Sun Exposure and Its Effects on Human Health: Mechanisms through Which Sun Exposure Could Reduce the Risk of Developing Obesity and Cardiometabolic Dysfunction
Abstract
:1. Introduction
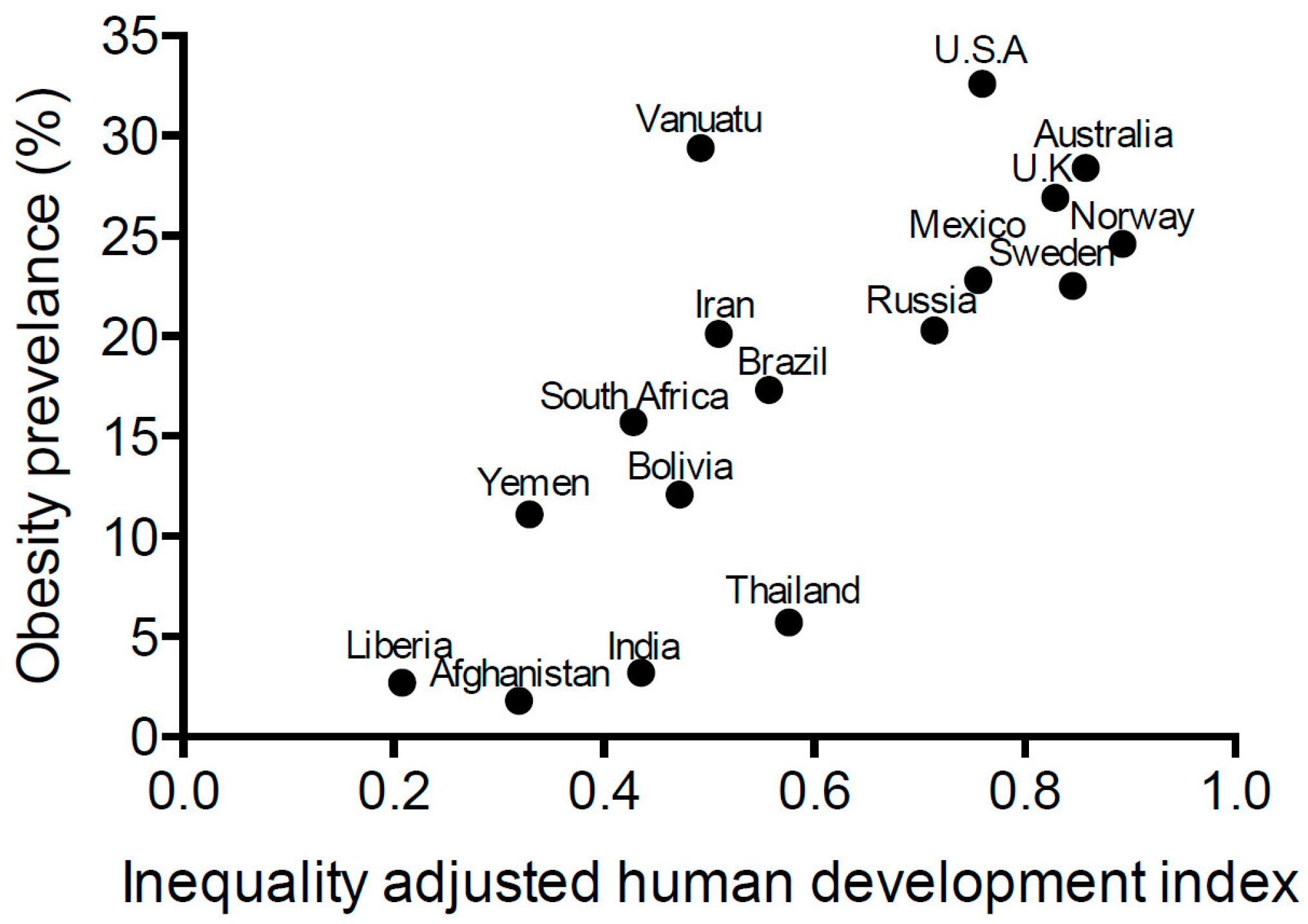
2. Diagnosis of Obesity
3. Metabolic Syndrome
4. Type-2 Diabetes
5. Non-Alcoholic Fatty Liver Disease (NAFLD)
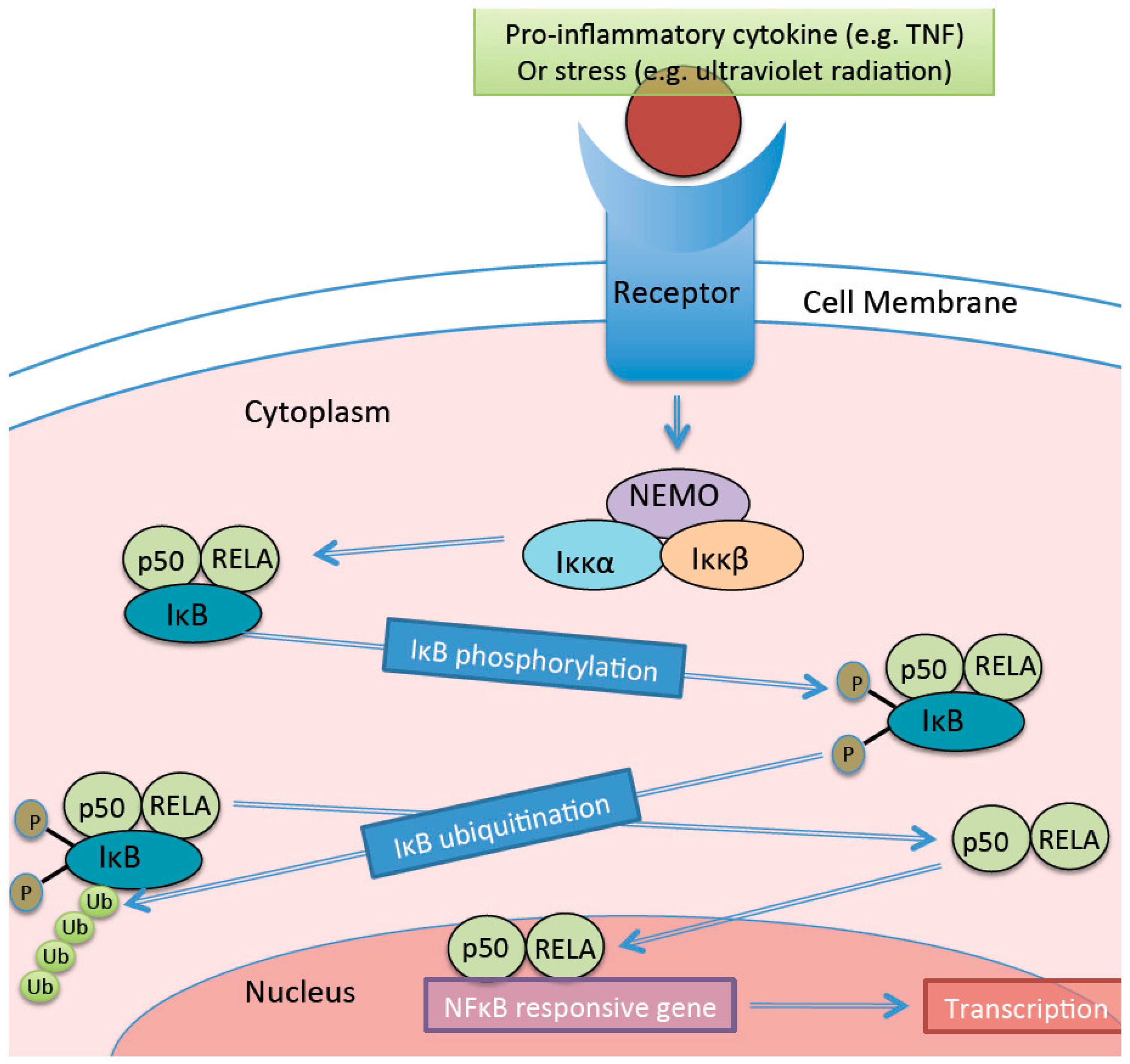
6. Chronic Low-Grade Inflammation Is Associated with Obesity and Cardiometabolic Disease
7. Sunlight and Ultraviolet Radiation
7.1. Negative Consequences of UVR Exposure
7.2. Health Benefits of Sun Exposure
7.3. Autoimmune and Allergic Disease
7.4. UVR Modulation of Obesity and Related Metabolic Disorders
7.4.1. Animal Studies
7.4.2. Human Studies
Human Observational Studies
Human Experimental Studies
7.5. Pathways through Which UVR Could Suppress Obesity and Signs of Cardiometabolic Dysfunction
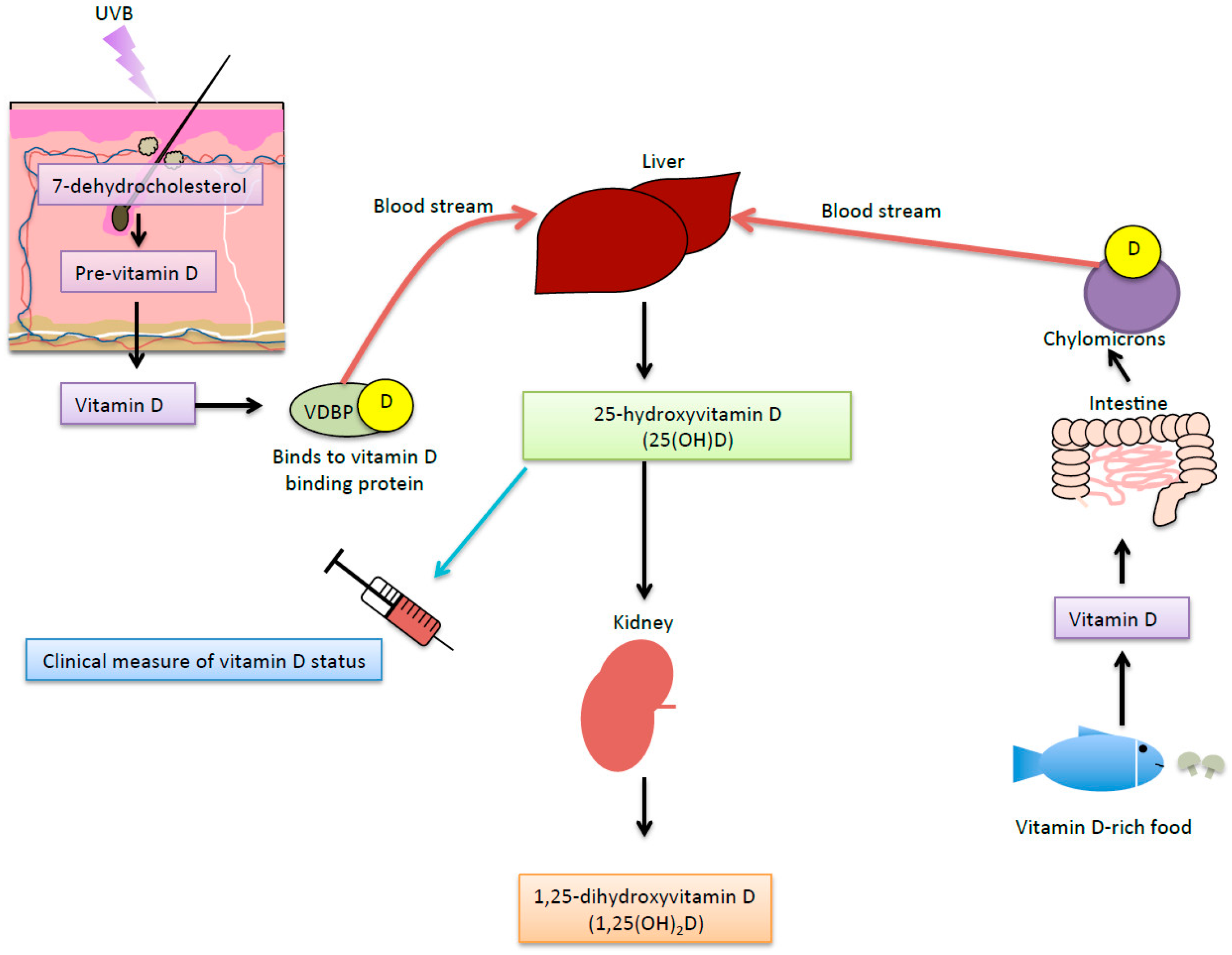
7.5.1. UVR-Induced Vitamin D
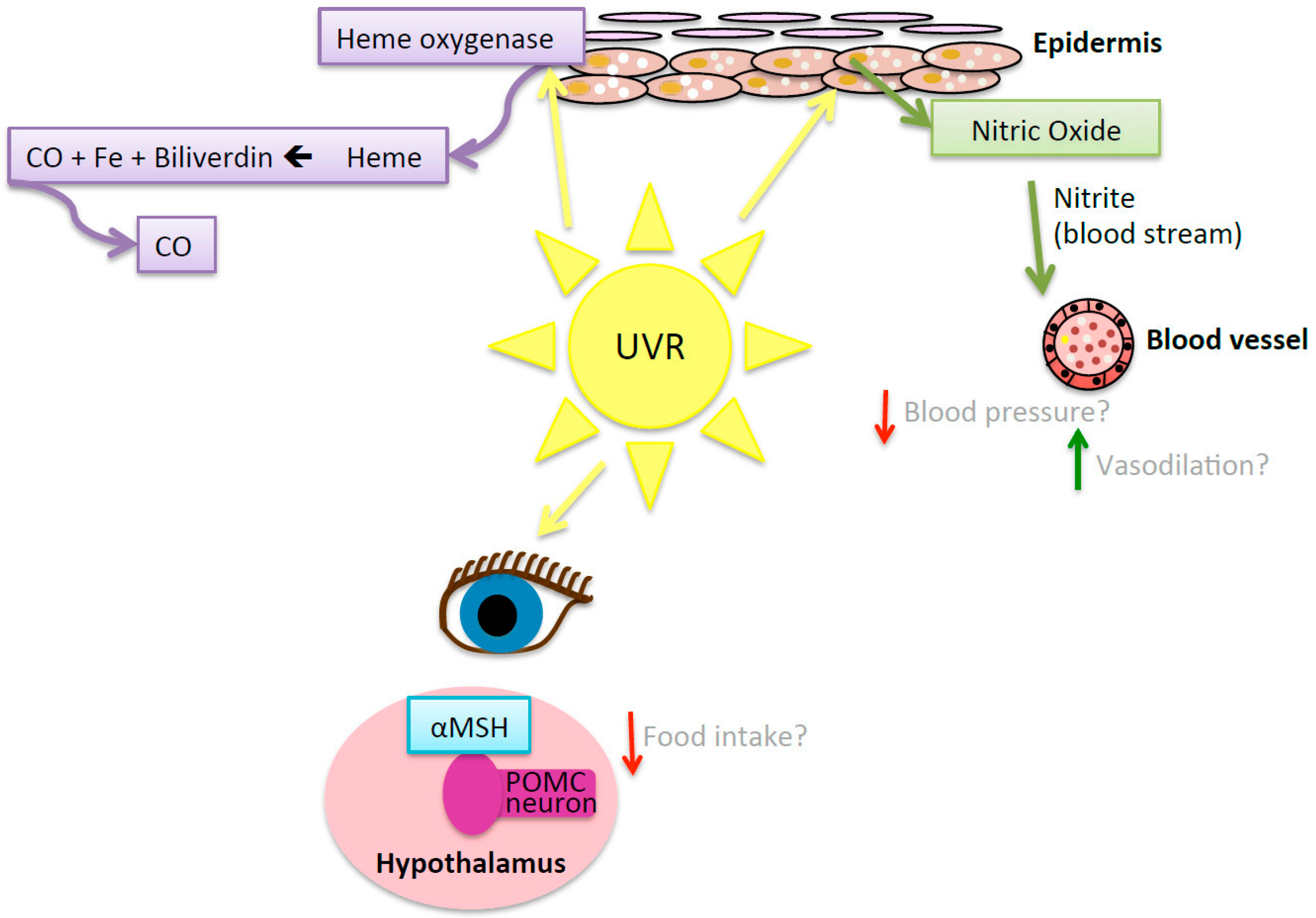
7.5.2. UVR-Induced Nitric Oxide
7.5.3. Pro-Opiomelanocortin Pathway
7.5.4. Heme-Oxygenase
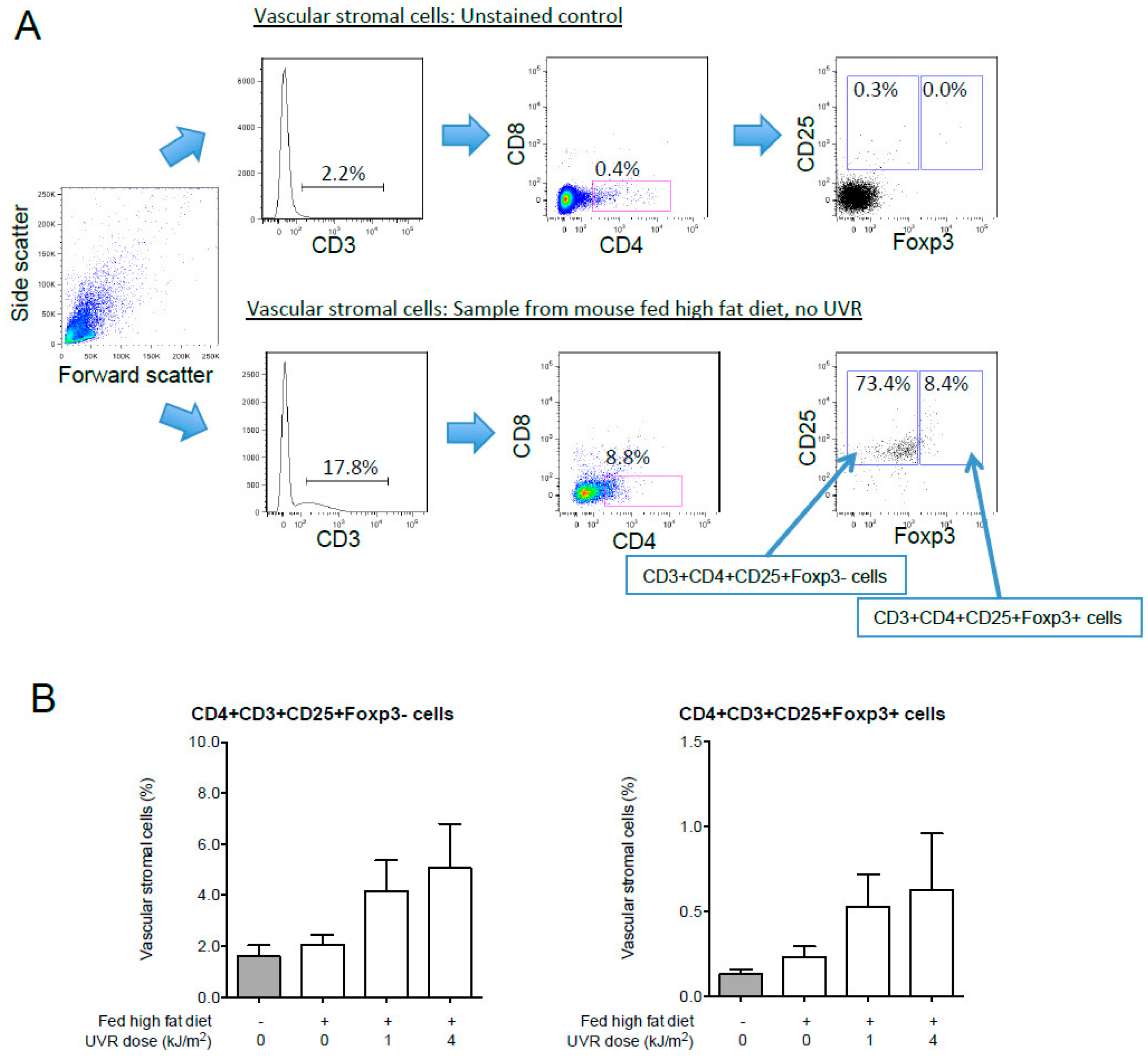
7.5.5. UVR-Induced Regulatory T Cells
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- NCD Risk Factor Collaboration (NCD-RisC). Trends in adult body-mass index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet 2016, 387, 1377–1396. [Google Scholar]
