A Review on the Respiratory System Toxicity of Carbon Nanoparticles
Abstract
:1. Introduction
2. Are CNT and CNF Structurally Different?
3. CNTs and CNFs as Bio-Scaffold and Bio-Sensor Materials
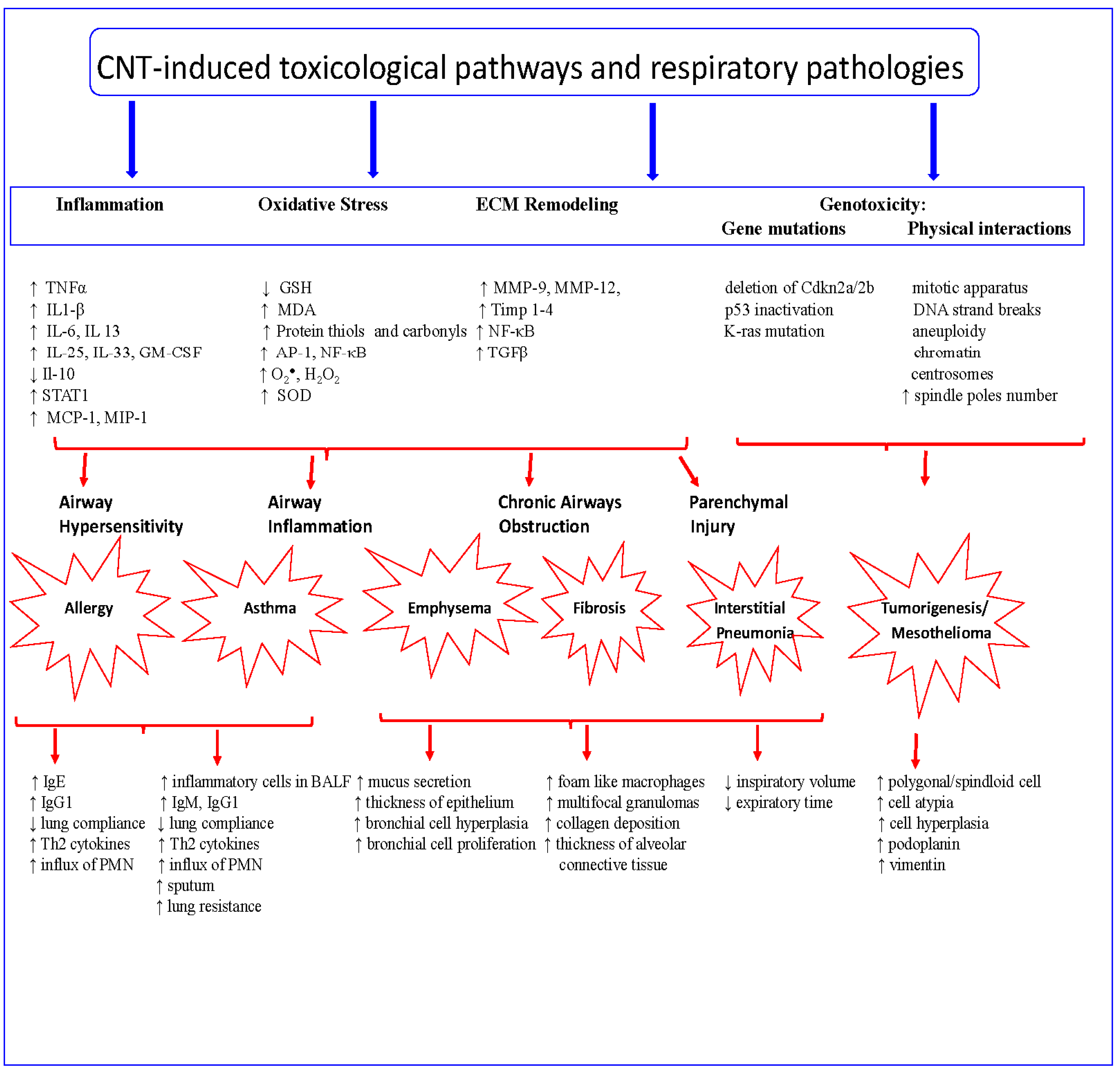
4. CNT Toxicological Pathways
4.1. Oxidative Stress
4.2. Extracellular Matrix Remodeling/Tissue Remodeling
4.3. Genotoxicity
4.4. MicroRNA Regulation
5. CNT-Induced Toxicological and Pathological Responses
5.1. Airway Hypersensitivity
5.2. Inflammatory Responses
5.3. Fibrogenic Responses
5.4. Tumorigenic Responses
6. Human Exposure to CNTs/CNFs
7. Conclusions and Perspectives on CNTs
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ijima, S.; Ichihashi, T. Single-shell carbon nanotubes of 1-nm diameter. Nature 1993, 363, 603–605. [Google Scholar] [CrossRef]
- Baughman, R.H.; Zakhidov, A.A.; de Heer, W.A. Carbon nanotubes-the route toward applications. Science 2002, 297, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Huizar, I.; Malur, A.; Patel, J.; McPeek, M.; Dobbs, L.; Wingard, C.; Barna, B.; Thomassen, M.J. The role of PPARγ in carbon nanotube-elicited granulomatous lung inflammation. Resp. Res. 2013, 14, 14–17. [Google Scholar] [CrossRef] [PubMed]
- Jensen, E.K.; Larsen, S.Y.; Nygaard, U.C.; Marioara, C.D.; Syversen, T. Early combination of material characteristics and toxicology is useful in the design of low toxicity carbon nanofiber. Materials 2012, 5, 1560–1580. [Google Scholar] [CrossRef]
- Donaldson, K.; Poland, C.A.; Murphy, F.A.; MacFarlane, M.; Chernova, T.; Schinwald, A. Pulmonary toxicity of carbon nanotubes and asbestos-similarities and differences. Adv. Drug Deliv. Rev. 2013, 65, 2078–2086. [Google Scholar] [CrossRef] [PubMed]
- Johnston, H.J.; Hutchison, G.R.; Christensen, F.M.; Peters, S.; Hankin, S.; Aschberger, K.; Stone, V. A critical review of the biological mechanisms underlying the in vivo and in vitro toxicity of carbon nanotubes: The contribution of physico-chemical characteristics. Nanotoxicology 2010, 4, 207–246. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization (ISO). Nanotechnologies—Terminology and Definitions for Nano-object—Nanoparticle, Nanofibre and Nanoplate; ISO/TS 27687:2008; International Organization for Standardization (ISO): Geneva, Switzerland, 2008. [Google Scholar]
- De Jong, K.; Geu, J. Carbon nanofibers: Catalytic synthesis and applications. Catal. Rev. Sci. Eng. 2000, 42, 481–510. [Google Scholar] [CrossRef]
- Kane, A.B. Epidemiology and pathology of asbestos-related diseases. Rev. Mineral. 1993, 18, 347–359. [Google Scholar]
- Shvedova, A.A.; Kisin, E.R.; Yanamala, N.; Tkach, A.V.; Gutkin, D.W.; Star, A.; Shuring, G.V.; Kagan, V.E.; Shurin, M.R. MDSC and TGFbeta are required for facilitation of tumor growth in the lungs of mice exposed to carbon nanotubes. Cancer Res. 2015, 75, 1615–1623. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Webster, T.J.; Sharma, A.; Basu, B. Intracellular reactive oxidative stress, cell proliferation and apoptosis of Schwann cells on carbon nanofibrous substrates. Biomaterials 2013, 34, 4891–4901. [Google Scholar] [CrossRef] [PubMed]
- Yokoama, A.; Sato, Y.; Nodasaka, Y.; Yamamoto, S.; Kawasaki, T.; Shindoh, M.; Kohgo, T.; Akasaka, T.; Uo, M.; Watari, F.; et al. Biological behavior of hat-stacked carbon nanofibers in the subcutaneous tissue in rats. Nano Lett. 2005, 5, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Khang, D.; Sato, M.; Price, R.L.; Ribbe, A.; Webster, T.J. Selective adhesion and mineral deposition by osteoblasts on carbon nanofiber patterns. Intern. J. Nanomed. 2006, 1, 65–72. [Google Scholar] [CrossRef]
- Price, R.L.; Ellison, K.; Haberstroh, K.W.; Webster, T.J. Nanometer surface roughness increases select ostoeblast adhesion on carbon nanofiber compacts. J. Biomed. Mater. Res. 2004, 70, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Ostrovidov, S.; Shi, X.; Zhang, L.; Liang, X.; Kim, S.B.; Fujie, T.; Ramalingam, M.; Chen, M.; Nakajima, K.; Al-Hazmi, F.; et al. Myotube formation on gelatin nanofibers-Multi-walled carbon nanotubes hybrid scaffolds. Biomaterials 2014, 35, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, C.B.; Peairs, J.; Venton, B.J. Review: Carbon nanotube based electrochemical sensors for biomolecules. Anal. Chim. Acta 2010, 662, 105–127. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Ahlawat, W.; Kumar, R.; Dilbaghi, N. Graphene, carbon nanotubes, zinc oxide and gold as elite nanomaterials for fabrication of biosensors for healthcare. Biosens. Bioelectron. 