Overview of Cotinine Cutoff Values for Smoking Status Classification
Abstract
:1. Introduction
2. Materials and Methods
3. Results
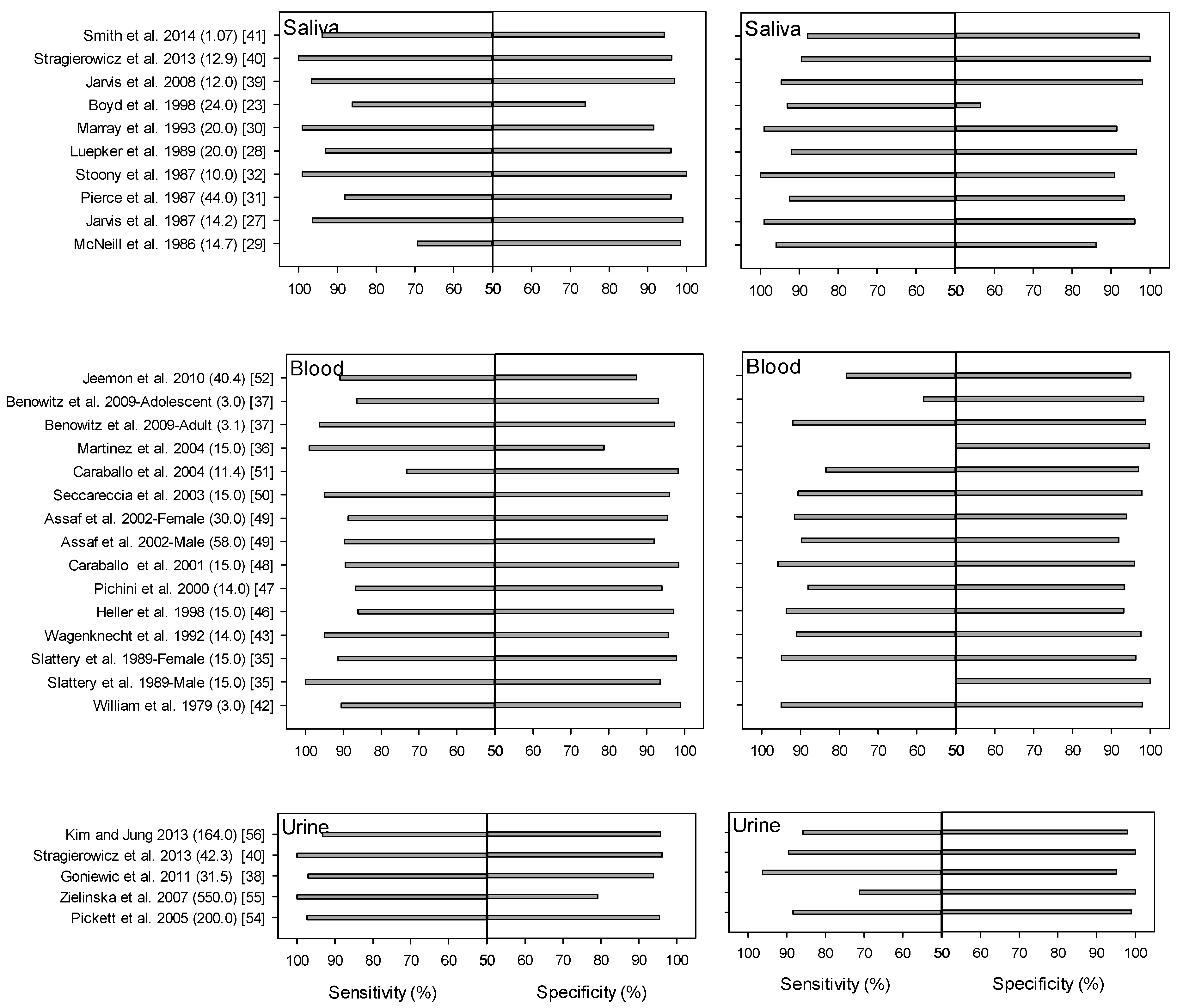
3.1. Salivary Cotinine
3.2. Serum Cotinine
3.3. Urinary Cotinine
4. Discussion
4.1. Issues in Determining Cutoffs
4.2. Drop in Cutoff Values over the Last 20 Years
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- International Agency for Research on Cancer IARC. Monographs on the Evaluation of Carcinogenic Risks to Humans: Tobacco Smoke and Involuntary Smoking; International Agency for Research on Cancer: Lyon, France, 1986; Volume 38. [Google Scholar]
- National Research Council. Environmental Tobacco Smoke: Measuring Exposures and Assessing Health Effects; National Academy Press: Washington, DC, USA, 1986. [Google Scholar]
- U.S. Department of Health and Human Services. The Health Consequences of Involuntary Smoking. A Report of the Surgeon General; (CDC) 87-8398; U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, Center for Health Promotion and Education, Office on Smoking and Health: Rockville, MD, USA, 1986. [Google Scholar]
- Warren, C.W.; Jones, N.R.; Eriksen, M.P.; Asma, S. Patterns of global tobacco use in young people and implications for future chronic disease burden in adults. Lancet 2006, 367, 749–753. [Google Scholar] [CrossRef]
- Kawachi, I.; Colditz, G.A. Invited commentary: Confounding, measurement error, and publication bias in studies of passive smoking. Am. J. Epidemiol. 1996, 144, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Wells, A.J.; English, P.B.; Posner, S.F.; Wagenknecht, L.E.; Perez-Stable, E.J. Misclassification rates for current smokers misclassified as nonsmokers. Am. J. Public Health 1998, 88, 1503–1509. [Google Scholar] [CrossRef] [PubMed]
- Benowitz, N.L. The use of biologic fluid samples in assessing tobacco smoke consumption. In Measurement in the Analysis and Treatment of Smoking Behavior; NIDA Research Monograph, No. 48; National Institute on Drug Abuse: Rockville, MD, USA, 1983. [Google Scholar]
- Benowitz, N.L. Biomarkers of environmental tobacco smoke exposure. Environ. Health Perspect. 1999, 107 (Suppl. S2), 349–355. [Google Scholar] [CrossRef] [PubMed]
- Coultas, D.B.; Howard, C.A.; Peake, G.T.; Skipper, B.J.; Samet, J.M. Salivary cotinine levels and involuntary tobacco smoke exposure in children and adults in New Mexico. Am. Rev. Respir. Dis. 1987, 136, 305–309. [Google Scholar] [CrossRef] [PubMed]
- DeLorenze, G.N.; Kharrazi, M.; Kaufman, F.L.; Eskenazi, B.; Bernert, J.T. Exposure to environmental tobacco smoke in pregnant women: The association between self-report and serum cotinine. Environ. Res. 2002, 90, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Eliopoulos, C.; Klein, J.; Koren, G. Validation of self-reported smoking by analysis of hair for nicotine and cotinine. Ther. Drug Monit. 1996, 18, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Etzel, R.A. A review of the use of saliva cotinine as a marker of tobacco smoke exposure. Prev. Med. 1990, 19, 190–197. [Google Scholar] [CrossRef]
