children’s Emergency Fund |
| IMAM | Integrated Management of Acute Malnutrition |
| WHZ | Weight-for-Height Z-score |
| WLZ | Weight-for-Length Z-score |
| RUTF | Ready-to-use Therapeutic Food |
| ID number | Identification number |
| HEPS | High energy protein supplement |
| CSB | Corn soya blended food |
| SPSS | Statistical packages for social sciences |
| TDRC | Tropical Diseases Research Centre |
| UNHCR | United Nations High Commissioner for Refugees |
| VCT | Voluntary counseling and testing |
| ARV | Antiretroviral |
| ART | Antiretroviral Therapy |
| TB | Tuberculosis |
References
- Zambia WHO Country Office. Zambia: WHO Statistical Profile. Available online: http://afro.who.int/en/zambia/who-country-office-zambia.html (accessed on 25 February 2016).
- Central Statistical Office (CSO) (Zambia), Ministry of Health (MOH) (Zambia), ICF International. Zambia Demographic and Health Survey 2013-14; Central Statistical Office, Ministry of Health, and ICF International: Rockville, MD, USA, 2014. [Google Scholar]
- Victora, C.G.; Adair, L.; Fall, C.; Hallal, P.C.; Martorell, R.; Richter, L.; Sachdev, H.S.; Maternal and Child Undernutrition Study Group. Maternal and child undernutrition: Consequences for adult health and human capital. Lancet 2008, 371, 340–357. [Google Scholar] [CrossRef]
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; de Onis, M.; Ezzati, M.; Grantham-McGregor, S.; Katz, J.; Martorell, R.; et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef]
- Charles Michelo, R.M. Set criteria might have high potentials in the management of severely malnourished children in Zambia, evidence from an evaluation of an “Outpatient Therapeutic Care Program (OTP)” in selected communities in Lusaka urban, Zambia. Med. J. Zambia 2012, 39, 6–12. [Google Scholar]
- Arpadi, S.M. Growth Failure in HIV-Infected Children. Available online: http://www.who.int/nutrition/topics/Paper%20Number%204%20-%20Growth%20failure.pdf (accessed on 20 June 2016).
- Heikens, G.T.; Bunn, J.; Amadi, B.; Manary, M.; Chhagan, M.; Berkley, J.A.; Rollins, N.; Kelly, P.; Adamczick, C.; Maitland, K.; et al. Case management of HIV-infected severely malnourished children: Challenges in the area of highest prevalence. Lancet 2008, 371, 1305–1307. [Google Scholar] [CrossRef]
- Fergusson, P.; Tomkins, A. HIV prevalence and mortality among children undergoing treatment for severe acute malnutrition in Sub-Saharan Africa: A systematic review and meta-analysis. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Buonomo, E.; Scarcella, P.; Tembo, D.; Giuliano, M.; Moramarco, S.; Palombi, L.; Nielsen-Saines, K.; Mancinelli, S.; Marazzi, M.C. Malnutrition decreases the odds of attaining motor milestones in HIV exposed children: Results from a paediatric DREAM cohort. Epidemiol. Prev. 2015, 39, 108–112. [Google Scholar] [PubMed]
- Ministry of Health Republic of Zambia. Sixth National Development Plan 2011–2015. Available online: http://siteresources.worldbank.org/INTZAMBIA/Resources/SNDP_Final_Draft__20_01_2011.pdf (accessed on 25 February 2016).
- Bachmann, M.O. Cost effectiveness of community-based therapeutic care for children with severe acute malnutrition in Zambia: Decision tree model. Cost Eff. Resour. Alloc. 2009, 15. [Google Scholar] [CrossRef] [PubMed]
- WHO, UNICEF, WF, UN System Standing Committee on Nutrition. Community-Based Management of Severe Acute Malnutrition. A Joint Statement by the World Health Organization, the World Food Programme, the United Nations System Standing Committee on Nutrition and the United Nations Children’s Fund. Available online: http://www.who.int/nutrition/topics/Statement_community_based_man_sev_acute_mal_eng.pdf (accessed on 25 February 2016).
- World Health Organization. Management of Severe Malnutrition: A Manual for Physicians and Other Senior Health Workers. Geneva 1999. Available online: http://whqlibdoc.who.int/hq/1999/a57361.pdf (accessed on 25 February 2016).
