Pyrethroid Pesticide Metabolite in Urine and Microelements in Hair of Children Affected by Autism Spectrum Disorders: A Preliminary Investigation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Subjects
2.3. First Group of Children: Assessment of 3-PBA in Urine
Urine Collection and Analysis of 3-PBA
2.4. Second Group of Children: Assessment of Metals and Microelements in Hair
2.4.1. Hair Samples
2.4.2. Analytical Methods
2.5. Statistical Analysis
Principal Components Analysis and Self-Organizing Maps
3. Results
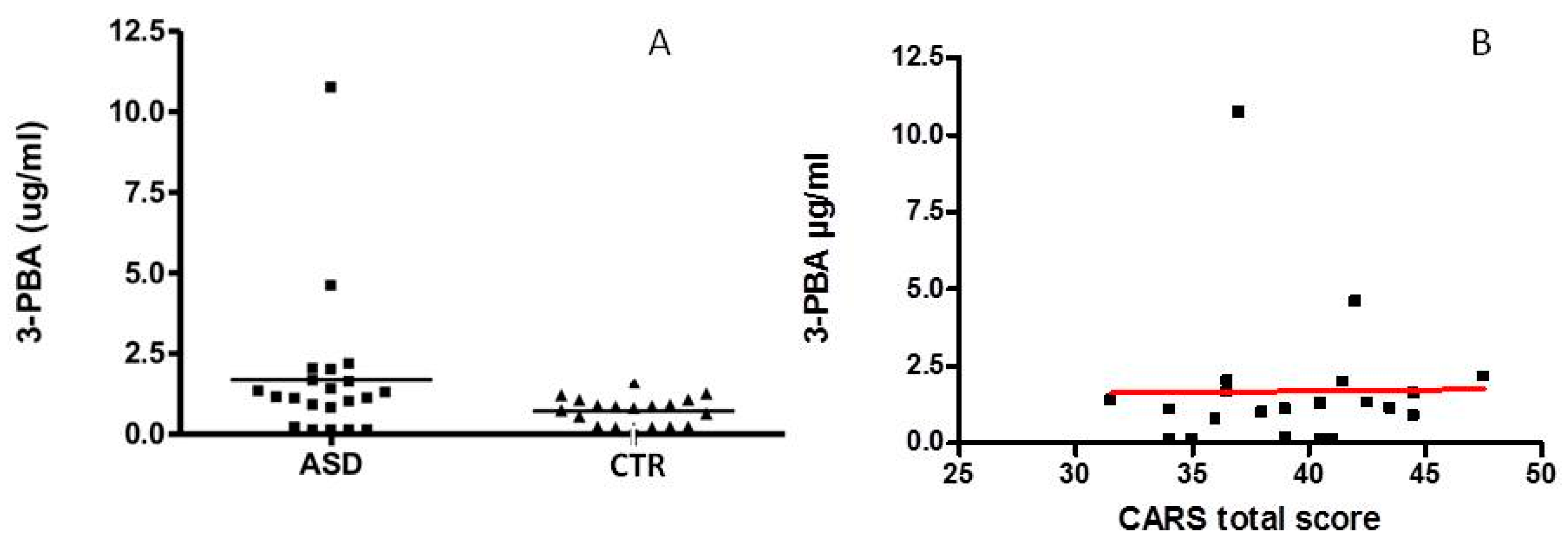
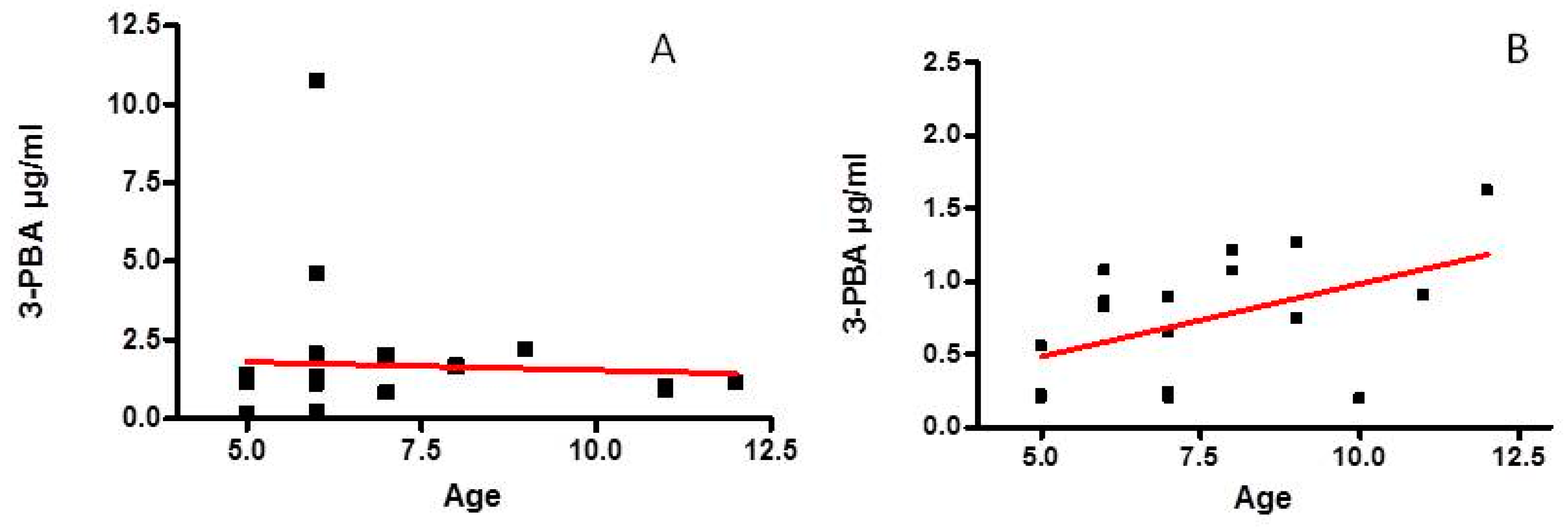
3.1. 3-PBA Level in Urine of ASD and Control Children (CTR) in the First Group
3.2. Analysis of Metals and Microelements in Hair Samples in the Second Group
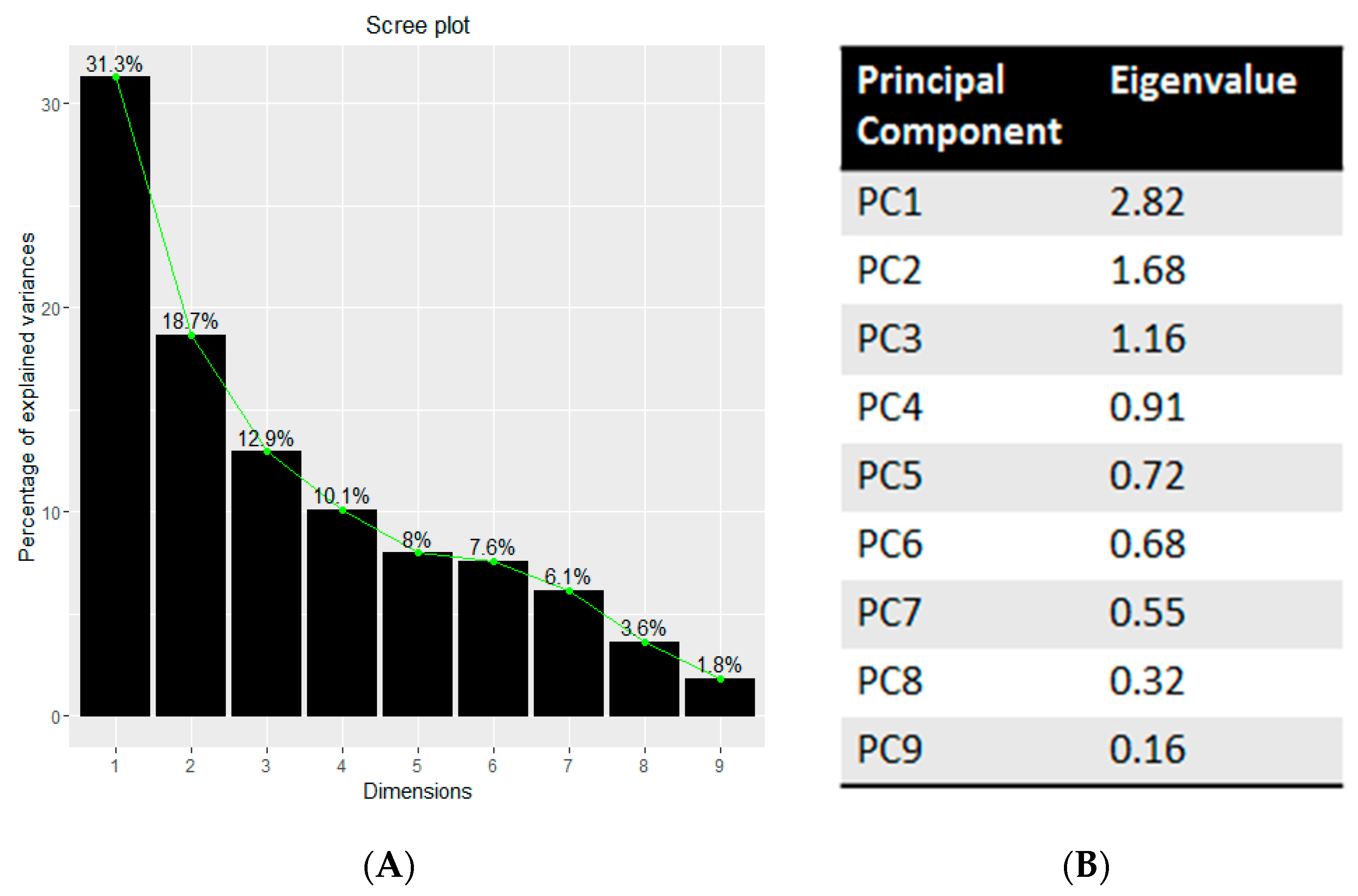
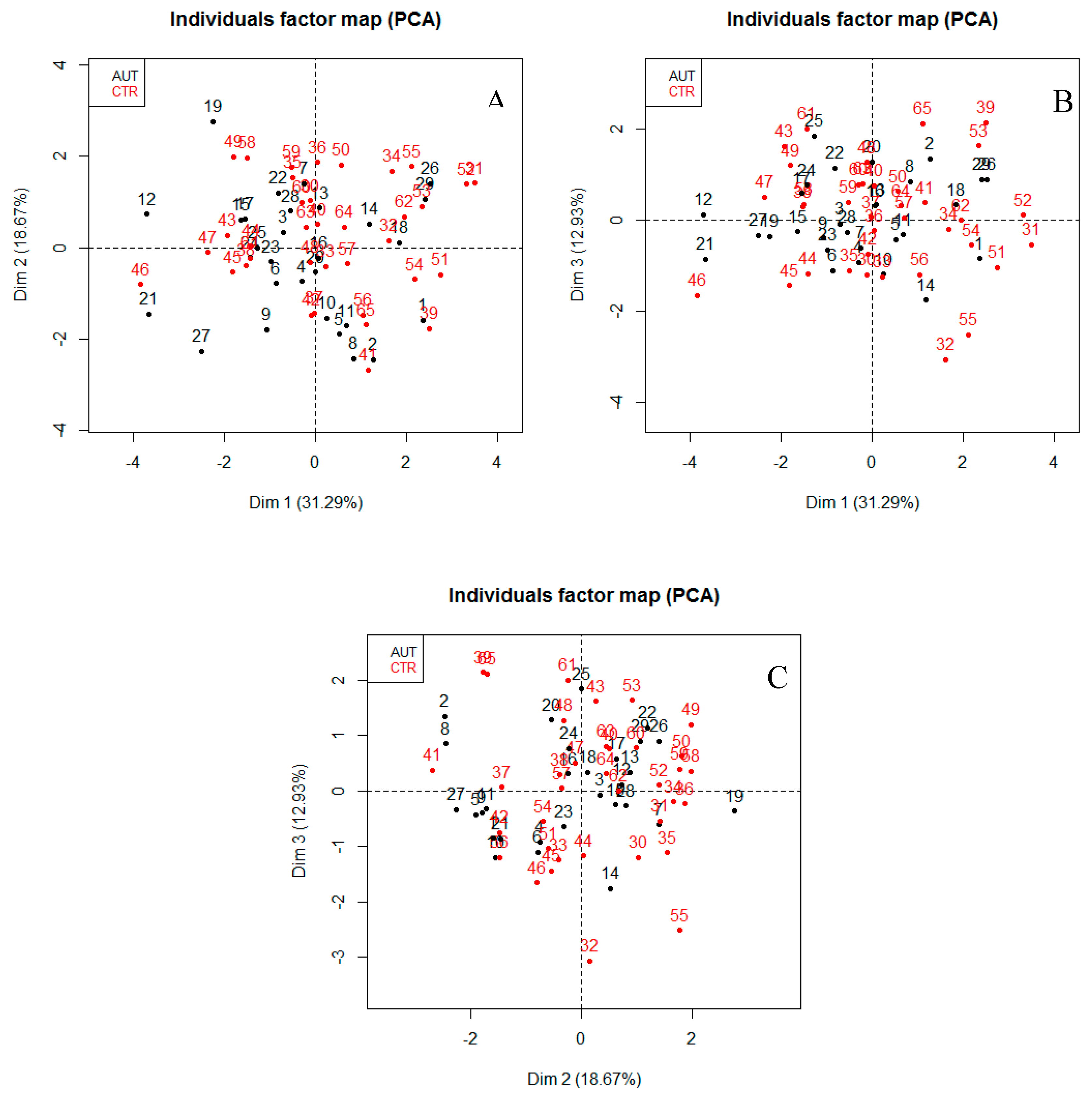
PCA Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ASD | Autism Spectrum Disorder |
| GC-MD | Gas chromatography-coupled trap mass detector |
| ICP-OES | Optical Emission Spectrometry |
| 3-PBA | 3-phenoxybenzoic acid |
| CTR | Control |
| PCA | Principal component analysis |
References
- Ambeskovic, M.; Fuchs, E.; Beaumier, P.; Gerken, M.; Metz, G.A. Hair trace elementary profiles in aging rodents and primates: Links to altered cell homeodynamics and disease. Biogerontology 2013, 14, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Bader, M.; Dietz, M.C.; Ihrig, A.; Triebig, G. Biomonitoring of manganese in blood, urine and axillary hair following low-dose exposure during the manufacture of dry cell batteries. Int. Arch. Occup. Environ. Health 1999, 72, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Nasuti, C.; Ferraro, S.; Giovannetti, R.; Piangerelli, M.; Gabbianelli, R. Metal and Microelement Biomarkers of Neurodegeneration in Early Life Permethrin-Treated Rats. Toxics 2016, 4. [Google Scholar] [CrossRef]
- Geier, D.A.; Kern, J.K.; King, P.G.; Sykes, L.K.; Geier, M.R. Hair toxic metal concentrations and autism spectrum disorder severity in young children. Int. J. Environ. Res. Public Health 2012, 9, 4486–4497. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, H.; Tsutsui, T. Assessment of infantile mineral imbalances in autism spectrum disorders (ASDs). Int. J. Environ. Res. Public Health 2013, 10, 6027–6043. [Google Scholar] [CrossRef] [PubMed]
- Vela, G.; Stark, P.; Socha, M.; Sauer, A.K.; Hagmeyer, S.; Grabrucker, A.M. Zinc in gut-brain interaction in autism and neurological disorders. Neural Plast. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Blaurock-Busch, E.; Amin, O.R.; Dessoki, H.H.; Rabah, T. Toxic Metals and Essential Elements in Hair and Severity of Symptoms among Children with Autism. Maedica 2012, 7, 38–48. [Google Scholar] [PubMed]
- Lubkowska, A.; Sobieraj, W. Hair concentrations of magnesium, calcium, iron, selenium, zinc and copper in autistic children. Trace Elem. Electrolytes 2009, 26, 72–77. [Google Scholar] [CrossRef]
- Adams, J.B.; Holloway, C.E.; George, F.; Quig, D. Analyses of toxic metals and essential minerals in the hair of Arizona children with autism and associated conditions, and their mothers. Biol. Trace Elem. Res. 2006, 110, 193–209. [Google Scholar] [CrossRef]