2015, 70, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Periyakaruppan, A.; Gandhiraman, R.P.; Meyyappan, M.; Koehne, J.E. Label-free detection of cardiac troponin-I using carbon nanofiber based nanoelectrode arrays. Anal. Chem. 2013, 85, 3858–3863. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Periyakaruppan, A.; Meyyappan, M.; Koehne, J.E. Label-free detection of C-reactive protein using a carbon nanofiber based biosensor. Biosens. Bioelectron. 2014, 59, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.; Kato, D.; Kamata, T.; Guo, Q.; You, T.; Niwa, O. Human cytochrome P450 3A4 and a carbon nanofiber modified film electrode as a platform for the simple evaluation of drug metabolism and inhibition reactions. Analyst 2013, 138, 6463–6468. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.; Li, L.; Yang, G.; Li, Y.; Luo, C.; Gong, T.; Zhou, S. Controlled green tea polyphenols release from electrospun PCL/MWCNTs composite nanofibers. Pharm. Nanotechnol. 2011, 421, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Pang, Z.; Chen, X.; Luo, L.; Cai, Y.; Wei, Q. A catechol biosensor based on electrospun carbon nanofibers. Beilstein J. Nanotechnol. 2014, 5, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Peng, J.; Wang, J.; Tang, H.; Tan, L.; Xie, Q. Carbon nanotube-based label-free electrochemical biosensor for sensitive detection of miRNA-24. Biosens. Bioelectron. 2014, 54, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Peng, Y.; Ren, J.; Qu, X. Carboxyl-modified single-walled carbon nanotubes selectively induce human telomeric i-motif formation. Proc. Natl. Acad. Sci. USA 2006, 103, 19658–19663. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, M.; Singh, A.; Kaur, S.; Arora, K. Enhancing lung cancer diagnosis: Electrochemical simultaneous bianalyte immunosensing using carbon nanotubes-chitosan nanocomposite. Appl. Biochem. Biotechnol. 2014, 174, 1188–1200. [Google Scholar] [CrossRef] [PubMed]
- Tian, Q.; Wang, Y.; Deng, R.; Lin, L.; Liu, Y.; Li, J. Carbon nanotube enhanced laber-free detection of microRNAs based on hairpin probe triggered solid-phase rolling-circle amplification. Nanoscale 2015, 7, 987–993. [Google Scholar] [CrossRef] [PubMed]
- Madani, S.Y.; Mandel, A.; Seifalian, A.M. A concise review of carbon nanotube’s toxicology. Nano Rev. 2013, 4. [Google Scholar] [CrossRef]
- Møller, P.; Christophersen, D.V.; Jensen, D.M.; Kermanizadeh, A.; Roursgaard, M.; Jacobsen, N.R.; Hemmingsen, J.G.; Danielsen, P.H.; Cao, Y.; Jantzen, K.; et al. Role of oxidative stress in carbon nanotube-generated health effect. Arch. Toxicol. 2014, 88, 1939–1964. [Google Scholar] [CrossRef] [PubMed]
- Schins, R.P. Mechanisms of genotoxicity of particles and fibers. Inhal. Toxicol. 2002, 14, 57–78. [Google Scholar] [CrossRef] [PubMed]
- Pacurari, M.; Schwegler-Berry, D.; Friend, D.; Vallyathan, V.; Castronova, V. Raw single-walled carbon nanotube-induced cytotoxic effects in human bronchial epithelial cells: Comparison to asbestos. Toxicol. Environ. Chem. 2011, 93, 1045–1072. [Google Scholar] [CrossRef]
- Pacurari, M.; Yin, X.; Ding, M.; Schwegler-Berry, D.; Morinobu, E.; Castranova, V.; Vallyathan, V. Oxidative and molecular interactions of multi-wall carbon nanoutbes (MWCNT) in normal and malignant human mesothelial cells. Nanotoxicology 2008, 3, 155–170. [Google Scholar] [CrossRef]
- Tsukahara, T.; Haniu, H. Cellular cytotoxic response induced by highly purified multi-wall carbon nanotube in human lung cells. Mol. Cell. Biochem. 2011, 352, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Pacurari, M.; Yin, X.; Zhao, J.; Ding, M.; Leonard, S.; Schwegler-Berry, D.; Ducatman, B.; Sbara, D.; Hoover, M.; Castranova, V.; et al. Raw single-wall carbon nanotubes induce oxidative stress and activate MAPKs, AP-1, NF-kB, and Akt in normal and malignant human mesothelial cells. Environ. Health Perspect. 2008, 116, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Khaliullin, T.O.; Shvedova, A.A.; Kisin, E.R.; Zalyalov, R.R.; Fatkhutdinova, L.M. Evaluation of fibrogenic potenatial of industrial multi-walled carbon nanotubes in acute aspiration experiment. Bull. Exp. Biol. Med. 2015, 158, 684–687. [Google Scholar] [CrossRef] [PubMed]
