Protective Effect of Onion Extract on Bleomycin-Induced Cytotoxicity and Genotoxicity in Human Lymphocytes
Abstract
:1. Introduction
2. Experimental Section
2.1. Cell Isolation and Culture
2.2. Preparation of Onion Extracts
2.3. Cell Treatment
2.4. Cell Viability Assay
2.5. Cytokinesis Block Micronucleus Assay
2.6. Single-Cell Gel Electrophoresis (Comet Assay)
2.7. Quantitative Analysis of Quercetin by High Performance Liquid Chromatography (HPLC)
2.8. Statistical Analysis
3. Results
3.1. Cell Viability with Onion Extracts and BLM Treatment
3.2. Protective Effect of Onion Extracts on the Frequency of BLM-Induced MN
3.3. Protective Effect of the Onion Extract on BLM-Induced DNA Damage
3.4. Quantitative Analysis of Quercetin
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anzai, K.; Ban, N.; Ozawa, T.; Tokonami, S. Fukushima daiichi nuclear power plant accident: Facts, environmental contamination, possible biological effects, and countermeasures. J. Clin. Biochem. Nutr. 2012, 50, 2–8. [Google Scholar] [PubMed]
- Strzelczyk, J.J.; Damilakis, J.; Marx, M.V.; Macura, K.J. Facts and controversies about radiation exposure, part 2: Low-level exposures and cancer risk. JACR 2007, 4, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Hevia, D.; Patchva, S.; Park, B.; Koh, W.; Aggarwal, B.B. Upsides and downsides of reactive oxygen species for cancer: The roles of reactive oxygen species in tumorigenesis, prevention, and therapy. Antioxid. Redox Signal. 2012, 16, 1295–1322. [Google Scholar] [CrossRef] [PubMed]
- Mira, A.; Gimenez, E.M.; Bolzan, A.D.; Bianchi, M.S.; Lopez-Larraza, D.M. Effect of thiol compounds on bleomycin-induced DNA and chromosome damage in human cells. Arch. Environ. Occup. Health 2013, 68, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Dresp, J.; Schmid, E.; Bauchinger, M. The cytogenetic effect of bleomycin on human peripheral lymphocytes in vitro and in vivo. Mutat. Res. 1978, 56, 341–353. [Google Scholar] [CrossRef]
- Povirk, L.F.; Austin, M.J. Genotoxicity of bleomycin. Mutat. Res. 1991, 257, 127–143. [Google Scholar] [CrossRef]