- Fido, A.; Al-Saad, S. Toxic trace elements in the hair of children with autism. Autism 2005, 9, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Lakshmi Priya, M.D.; Geetha, A. Level of trace elements (copper, zinc, magnesium and selenium) and toxic elements (lead and mercury) in the hair and nail of children with autism. Biol. Trace Elem. Res. 2011, 142, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Kern, J.K.; Grannemann, B.D.; Trivedi, M.H.; Adams, J.B. Sulfhydryl-reactive metals in autism. J. Toxicol. Environ. Health A 2007, 70, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, F.E.B.; Zaky, E.A.; El-Sayed, A.B.; Elhossieny, R.M.; Zahra, S.S.; Salah Eldin, W. Assessment of Hair Aluminum, Lead, and Mercury in a Sample of Autistic Egyptian Children: Environmental Risk Factors of Heavy Metals in Autism. Behav. Neurol. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Eriksson, P.; Fredriksson, A.; Buratovic, S.; Viberg, H. Developmental neurotoxic effects of two pesticides: Behavior and neuroprotein studies on endosulfan and cypermethrin. Toxicology 2015, 335, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Holzman, D.C. Pesticides and autism spectrum disorders: New findings from the CHARGE study. Environ. Health Perspect. 2014, 122. [Google Scholar] [CrossRef] [PubMed]
- Keil, A.P.; Daniels, J.L.; Hertz-Picciotto, I. Autism spectrum disorder, flea and tick medication, and adjustments for exposure misclassification: The CHARGE (Childhood Autism Risks from Genetics and Environment) case-control study. Environ. Health 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Caudle, W.M. Vulnerability of synapses in the frontal cortex of mice developmentally exposed to an insecticide: Potential contribution to neuropsychiatric disease. Neurotransmitter 2015, 2. [Google Scholar] [CrossRef]