- Shvedova, A.A.; Kisin, E.; Murray, A.R.; Johnson, V.J.; Gorelik, O.; Arepalli, S.; Hubbs, A.F.; Mercer, R.R.; Keohavong, P.; Sussman, N.; et al. Inhalation vs. aspiration of single-walled carbon nanotubes in C57BL/6 mice: Inflammation, fibrosis, oxidative stress, and mutagenesis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 295, L552–L556. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, T.; O’Reilly, P.; Antony, V.B.; Gaggar, A.; Thannickal, V.J. Matrix remodeling in pulmonary fibrosis and emphysema. Am. J. Respir. Cell Mol. Biol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Mercer, R.R.; Hubbs, A.F.; Scabilloni, J.F.; Wang, L.; Battelli, L.A.; Friend, S.; Castranova, V.; Porter, D.W. Pulmonary fibrotic response to aspiration of multi-walled carbon nanotubes. Part. Fibre Toxicol. 2011, 8, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Pacurari, M.; Qian, Y.; Schegler-Berry, D.; Castranova, V.; Guo, N.L. Cell permeability, migration, and reactive oxygen species induced by multi-walled carbon nanotubes in human microvascular endothelial cells. J. Toxicol. Environ. Health A 2012, 75, 112–128. [Google Scholar] [CrossRef] [PubMed]
- Shvedova, A.A.; Tkach, A.V.; Kisin, E.R.; Khaliullin, T.; Stanley, S.; Gutkin, D.W.; Star, A.; Chen, Y.; Shurin, G.V.; Kagan, V.E.; Shuring, M.R. Carbon nanotubes enhance metastatic growth of lung carcinoma via up-regulation of myeloid-derived suppressor cells. Small 2013, 9, 1691–1965. [Google Scholar] [CrossRef] [PubMed]
- Thompson, E.A.; Sayers, B.C.; Glista-Baker, E.E.; Shipkowski, K.A.; Ihrie, M.D.; Duke, K.S.; Taylor, A.J.; Bonner, J.C. Role of signal transducer and activator of transcription 1 in murine allergen-induced airway remodeling and exarcerbation by carbon nanotubes. Am. J. Respir. Cell Mol. Biol. 2015, 53, 625–636. [Google Scholar] [CrossRef] [PubMed]
- Umemoto, E.Y.; Speck, M.; Shimoda, L.M.; Kahue, K.; Sung, C.; Stokes, A.J.; Turner, H. Single-walled carbon nanotube exposure induces membrane re-arrangement and suppression of receptor-mediated signaling pathways in model mast cells. Toxicol. Lett. 2014, 229, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Manke, A.; Luanpitpong, S.; Dong, C.; Wang, L.; He, X.; Battelli, L.; Derk, R.; Stueckle, T.A.; Porter, D.W.; Sager, T.; et al. Effect of fiber length on carbon nanotube-induced fibrogenesis. Int. J. Mol. Sci. 2014, 15, 7444–7461. [Google Scholar] [CrossRef] [PubMed]
- Ronzani, C.; Casset, A.; Pons, F. Exposure to multi-walled carbon nanotubes results in aggravation of airways inflammation and remodeling and in increased production of epithelium-derived innate cytokines in a mouse model of asthma. Arch. Toxicol. 2014, 88, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Li, X.; Wang, C.; Guo, H.; Liu, J.; Xu, H. Carbon nanotubes activate macrophages into a M1/M2 mixed status: Recruiting naїve macrophages and supporting angiogenesis. ACS Appl. Mater. Interfaces 2015, 7, 3180–3188. [Google Scholar] [CrossRef] [PubMed]
- Lohcharoenkal, W.; Wang, L.; Stueckle, T.A.; Dinu, C.Z.; Castranova, V.; Liu, Y.; Rojanasakul, Y. Chronic exposure to carbon nanotubes induces invasion of human mesothelial cells through matrix metalloproteinase-2. ACS Nano 2013, 7, 7711–7723. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, S.S.; Saber, A.T.; Williams, A.; Andersen, O.; Kobler, C.; Alturi, R.; Pozzebon, M.E.; Mucelli, S.P.; Simion, M.; Rickerby, D.; et al. MWCNTs of different physicochemical properties cause similar inflammatory responses, but differences in transcriptional and histological markers of fibrosis in mouse lungs. Toxicol. Appl. Pharmacol. 2015, 284, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Sayers, B.C.; Taylor, A.J.; Glista-Baker, E.E.; Shipley-Phillips, J.K.; Dackor, R.T.; Edin, M.L.; Lih, F.B.; Tomer, K.B.; Zeldin, D.C.; Langenbach, R.; et al. Role of cyclooxygenase-2 in exarcerbation of allergen-induced airway remodeling by multiwalled carbon nanotubes. Am. J. Respir. Cell Mol. Biol. 2013, 49, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Mizutani, N.; Nabe, T.; Yoshino, S. Exposure to multiwalled carbon nanotubes and allergen promotes early- and late-phase increases in airway resistance in mice. Biol. Pharm. Bull. 2012, 35, 2133–2140. [Google Scholar] [CrossRef] [PubMed]