- Wallach-Dayan, S.B.; Izbicki, G.; Cohen, P.Y.; Gerstl-Golan, R.; Fine, A.; Breuer, R. Bleomycin initiates apoptosis of lung epithelial cells by ros but not by fas/fasl pathway. Am. J. Physiol. Lung Cell. Mol. Physiol. 2006, 290, L790–L796. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.A.; Kasum, C.M. Dietary flavonoids: Bioavailability, metabolic effects, and safety. Ann. Rev. Nutr. 2002, 22, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Miean, K.H.; Mohamed, S. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. J. Agric. Food Chem. 2001, 49, 3106–3112. [Google Scholar] [CrossRef] [PubMed]
- Nijveldt, R.J.; Van Nood, E.; Van Hoorn, D.E.; Boelens, P.G.; Van Norren, K.; Van Leeuwen, P.A. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar] [PubMed]
- Fauconneau, B.; Waffo-Teguo, P.; Huguet, F.; Barrier, L.; Decendit, A.; Merillon, J.M. Comparative study of radical scavenger and antioxidant properties of phenolic compounds from vitis vinifera cell cultures using in vitro tests. Life Sci. 1997, 61, 2103–2110. [Google Scholar] [CrossRef]
- RiceEvans, C.A.; Miller, J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Liu, R.H. Potential synergy of phytochemicals in cancer prevention: Mechanism of action. J. Nutr. 2004, 134, 3479S–3485S. [Google Scholar] [PubMed]
- Hertog, M.G.L.; Hollman, P.C.H.; Katan, M.B. Content of potentially anticarcinogenic flavonoids of 28 vegetables and 9 fruits commonly consumed in the netherlands. J. Agric. Food Chem. 1992, 40, 2379–2383. [Google Scholar] [CrossRef]
- Slimestad, R.; Fossen, T.; Vagen, I.M. Onions: A source of unique dietary flavonoids. J. Agr. Food Chem. 2007, 55, 10067–10080. [Google Scholar] [CrossRef] [PubMed]
- Prakash, D.; Singh, B.N.; Upadhyay, G. Antioxidant and free radical scavenging activities of phenols from onion (Allium cepa). Food Chem. 2007, 102, 1389–1393. [Google Scholar] [CrossRef]
- Hosseinimehr, S.J. Flavonoids and genomic instability induced by ionizing radiation. Drug. Discov. Today 2010, 15, 907–918. [Google Scholar] [CrossRef] [PubMed]
- Fenech, M.; Chang, W.P.; Kirsch-Volders, M.; Holland, N.; Bonassi, S.; Zeiger, E.; project, H.U.M. Humn project: Detailed description of the scoring criteria for the cytokinesis-block micronucleus assay using isolated human lymphocyte cultures. Mutat. Res. 2003, 534, 65–75. [Google Scholar] [CrossRef]
- Tice, R.R.; Agurell, E.; Anderson, D.; Burlinson, B.; Hartmann, A.; Kobayashi, H.; Miyamae, Y.; Rojas, E.; Ryu, J.C.; Sasaki, Y.F. Single cell gel/comet assay: Guidelines for in vitro and in vivo genetic toxicology testing. Environ. Mol. Mutagen. 2000, 35, 206–221. [Google Scholar] [CrossRef]