- National Research Council. Biologic Markers of Pulmonay Toxicology; National Academy Press: Washington, DC, USA, 1989. [Google Scholar]
- Benowitz, N.L.; Jacob, P., III; Fong, I.; Gupta, S. Nicotine metabolic profile in man: Comparison of cigarette smoking and transdermal nicotine. J. Pharmacol. Exp. Ther. 1994, 268, 296–303. [Google Scholar] [PubMed]
- Benowitz, N.L.; Jacob, P., III. Metabolism of nicotine to cotinine studied by a dual stable isotope method. Clin. Pharmacol. Ther. 1994, 56, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Jacob, P.I.; Byrd, G.D. Use of gas chromatographic and mass spectrometric techniques for the determination of nicotine and its metabolites. In Analytical Determination of Nicotine and Related Compounds and Their Metabolites; Gorrod, J.W., Jacob, P.I., Eds.; Elsevier Science: Amsterdam, The Netherlands, 1999; pp. 191–224. [Google Scholar]
- Benner, C.L.; Bayona, J.M.; Caka, F.M.; Tang, H.; Kewis, L.; Crawford, J.; Lamb, J.D.; Lee, M.L.; Lewis, E.A.; Hansen, L.D.; et al. Chemical composition of environmental tobacco smoke. 2. Particulate-phase compounds. Environ. Sci. Technol. 1986, 23, 688–699. [Google Scholar] [CrossRef]
- Ogden, M.W.; Maiolo, K.C.; Nelson, P.R.; Heavner, D.L.; Green, C.R. Artifacts in determining the vapor-particulate phase distribution of environmental tobacco smoke nicotine. Environ. Technol. 1993, 14, 779–785. [Google Scholar] [CrossRef]
- Benowitz, N.L. Cotinine as a biomarker of environmental tobacco smoke exposure. Epidemiol. Rev. 1996, 18, 188–204. [Google Scholar] [CrossRef] [PubMed]
- Hammond, S.K.; Leaderer, B.P. A diffusion monitor to measure exposure to passsive smoking. Environ. Sci. Technol. 1987, 21, 494–497. [Google Scholar] [CrossRef] [PubMed]
- Jaakkola, M.S.; Jaakkola, J.J. Assessment of exposure to environmental tobacco smoke. Eur. Respir. J. 1997, 10, 2384–2397. [Google Scholar] [CrossRef] [PubMed]
- Flegal, K.M.; Brownie, C.; Haas, J.D. The effects of exposure misclassification on estimates of relative risk. Am. J. Epidemiol. 1986, 123, 736–751. [Google Scholar] [PubMed]
- Boyd, N.R.; Windsor, R.A.; Perkins, L.L.; Lowe, J.B. Quality of measurement of smoking status by self-report and saliva cotinine among pregnant women. Matern. Child Health J. 1998, 2, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Etter, J.F.; Vu, D.T.; Perneger, T.V. Saliva cotinine levels in smokers and nonsmokers. Am. J. Epidemiol. 2000, 151, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Hegaard, H.K.; Kjaergaard, H.; Moller, L.F.; Wachmann, H.; Ottesen, B. Determination of a saliva cotinine cut-off to distinguish pregnant smokers from pregnant non-smokers. Acta Obstet. Gynecol. Scand. 2007, 86, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, M.; Tunstall-Pedoe, H.; Feyerabend, C.; Vesey, C.; Salloojee, Y. Biochemical markers of smoke absorption and self reported exposure to passive smoking. J. Epidemiol. Community Health 1984, 38, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, M.J.; Tunstall-Pedoe, H.; Feyerabend, C.; Vesey, C.; Saloojee, Y. Comparison of tests used to distinguish smokers from nonsmokers. Am. J. Public Health 1987, 77, 1435–1438. [Google Scholar] [CrossRef] [PubMed]