- Mercer, R.R.; Scabolloni, J.F.; Hubbs, A.F.; Battelli, L.A.; McKinney, W.; Friend, S.; Wolfarth, M.G.; Andrew, M.; Castranova, V.; Porter, D.W. Distribution and fibrotic response following inhablation exposure to multi-walled carbon nanotubes. Part. Fibre Toxicol. 2013, 10, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Shannahan, J.H.; Bronw, J.M. IL-33 modulates chronic airway resistance changes induced by multiwalled carbon nanotubes. Inhal. Toxicol. 2014, 26, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Beamer, C.A.; Girtsman, T.A.; Seaver, B.P.; Finsaas, K.J.; Migliaccio, C.T.; Perry, K.V.; Rottman, B.J.; Smith, E.D.; Holian, A. IL-33 mediates multi-walled carbon nanoutubes (MWCNT)-induced airway hyper-reactivity via the mobilization of innate helper cells in the lung. Nanotoxicology 2013, 7, 1070–1081. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, H.K.; Falck, G.C.; Suhonen, S.; Vippola, M.; Vanhala, E.; Catalán, J. Genotoxicity of nanomaterials: DNA damage and micronuclei induced by carbon nanotubes and graphite nanofibres in human bronchial epithelial cells in vitro. Toxicol. Lett. 2009, 186, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Oesch, F.; Landsiedel, R. Genotoxicity investigations on nanomaterials. Arch. Toxicol. 2012, 86, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Toyokuni, S. Genotoxicity and carcinogenicity risk of carbon nanotubes. Adv. Drug Deliv. Rev. 2013, 65, 2098–2110. [Google Scholar] [CrossRef] [PubMed]
- Naya, M.; Kobayashi, N.; Endoh, S.; Mary, J.; Honda, K.; Ema, M.; Tanaka, J.; Fukumuro, M.; Hasegawa, K.; Nakajima, M.; et al. In vivo genotoxicity study of single-wall carbon nanotubes using comet assay following intratracheal instillation in rats. Regul. Toxicol. Pharmacol. 2012, 64, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Shvedova, A.A.; Yanamala, N.; Kisin, E.R.; Tkach, A.V.; Murray, A.R.; Hubbs, A.; Chirila, M.M.; Castranova, V. Long term effects of carbon containing engineered nanomaterials and asbestos in the lung: One year postexposure comparisons. Am. J. Physiol. Lung Cell. Mol. Physiol. 2014, 306, L170–L182. [Google Scholar] [CrossRef] [PubMed]
- Sargent, L.M.; Hubbs, A.F.; Young, S.H.; Kashon, M.L.; Dinu, C.Z.; Salisbury, J.L.; Benkovic, S.A.; Lowry, D.T.; Murray, A.R.; Kisin, E.R.; et al. Single-walled carbon nanotube-induced mitotic disruption. Mutat. Res. 2012, 745, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Sargent, L.M.; Porter, D.E.; Staska, L.M.; Hubbs, A.F.; Lowry, D.T.; Battelli, L.; Siegrist, K.J.; Kashon, M.L.; Mercer, R.R.; Castronava, V.; et al. Promotion of lung adenocarcinoma following inhalation exposure to multi-walled carbon nanotubes. Part. Fibre Toxicol. 2014, 11, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Hecht, S.S. Carcinogenicity studies of inhaled cigarette smoke in laboratory animals: Old and new. Carcinogenesis 2005, 26, 1488–1492. [Google Scholar] [CrossRef] [PubMed]
- Ulfvarson, U.; Alexandersson, R.; Aringer, L.; Svensson, E.; Hedenstierna, G.; Hogstedt, C.; Holmberg, B.; Rosen, G.; Sorsa, M. Effects of exposure to vehicle exhaust on health. Scand. J. Work Environ. Health 1987, 13, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Kisin, E.R.; Murray, A.R.; Sargent, L.; Lowry, D.; Chirila, M.; Siegrist, K.J.; Schwegler-Berry, D.; Leonard, S.; Castranova, V.; Fadeel, B.; et al. Genotoxicity of carbon nanofibers: Are they potentially more or less dangerous than carbon nanotubes or asbestos? Toxicol. Appl. Pharmacol. 2011, 252, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Di Giorgio, M.L.; Di Bucchianico, S.; Ragnelli, A.M.; Aimola, P.; Santucci, S.; Poma, A. Single and and multi walled carbon nanotubes on macrophages: Cyto and genotoxicity and electron microscopy. Mutat. Res. 2011, 722, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Totsuka, Y.; Ishino, K.; Matsumoto, Y.; Tada, Y.; Nakae, D.; Goto, S.; Masuda, S.; Ogo, S.; Kawanishi, M. Genotoxicity of multi-walled carbon nanotubes in both in vitro and in vivo assay systems. Nanotoxicology 2013, 7, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Monteiro-Riviere, N.A.; Nemanich, R.J.; Inman, A.O.; Wang, Y.Y.; Riviere, J.E. Multi-walled carbon nanotube interactions with human epidermal keratinocytes. Toxicol. Lett. 2005, 155, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Muller, J.; Decordier, I.; Hoet, P.H.; Lombaert, N.; Thomassen, L.; Huaux, F.; Lison, D.; Kirsch-Volders, M. Clastogenic and aneugenic effects of multi-wall carbon nanotubes in epithelial cells. Carcinogenesis 2008, 29, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Van Berlo, D.; Clift, M.J.; Albrecht, C.; Schins, R.P. Carbon nanotubes: An insight into the mechanism of their potential genotoxicity. Swiss Med. Wkly. 2012, 142, 13698–13704. [Google Scholar]