- Boyum, A. Separation of lymphocytes, granulocytes, and monocytes from human blood using iodinated density gradient media. Meth. Enzymol. 1984, 108, 88–102. [Google Scholar] [PubMed]
- Strober, W. Trypan blue exclusion test of cell viability. Curr. Protoc. Immunol. 2001. [Google Scholar] [CrossRef]
- Fenech, M. The in vitro micronucleus technique. Mutat. Res. 2000, 455, 81–95. [Google Scholar] [CrossRef]
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res. 1988, 175, 184–191. [Google Scholar] [CrossRef]
- Olive, P.L.; Banath, J.P. The comet assay: A method to measure DNA damage in individual cells. Nat. Protoc. 2006, 1, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Kumaravel, T.S.; Jha, A.N. Reliable comet assay measurements for detecting DNA damage induced by ionising radiation and chemicals. Mutat. Res. 2006, 605, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Olive, P.L.; Banath, J.P.; Durand, R.E. Heterogeneity in radiation-induced DNA damage and repair in tumor and normal cells measured using the ”comet” assay. Radiat. Res. 1990, 122, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Van Deventer, E.; Del Rosario, P.M.; Tritscher, A.; Fukushima, K.; Carr, Z. Who’ s public health agenda in response to the fukushima daiichi nuclear accident. J. Radiol. Prot. 2012, 32, N119–N122. [Google Scholar] [CrossRef] [PubMed]
- Akiba, S. Epidemiological studies of fukushima residents exposed to ionising radiation from the fukushima daiichi nuclear power plant prefecture—A preliminary review of current plans. J. Radiol. Prot. 2012, 32, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tsushima, H.; Iwanaga, M.; Miyazaki, Y. Late effect of atomic bomb radiation on myeloid disorders: Leukemia and myelodysplastic syndromes. Int. J. Hematol. 2012, 95, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Sauvaget, C.; Lagarde, F.; Nagano, J.; Soda, M.; Koyama, K.; Kodama, K. Lifestyle factors, radiation and gastric cancer in atomic-bomb survivors (Japan). Cancer Causes Control 2005, 16, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Sauvaget, C.; Kasagi, F.; Waldren, C.A. Dietary factors and cancer mortality among atomic-bomb survivors. Mutat. Res. Fund Mol. Mech. 2004, 551, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Nagano, J.; Kono, S.; Preston, D.L.; Moriwaki, H.; Sharp, G.B.; Koyama, K.; Mabuchi, K. Bladder-cancer incidence in relation to vegetable and fruit consumption: A prospective study of atomic-bomb survivors. Int. J. Cancer 2000, 86, 132–138. [Google Scholar] [CrossRef]