- Luepker, R.V.; Pallonen, U.E.; Murray, D.M.; Pirie, P.L. Validity of telephone surveys in assessing cigarette smoking in young adults. Am. J. Public Health 1989, 79, 202–204. [Google Scholar] [CrossRef] [PubMed]
- McNeill, A.D.; West, R.J.; Jarvis, M.; Jackson, P.; Bryant, A. Cigarette withdrawal symptoms in adolescent smokers. Psychopharmacology 1986, 90, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Murray, R.P.; Connett, J.E.; Lauger, G.G.; Voelker, H.T. Error in smoking measures: Effects of intervention on relations of cotinine and carbon monoxide to self-reported smoking. The Lung Health Study Research Group. Am. J. Public Health 1993, 83, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Pierce, J.P.; Dwyer, T.; DiGiusto, E.; Carpenter, T.; Hannam, C.; Amin, A.; Yong, C.; Sarfaty, G.; Shaw, J.; Burke, N. Cotinine validation of self-reported smoking in commercially run community surveys. J. Chronic Dis. 1987, 40, 689–695. [Google Scholar] [CrossRef]
- Stookey, G.K.; Katz, B.P.; Olson, B.L.; Drook, C.A.; Cohen, S.J. Evaluation of biochemical validation measures in determination of smoking status. J. Dent. Res. 1987, 66, 1597–1601. [Google Scholar] [CrossRef] [PubMed]
- SRNT Subcommittee on Biochemical Verification. Biochemical verification of tobacco use and cessation. Nicotine Tob. Res. 2002, 4, 149–159. [Google Scholar]
- Hoffmann, D.; Haley, N.J.; Adams, J.D.; Brunnemann, K.D. Tobacco sidestream smoke: Uptake by nonsmokers. Prev. Med. 1984, 13, 608–617. [Google Scholar] [CrossRef]
- Slattery, M.L.; Hunt, S.C.; French, T.K.; Ford, M.H.; Williams, R.R. Validity of cigarette smoking habits in three epidemiologic studies in Utah. Prev. Med. 1989, 18, 11–19. [Google Scholar] [CrossRef]
- Martinez, M.E.; Reid, M.; Jiang, R.; Einspahr, J.; Alberts, D.S. Accuracy of self-reported smoking status among participants in a chemoprevention trial. Prev. Med. 2004, 38, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Benowitz, N.L.; Bernert, J.T.; Caraballo, R.S.; Holiday, D.B.; Wang, J. Optimal serum cotinine levels for distinguishing cigarette smokers and nonsmokers within different racial/ethnic groups in the United States between 1999 and 2004. Am. J. Epidemiol. 2009, 169, 236–248. [Google Scholar] [CrossRef] [PubMed]
- Goniewicz, M.L.; Eisner, M.D.; Lazcano-Ponce, E.; Zielinska-Danch, W.; Koszowski, B.; Sobczak, A.; Havel, C.; Jacob, P.; Benowitz, N.L. Comparison of urine cotinine and the tobacco-specific nitrosamine metabolite 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) and their ratio to discriminate active from passive smoking. Nicotine Tob. Res. 2011, 13, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, M.J.; Fidler, J.; Mindell, J.; Feyerabend, C.; West, R. Assessing smoking status in children, adolescents and adults: Cotinine cut-points revisited. Addiction 2008, 103, 1553–1561. [Google Scholar] [CrossRef] [PubMed]
