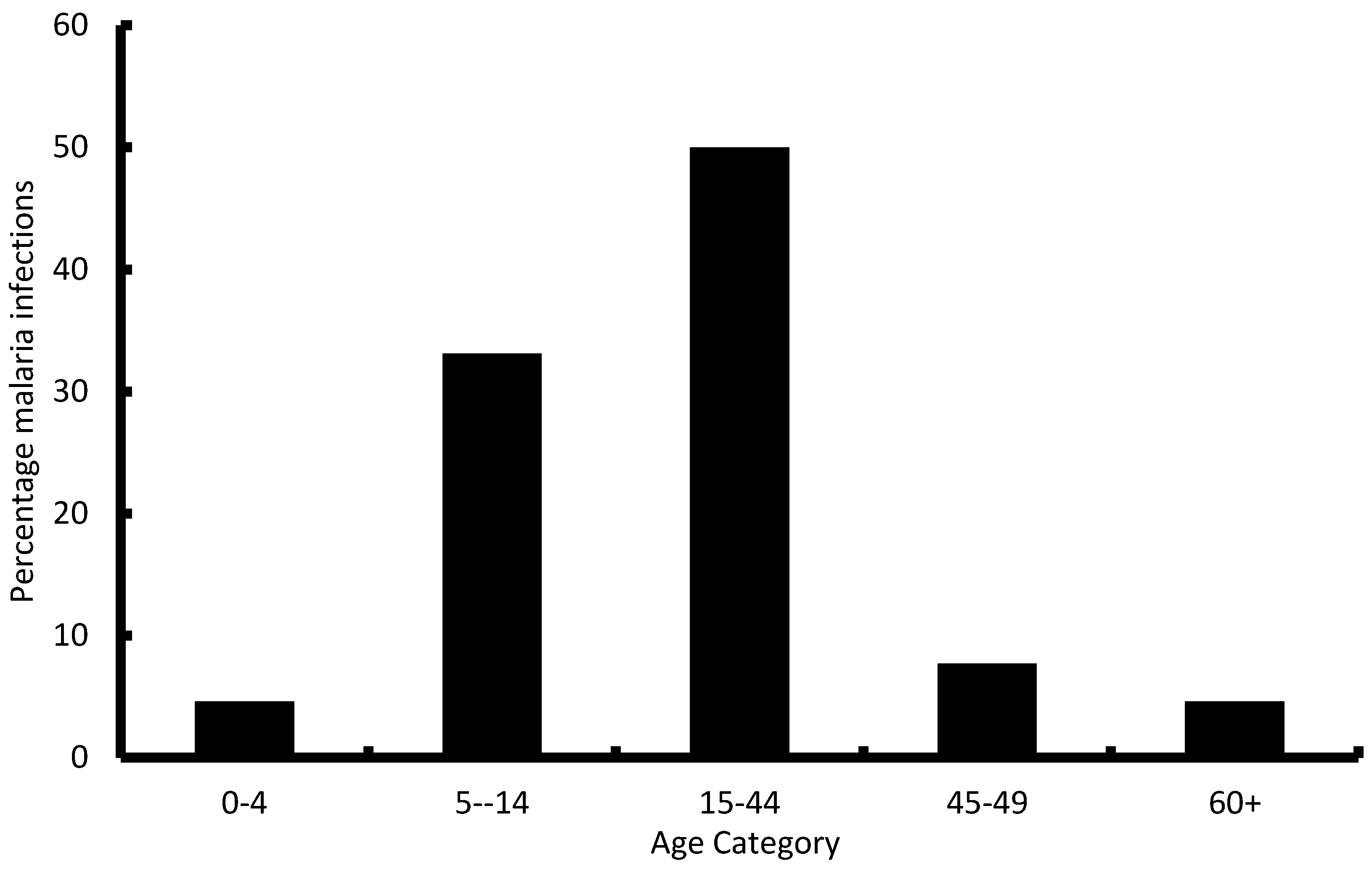1. Introduction
Malaria remains a challenge to public health in sub-Saharan Africa and is a leading cause of morbidity and mortality [
1,
2,
3]. It is one of the most prevalent parasitic diseases worldwide, with thousands of people suffering every year [
4]. According to the WHO World Malaria Report [
5], a total of 104 countries are considered malaria endemic and at least 3.2 billion people are at risk. The same report states that a total of 198 million cases occurred globally in 2013 with most deaths occurred in Africa in children under the age of 5 years. Malaria contributes to a large public health burden with over 75% of the clinical episodes worldwide found in Africa [
6]. In sub-Saharan Africa alone, mortality and morbidity due to malaria accounts for a loss of 35.4 million Disability Adjusted Life Years (DALYs) [
7,
8].