- Little, J.B. Radiation-induced genomic instability. Int. J. Radiation Biol. 1998, 74, 663–671. [Google Scholar] [CrossRef]
- Trachootham, D.; Alexandre, J.; Huang, P. Targeting cancer cells by ros-mediated mechanisms: A radical therapeutic approach? Nat. Rev. Drug Discov. 2009, 8, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Knekt, P.; Jarvinen, R.; Reunanen, A.; Maatela, J. Flavonoid intake and coronary mortality in Finland: A cohort study. BMJ 1996, 312, 478–481. [Google Scholar] [CrossRef] [PubMed]
- Shon, M.Y.; Choi, S.D.; Kahng, G.G.; Nam, S.H.; Sung, N.J. Antimutagenic, antioxidant and free radical scavenging activity of ethyl acetate extracts from white, yellow and red onions. Food Chem. Toxicol. 2004, 42, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Alpsoy, S.; Kanter, M.; Aktas, C.; Erboga, M.; Akyuz, A.; Akkoyun, D.C.; Oran, M. Protective effects of onion extract on cadmium-induced oxidative stress, histological damage, and apoptosis in rat heart. Biol. Trace Elem. Res. 2014, 159, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Mete, R.; Oran, M.; Topcu, B.; Oznur, M.; Seber, E.S.; Gedikbasi, A.; Yetisyigit, T. Protective effects of onion (Allium cepa) extract against doxorubicin-induced hepatotoxicity in rats. Toxicol. Ind. Health 2013. [Google Scholar] [CrossRef] [PubMed]
- Shimoi, K.; Masuda, S.; Furugori, M.; Esaki, S.; Kinae, N. Radioprotective effect of antioxidative flavonoids in gamma-ray irradiated mice. Carcinogenesis 1994, 15, 2669–2672. [Google Scholar] [CrossRef] [PubMed]
- Del Bano, M.J.; Castillo, J.; Benavente-Garcia, O.; Lorente, J.; Martin-Gil, R.; Acevedo, C.; Alcaraz, M. Radioprotective-antimutagenic effects of rosemary phenolics against chromosomal damage induced in human lymphocytes by gamma-rays. J. Agric. Food Chem. 2006, 54, 2064–2068. [Google Scholar] [CrossRef] [PubMed]
- Heo, M.Y.; Lee, S.J.; Kwon, C.H.; Kim, S.W.; Sohn, D.H.; Au, W.W. Anticlastogenic effects of galangin against bleomycin-induced chromosomal aberrations in mouse spleen lymphocytes. Mutat. Res. 1994, 311, 225–229. [Google Scholar] [CrossRef]