- Shelton, J.F.; Geraghty, E.M.; Tancredi, D.J.; Delwiche, L.D.; Schmidt, R.J.; Ritz, B.; Hansen, R.L.; Hertz-Picciotto, I. Neurodevelopmental disorders and prenatal residential proximity to agricultural pesticides: The CHARGE study. Environ. Health Perspect. 2014, 122. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations Statistics Division. Available online: http://faostat3.fao.org (accessed on 28 March 2016).
- Zartarian, V.; Xue, J.; Glen, G.; Smith, L.; Tulve, N.; Tornero-Velez, R. Quantifying children’s aggregate (dietary and residential) exposure and dose to permethrin: Application and evaluation of EPA’s probabilistic SHEDS-Multimedia model. J. Expo. Sci. Environ. Epidemiol. 2012, 22, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Barr, D.B.; Olsson, A.O.; Wong, L.Y.; Udunka, S.; Baker, S.E.; Whitehead, R.D.; Magsumbol, M.S.; Williams, B.L.; Needham, L.L. Urinary concentrations of metabolites of pyrethroid insecticides in the general U.S. population: National Health and Nutrition Examination Survey 1999–2002. Environ. Health Perspect. 2010, 118, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Bradberry, S.M.; Cage, S.A.; Proudfoot, A.T.; Vale, J.A. Poisoning due to pyrethroids. Toxicol. Rev. 2005, 24, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, D.A.; Genuis, S.J.; Frye, R.E. Environmental toxicants and autism spectrum disorders: A systematic review. Transl. Psychiatry 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 2000. [Google Scholar]