- Finucane, M.M.; Stevens, G.A.; Cowan, M.J.; Danaei, G.; Lin, J.K.; Paciorek, C.J.; Singh, G.M.; Gutierrez, H.R.; Lu, Y.; Bahalim, A.N.; et al. National, regional, and global trends in body-mass index since 1980: Systemic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet 2011, 377, 557–567. [Google Scholar] [CrossRef]
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Gakidou, E. Global, regional and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the global burden of disease study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef]
- Jahan, S. Human Development Report 2015—Work for Human Development; United Nations Development Programme: New York, NY, USA, 2015. [Google Scholar]
- Jahan, S. International Human Development Indicators. Available online: http://hdr.undp.org/en/countries (accessed on 4 May 2016).
- World Health Organization (WHO). Prevalence of Obesity, Ages 18+, 2010–2014 (Age Standardized Estimate). Available online: http://gamapserver.who.int/gho/interactive_charts/ncd/risk_factors/obesity/atlas.html (accessed on 4 May 2016).
- Rothman, K.J. Bmi-related errors in the measurement of obesity. Int. J. Obes. 2008, 32 (Suppl. S3), S56–S59. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Ford, E.S.; Li, C.; Tsai, J.; Dhingra, S.; Balluz, L.S. Waist circumference, abdominal obesity, and depression among overweight and obese U.S. Adults: National health and nutrition examination survey 2005–2006. BMC Psychiatry 2011, 11, 130. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ford, E.S.; McGuire, L.C.; Mokdad, A.H. Increasing trends in waist circumference and abdominal obesity among US adults. Obesity 2007, 15, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.; Allison, D.B.; Heymsfield, S.B.; Kelley, D.E.; Leibel, R.L.; Nonas, C.; Kahn, R. Waist circumference and cardiometabolic risk: A consensus statement from shaping America’s health: Association for weight management and obesity prevention; NASSO, the obesity society; the American society for nutrition and the American diabetes association. Obesity 2007, 15, 1061–1067. [Google Scholar] [PubMed]
- Erselcan, T.; Candan, F.; Saruhan, S.; Ayca, T. Comparison of body composition analysis methods in clinical routine. Ann. Nutr. Metab. 2000, 44, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Minderico, C.S.; Silva, A.M.; Teixeira, P.J.; Sardinha, L.B.; Hull, H.R.; Fields, D.A. Validity of air-displacement plethysmography in the assessment of body composition changes in a 16-month weight loss program. Nutr. Metab. 2006, 3, 32. [Google Scholar] [CrossRef] [PubMed]
- Larkin, A.; Sheahan, N.; O’Connor, U.; Gray, L.; Dowling, A.; Vano, E.; Torbica, P.; Salat, D.; Schreiner, A.; Neofotistou, V.; et al. QA/acceptance testing of DEXA X-ray systems used in bone mineral densitometry. Radiat. Prot. Dosim. 2008, 129, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Metabolic syndrome pandemic. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Lepor, N.E.; Vogel, R.E.; National Cholesterol Education Program Adult Treatment Panel III. Summary of the third report of the national cholesterol education program adult treatment panel III. Rev. Cardiovasc. Med. 2001, 2, 160–165. [Google Scholar] [PubMed]