- Siegrist, K.J.; Reynolds, S.H.; Kashon, M.L.; Lowry, D.T.; Dong, C.; Hubbs, A.F.; Young, S.H.; Salisbury, J.L.; Porter, D.W.; Benkovic, S.A.; et al. Genotoxicity of multi-walled carbon nanotubes at occupationally relevant doses. Part. Fibre Toxicol. 2014, 11. [Google Scholar] [CrossRef] [PubMed]
- Sargent, L.M.; Shevedova, A.A.; Hubbs, A.F.; Salisbury, J.L.; Benkovic, S.A.; Kashon, M.L.; Lowry, D.T.; Murray, A.R.; Kisin, E.R.; Friend, S.; et al. Induction of aneuploidy by single-walled carbon nanotubes. Environ. Mol. Mutagen. 2009, 50, 708–717. [Google Scholar] [CrossRef] [PubMed]
- Pacurari, M.; Qian, Y.; Porter, D.W.; Wolfarth, M.; Wan, Y.; Luo, D.; Ding, M.; Castranova, V.; Guo, N.L. Multi-walled carbon nanoutube-induced gene expression in the mouse lung: association with lung pathology. Toxicol. Appl. Pharmacol. 2011, 255, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, D.; Fanizza, C.; Ursini, C.L.; Casciardi, S.; Paba, E.; Ciervo, A. Multi-walled carbon nanotubes induce cytotoxicity and genotoxicity in human lung epithelial cells. J. Appl. Toxicol. 2012, 32, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Rittinghausen, S.; Hackbarth, A.; Creutzenberg, O.; Ernst, H.; Heinrich, U.; Leonhardt, A.; Schaudien, D. The carcinogenic effect of various multi-walled carbon nanotubes (MWCNT) after intraperitoneal injection in rats. Part. Fibre Toxicol. 2014, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Nagai, H.; Okazaki, Y.; Chew, S.H.; Misawa, N.; Yamashita, Y.; Akatsuka, S.; Ishihara, T.; Yamashita, K.; Yoshikawa, Y.; Yasui, H.; et al. Diameter and rigidity of multiwalled carbon nanotubes are critical factors in mesothelial injury and carcinogenesis. Proc. Natl. Acad. Sci. USA 2011, 108, E1330–E1338. [Google Scholar] [CrossRef] [PubMed]
- Bartel, C.P. MicroRNAs. Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Zhao, Y.; Wu, Q.; Abdelli, Y.L.; Nouara, A.; Jia, R.; Wang, D. In vivo translocation and toxicity of multi-walled carbon nanotubes are regulated by microRNAs. Nanoscale 2014, 6, 4275–4284. [Google Scholar] [CrossRef] [PubMed]
- Nymark, P.; Wijshoff, P.; Cavill, R.; van Herwijnen, M.; Coonen, M.L.; Claessen, S.; Catalan, J.; Norppa, H.; Kleinjans, J.C.; Birede, J.J. Extensive temporal transcriptome and microRNA analyses identify molecular mechanisms underlying mitochondrial dysfunction induced by multi-walled carbon nanotubes in human lung cells. Nanotoxicology 2015, 1, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Alessandrini, F.; Schulz, H.; Takenaka, S.; Lentner, B.; Karg, E.; Behrendt, H.; Jakob, T. Effects of ultrafine carbon particle inhalation on allergic inflammation of the lung. Allergy Clin. Immunol. 2006, 17, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Alessandrini, F.; Semmler-Behnke, M.; Jakob, T.; Schulz, H.; Behrendt, H.; Kreyling, W. Total and regional deposition of ultrafine particles in a mouse model of allergic inflammation of the lung. Inhal. Toxicol. 2008, 20, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Cho, W.S.; Jeong, J.Y.J.; Choi, K.; Park, K. Pro-inflammatory and potential allergic responses resulting from B cell activation in mice treated with multi-walled carbon nanotubes by intratracheal instillation. Toxicology 2009, 259, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Koike, E.; Yanagisawa, R.; Hirano, S.; Nishikawa, M.; Takano, H. Effects of multi-walled carbon nanotubes on a murine allergic airway inflammation model. Toxicol. Appl. Pharmacol. 2009, 237, 306–316. [Google Scholar] [CrossRef] [PubMed]
- Rydman, E.M.; Ilves, M.; Koivisto, A.J.; Kinaret, P.A.; Fortino, V.; Savinko, T.S.; Lehto, M.T.; Pulkkinen, V.; Vippola, M.; Hameri, K.J.; et al. Inhalation of rod-like carbon nanotubes causes unconventional allergic airway inflammation. Part. Fibre Toxicol. 2014, 11, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, W.Y.; Chou, C.C.; Ho, C.C.; Yu, S.L.; Chen, H.Y.; Chou, H.Y.; Chen, J.W.J.; Yang, P.C. Single-walled carbon nanotubes induce airway hypereactivity and parenchymal injury in mice. Am. J. Respir. Cell Mol. Biol. 2011, 46, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Yanagisawa, R.; Koike, E.; Nishikawa, M.; Takano, H. Repeated pulmonary exposure to single-walled carbon nanotubes exacerbates allergic inflammation of the airway: Possible role of oxidative stress. Free Radic. Biol. Med. 2010, 48, 924–934. [Google Scholar] [CrossRef] [PubMed]