- Valid International. Community-Based Therapeutic Care (CTC): A Field Manual. Available online: http://www.concernusa.org/content/uploads/2015/08/ctc_manual_v1_oct06.pdf (accessed on 25 February 2016).
- Kennedy, E.; Branca, F.; Webb, P.; Bhutta, Z.; Brown, R. Setting the scene: An overview of issues related to policies and programs for moderate and severe acute malnutrition. Food Nutr. Bull. 2015, 36, S9–S14. [Google Scholar] [CrossRef] [PubMed]
- Moramarco, S.; Amerio, G.; Gozza Maradini, G.; Garuti, E. Rainbow project: A model to fight child malnutrition in Zambia. In Turning Rapid Growth into Meaningful Growth: Sustaining the Commitment to Nutrition in Zambia; Harris, J., Haddad, L., Grütz, S.S., Eds.; Publisher: IDS, Brighton, UK, 2014; pp. 57–61. [Google Scholar]
- World Health Organization, United Nations Children’s Fund. Child Growth Standard and Identification of Severe Acute Malnutrition in Infant and Children. A Joint Statement; WHO: Geneva, Switzerland, 2009; Available online: http://apps.who.int/iris/bitstream/10665/44129/1/9789241598163_eng.pdf?ua=1 (accessed on 25 February 2016).
- Zambian Ministry of Health. DRAFT Integrated Management of Acute Malnutrition Zambia Guidelines; Zambian Ministry of Health: Lusaka, Zambia, 2012. [Google Scholar]
- WHO. Guideline: Updates on the Management of Severe Acute Malnutrition in Infants and Children. 2013. Available online: http://apps.who.int/iris/bitstream/10665/95584/1/9789241506328_eng.pdf?ua=1 (accessed on 25 February 2016).
- World Health Organization. WHO Child Growth Standards: Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-age: Methods and Development. Geneva 2006. Available online: http://www.who.int/childgrowth/standards/Technical_report.pdf (accessed on 19 October 2015).
- The SPHERE Project Geneva. The Sphere Handbook: Humanitarian Charter and Minimum Standards in Disaster Response, 3rd ed.; The SPHERE Project Geneva: Geneva, Switzerland, 2011. [Google Scholar]
- UNHCR. Guidelines for Selective Feeding: The Management of Malnutrition in Emergencies. January 2011. Available online: http://www.unhcr.org/4b7421fd20.pdf (accessed on 25 February 2016).
- Management of Moderate Acute Malnutrition. Available online: http://www.unscn.org/en/gnc_htp/modul.php?modID=17 (accessed on 20 June 2016).
- Golden, M.H. Proposed recommended nutrient densities for moderately malnourished children. Food Nutr. Bull. 2009, 30, S267–S343. [Google Scholar] [CrossRef] [PubMed]
- Somassé, Y.E.; Bahwere, P.; Laokri, S.; Elmoussaoui, N.; Donnen, P. Sustainability and scaling-up analysis of community- based management of acute malnutrition: Lessons learned from Burkina Faso. Food Nutr. Bull. 2013, 34, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, A.; Ferguson, E. Dietary counseling in the management of moderate malnourishment in children. Food Nutr. Bull. 2009, 30, S405–S433. [Google Scholar] [CrossRef] [PubMed]
- Ackatia-Armah, R.S.; McDonald, C.M.; Doumbia, S.; Erhardt, J.G.; Hamer, D.H.; Brown, K.H. Malian children with moderate acute malnutrition who are treated with lipid- based dietary supplements have greater weight gains and recovery rates than those treated with locally produced cereal-legume products: A community-based, cluster-randomized trial. Am. J. Clin. Nutr. 2015, 101, 632–645. [Google Scholar] [CrossRef] [PubMed]
- Karakochuk, C.; Van den Briel, T.; Stephens, D.; Zlotkin, S. Treatment of moderate acute malnutrition with ready-to-use supplementary food results in higher overall recovery rates compared with a corn-soya blend in children in southern Ethiopia: An operations research trial. Am. J. Clin. Nutr. 2012, 96, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Munthali, T.; Jacobs, C.; Sitali, L.; Dambe, R.; Michelo, C. Mortality and morbidity patterns in under-five children with severe acute malnutrition (SAM) in Zambia: A five-year retrospective review of hospital-based records (2009–2013). Arch. Public Health 2015, 73. [Google Scholar] [CrossRef] [PubMed]
- UNAIDS. Zambia Country Report. 2014. Available online: http://www.unaids.org/en/regionscountries/countries/zambia (accessed on 31 May 2016).