- Alberti, K.G.; Zimmet, P.; Shaw, J. Metabolic syndrome—A new world-wide definition. A consensus statement from the international diabetes federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Definition, Diagnosis and Classification of Diabetes Mellitus and Its Complications; WHO: Geneva, Switzerland, 1999. [Google Scholar]
- Dandona, P.; Aljada, A.; Chaudhuri, A.; Mohanty, P.; Garg, R. Metabolic syndrome: A comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation 2005, 111, 1448–1454. [Google Scholar] [CrossRef] [PubMed]
- Siniako, A.R.; Caprio, S. Insulin resistance. J. Pediatr. 2012, 161, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.W.; Adams, L.A. Non-alcoholic fatty liver disease. Crit. Rev. Clin. Lab. Sci. 2011, 48, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Zezos, P.; Renner, E.L. Liver transplantation and non-alcoholic fatty liver disease. World J. Gastroenterol. 2014, 20, 15532–15538. [Google Scholar] [CrossRef] [PubMed]
- Cowin, G.J.; Jonsson, J.R.; Bauer, J.D.; Ash, S.; Ali, A.; Osland, E.J.; Purdie, D.M.; Cloustan, A.D.; Powell, E.E.; Galloway, G.J. Magnetic resonance imaging and spectroscopy for monitoring liver steatosis. J. Magn. Reson. Imaging 2008, 28, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Hernandez, H.; Simental-Mendia, L.E.; Rodriguez-Ramirez, G.; Reyes-Romero, M.A. Obesity and Inflammation: Epidemiology, Risk Factors, and Markers of Inflammation. Available online: https://www.hindawi.com/journals/ije/2013/678159/ (accessed on 4 May 2016).