- Stragierowicz, J.; Mikołajewska, K.; Zawadzka-Stolarz, M.; Polańska, K.; Ligocka, D. Estimation of cutoff values of cotinine in urine and saliva for pregnant women in Poland. Biomed Res. Int. 2013, 2013, 386784. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.J.; Robinson, R.F.; Khan, B.A.; Sosnoff, C.S.; Dillard, D.A. Estimating cotinine associations and a saliva cotinine level to identify active cigarette smoking in Alaska native pregnant women. Matern. Child Health J. 2014, 18, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.L.; Eng, A.; Botvin, G.J.; Hill, P.; Wynder, E.L. Validation of students’ self-reported cigarette smoking status with plasma cotinine levels. Am. J. Public Health 1979, 69, 1272–1274. [Google Scholar] [CrossRef] [PubMed]
- Wagenknecht, L.E.; Burke, G.L.; Perkins, L.L.; Haley, N.J.; Friedman, G.D. Misclassification of smoking status in the CARDIA study: A comparison of self-report with serum cotinine levels. Am. J. Public Health 1992, 82, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Pirkle, J.L.; Flegal, K.M.; Bernert, J.T.; Brody, D.J.; Etzel, R.A.; Maurer, K.R. Exposure of the U.S. population to environmental tobacco smoke: The Third National Health and Nutrition Examination Survey, 1988 to 1991. JAMA 1996, 275, 1233–1240. [Google Scholar] [CrossRef] [PubMed]
- Nafstad, P.; Kongerud, J.; Botten, G.; Urdal, P.; Silsand, T.; Pedersen, B.S.; Jaakkola, J.J. Fetal exposure to tobacco smoke products: A comparison between self-reported maternal smoking and concentrations of cotinine and thiocyanate in cord serum. Acta Obstet. Gynecol. Scand. 1996, 75, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Heller, W.D.; Scherer, G.; Sennewald, E.; Adlkofer, F. Misclassification of smoking in a follow-up population study in southern Germany. J. Clin. Epidemiol. 1998, 51, 211–218. [Google Scholar] [CrossRef]
- Pichini, S.; Basagana, X.B.; Pacifici, R.; Garcia, O.; Puig, C.; Vall, O.; Harris, J.; Zuccaro, P.; Segura, J.; Sunyer, J. Cord serum cotinine as a biomarker of fetal exposure to cigarette smoke at the end of pregnancy. Environ. Health Perspect. 2000, 108, 1079–1083. [Google Scholar] [CrossRef] [PubMed]
- Caraballo, R.S.; Giovino, G.A.; Pechacek, T.F.; Mowery, P.D. Factors associated with discrepancies between self-reports on cigarette smoking and measured serum cotinine levels among persons aged 17 years or older: Third National Health and Nutrition Examination Survey, 1988–1994. Am. J. Epidemiol. 2001, 153, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Assaf, A.R.; Parker, D.; Lapane, K.L.; McKenney, L., Jr.; Carleton, R.A. Are there gender differences in self-reported smoking practices? Correlation with thiocyanate and cotinine levels in smokers and nonsmokers from the Pawtucket Heart Health Program. J. Women’s Health 2002, 11, 899–906. [Google Scholar] [CrossRef]
- Seccareccia, F.; Zuccaro, P.; Pacifici, R.; Meli, P.; Pannozzo, F.; Freeman, K.M.; Santaquilani, A.; Giampaoli, S. Serum cotinine as a marker of environmental tobacco smoke exposure in epidemiological studies: The experience of the MATISS project. Eur. J. Epidemiol. 2003, 18, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Caraballo, R.S.; Giovino, G.A.; Pechacek, T.F. Self-reported cigarette smoking vs. serum cotinine among U.S. adolescents. Nicotine Tob. Res. 2004, 6, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Jeemon, P.; Agarwal, S.; Ramakrishnan, L.; Gupta, R.; Snehi, U.; Chaturvedi, V.; Reddy, K.S.; Prabhakaran, D. Validation of self-reported smoking status by measuring serum cotinine levels: An Indian perspective. Natl. Med. J. India 2010, 23, 134–136. [Google Scholar] [PubMed]