- Buonomo, E.; De Luca, S.; Tembo, D.; Scarcella, P.; Germano, P.; Doro Altan, A.M.; Palombi, L.; Liotta, G.; Nielsen-Saines, K.; Erba, F.; et al. Nutritional Rehabilitation of HIV-Exposed Infants in Malawi: Results from the Drug Resources Enhancement against AIDS and Malnutrition Program. Int. J. Environ. Res. Public Health 2012, 9, 421–434. [Google Scholar] [CrossRef] [PubMed]
- Government of the Republic of Zambia Ministry of Health. Lifelong Antiretroviral Drugs (ARV’s) for All HIV Positive Pregnant Women in Zambia—Policy Guidelines for Health Facilities in Zambia, 2013. Available online: http://www.emtct-iatt.org/wp-content/uploads/2013/02/Policy-guidelines-for-eMTCT-Option-B+_Zambia-2013-signed2.pdf (accessed on 25 February 2016).
| Variables (n = 858) | Value |
|---|---|
| Male, n. (%) | 428 (49.9) |
| Age in months, mean (SD) | 19 ± 9.4 |
| <18 months of age | 455 (53) |
| >18 months of age | 403 (47) |
| Urban area, n. (%) | 695 (81) |
| Rural area, n. (%) | 163 (19) |
| Orphans, n. (%) | 48 (5.6) |
| Orphans of both parents, n. (%) | 15 (1.7) |
| Disability, n. (%) * | 8 (6) |
| Twins, n. (%) | 62 (7.2) |
| Caregiver’s age, mean (SD) | 29 ± 10 |
| (min–max) | (13–78) |
| Referred from, n. (%) | |
| Hospital | 46 (5.4) |
| Local health facility | 279 (32.5) |
| Community | 533 (62.1) |
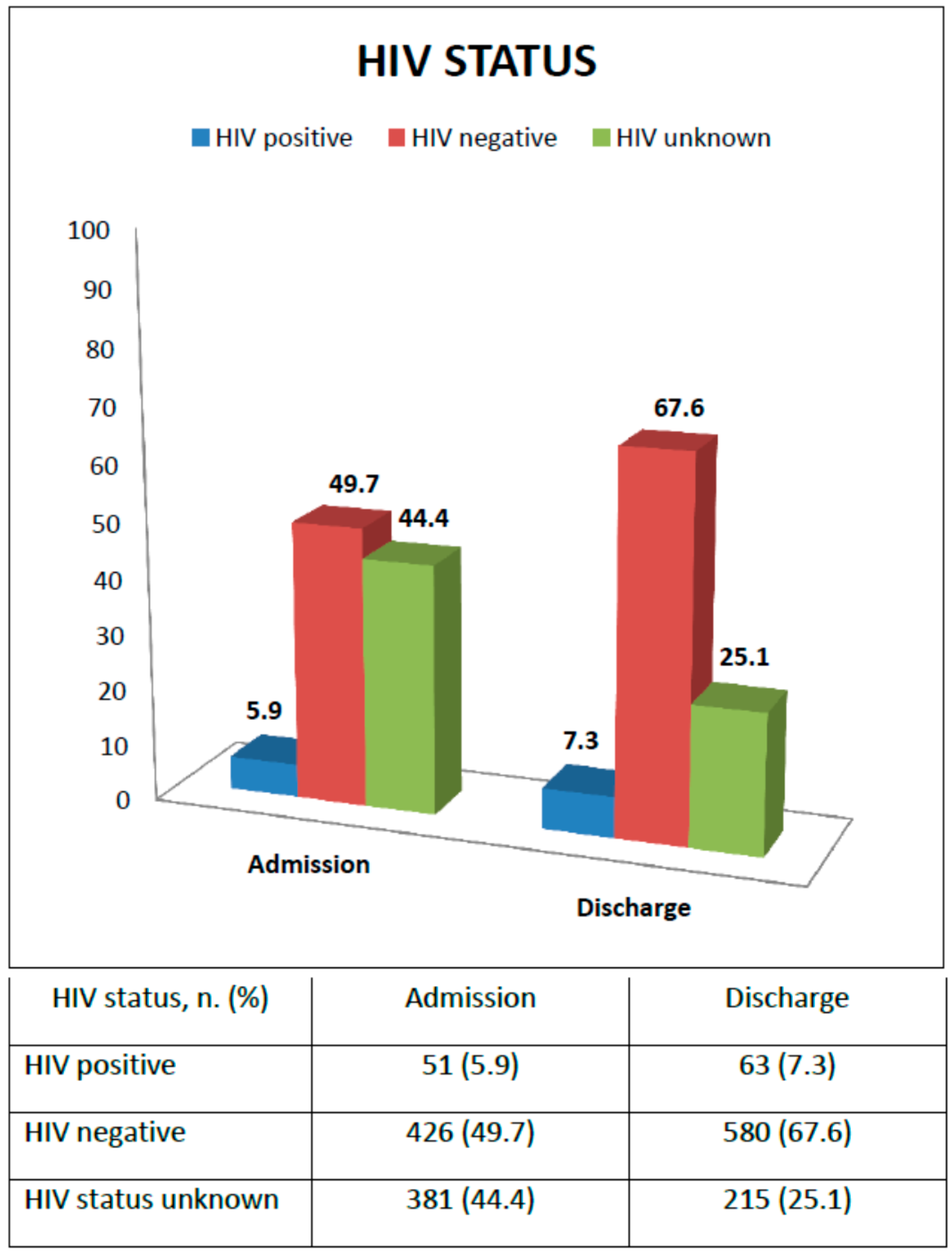
| HIV status, n. (%) | |
| Infected | 51 (5.9) |
| Uninfected | 426 (49.7) |
| Status unknown | 381 (44.4) |
| Admission criteria: MAM, n. (%) | 323 (37.6) |
| <18 months of age | 195 (60.4) |
| >18 months of age | 128 (39.6) |
| Admission criteria: SAM, n. (%) | 241 (28.1) |
| <18 months of age | 146 (60.6) |
| >18 months of age | 95 (39.4) |
| Admission criteria: Underweight, n. (%) | 294 (34.3) |
| <18 months of age | 114 (38.8) |
| >18 months of age | 180 (61.2) |
| Presence of edema, n. (%) | 77 (9) |
| Relapses of malnutrition event, n. (%) | 68 (7.9) |
| Weight (kg), mean (SD) | 7.8 ± 1.6 |
| WAZ, mean (SD) | −2.8 ± 1.1 |
| MUAC (cm), mean (SD) | 12.4 ± 1 |
| Health problems, n. (%) | 299 (34.9) |