- Ye, J.; McGuinness, O.P. Inflammation during obesity is not all bad: Evidence from animal and human studies. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E466–E477. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S.; Shargill, N.S.; Spiegelman, B.M. Adipose expression of tumor necrosis factor-alpha: Direct role in obesity-linked insulin resistance. Science 1993, 259, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Balistreri, C.R.; Caruso, C.; Candore, G. The role of adipose tissue and adipokines in obesity-related inflammatory diseases. Med. Inflamm. 2010, 2010, 802078. [Google Scholar] [CrossRef] [PubMed]
- Akash, M.S.; Rehman, K.; Chen, S. Role of inflammatory mechanisms in pathogenesis of type 2 diabetes mellitus. J. Cell. Biochem. 2013, 114, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef] [PubMed]
- De Heredia, F.P.; Gomez-Martinez, S.; Marcos, A. Obesity, inflammation and the immune system. Proc. Nutr. Soc. 2012, 71, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [PubMed]
- Cha-Molstad, H.; Agrawal, A.; Zhang, D.; Samols, D.; Kushner, I. The rel family member p50 mediated cytokine-induced C-reactive protein expression by a novel mechanism. J. Immunol. 2000, 165, 4592–4597. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Cha-Molstad, H.; Samols, D.; Kushner, I. Overexpressed nuclear factor-kappaB can participated in endogenous c-reactive protein induction, and enhances the effects of C/EBPbeta and signal transducer and activator of transcription 3. Immunology 2003, 108, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Day, C.P. Non-alcoholic fatty liver disease: Current concepts and management strategies. Clin. Med. 2006, 6, 19–25. [Google Scholar] [CrossRef]
- Cai, D.; Yuan, M.; Frantz, D.F.; Melendez, P.A.; Hansen, L.; Lee, J.; Shoelson, S.E. Local and systemic insulin resistance resulting from hepatic activation of IKKK-B and NK-kB. Nat. Med. 2005, 11, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, H.; Haugen, F.; Zadelaar, S.; Kleeman, R.; Kooistra, T.; Drevon, C.A.; Blomhoff, R. Diet-induced obesity increases NF-kB signalling in reporter mice. Genes Nutr. 2009, 4, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Gerondakis, S.; Fulford, T.S.; Messina, N.L.; Grumont, R.J. NF-kB control of T cell development. Nat. Immunol. 2014, 15, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Ilan, Y. Leaky gut and the liver: A role for bacterial translocation in nonalcoholic steatohepatitis. World J. Gastroenterol. 2012, 18, 2609–2618. [Google Scholar] [CrossRef] [PubMed]
- Halliday, G.M.; Lyons, J.G. Inflammatory doses of UV may not be necessary for skin carcinogenesis. Photochem. Photobiol. 2008, 84, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Hart, P.H.; Gorman, S.; Finlay-Jones, J.J. Modulation of the immune system by UV radiation: More than just the effects of vitamin D? Nat. Rev. Immunol. 2011, 11, 584–596. [Google Scholar] [CrossRef] [PubMed]
- Van der Leun, J.C. The ozone layer. Photodermatol. Photoimmunol. Photomed. 2004, 20, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Agar, N.S.; Halliday, G.M.; Barnetson, R.S.; Ananthaswamy, H.N.; Wheeler, M.; Jones, A.M. The basal layer in human squamous tumors harbors more UVA than UVB fingerprint mutations: A role for UVA in human skin carcinogenesis. Proc. Natl. Acad. Sci. USA 2004, 101, 4954–4959. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.M.; Norval, M.; Neale, R.E.; Young, A.R.; de Gruijl, F.R.; Takizawa, Y.; van der Leun, J.C. The consequences for human health of stratospheric ozone depletion in association with other environmental factors. Photochem. Photobiol. Sci. 2015, 14, 53–87. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, R.P.; Lee, T.K. Adverse effects of ultraviolet radiation: A brief review. Prog. Biophys. Mol. Biol 2006, 92, 119–131. [Google Scholar] [CrossRef] [PubMed]
- McMillan, T.J.; Leatherman, E.; Ridley, A.; Shorrocks, J.; Tobi, S.E.; Whiteside, J.R. Cellular effects of long wavelength UV light (UVA) in mammalian cells. J. Pharm. Pharmacol. 2008, 60, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Mouret, S.; Baudouin, C.; Charveron, M.; Favier, A.; Cadet, J.; Douki, T. Cyclobutane pyrimidine dimers are the predominant DNA lesions in whole human skin exposed to UVA radiation. Proc. Natl. Acad. Sci. USA 2006, 103, 13765–13770. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, D.L.; Saladi, R.N.; Fox, J.L. Ultraviolet radiation and skin cancer. Int. J. Dermatol. 2010, 49, 978–986. [Google Scholar] [CrossRef] [PubMed]
- Varma, S.D.; Kovtun, S.; Hegde, K.R. Role of ultraviolet irradiation and oxidative stress in cataract formation-medical prevention by nutritional antioxidants and metabolic agonists. Eye Contact Lens 2011, 37, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.R.; DeCosta, B.R.; Holick, M.F. Sunlight regulates the cutaneous production of vitamin D3 by causing its photodegradation. J. Clin. Endocrinol. Metab. 1989, 68, 882–887. [Google Scholar] [CrossRef] [PubMed]