- Masella, R.; Di Benedetto, R.; Vari, R.; Filesi, C.; Giovannini, C. Novel mechanisms of natural antioxidant compounds in biological systems: Involvement of glutathione and glutathione-related enzymes. J. Nutr. Biochem. 2005, 16, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Moskaug, J.O.; Carlsen, H.; Myhrstad, M.C.; Blomhoff, R. Polyphenols and glutathione synthesis regulation. Am. J. Clin. Nutr. 2005, 81, 277S–283S. [Google Scholar] [PubMed]
- Griffiths, G.; Trueman, L.; Crowther, T.; Thomas, B.; Smith, B. Onions—A global benefit to health. Phytother. Res. 2002, 16, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.P.; Calomme, M.; Lasure, A.; De Bruyne, T.; Pieters, L.; Vlietinck, A.; Van den Berghe, D.A. Structure-activity relationship of flavonoids with superoxide scavenging activity. Biol. Trace Element Res. 1995, 47, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Magnani, L.; Gaydou, E.M.; Hubaud, J.C. Spectrophotometric measurement of antioxidant properties of flavones and flavonols against superoxide anion. Anal. Chim. Acta 2000, 411, 209–216. [Google Scholar] [CrossRef]
- Inal, M.E.; Kahraman, A. The protective effect of flavonol quercetin against ultraviolet a induced oxidative stress in rats. Toxicology 2000, 154, 21–29. [Google Scholar] [CrossRef]
- Afanasev, I.B.; Dorozhko, A.I.; Brodskii, A.V.; Kostyuk, V.A.; Potapovitch, A.I. Chelating and free-radical scavenging mechanisms of inhibitory-action of rutin and quercetin in lipid-peroxidation. Biochem. Pharmacol. 1989, 38, 1763–1769. [Google Scholar] [CrossRef]
- Formica, J.V.; Regelson, W. Review of the biology of quercetin and related bioflavonoids. Food Chem. Toxicol. 1995, 33, 1061–1080. [Google Scholar] [CrossRef]
- Anderson, R.F.; Amarasinghe, C.; Fisher, L.J.; Mak, W.B.; Packer, J.E. Reduction in free-radical-induced DNA strand breaks and base damage through fast chemical repair by flavonoids. Free Radical. Res. 2000, 33, 91–103. [Google Scholar] [CrossRef]
- Charles, C.; Nachtergael, A.; Ouedraogo, M.; Belayew, A.; Duez, P. Effects of chemopreventive natural products on non-homologous end-joining DNA double-strand break repair. Mutat. Res. Gen. Tox. Environ. Mutagen. 2014, 768, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Charles, C.; Chemais, M.; Stevigny, C.; Dubois, J.; Nachergael, A.; Duez, P. Measurement of the influence of flavonoids on DNA repair kinetics using the comet assay. Food Chem. 2012, 135, 2974–2981. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, K.; King, J.W.; Howard, L.R.; Monrad, J.K. Solubility and solution thermodynamic properties of quercetin and quercetin dihydrate in subcritical water. J. Food Eng. 2010, 100, 208–218. [Google Scholar] [CrossRef]