- Riboli, E.; Preston-Martin, S.; Saracci, R.; Haley, N.J.; Trichopoulos, D.; Becher, H.; Burch, J.D.; Fontham, E.T.; Gao, Y.T.; Jindal, S.K. Exposure of nonsmoking women to environmental tobacco smoke: A 10-country collaborative study. Cancer Causes Control 1990, 1, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Pickett, K.E.; Rathouz, P.J.; Kasza, K.; Wakschlag, L.S.; Wright, R. Self-reported smoking, cotinine levels, and patterns of smoking in pregnancy. Paediatr. Perinat. Epidemiol. 2005, 19, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Zielinska-Danch, W.; Wardas, W.; Sobczak, A.; Szoltysek-Boldys, I. Estimation of urinary cotinine cut-off points distinguishing non-smokers, passive and active smokers. Biomarkers 2007, 12, 484–496. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jung, A. Optimum cutoff value of urinary cotinine distinguishing South Korean adult smokers from nonsmokers using data from the KNHANES (2008–2010). Nicotine Tob. Res. 2013, 15, 1608–1616. [Google Scholar] [CrossRef] [PubMed]
- Frendrich, M.; Mackesy-Amiti, M.E.; Johnson, T.P.; Hubbell, A.; Wislar, J.S. Tobacco-reporting validity in an epidemiological drug-use survey. Addict. Behav. 2005, 30, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Perez-Stable, E.J.; Marin, B.V.; Marin, G.; Brody, D.J.; Benowitz, N.L. Apparent underreporting of cigarette consumption among Mexican American smokers. Am. J. Public Health 1990, 80, 1057–1061. [Google Scholar] [CrossRef] [PubMed]
- Rebagliato, M.; Bolumar, F.; Florey, C.V.; Jarvis, M.J.; Perez-Hoyos, S.; Hernandez-Aguado, I.; Avino, M.J. Variations in cotinine levels in smokers during and after pregnancy. Am. J. Obstet. Gynecol. 1998, 178, 568–571. [Google Scholar] [CrossRef]
- World Health Organization. WHO Report on the Global Tobacco Epidemic, 2008: The MPOWER Package; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Stoner, W.I.; Foley, B.X. Current tobacco control policy trends in the United States. Clin. Occup. Environ. Med. 2006, 5, 85–99. [Google Scholar] [PubMed]
- Protano, C.; Vitali, M. The new danger of thirdhand smoke: Why passive smoking does not stop at secondhand smoke. Environ. Health Perspect. 2011, 119, A422. [Google Scholar] [CrossRef] [PubMed]
| Author | Year | Ref. | Study Population Characteristics | Cutoff Value (ng/mL) | Number of Self-Reported | Method for Cutoff Determination | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Description | N | Age | Female (%) | Smokers | Non-Smokers | |||||
| McNeill et al. | 1986 | [29] | Students attending a girls’ comprehensive school in London, UK | 508 | 11–16 years | 100 | 14.7 | 173 | 335 | 2 × 2 table |