- Nygaard, U.C.; Samuelsen, M.; Marioara, C.D.; Løvik, M. Carbon nanofibers have IgE adjuvant capacity but are less potent than nanotubes in promoting allergic airway responses. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, R.F.; Wu, Z.; Mitra, S.; Shaw, P.K.; Holian, A. Effect of MWCNT size, carboxylation, and purification on in vitro and in vivo toxicity, inflammation and lung pathology. Part. Fibre Toxicol. 2013, 13, 13–57. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.W.; Kadoya, C.; Horie, M.; Mizuguchi, Y.; Hashiba, M.; Kambara, T.; Okada, T.; Myoho, T.; Oyabu, T.; Ogami, A.; et al. Analysis of pulmonary surfactant in rat lungs after intratracheal instillation of short and long multi-walled carbon nanotubes. Inhal. Toxicol. 2013, 25, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Ma-Hock, L.; Treumann, S.; Strauss, V.; Brill, S.; Luizi, F.; Mertler, M.; Wiench, K.; Gamer, A.O.; van Landdiedel, R. Inhalation toxicity of multiwall carbon nanotubes in rats exposed for 3 months. Toxicol. Sci. 2009, 112, 468–481. [Google Scholar] [CrossRef] [PubMed]
- Mercer, R.R.; Scabilloni, J.; Wang, L.; Kisin, E.; Murray, A.R.; Schwegler-Berry, D.; Shvedova, A.A.; Castranova, V. Alteration of deposition pattern and pulmonary response as a result of improved dispersion of aspirated single-walled carbon nanotubes in a mouse model. Am. J. Physiol. Cell Mol. Physiol. 2008, 294, L87–L97. [Google Scholar] [CrossRef] [PubMed]
- Muller, J.; Delos, M.; Panin, N.; Rabolli, V.; Huaux, F.; Lison, D. Absence of carcinogenic response to multiwall carbon nanotubes in a 2-year bioassay in the peritoneal cavity of the rat. Toxicol. Sci. 2009, 110, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Lam, C.W.; James, J.T.; McCluskey, R.; Hunter, R.L. Pulmonary toxicity of single-wall carbon nanotubes in mice 7 and 90 days after intratracheal instillation. Toxicol. Sci. 2004, 77, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Warheit, D.B.; Laurence, B.R.; Reed, K.L.; Roach, D.H.; Reynolds, G.A.; Webb, T.R. Comparative pulmonary toxicity assessment of single-wall carbon nanotubes in rats. Toxicol. Sci. 2004, 77, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Stueckle, T.A.; Mishra, A.; Derk, R.; Meighan, T.; Castranova, V.; Rojanasakul, Y. Neoplastic-like transformation effect of single-walled carbon nanotubes compared to asbestos on human lung small airway epithelial cells. Nanotoxicology 2014, 8, 485–507. [Google Scholar] [CrossRef] [PubMed]
- Luanpitpong, S.; Wang, L.; Manke, A.; Martin, K.H.; Ammer, A.G.; Castranova, V.; Yang, Y.; Rojansakul, Y. Induction of stemlike cells with fibrogenic properties by carbon nanotubes and its role in fibrogenesis. Nano Lett. 2014, 14, 3110–3116. [Google Scholar] [CrossRef] [PubMed]
- Nagai, H.; Okazaki, Y.; Chew, S.H.; Misawa, N.; Miyata, Y.; Shinohara, H.; Toyokuni, S. Intraperitoneal administration of tangled multiwalled carbon nanotubes of 15 nm in diameter does not induce mesothelial carcinogenesis in rats. Pathol. Int. 2013, 63, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Takagi, A.; Hirose, A.; Nishimura, T.; Fukumori, N.; Ogata, A.; Ohashi, N.; Kitajima, S.; Kanno, J. Induction of mesothelioma in p53+/− mouse by intraperitoneal application of multi-wall carbon nanotube. J. Toxicol. Sci. 2008, 33, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Luanpitpong, S.; Wang, L.; Castranova, V.; Rojanasakul, Y. Induction of stem-like cells with malignant properties by chronic exposure of human lung epithelial cells to single-walled carbon nanotubes. Part. Fibre Toxicol. 2014, 11, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Luanpitpong, S.; Wang, L.; Suteckle, T.A.; Tse, W.; Chen, Y.C.; Rojanasakul, Y. Caveolin-1 regulates lung cancer stem-like cell induction and p53 inactivation in carbon nanotube-driven tumorigenesis. Oncotarget 2014, 5, 3541–3554. [Google Scholar] [CrossRef] [PubMed]
- Dahm, M.M.; Evans, D.E.; Schubauer-Berigan, M.K.; Birch, M.E.; Fernback, J.E. Occupational exposure assessment in carbon nanotube and nanofiber primary and secondary manufacturers. Ann. Occup. Hyg. 2014, 56, 542–556. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Occupational Safety and Health (NIOSH). Draft Current Intelligence Bulletin Occupational Exposure to Carbon Nanotubes and Nanofibers; U.S. Department of Health and Human Services, Centers for Disease Control, National Institute for Occupational Safety and Health: Cincinnati, OH, USA, 2010. [Google Scholar]