- Lord, C.; Rutter, M.; DiLavore, P.C.; Risi, S. Autism Diagnostic Observation Schedule—Generic; Western Psychological Service: Los Angeles, CA, USA, 1999. [Google Scholar]
- Schopler, E.; Reichler, R.J.; Renner, B.R. The Childhood Autism Rating Scale (CARS); Western Psychological Services: Los Angeles, CA, USA, 1988. [Google Scholar]
- Schopler, E.; Lansing, M.D.; Reichler, R.J.; Marcus, L.M. Psychoeducational Profile, 3rd ed.; (PEP-3); Western Psychological Services: Torrance, CA, USA, 2005. [Google Scholar]
- Roid, G.M.; Miller, L.J. Leiter International Performance Scale-Revised: Examiners Manual; Stoelting Co.: Wood Dale, IL, USA, 1997. [Google Scholar]
- Kohonen, T. Self-organized formation of topologically correct feature maps. Biol. Cybern. 1982, 43, 59–69. [Google Scholar] [CrossRef]
- Kohonen, T. Self-Organizing Maps, 3rd ed.; Springer: New York, NJ, USA, 2001. [Google Scholar]
- Kohonen, T. Self-Organization and Associative Memory; Springer Verlag: Berlin, Germany, 1988. [Google Scholar]
- Ballabio, D.; Consonni, V.; Todeschini, R. The Kohonen and CP-ANN toolbox: A collection of MATLAB modules for Self Organizing Maps and Counterpropagation. Chemometr. Intell. Lab. Syst. 2009, 98, 115–122. [Google Scholar] [CrossRef]
- Ballabio, D.; Vasighi, M.A. MATLAB Toolbox for Self Organizing Maps and supervised neural network learning strategies. Chemometr. Intell. Lab. Syst. 2012, 118, 24–32. [Google Scholar] [CrossRef]
- Milano Chemometrics and QSAR Research Group. Available online: http://michem.disat.unimib.it/chm/download/kohoneninfo.htm (accessed on 28 March 2016).