| Jarvis et al. | 1987 | [27] | Outpatients at St. Mary’s Hospital, London, UK | 211 | Mean age: 55 years | 24.6 | 14.2 | 111 | 100 | 2 × 2 table: The cutoff value providing the highest numbers of correctly classified smokers and nonsmokers |
| Pierce et al. | 1987 | [31] | Residents randomly selected in a community, Australia | 975 | 14 years or older | 49.2 | 44 | 353 | 622 | 2 × 2 table |
| Stookey et al. | 1987 | [32] | Participants in a clinical trial for evaluating measures to facilitate smoking cessation, USA | 236 | NA | NA | 10 | 216 | 20 | Cutoff value was adopted from the study results of Benowitz (1983) [7]: “there was no nonsmoker with blood cotinine values greater than 10 ng/mL” (page 21) |
| Luepker et al. | 1989 | [28] | High school students randomly selected for survey in Minneapolis, MN, USA | 263 | 17–21 years | NA | 20 | 87 | 176 | 2 × 2 table |
| Etzel * | 1990 | [12] | Participants in 22 studies published between 1973 and 1989 | NA | NA | NA | 10 | NA | NA | Cutoff value was selected after comparisons of the distributions of salivary cotinine concentrations with respect to smoking status between 22 study papers published between 1973 and 1989 |
| Murray et al. | 1993 | [30] | Participants under routine care in a clinical trial of “Lung Health Study” in USA and Canada with evidence of early stage chronic obstructive lung disease | 1498 | 35–60 years (mean: 48.5 years) | 36 | 20 | 1345 | 153 | Cutoff value was selected as the salivary cotinine level that provided the highest percentage of correctly classified smoking status |
| Boyd et al. | 1998 | [23] | Pregnant women in the Birmingham Trial II conducted at four public health maternity clinics in Birmingham, AL, USA | 548 | Mean: 24.6 years | 100 | 24 | 441 | 107 | ROC (receiver operating characteristics curve): The cutoff value providing the greatest percentage of correctly classified smoking status |
| Jarvis et al. | 2008 | [39] | Participants in the Health Survey for England: 1996–2004 | 24,332 | All adults | NA | 12 | 8808 | 15,524 | ROC |
| Stragierowicz et al. | 2013 | [40] | Pregnant women in Polish Mother and Child Cohort Study | 69 | Mean: 26.4 years | 100 | 12.9 | 19 | 50 | ROC |
| Smith et al. | 2014 | [41] | Pregnant women in Southcentral Foundation’s Anchorage Primary Health Care Center: 2006–2010 | 376 | Mean: 26.9 years | 100 | 1.07 | 116 | 260 | ROC |
| Author | Year | Ref. | Study Population Characteristics | Cutoff Value (ng/mL) | Number of Self-Reported | Method for Cutoff Determination | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Description | N | Age | Female (%) | Smokers | Non-Smokers | |||||
| Williams et al. | 1979 | [42] | High school students participating in a school health education program. Blinded for purpose of blood collection in USA | 118 | 14–17 years | 53 | 3.0 | 21 | 97 | 2 × 2 table |
| Benowitz | 1983 | [7] | Participants in a test evaluating an elimination half-life for cotinine. They stopped smoking in a research ward (Average, 19.1 h; range, 10.9 to 37.0 h) | 16 | NA | NA | 10 | NA | NA | Authors selected the cutoff value from a range of concentrations among nonsmokers, and reported that “no nonsmoker had blood cotinine values higher than 10 ng/mL” |