The Disability-Adjusted Life Year (DALY) is a metric measure for burden of disease [
9] developed by the World Health Organization (WHO), World Bank and the Harvard School of Public Health researchers [
10,
11,
12]. The 2010 Global Burden of Disease (GBD) study showed malaria as the seventh leading cause of DALYs lost globally [
13]. The DALY not only looks at premature death due to disease, but also takes into account disability caused by disease. It combines both time lost due to premature mortality and non-fatal conditions. In the DALY approach, a disability weighted zero indicates perfect health (no disability), and one weighted 1 indicates death [
11]. The DALY is a time-based measure that combines years of life lost due to premature mortality and years of life lost due to time lived in health states less than ideal health [
12]. One DALY is defined as one lost year of “healthy” life, and the burden of disease (BoD) is a measurement of the gap between current health status and an ideal situation where everyone lives into old age, free of disease and disability [
11]. Use of the DALY metric in assessing disease burden is essential as it is a measure used by most organisations to determine funding for malaria control in Africa [
14,
15]. Although the DALY is a measure of disease burden, it is also commonly used in the analysis of cost-effectiveness of interventions [
16]. It therefore helps to indicate the socio-economic impact of disease burden unlike the conventional methods which only show the prevalence and mortality rates of the disease.
About 50% of the population in Zimbabwe lives in malarious areas therefore, nearly 6 million people are at risk of malaria [
5,
17]. The country experiences seasonal malaria transmission which is potentially epidemic [
18]. Although there are on-going malaria control efforts in the country, the disease still remains a public health challenge [
19,
20] and continues to be a major cause of mortality and morbidity [
21]. Resistance of the malaria vectors to insecticides [
19,
22] as well as resistance to anti-malarial drugs [
23] contribute to the challenges in control of malaria in Zimbabwe.
In addition to receiving government funding, the national malaria control programme in Zimbabwe also receives support from the Roll Back Malaria Partnership, the Global Fund to Fight AIDS, Tuberculosis and Malaria, and The President’s Malaria Initiative [
5,
24,
25]. The Roll Back Malaria Programme was launched in Zimbabwe in 2001 and was subsequently scaled up [
21]. The Global Fund to Fight AIDS, Tuberculosis and Malaria, and The President’s Malaria Initiative were launched in 2008 and 2009 respectively [
24]. The recommended first-line treatment regimen changed from chloroquine or a combination of chloroquine plus sulphadoxine/pyrimethamine to artemisinin-based combination therapy, and the latter adopted by all health clinics by 2010 [
21]. Diagnostic capacity improved, with rapid diagnostic tests (RDTs) available in health clinics by 2008. Mosquito control consists of indoor residual spraying and use of long-lasting insecticidal nets (LLINs) [
21].
In order to assess the effectiveness of the current interventions to reduce malaria, there is need for in-country estimates of the national burden for the disease. This enables quantification of the needs for interventions to control and prevent malaria transmission such as indoor residual spraying and use of insecticide-treated bed nets in the population at risk. Estimates of the burden of disease also guide national disease surveillance strategies [
26] and provide information on disease severity [
26]. This assists in resource allocation as well as in targeting high priority areas. Most African countries have health budgetary constraints due to many competing health challenges and health system that are weak or dysfunctional [
27]. It is therefore crucial that policy makers are provided with information on the economic benefits of malaria control and prevention.
Limitations of the available data sources in many countries have led either to disease burden not being estimated at all or estimated using inaccurate methods [
26]. It is important that calculations for disease burden are done at both the population and individual levels. A person may carry an important individual burden of disease, even though the population burden may be negligible [
28]. Likewise, certain sub-populations may suffer a higher burden of disease than the average population burden. Previous studies on burden of disease in Zimbabwe focused on all-cause burden at national level [
13,
29]. The present study is the first to measure burden of malaria in Zimbabwe in terms of DALYs at household and district levels using locally-derived epidemiological data. This study was carried out as part of a larger study on malaria and schistosomiasis in Gwanda district, Zimbabwe.