- Stalikas, C.D. Extraction, separation, and detection methods for phenolic acids and flavonoids. J. Sep. Sci. 2007, 30, 3268–3295. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.L.; Haley, S.; Perret, J.; Harris, M.; Wilson, J.; Qian, M. Free radical scavenging properties of wheat extracts. J. Agric. Food Chem. 2002, 50, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Jo, S.H.; Jang, H.D.; Lee, M.S.; Kwon, Y.I. Antioxidant activity and alpha-glucosidase inhibitory potential of onion (Allium cepa L.) extracts. Food Sci. Biotechnol. 2010, 19, 159–164. [Google Scholar] [CrossRef]
- Hollman, P.C.; Cassidy, A.; Comte, B.; Heinonen, M.; Richelle, M.; Richling, E.; Serafini, M.; Scalbert, A.; Sies, H.; Vidry, S. The biological relevance of direct antioxidant effects of polyphenols for cardiovascular health in humans is not established. J. Nutr. 2011, 141, 989S–1009S. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [PubMed]
- Anson, N.M.; Van den Berg, R.; Havenaar, R.; Bast, A.; Haenen, G.R.M.M. Bioavailability of ferulic acid is determined by its bioaccessibility. J. Cereal. Sci. 2009, 49, 296–300. [Google Scholar] [CrossRef]
- Barve, A.; Chen, C.; Hebbar, V.; Desiderio, J.; Saw, C.L.L.; Kong, A.N. Metabolism, oral bioavailability and pharmacokinetics of chemopreventive kaempferol in rats. Biopharm. Drug Dispos. 2009, 30, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Caridi, D.; Trenerry, V.C.; Rochfort, S.; Duong, S.; Laugher, D.; Jones, R. Profiling and quantifying quercetin glucosides in onion (Allium cepa L.) varieties using capillary zone electrophoresis and high performance liquid chromatography. Food Chem. 2007, 105, 691–699. [Google Scholar] [CrossRef]
- Leighton, T.; Ginther, C.; Fluss, L.; Harter, W.K.; Cansado, J.; Notario, V. Molecular characterization of quercetin and quercetin glycosides in allium vegetables—Their effects on malignant-cell transformation. Acs. Sym. Ser. 1992, 507, 220–238. [Google Scholar]
- Lombard, K.A.; Geoffriau, E.; Peffley, E. Flavonoid quantification in onion by spectrophotometric and high performance liquid chromatography analysis. Hortscience 2002, 37, 682–685. [Google Scholar]