| Slattery et al. | 1989 | [35] | Participants in (1) a cross-sectional study on dietary intake and hormone; | (1): 112 | 17 years or older | (1): male only | 15 | (1): 3 | (1): 109 | Authors selected the cutoff value by calculating 6% of the mean serum cotinine levels in smokers in the study |
| (2) a case control study of squamous cell cervical cancer in Utah, USA | (2): 547 | (2): female only | (2): 163 | (2): 379 | ||||||
| Wagenknecht et al. | 1992 | [43] | Young adults in a cohort of cardiovascular disease study in USA | 4984 | 17–30 years | NA | 14 | 1540 | 3444 | ROC |
| Pirkle et al. * | 1996 | [44] | Participants in the third National Health and Nutrition Examination Survey (NHANES), USA | 10,270 | 4 years or older | 50 | 15 | NA | NA | Authors selected the cutoff value from a separation point in the bimodal distribution of serum cotinine in tobacco users and nonusers |
| Nafstad et al. * | 1996 | [45] | Pregnant women in the Oslo Birth Cohort, Norway | 202 | Mean: 30 years (Range: 19–43) | 100 | 14 | 42 regular + 24 occasional | 136 | The traditionally used cutoff value (14 ng/mL) was chosen at the authors’ discretion |
| Heller et al. | 1998 | [46] | Followers among participants in the WHO MONICA (Monitoring trends and determinants in cardiovascular disease) project in 1987–1988, Germany | 3661 | TBA | 50.9 | 15 | 1227 | 2434 | Cutoff value was adopted from the study results of Wagenknecht et al., 1992 [43] |
| Pichini et al. | 2000 | [47] | Pregnant women attending the Hospital del Mar in Barcelona, Spain | 404 | TBA | 100 | 14 | 136 | 268 | Cutoff value was adopted from the study results of Nafstad et al., 1996 [42] |
| Caraballo et al. | 2001 | [48] | Adults in the third National Health and Nutrition Examination Survey (NHANES) 1988–1994, USA | 15,357 | 17 years or older | 53.8 | 15 | 4274 | 11,083 | Cutoff value was adopted from the study results of Pirkle et al., 1996 [44] |
| Assaf et al. | 2002 | [49] | Adults those who conducted cotinine tests in Pawtucket Heart Health Program 1985–1986, USA | 784 | 18–65 years | 57.5 | 58 for male (M) | 131 (M) | 172 (M) | ROC |
| 30 for female (F) | 141 (F) | 279 (F) | ||||||||
| Seccareccia et al. | 2003 | [50] | Providers of serum samples among participants in the project of MATISS (Malattie cardiovascular Aterosclerotiche, Istituto Superiore di Sanità), Italy | 3379 | 20–79 years | 39.5 | 15 | 977 | 2402 | ROC |
| Caraballo et al. | 2004 | [51] | Adolescents in the third National Health and Nutrition Examination Survey (NHANES) 1988–1994, USA | 2107 | 12–17 years | 53.8 | 11.4 | 213 | 1894 | ROC |
| Martinez et al. | 2004 | [36] | Participants in a dietary trial on adenoma recurrence, Phoenix, AZ, USA | 824 | 40–80 years | 31 | 20 | 95 | 729 | 2 × 2 table |
| Benowitz et al. | 2009 | [37] | Participants in the National Health and Nutrition Examination Survey (NHANES) for 1999–2004, USA | 9901 for adults, | 20 years or older | 50.6 for adults | 3.08 for adults | 2340 | 7561 | ROC |
| 5138 for adolescents | 12–19 years | 49.6 for adolescents | 2.99 for adolescents | 515 | 4623 | |||||