- Lee, J.S.; Choi, Y.C.; Shin, J.H.; Lee, J.H.; Lee, Y.; Park, S.Y.; Baek, J.E.; Park, J.D.; Ahn, K.; Yu, I.J. Health surveillance study of workers who manufacture multi-walled carbon nanotubes. Nanotoxicology 2015, 9, 802–811. [Google Scholar] [CrossRef] [PubMed]
- Kuijpers, E.; Bekker, C.; Fransman, W.; Brouwer, D.; Tromp, P.; Vlaanderen, J.; Godderis, L.; Hoet, P.; Lan, Q.; Silverman, D.; et al. Occupational exposure to multwalled carbon nanotubes during commercial production synthesis and handling. Ann. Occup. Hyg. 2015. [Google Scholar] [CrossRef]
- Howard, J. Current Intelligence Bulletin 65: Occupational Exposure to Carbon Nanotubes and Nanofibers; DHHS (NIOSH) Publication: Washington, DC, USA, 2013. [Google Scholar]
- Han, J.H.; Lee, E.J.; Lee, J.H.; So, K.P.; Lee, Y.H.; Bae, G.N.; Lee, S.B.; Ji, J.H.; Cho, M.H.; Yu, I.J. Monitoring multiwalled carbon nanotube exposure in carbon nanotube research facility. Inhal. Toxicol. 2008, 20, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Methner, M.M. Effectiveness of local exhaust ventilation (LEV) in controlling engineered nanomaterial emissions during rector clean out operations. J. Occup. Environ. Hyg. 2008, 5, D63–D69. [Google Scholar] [PubMed]
- Regasamy, S.; King, W.; Eimer, B.; Shaffer, R. Filtration performance of NIOSH-approved N95 and PI00 filtering facemasks respirators against 4 to 30 nm-size nanoparticles. J. Occup. Environ. Hyg. 2008, 5, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Grosse, Y.; Loomis, D.; Guyton, K.Z.; Laube-Secretan, B.; Ghissassi, F.E.; Bouvard, V.; Benbrahim-Talla, L.; Guha, N.; Scoccianti, C.; Mattock, H.; et al. Carcinogenicity of flouro-dedenite, silican carbide fibres, and whiskers, and carbon nanoutbes. Lancet Oncol. 2014, 15, 1427–1428. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Fluoro-Edenite, Silicon Carbide Fibres and Whiskers, and Single-Walled and Multi-Walled Carbon Nanotubes IARC Working Group; IARC: Lyon, France, 2014; Volume 111. [Google Scholar]
- Kobayashi, N.; Naya, M.; Ema, M.; Endoh, S.; Maru, J.; Mizuno, K.; Nakanishi, J. Biological response and morphological assessment of individually dispersed multi-wall carbon nanotubes in the lung after intratracheal instillation in rats. Toxicology 2010, 276, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Kasai, T.; Umeda, Y.; Ohnishi, M.; Kondo, H.; Takeuchi, T.; Nisizawa, T.; Matsumoto, M.; Fukushima, S. Thirteen-week study of toxicity of fiber-like multi-walled carbon nanotubes with whole-body inhalation exposure in rats. Nanotoxicology 2015, 9, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Fukuda, M.; Endoh, S.; Maru, J.; Kato, H.; Nakamura, A.; Shinohara, N.; Uchino, K.; Honda, K. Size effects of single-walled carbon nanotubes on in vivo and in vitro pulmonary toxicity. Inhal. Toxicol. 2015, 27, 107–123. [Google Scholar] [CrossRef] [PubMed]
- Martino, E.; Prandi, L.; Fenoglio, I.; Bonfante, P.; Perotto, S. Soil fungal hyphae bind and attack asbestos fibers. Angew. Chem. Int. Ed. 2003, 42, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Favero-Longo, S.E.; Turci, F.; Tomatis, M.; Castelli, D.; Bonfante, P. Chrysotile asbestos is progressively converted into a non-fibrous amorphous material by the chelating action of lichen metabolites. J. Environ. Monit. 2005, 7, 764–766. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.T.; Wang, H.; Wang, Y.; Wang, Y.; Nie, H.; Liu, Y. Removal of carbon nanotubes from aqeous environment with filter paper. Chemosphere 2011, 82, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, M.; Xu, X.; Liu, N. Effects of carbon nanotubes on atrazine biodegradation by Arthrobacter sp. J. Hazard. Mater. 2015, 287, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Lambreva, M.D.; Lavecchia, T.; Tyystjarvia, E.; Antal, T.K.; Orlanducci, S.; Margonelli, A.; Rea, G. Potential of carbon nanotubes in algal biotechnology. Photosynth. Res. 2015, 125, 451–471. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, V.C.; Pietruska, J.R.; Miselis, N.R.; Hurt, R.H.; Kane, A.B. Biopersistence and potential adverse health impacts of fibrous nanomaterials: What have we learned from asbestos? Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2009, 1, 511–529. [Google Scholar] [CrossRef] [PubMed]
| Year | Types of CNTs/Sources | CNTs Length | Assay Type | Mode of Administration | Dose | Exposure Time | Outcome | Reference |
|---|---|---|---|---|---|---|---|---|