| Fever | 73 (8.5) |
| Diarrhea | 71 (8.3) |
| Lack of appetite | 66 (7.7) |
| Cough | 28 (3.3) |
| Malaria | 7 (0.8) |
| Others | 54 (6.3) |
| Indicators | Rainbow Project SFPs Outcomes | International Standards | |||
|---|---|---|---|---|---|
| Total (n = 858) | MAM (n = 323) | SAM (n = 241) | Acceptable | Alarming | |
| Recovered (%) | 709 (82.6) | 278 (86.1) | 177 (73.5) | >75% * | <50% * |
| Defaulters (%) | 101 (11.8) | 36 (11.1) | 34 (14.1) | <15% * | >30% * |
| Deaths (%) | 48 (5.6) | 9 (2.8) | 30 (12.4) | <3% for SFPs * <10% for TFPs * | >10% * |
| Mean length of stay, weeks (SD) | 19.3 ± 11.5 | 19.3 ± 11.9 | 22 ± 11.8 | <12 weeks for SFPs (depending on the national guidelines) § <3–4 weeks for TFPs (IC till full recovery) § <60 days for TFPs (IC and OTP combined) § | |
| Average weight gain, g/kg/day (SD) | 1.7 ± 1.2 | 1.7 ± 1 | 2 ± 1.3 | ≥3 g/kg/day for SFPs § ≥8 g/kg/day for TFPs (IC till full recovery) § ≥4 g/kg/day for TFPs (IC and OTP combined) § | |
| Admission | Discharge | Differences between Means | 95% Confidence Intervals (CI) | Student t-Test | p-Value | ||
|---|---|---|---|---|---|---|---|
| Lower | Upper | ||||||
| Mean weight (kg), (SD) | 7.8 ± 1.6 | 9.2 ± 1.6 | −1.4 ± 0.8 | 1.37822 | 1.50302 | 45.327 | 0.000 |
| Mean WAZ, (SD) | −2.8 ± 1.1 | −1.9 ± 0.9 | −0.8 ± 0.8 | 0.714137 | 0.832647 | 25.625 | 0.000 |
| Mean MUAC (cm), (SD) | 12.4 ± 1 | 13.7 ± 0.8 | −1.3 ± 0.9 | 1.2356 | 1.3689 | 38.351 | 0.000 |
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| Predictors of Mortality | HR | 95% CI | p | HR Exp (B) | 95% CI | p |
| SAM | 3.8 | 2.1–6.8 | <0.001 | 4.3 | 2.3–7.9 | <0.001 |
| HIV infection | 3.1 | 1.7–5.5 | <0.001 | 3.3 | 1.8–5.9 | <0.001 |
| Low weight gain | 2.1 | 0.9–5.1 | NS (0.08) | 3.1 | 1.3–7.5 | 0.009 |
| WAZ < −3 at admission | 3.1 | 1.6–5.7 | <0.001 | NS | ||
| Age < 18 months | 1.0 | 0.5–1.7 | NS (0.9) | NS | ||
| Frequency of health problems | 0.9 | 0.5–1.7 | NS (0.9) | NS | ||
| Health problems at admission | 1.7 | 0.9–3.1 | NS (0.07) | NS | ||