- Freedman, D.M.; Dosemeci, M.; McGlynn, K. Sunlight and mortality form breast, ovarian, colon, prostate and non-melanoma skin cancer: A composite death certificate based case-control study. Occup. Environ. Med. 2002, 59, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, R.; Metcalfe, C.; Oliver, S.E.; Whiteman, D.C.; Bain, C.; Ness, A.; Donovan, J.; Hamdy, F.; Neal, D.E.; Lane, J.A.; et al. Life course sun exposure and risk of prostate cancer: Population-based nested case-control study (protect) and meta-analysis. Int. J. Cancer 2009, 125, 1414–1423. [Google Scholar] [CrossRef] [PubMed]
- John, E.M.; Scwartz, G.G.; Koo, J.; Wang, W.; Ingles, S.A. Sun exposure, vitamin D receptor gene polymorphisms, and breast cancer risk in a multiethnic population. Am. J. Epidemiol. 2007, 166, 1409–1419. [Google Scholar] [CrossRef] [PubMed]
- Kricker, A.; Armstrong, B.K.; Hughes, A.M.; Goumas, C.; Smedby, E.; Zheng, T.; Spinelli, J.J.; De Sanjose, S.; Hartge, P.; Melbye, M.; et al. Personal sun exposure and risk of non hodgkin lymphoma: A pooled analysis from the interlymph consortium. Int. J. Cancer 2008, 122, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Van der Rhee, H.; Coebergh, J.W.; de Vries, E. Is prevention of cancer by sun exposure more than just the effect of vitamin D? A systematic review of epidemiological studies. Eur. J. Cancer 2013, 49, 1422–1436. [Google Scholar] [CrossRef] [PubMed]
- Berwick, M.; Armstrong, B.K.; Ben-Porat, L.; Fine, J.; Kricker, A.; Eberle, C.; Barnhill, R. Sun exposure and mortality from melanoma. J. Natl. Cancer Inst. 2005, 97, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Roychoudhuri, R.; Peto, J.; Schwartz, G.G.; Baade, P.; Moller, H. Cancer survival is dependent on season of diagnosis and sunlight exposure. Int. J. Cancer 2006, 119, 1530–1536. [Google Scholar] [CrossRef] [PubMed]
- Porojnicu, A.C.; Dahlback, A.; Moan, J. Sun exposure and cancer survial in Norway: Changes in the risk of death with season of diagnosis and latitude. Adv. Exp. Med. Biol. 2008, 624, 43–54. [Google Scholar] [PubMed]
- Koh, G.C.; Hawthorne, G.; Turner, A.M.; Kunst, H.; Dedicoat, M. Tuberculosis incidence correlates with sunshine: An ecological 28-year time series study. PLoS ONE 2013, 8, e57752. [Google Scholar] [CrossRef] [PubMed]
- Laaksi, I.; Ruohola, J.P.; Tuohimaa, P.; Auvinen, A.; Haataja, R.; Pihlajamaki, H.; Ylikomi, T. An association of serum vitamin D concentrations <40 nmol/L with acute respiratory tract infection in young Finnish men. Am. J. Clin. Nutr. 2007, 86, 714–717. [Google Scholar] [PubMed]
- Ding, C.; Cicuttini, F.; Parameswaran, V.; Burgess, J.; Quinn, S.; Jones, G. Serum levels of vitamin D, sunlight exposure, and knee cartilage loss in older adults: The Tasmanian older adult cohort study. Arthritis Rheum. 2009, 60, 1381–1389. [Google Scholar] [CrossRef] [PubMed]
- Khalili, H.; Huang, E.S.; Ananthakrishnan, A.N.; Higuchi, L.; Richter, J.M.; Fuchs, C.S.; Chan, A.T. Geographical variation and incidence of inflammatory bowel disease among US women. Gut 2012, 61, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Nerich, V.; Jantchou, P.; Boutron-Ruault, M.C.; Monnet, E.; Weill, A.; Vanbockstael, V.; Auleley, G.R.; Balaire, C.; Dubost, P.; Rican, S.; et al. Low exposure to sunlight is a risk factor for Cohn’s disease. Aliment. Pharmacol. Ther. 2011, 33, 940–945. [Google Scholar] [CrossRef] [PubMed]
- Baarnhielm, M.; Hedstrom, A.K.; Kockum, I.; Sundqvist, E.; Gustafsson, S.A.; Hillert, J.; Olsson, T.; Alfredson, L. Sunlight is associated with decreased multiple sclerosis risk: No interaction with human leukocyte antigen-DRB1*15. Eur. J. Neurol. 2012, 19, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Bjornevik, K.; Riise, T.; Casetta, I.; Drulovic, J.; Granieri, E.; Holmoy, T.; Kampman, M.T.; Landtblom, A.M.; Lauer, K.; Lossius, A.; et al. Sun exposure and multiple sclerosis risk in Norway and Italy: The EnviMS study. Mult. Scler. 2014, 20, 1042–1049. [Google Scholar] [CrossRef] [PubMed]
- Islam, T.; Gauderman, J.; Cozen, W.; Mack, T.M. Childhood sun exposure influences risk of multiple sclerosis in monozygotic twins. Neurology 2007, 69, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Kampman, M.T.; Wilsgaard, T.; Mellgren, I. Outdoor activities and diet in childhood and adolescence relate to MS risk above the Arctic Circle. J. Neurol. 2007, 254, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Orton, S.M.; Wald, L.; Confavreux, C.; Vuksic, S.; Krohn, J.P.; Ramagopalan, S.V.; Herrera, B.M.; Sadovnick, A.D.; Ebers, G.C. Association of UV radiation with multiple sclerosis prevalence and sex ratio in France. Neurology 2011, 76, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Simpson, S.; Taylor, B.; Blizzard, L.; Ponsonby, A.; Pittas, F.; Tremlett, H.; Dwyer, T.; Gies, P.; van der Mei, I. Higher 25-hydroxyvitamin D is associated with lower relapse risk in multiple sclerosis. Ann. Neurol. 2010, 68, 193–203. [Google Scholar] [PubMed]
- Sloka, J.S.; Pryse-Phillips, W.E.; Stefanelli, M. The relation of ultraviolet radiation and multiple sclerosis in Newfoundland. Can. J. Neurol. Sci. 2008, 35, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Van der Mei, I.A.; Ponsonby, A.L.; Dwyer, T.; Blizzard, L.; Simmons, R.; Taylor, B.V.; Butzkueven, H.; Kilpatrick, T. Past exposure to sun, skin phenotype, and risk of multiple sclerosis: Case-control study. BMJ 2003, 327, 316. [Google Scholar] [CrossRef] [PubMed]