4. Discussion
Our study is the first in Zimbabwe to use local sources of information to assess burden of malaria at household and district levels. Most countries have estimated all-cause burden of disease at national level rather than disease-specific burden. One such study was carried in Zimbabwe [
29]. Our study focused on malaria burden for the period 2011to 2015 because before 2011, reported cases were mostly based on clinical symptoms without confirmation through a rapid test or a blood slide. This meant that the number of reported malaria cases before 2011 in Zimbabwe were likely to be overstated. Hence malaria cases from 2011 onwards give a more accurate picture of the burden of malaria as all reported cases were confirmed.
The results of this study show a general decline in the calculated number of DALYs lost due to malaria in Gwanda. This decline may be explained by the intensification of the malaria control intervention programmes in the district including case treatment, distribution of insecticide treated nets and indoor residual spraying (IRS). A total of 96% of cases reported led to remission of the diseases. Cases that led to deaths were mostly as a result of failure by the patient to present at a health facility before complications set in. It is also possible that some of the people who presented late at health facilities would have tried to seek treatment elsewhere. Alternative sources of treatment reported included traditional healers and faith-based healers. The high number of cases leading to remission shows the effectiveness of the current malaria treatment regimen.
Although the number of deaths from malaria was low, the few malaria deaths contributed to the bulk of the DALYs lost due to malaria through YLLs. A malaria episode lasted for 7 days on average. This short disease duration resulted in lower values for YLDs compared to YLLs. However, the burden of malaria in terms of YLDs would actually be higher if other possible consequences of malaria, such as renal complications, are taken into consideration [
39].
Mortality was therefore a major contributor to DALYs for malaria. Malaria interventions that avert malaria deaths are therefore very critical as they significantly reduce the disease burden.
Some previous studies on malaria burden reported higher burden in the below 5 years age category [
39,
40]. However, our results show a different scenario with most of the reported cases falling in the 5 years and above age group. This could be as a result of the many malaria interventions in the country targeting the below 5 years age group thereby resulting in very low number of cases in that group. This study showed that the majority of cases fall in the economically productive age groups (15–45 years) indicating that malaria has an economic impact on affected families as they lose productive time during sickness. There is therefore need for good investment in malaria control, not only to reduce the public health problem, but to also reduce the economic strain that malaria exerts on households.
DALYs are estimates and not definitive numbers but they indicate the relative size of the burden [
41] and hence allow comparisons across different settings and geographical regions. Although mortality and morbidity due to malaria are showing a downward trend globally [
5], it is important to have burden estimates for the areas that are still affected by the disease. The DALYs for the district showed variations between 2011 and 2015. If these DALYs were calculated over a longer period than the one in this study, they will have provided insight on the general trend of the burden of malaria in the area. This information will be important for policy makers as they plan for future control efforts. This shows the need for continued close monitoring of the burden of malaria as the variations from this study indicate that malaria control efforts are fragile. Outbreaks can easily occur if there are inconsistencies in the interventions for prevention of malaria. Since malaria is a focal disease, it is advisable for policy makers to continually implement geographically-targeted interventions for high burden areas as this may have a favourable cost-benefit ratio [
42]. This will ensure that the disease burden is reduced and the possibilities of outbreaks are minimised.
Other previous studies that utilised the DALYs in assessing burden of malaria and other neglected tropical diseases are shown in
Table 8. The major difference with our study is that most of these studies applied age weighting and discounting in their DALY calculations [
42,
43,
44,
45,
46,
47,
48,
49] and the studies were done at national level [
39,
42,
44,
46,
48,
50].
Malaria is a notifiable disease in Zimbabwe and the majority of malaria episodes are captured in the DHIS. The DHIS is therefore a good representation of the malaria situation in the district. However, it is possible that some malaria cases may have not presented at the health facilities and hence not be included in the DHIS. Health facility-based data is also affected by many factors including accessibility, perceptions of care and whether or not self-treatment is available [
52]. There is also a possibility of health workers not ascertaining malaria or failing to record cases even if they are diagnosed. Although the household census did not indicate any form of under-reporting by health workers it could still be a source of bias in the DHIS figures. We could not make direct comparisons between the household and district level DALYs because the data for the district was not disaggregated by age groups. There were only two broad groups of below 5 years and above 5 years. It was not logistically possible to collect age-specific data for the whole district during this study. Also, the data for the district did not classify deaths by sex whereas we were able to do in the household survey. This means that although our DALY estimates for the whole district gave us a good picture of the burden; the estimates from the household level data gave a better representation of the malaria burden.