- Grant, E.J.; Ozasa, K.; Preston, D.L.; Suyama, A.; Shimizu, Y.; Sakata, R.; Sugiyama, H.; Pham, T.M.; Cologne, J.; Yamada, M.; et al. Effects of radiation and lifestyle factors on risks of urothelial carcinoma in the life span study of atomic bomb survivors. Radiat. Res. 2012, 178, 86–98. [Google Scholar] [CrossRef] [PubMed]
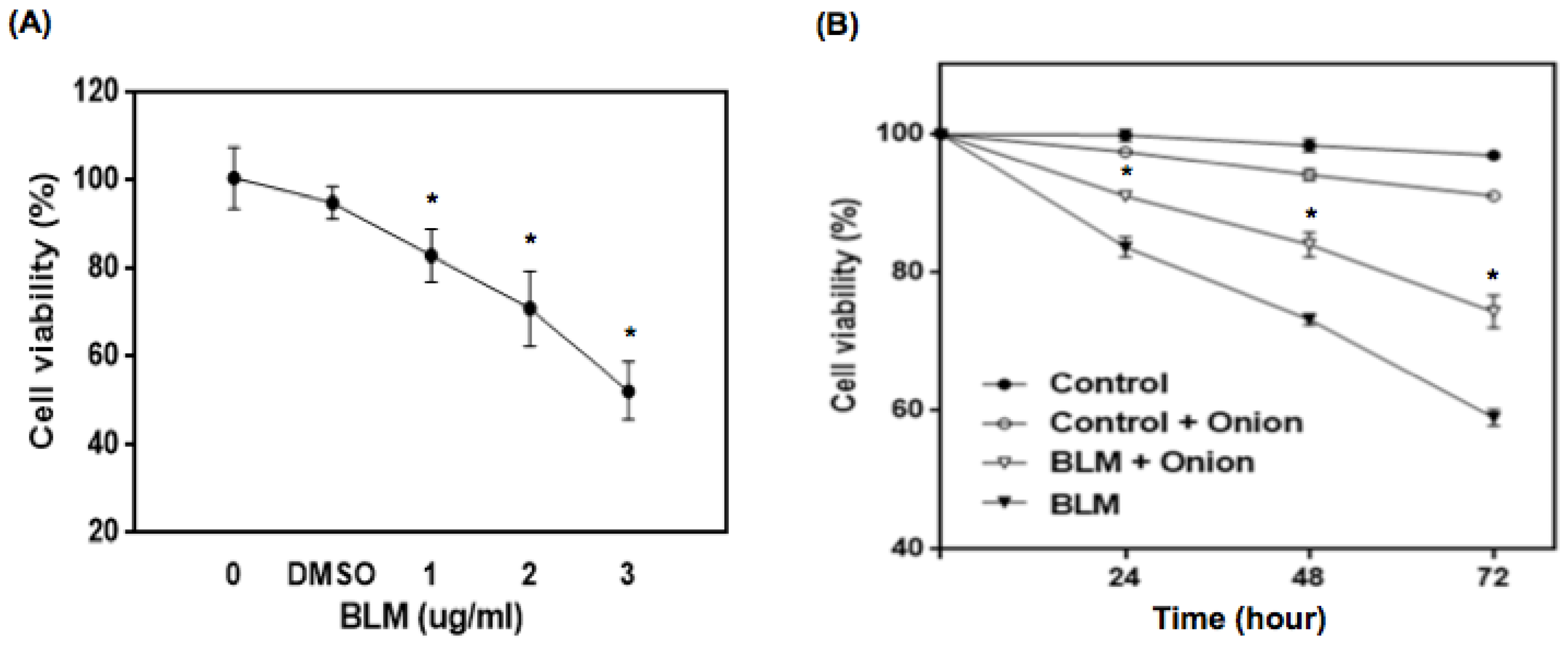
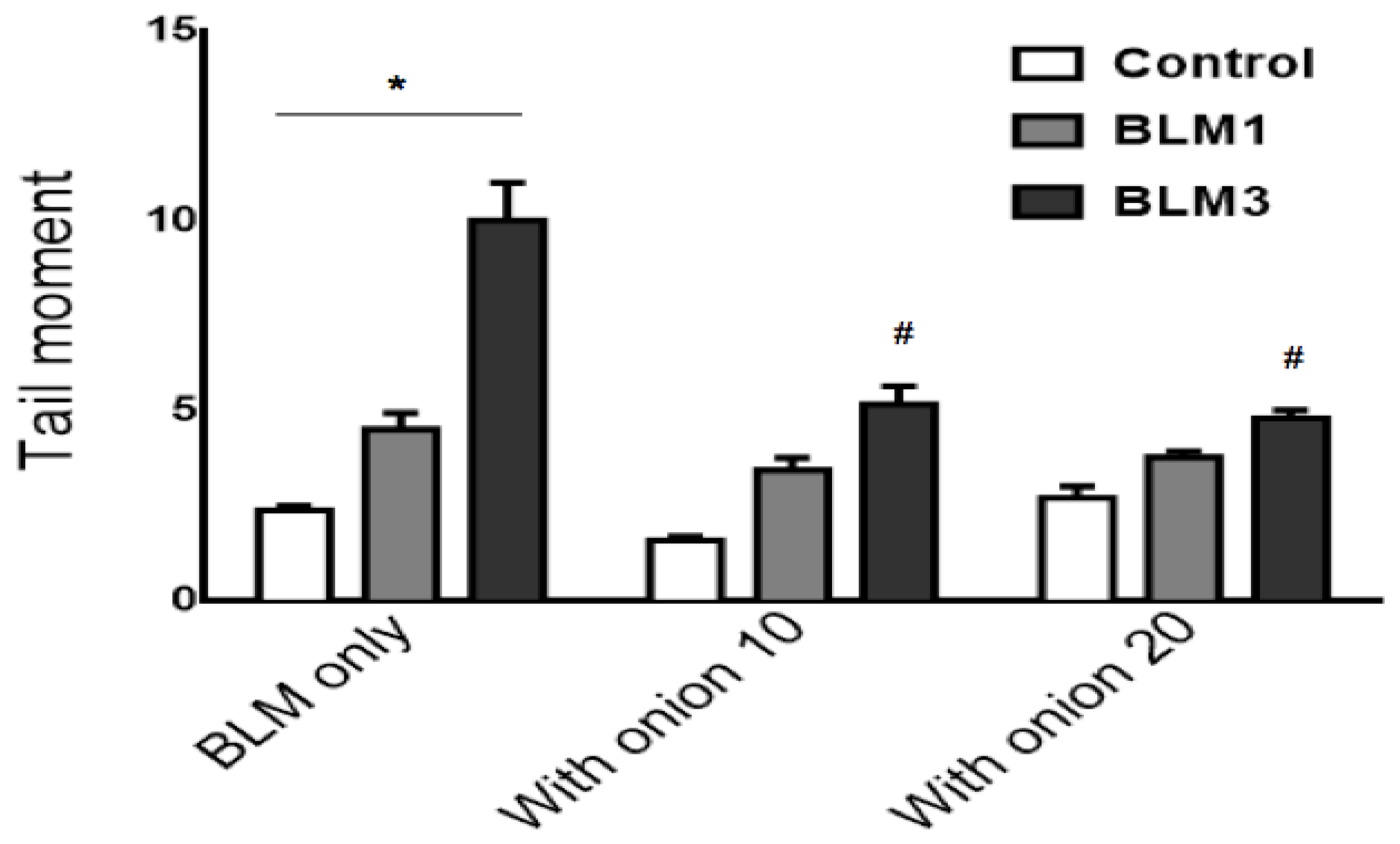
| Treatment | No. of MNCB a Cells/2000 BN Cells | Multi MNCB b/MNCB Cells (%) † | Total No. of MN/2000 BN Cells |
|---|---|---|---|
| Control | 17 ± 2.65 | 5.99 ± 0.01 | 18 ± 2.65 |
| Control + onion 10 c | 13 ± 2.52 | 7.80 ± 0.07 | 14 ± 3.61 |
| Control + onion 20 d | 14 ± 1.73 | 14.9 ± 0.09 | 16 ± 1.00 |
| BLM 1 e | 32 ± 6.56 | 12.8 ± 0.09 | 36 ± 6.24 |
| BLM 1 + onion 10 | 22 ± 4.36 | 13.4 ± 0.03 | 25 ± 5.29 |
| BLM 1 + onion 20 | 18 ± 2.65 # | 11.3 ± 0.02 | 20 ± 2.65 # |
| BLM 3 f | 46 ± 2.65 * | 27.5 ± 0.02 | 60 ± 4.36 * |
| BLM 3 + onion 10 | 33 ± 3.61 | 15.0 ± 0.02 | 38 ± 4.36 # |
| BLM 3 + onion 20 | 29 ± 2.65 # | 24.0 ± 0.04 | 37 ± 2.65 # |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cho, Y.H.; Lee, J.W.; Woo, H.D.; Lee, S.; Kim, Y.J.; Lee, Y.; Shin, S.; Joung, H.; Chung, H.W. Protective Effect of Onion Extract on Bleomycin-Induced Cytotoxicity and Genotoxicity in Human Lymphocytes. Int. J. Environ. Res. Public Health 2016, 13, 227. https://doi.org/10.3390/ijerph13020227
Cho YH, Lee JW, Woo HD, Lee S, Kim YJ, Lee Y, Shin S, Joung H, Chung HW. Protective Effect of Onion Extract on Bleomycin-Induced Cytotoxicity and Genotoxicity in Human Lymphocytes. International Journal of Environmental Research and Public Health. 2016; 13(2):227. https://doi.org/10.3390/ijerph13020227
Chicago/Turabian StyleCho, Yoon Hee, Joong Won Lee, Hae Dong Woo, Sunyeong Lee, Yang Jee Kim, Younghyun Lee, Sangah Shin, Hyojee Joung, and Hai Won Chung. 2016. "Protective Effect of Onion Extract on Bleomycin-Induced Cytotoxicity and Genotoxicity in Human Lymphocytes" International Journal of Environmental Research and Public Health 13, no. 2: 227. https://doi.org/10.3390/ijerph13020227
APA StyleCho, Y. H., Lee, J. W., Woo, H. D., Lee, S., Kim, Y. J., Lee, Y., Shin, S., Joung, H., & Chung, H. W. (2016). Protective Effect of Onion Extract on Bleomycin-Induced Cytotoxicity and Genotoxicity in Human Lymphocytes. International Journal of Environmental Research and Public Health, 13(2), 227. https://doi.org/10.3390/ijerph13020227