| Jeemon et al. | 2010 | [52] | Participants in the cardiovascular disease surveillance program at New Delhi, India | 426 | 18 years or older | TBA | 40.35 | 142 | 284 | ROC |
| Author | Year | Ref. | Study Population Characteristics | Cutoff Value (ng/mL) | Number of Self-Reported | Method for Cutoff Determination | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Description | N | Age | Female (%) | Smokers | Non-Smokers | |||||
| Hoffmann et al. * | 1984 | [34] | Volunteers joined a study on uptake of sidestream smoke | NA | NA | NA | 55 | NA | NA | The value was obtained from urine samples that were collected when saliva nicotine levels returned to baseline levels (i.e., 5 h after study subjects were exposed to passive smoke in closed chamber (280 mg/m3 for air nicotine concentration) for 1 h) |
| Riboli et al. * | 1990 | [53] | Married nonsmoking women from 10 countries | 1369 | Age: 42–60 years | NA | 50 | NA | NA | Cutoff value was chosen as the value that provided 3.4% misclassification. It was also compared with the study results of Hoffmann et al. (1984) [34] |
| Pickett et al. | 2005 | [54] | Pregnant women attending the East Boston neighborhood health clinic, USA between 1986 and 1992 with allowance of multiple visits | 998 | 19 years or more | NA | 200 | 1272 | 3566 | ROC |
| Zielinska-Danch | 2007 | [55] | Volunteers living in Sosnowiec, Poland | 327 | 19–60 | 57.2 | 550 | 111 | 216 | Authors selected the cutoff value from a separation point in the bimodal distribution of urine cotinine in self-reported smokers and nonsmokers |
| Goniewicz et al. | 2011 | [38] | Smokers from three different studies conducted in; San Francisco, CA, USA, Silesia, Poland, and Pittsburgh, PA, USA Nonsmokers from the other three studies conducted in USA, Poland, and Mexico | 601 | 18 years or older | 52.7 | 31.5 | 373 | 228: passive smokers only | ROC |
| Stragierowicz et al. | 2013 | [40] | Pregnant women in Polish Mother and Child Cohort Study | 69 | Mean: 26.4 years | 100 | 53.0 | 17 | 52 | ROC |
| Kim and Jung | 2013 | [56] | Participants in Korea National Health and Nutrition Examination Survey (KNHANES) for 2008–2010, Korea | 11,629 | 19 years or older | 55.5 | 164 | 2547 | 9082 | ROC |
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S. Overview of Cotinine Cutoff Values for Smoking Status Classification. Int. J. Environ. Res. Public Health 2016, 13, 1236. https://doi.org/10.3390/ijerph13121236
Kim S. Overview of Cotinine Cutoff Values for Smoking Status Classification. International Journal of Environmental Research and Public Health. 2016; 13(12):1236. https://doi.org/10.3390/ijerph13121236
Chicago/Turabian StyleKim, Sungroul. 2016. "Overview of Cotinine Cutoff Values for Smoking Status Classification" International Journal of Environmental Research and Public Health 13, no. 12: 1236. https://doi.org/10.3390/ijerph13121236
APA StyleKim, S. (2016). Overview of Cotinine Cutoff Values for Smoking Status Classification. International Journal of Environmental Research and Public Health, 13(12), 1236. https://doi.org/10.3390/ijerph13121236