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| Predictors of Failure | HR | 95% CI | p | HR Exp (B) | 95% CI | p |
| SAM | 1.7 | 1.1−2.4 | 0.001 | 1.8 | 1.2−2.5 | 0.001 |
| HIV infection | 2.2 | 1.6−3.1 | <0.001 | 2.2 | 1.6−3.1 | <0.001 |
| Low weight gain | 1.6 | 1.0−2.5 | 0.041 | 1.9 | 1.2−3.0 | 0.006 |
| WAZ < −3 at admission | 0.9 | 0.6−1.4 | NS (0.6) | NS | ||
| Age < 18 months | 0.8 | 0.5−1.0 | NS (0.1) | NS | ||
| Frequency of health problems | 0.8 | 0.5−1.1 | NS (0.1) | NS | ||
| Health problems at admission | 1.1 | 0.7−1.6 | NS (0.06) | NS | ||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moramarco, S.; Amerio, G.; Ciarlantini, C.; Chipoma, J.K.; Simpungwe, M.K.; Nielsen-Saines, K.; Palombi, L.; Buonomo, E. Community-Based Management of Child Malnutrition in Zambia: HIV/AIDS Infection and Other Risk Factors on Child Survival. Int. J. Environ. Res. Public Health 2016, 13, 666. https://doi.org/10.3390/ijerph13070666
Moramarco S, Amerio G, Ciarlantini C, Chipoma JK, Simpungwe MK, Nielsen-Saines K, Palombi L, Buonomo E. Community-Based Management of Child Malnutrition in Zambia: HIV/AIDS Infection and Other Risk Factors on Child Survival. International Journal of Environmental Research and Public Health. 2016; 13(7):666. https://doi.org/10.3390/ijerph13070666
Chicago/Turabian StyleMoramarco, Stefania, Giulia Amerio, Clarice Ciarlantini, Jean Kasengele Chipoma, Matilda Kakungu Simpungwe, Karin Nielsen-Saines, Leonardo Palombi, and Ersilia Buonomo. 2016. "Community-Based Management of Child Malnutrition in Zambia: HIV/AIDS Infection and Other Risk Factors on Child Survival" International Journal of Environmental Research and Public Health 13, no. 7: 666. https://doi.org/10.3390/ijerph13070666
APA StyleMoramarco, S., Amerio, G., Ciarlantini, C., Chipoma, J. K., Simpungwe, M. K., Nielsen-Saines, K., Palombi, L., & Buonomo, E. (2016). Community-Based Management of Child Malnutrition in Zambia: HIV/AIDS Infection and Other Risk Factors on Child Survival. International Journal of Environmental Research and Public Health, 13(7), 666. https://doi.org/10.3390/ijerph13070666