- Shelton, J.F.; Hertz-Picciotto, I.; Pessah, I.N. Tipping the balance of autism risk: Potential mechanisms linking pesticides and autism. Environ. Health Perspect. 2012, 120, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Roberts, E.M.; English, P.B.; Grether, J.K.; Windham, G.C.; Somberg, L.; Wolff, C. Maternal Residence Near Agricultural Pesticide Applications and Autism Spectrum Disorders among Children in the California Central Valley. Environ. Health Perspect. 2007, 115, 1482–1489. [Google Scholar] [CrossRef] [PubMed]
- Nasuti, C.; Gabbianelli, R.; Falcioni, M.L.; Di Stefano, A.; Sozio, P.; Cantalamessa, F. Dopaminergic system modulation, behavioral changes, and oxidative stress after neonatal administration of pyrethroids. Toxicology 2007, 229, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Nasuti, C.; Carloni, M.; Fedeli, D.; Gabbianelli, R.; Di Stefano, A.; Serafina, C.L.; Silva, I.; Domingues, V.; Ciccocioppo, R. Effects of early life permethrin exposure on spatial working memory and on monoamine levels in different brain areas of pre-senescent rats. Toxicology 2013, 7, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Bordoni, L.; Nasuti, C.; Mirto, M.; Caradonna, F.; Gabbianelli, R. Intergenerational Effect of Early Life Exposure to Permethrin: Changes in Global DNA Methylation and in Nurr1 Gene Expression. Toxics 2015, 3, 451–461. [Google Scholar] [CrossRef]
- Oates, L.; Cohen, M. Assessing Diet as a Modifiable Risk Factor for Pesticide Exposure. Int. J. Environ. Res. Public Health 2011, 8, 1792–1804. [Google Scholar] [CrossRef] [PubMed]
| Metals and Macro/Micro-Elements | ASD Mean ± SEM µg/g (ppm) | CTR Mean ± SEM µg/g (ppm) |
|---|---|---|
| Calcium (Ca) | 409 ± 41.6 ° | 537 ± 71.0 |
| Magnesium (Mg) | 55.3 ± 11.0 * | 97.2 ± 18.4 |
| Ca/Mg | 9.93 ± 1.02 # | 7.86 ± 0.76 |
| Sodium (Na) | n.d. | n.d. |
| Potassium (K) | n.d. | n.d. |
| Copper (Cu) | 16.9 ± 3.01 | 25.3 ± 9.85 |
| Zinc (Zn) | 103 ± 9.31 § | 126 ± 9.57 |
| Iron (Fe) | 11.6 ± 1.16 | 15.2 ± 2.12 |
| Phosphorus (P) | 109 ± 6.71 | 121 ± 5.53 |
| Selenium (Se) | 1.41 ± 0.13 | 1.71 ± 0.21 |
| Manganese (Mn) | 0.19 ± 0.03 | 0.25 ± 0.03 |
| Chromium (Cr) | n.d. | n.d. |
| Cobalt (Co) | n.d. | n.d. |
| Molybdenum (Mo) | 0.28 ± 0.03 | 0.29 ± 0.01 |
| Germanium (Ge) | n.d. | n.d. |
| Sulfur (S) | 28614 ± 119 $ | 31098 ± 783 |
| Vanadium (V) | 0.05 ± 0.01 $ | 0.11 ± 0.03 |
| Barium (Ba) | n.d. | n.d. |
| Lithium (Li) | 0.03 ± 0.01 | 0.05 ± 0.02 |
| Strontium (Sr) | n.d. | n.d. |
| Tin (Sn) | n.d. | n.d. |