- Krstic, G. Asthma prevalence associated with geographical latitude and regional insolation in the United States of America and Australia. PLoS ONE 2011, 6, e18492. [Google Scholar] [CrossRef] [PubMed]
- Mullins, R.J.; Camargo, C.A. Latitude, sunlight, vitamin D, and childhood food allergy/anaphylaxis. Curr. Allergy Asthma Rep. 2012, 12, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Mullins, R.J.; Clark, S.; Camargo, C.A. Regional variation in epinephrine autoinjector prescriptions in Australia: More evidence for the vitamin D-anaphylaxis hypothesis. Ann. Allergy Asthma Immunol. 2009, 103, 488–495. [Google Scholar] [CrossRef]
- Simpson, S.; Blizzard, L.; Otahal, P.; van der Mei, I.; Taylor, B. Latitude is significantly associated with the prevalence of multiple sclerosis: A meta-analysis. J. Neurol. Neurosurg. Psychiatry 2011, 82, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Munger, K.L.; Levin, L.I.; Hollis, B.W.; Howard, N.S.; Ascherio, A. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA 2006, 296, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- Geldenhuys, S.; Hart, P.H.; Endersby, R.; Jacoby, P.; Feelisch, M.; Weller, R.B.; Matthews, V.; Gorman, S. Ultraviolet radiation suppresses obesity and symptoms of metabolic syndrome independently of vitamin D in mice fed a high-fat diet. Diabetes 2014, 63, 3759–3769. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Cheng, Y.F.; Lai, C.Y.; Hsu, L.W.; Chang, Y.C.; Deng, J.Y.; Huang, Y.Z.; Honda, H.; Chen, K.D.; Wang, C.C.; et al. Impact of artificial sunlight therapy on the progress of non-alcoholic fatty liver disease in rats. J. Hepatol. 2011, 55, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Gorman, S.; Robyn, L.; Allen-Hall, A.; Fleury, N.; Feelisch, M. Ultraviolet radiation, vitamin D and the development of obesity, metabolic syndrome and type-2 diabetes. Photochem. Photobiol. Sci. 2016. under review. [Google Scholar]
- Muldowney, S.; Lucey, A.J.; Paschos, G.; Martinez, J.A.; Bandarra, N.; Thorsdottir, I.; Cashman, K.D.; Kiely, M. Relationships between vitamin D status and cardio-metabolic risk factors in young European adults. Ann. Nutr. Metab. 2011, 58, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Woolcott, O.O.; Castillo, O.A.; Gutierrez, C.; Elashoff, R.M.; Stefanovski, D.; Bergman, R.N. Inverse association between diabetes and altitude: A cross-sectional study in the adult population of the United States. Obesity (Silver Spring) 2014, 22, 2080–2090. [Google Scholar] [CrossRef] [PubMed]
- Hirschler, V. Cardiometabolic risk factors in native populations living at high altitudes. Int. J. Clin. Pract. 2016, 70, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Young, T.K.; Szathmary, E.J.; Evers, S.; Wheatley, B. Geographical distribution of diabetes among the native population of Canada: A national survey. Soc. Sci. Med. 1990, 31, 129–139. [Google Scholar] [CrossRef]
- Sohmiya, M.; Kanazawa, I.; Kato, Y. Seasonal changes in body composition and blood HbA1c levels without weight change in male patients with type 2 diabetes treated with insulin. Diabetes Care 2004, 27, 1238–1239. [Google Scholar] [CrossRef] [PubMed]
- Visscher, T.L.; Seidell, J.C. Time trends (1993–1997) and seasonal variation in body mass index and waist circumference in the Netherlands. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1309–1316. [Google Scholar] [CrossRef] [PubMed]
- Doro, P.; Grant, W.B.; Benko, R.; Matuz, M.; Toth, T.; Soos, G. Vitamin D and the seasonality of type 2 diabetes. Med. Hypotheses 2008, 71, 317–318. [Google Scholar] [CrossRef] [PubMed]
- Shore-Lorenti, C.; Brennan, S.L.; Sanders, K.M.; Neale, R.E.; Lucas, R.M.; Ebeling, P.R. Shining the light on sunshine: A systematic review of the influence of sun exposure on type 2 diabetes mellitus-related outcomes. Clin. Endocrinol. 2014, 81, 799–811. [Google Scholar] [CrossRef] [PubMed]
- Algert, C.S.; Roberts, C.L.; Shand, A.W.; Morris, J.M.; Ford, J.B. Seasonal variation in pregnancy hypertension is correlated with sunlight intensity. Am. J. Obstet. Gynecol. 2010, 203, 215.e1–215.e5. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, P.G.; Olsson, H.; Landin-Olsson, M. Are active sun exposure habits related to lowering risk of type 2 diabetes mellitus in women, a prospective cohort study? Diabetes Res. Clin. Pract. 2010, 90, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, P.G.; Epstein, E.; Olsson, H. Does an active sun exposure habit lower the risk of venous thrombotic events? A D-lightful hypothesis. J. Thromb. Haemost. 2009, 7, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, P.G.; Epstein, E.; Landin-Olsson, M.; Ingvar, C.; Nielsen, K.; Stenbeck, M.; Olsson, H. Avoidance of sun exposure is a risk factor for all-cause mortality: Results from the melanoma in Southern Sweden Cohort. J. Intern. Med. 2014, 276, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Al-Daghri, N.M.; Alkharfy, K.M.; Al-Saleh, Y.; Al-Attas, O.S.; Alokail, M.S.; Al-Othman, A.; Moharram, O.; El-Kholie, E.; Sabico, S.; Kumar, S.; et al. Modest reversal of metabolic syndrome manifestations with vitamin D status correction: A 12-month prospective study. Metabolism 2012, 61, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Romani, J.; Caixas, A.; Carrascosa, J.M.; Ribera, M.; Rigla, M.; Luelmo, J. Effect of narrowband ultraviolet B therapy on inflammatory markers and body fat composition in moderate to severe psoriasis. Br. J. Dermatol. 2012, 166, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Krause, R.; Buhring, M.; Hopfenmuller, W.; Holick, M.F.; Sharma, A.M. Ultraviolet B and blood pressure. Lancet 1998, 352, 709–710. [Google Scholar] [CrossRef]