| MWCNTs negative results | ||||||||
| 2009 | MWCNT +/− defects | <1 μm | Wistar rats | I.P. | 2, 20 mg/animal | 24 months | No mesothelioma | [88] |
| 2010 | MWCNT (MWNT-7, Lot # T050831-01, Mitsui & Co. Ltd. 9, Tokyo, Japan) | 1.5 μm | SD rats | I.T. | 0.04, 0.2, 1 mg/kg | 3, 7 days; 1, 3, 6 months | Negative interstitial tissue, absence of fibrosis, MWCNT + Macrophages in alveoli. Lower case for macrophages. | [107] |
| MWCNTs positive results | ||||||||
| 2004 | CNT (Raw and Purified HiPco NTs, Rice University, Huston, TX, USA); CNT (CaboLex Inc., Lexington, KY, USA) | <1 μm | mice | I.T. | 0.1, 0.5 mg/animal | 7, 90 days | Epitheloid granulomas, interstitial inflammation, lung necrosis; high dose induced 60% death | [89] |
| 2009 | MWCNT (Bussan Nanotech Research, Ibaraki, Japan); SES Research (TX, USA) | 3–30 μm, several μm | ICR mice | I.T. | 25, 50 μg | 6 weeks | Exacerbation of allergic murine airway inflammation | [79] |
| 2012 | MWCNT (Mitsui MWCNT-7, No. 060125-01k, Tokyo, Japan) | <5 μm | p53 +/− mice | I.P. | 0.3 mg/animal | 1 year | Mesothelioma | [94] |
| 2013 | MWCNT (MWCNT-7, lot # 06122031, Hodogaya, Japan) | C57BL6J | Inhal. | 5 mg/m3, 5 h/d, 4 d/wk, 12 d | 1, 14, 84, 168, 336 days | Pulmonary inflammation, progressive collagen in alveolar regions after 336 days | [49] | |
| 2013 | CNF (Statoil and Elkem Carbon AS, Kristiansand, Norway) SWCNT (Sigma Aldrich, St. Louis, MO, USA, Cat. # 636797); MWCNT (Sigma-Aldrich, St. Louis, MO, USA, Cat. # 636487) | 5–10 0.5–100 0.5–200 μm | BALB/cAnNCrl Mice | Intra-nasal | 400 μg/mouse | 26 days | Modulation of airway responses to allergens; CNT more potent than CNF | [83] |
| 2014 | MWCNT (Mitsui-7, MWNT-7, lot #061220-31, Hodogaya, Japan) MCA, MWCNT, MCA + MWCNT | 0.5–5 μm | B6C3F1 | Inhal. | 5 mg/m3, 5 h/d, 5 d/wk, 15 d | 17 months | MCA potentiated MWCNT adenocarcinoma MWCNT alone did not induce adenocarcinoma | [58] |
| 2015 | MWCNT | 5.53–6.19 μm | F344 rats | Inhal. | 0.2, 1, 5 mg/m3 | 13 weeks | granulomatousa in females at 1 and 5 mg/m3; in males at 0.2 mg/m3 fibrosis | [108] |
| 2015 | MWCNT CNTsmall (NC700, Nanocyl, Sambreville, Belgium; 13% impurities); CNTlarge (NM-401, European Joint Research Centre, Ispra, Italy; 3% impurities) | 0.85, 4.05 μm | C57BL/6 mice, female | I.T. | 18, 54, 162 μg/animal | 1, 3, 28 days | Similar inflammatory and acute responses to both types; stronger fibrotic response to CNTlarge than CNTsmall | [46] |
| SWCNTs positive results | ||||||||
| 2010 | SWCNT: CNI, (USA); SES Res. (USA) | 1–15 μm | ICR mice | T. | 50 μg | 6 weeks | Exacerbation of allergic murine airway inflammation | [82] |
| 2014 | SWCNT (HiPco, Unidym, Sunnyvale, CA, USA); CNF (Pyrograf Prod., USA) Asbestos (UICC, USA) | 1–3 μm, 5–30 μm, 2–30 μm | C57BL/6 mice | Pharyngeal aspiration, inhalation | 40 μg/animal 40, 120 μg/animal 120 μg/animal 5 mg/m3, 5 h/4 d | 1 year | All particles induced chronic bronchopneumonia, pulmonary fibrosis. CNF > asbestos > SWCNT inflammation, SWCNT were the most fibrogenic, CNF, SWCNT induced K-ras mutations, No tumors | [56] |
| 2015 | SWCNTs (Tech. Res. Assoc. for SWCNT, Japan) | CNT-1 (0.51 μm, short); CNT-2 (1.67 μm, long/thick) | Wistar rats | I.T. | 0.18, 1.8 mg/kg | 1, 3, 7, 30, 90 days | Lung focal inflammation, neutrophil in alveoli, lung macrophages and cell derby in alveoli, higher CNT burden in CNT-1 than CNT-2 at 90 days | [109] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacurari, M.; Lowe, K.; Tchounwou, P.B.; Kafoury, R. A Review on the Respiratory System Toxicity of Carbon Nanoparticles. Int. J. Environ. Res. Public Health 2016, 13, 325. https://doi.org/10.3390/ijerph13030325
Pacurari M, Lowe K, Tchounwou PB, Kafoury R. A Review on the Respiratory System Toxicity of Carbon Nanoparticles. International Journal of Environmental Research and Public Health. 2016; 13(3):325. https://doi.org/10.3390/ijerph13030325
Chicago/Turabian StylePacurari, Maricica, Kristine Lowe, Paul B. Tchounwou, and Ramzi Kafoury. 2016. "A Review on the Respiratory System Toxicity of Carbon Nanoparticles" International Journal of Environmental Research and Public Health 13, no. 3: 325. https://doi.org/10.3390/ijerph13030325
APA StylePacurari, M., Lowe, K., Tchounwou, P. B., & Kafoury, R. (2016). A Review on the Respiratory System Toxicity of Carbon Nanoparticles. International Journal of Environmental Research and Public Health, 13(3), 325. https://doi.org/10.3390/ijerph13030325