| Titanium (Ti) | n.d. | n.d. |
| Zirconium (Zr) | n.d. | n.d. |
| Toxic Metals | ASD Mean ± SEM µg/g (ppm) | CTR Mean ± SEM µg/g (ppm) |
|---|---|---|
| Mercury (Hg) | 0.55 ± 0.13 | 0.74 ± 0.27 |
| Cadmium (Cd) | 0.02 ± 0.01 | 0.04 ± 0.01 |
| Lead (Pb) | 1.09 ± 0.17 | 1.05 ± 0.14 |
| Beryllium (Be) | n.d. | n.d. |
| Aluminum (Al) | 9.33 ± 1.33 | 10.4 ± 1.42 |
| Arsenic (As) | 0.62 ± 0.16 | 0.79 ± 0.21 |
| Uranium (U) | 0.51 ± 0.09 | 0.55 ± 0.09 |
| Palladium (Pd) | n.d. | n.d. |
| Rhodium (Rh) | n.d. | n.d. |
| Gadolinium (Gd) | n.d. | n.d. |
| Antimony (Sb) | 0.11 ± 0.01 | 0.13 ± 0.02 |
| Gold (Au) | n.d. | n.d. |
| Nickel (Ni) | 0.29 ± 0.05 | 0.27 ± 0.04 |
| Platinum (Pt) | n.d. | n.d. |
| Silver (Ag) | n.d. | n.d. |
| Tungsten (W) | n.d. | n.d. |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domingues, V.F.; Nasuti, C.; Piangerelli, M.; Correia-Sá, L.; Ghezzo, A.; Marini, M.; Abruzzo, P.M.; Visconti, P.; Giustozzi, M.; Rossi, G.; et al. Pyrethroid Pesticide Metabolite in Urine and Microelements in Hair of Children Affected by Autism Spectrum Disorders: A Preliminary Investigation. Int. J. Environ. Res. Public Health 2016, 13, 388. https://doi.org/10.3390/ijerph13040388
Domingues VF, Nasuti C, Piangerelli M, Correia-Sá L, Ghezzo A, Marini M, Abruzzo PM, Visconti P, Giustozzi M, Rossi G, et al. Pyrethroid Pesticide Metabolite in Urine and Microelements in Hair of Children Affected by Autism Spectrum Disorders: A Preliminary Investigation. International Journal of Environmental Research and Public Health. 2016; 13(4):388. https://doi.org/10.3390/ijerph13040388
Chicago/Turabian StyleDomingues, Valentina F., Cinzia Nasuti, Marco Piangerelli, Luísa Correia-Sá, Alessandro Ghezzo, Marina Marini, Provvidenza M. Abruzzo, Paola Visconti, Marcello Giustozzi, Gerardo Rossi, and et al. 2016. "Pyrethroid Pesticide Metabolite in Urine and Microelements in Hair of Children Affected by Autism Spectrum Disorders: A Preliminary Investigation" International Journal of Environmental Research and Public Health 13, no. 4: 388. https://doi.org/10.3390/ijerph13040388
APA StyleDomingues, V. F., Nasuti, C., Piangerelli, M., Correia-Sá, L., Ghezzo, A., Marini, M., Abruzzo, P. M., Visconti, P., Giustozzi, M., Rossi, G., & Gabbianelli, R. (2016). Pyrethroid Pesticide Metabolite in Urine and Microelements in Hair of Children Affected by Autism Spectrum Disorders: A Preliminary Investigation. International Journal of Environmental Research and Public Health, 13(4), 388. https://doi.org/10.3390/ijerph13040388