- Kokot, F.; Schmidt-Gayk, H.; Wiecek, A.; Mleczko, Z.; Bracel, B. Influence of ultraviolet irradiation on plasma vitamin D and calcitonin levels in humans. Kidney Int. Suppl. 1989, 27, 143–146. [Google Scholar]
- Oplander, C.; Volkmar, C.M.; Paunel-Gorgulu, A.; van Faassen, E.E.; Heiss, C.; Kelm, M.; Halmer, D.; Murtz, M.; Pallua, N.; Suschek, C.V. Whole body UVA irradiation lowers systemic blood pressure by release of nitric oxide from intracutaneous photolabile nitric oxide derivates. Circ. Res. 2009, 105, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Deeb, K.K.; Trump, D.L.; Johnson, C.S. Vitamin d signalling pathways in cancer: Potential for anticancer therapeutics. Nat. Rev. Cancer 2007, 7, 684–700. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. The role of vitamin D for bone health and fracture prevention. Curr. Osteoporos. Rep. 2006, 4, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Carlin, A.M.; Rao, D.S.; Meslemani, A.M.; Genaw, J.A.; Parikh, N.J.; Levy, S.; Bhan, A.; Talpos, G.B. Prevalence of vitamin D depletion among morbidly obese patients seeking gastric bypass surgery. Surg. Obes. Relat. Dis. 2006, 2, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Barchetta, I.; Angelico, F.; Del Ben, M.; Baroni, M.G.; Pozzilli, P.; Morini, S.; Cavallo, M.G. Strong association between non alcoholic fatty liver disease (NAFLD) and low 25(OH) vitamin D levels in an adult population with normal serum liver enzymes. BMC Med. 2011, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Eliades, M.; Spryou, E.; Agrawal, N.; Lazo, M.; Brancati, F.L.; Potter, J.J.; Koteish, A.A.; Clark, J.M.; Guallar, E.; Hernaez, R. Meta-analysis: Vitamin D and non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2013, 38, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Ford, E.S.; Ajani, U.A.; McGuire, L.C.; Liu, S. Concentrations of serum vitamin D and the metabolic syndrome among U.S. Adults. Diabetes Care 2005, 28, 1228–1230. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Lau, J.; Hu, F.B.; Dawson-Hughes, B. The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2007, 92, 2017–2029. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Pencina, M.J.; Booth, S.L.; Jacques, P.F.; Ingelsson, E.; Lanier, K.; Benjamin, E.J.; D’Agostino, R.B.; Wolf, M.; Vasan, R.S. Vitamin D deficiency and risk of cardiovascular disease. Circulation 2008, 117, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Kull, M.; Kallikorm, R.; Lember, M. Body mass index determines sunbathing habits: Implications on vitamin D levels. Intern. Med. J. 2009, 39, 256–258. [Google Scholar] [CrossRef] [PubMed]
- Wamberg, L.; Christiansen, T.; Paulsen, S.K.; Fisker, S.; Rask, P.; Rejnmark, L.; Richelsen, B.; Pedersen, S.B. Expression of vitamin D-metabolizing enzymes in human adipose tissue—The effect of obesity and diet-induced weight loss. Int. J. Obes. 2013, 37, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Rosenstreich, S.J.; Rich, C.; Volwiler, W. Deposition and release of vitamin D3 from body fat: Evidence for a storage site in the rat. J. Clin. Investig. 1971, 50, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Gao, D.; Wilding, J.; Trayhurn, P.; Bing, C. Vitamin D signalling in adipose tissue. Br. J. Nutr. 2012, 108, 1915–1923. [Google Scholar] [CrossRef] [PubMed]
- Bang, U.C.; Novovic, S.; Andersen, A.M.; Fenger, M.; Hansen, M.B.; Jensen, J.E. Variations in serum 25-hydroxyvitamin D during acute pancreatitis: An exploratory longitudinal study. Endocr. Res. 2011, 36, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Reid, D.; Toole, B.J.; Knox, S.; Talwar, D.; Harten, J.; O’Reilly, D.S.; Blackwell, S.; Kinsella, J.; McMillan, D.C.; Wallace, A.M. The relation between acute changes in the systemic inflammatory response and plasma 25-hydroxyvitamin D concentrations after elective knee arthroplasty. Am. J. Clin. Nutr. 2011, 93, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Gorman, S.; Black, L.J.; Feelisch, M.; Hart, P.H.; Weller, R. Can skin exposure to sunlight prevent liver inflammation? Nutrients 2015, 7, 3219–3239. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Fernandez, B.O.; Hamilton, A.; Lang, N.N.; Gallagher, J.M.; Newby, D.E.; Feelisch, M.; Weller, R.B. UVA irradiation of human skin vasodilates arterial vasculature and lowers blood pressure independently of nitric oxide synthase. J. Investig. Dermatol. 2014, 134, 1839–1846. [Google Scholar] [CrossRef] [PubMed]
- Mowbray, M.; McLintock, S.; Weerakoon, R.; Lomatschinsky, N.; Jones, S.; Rossi, A.G.; Weller, R.B. Enzyme-independent no stores in human skin: Quantification and influence of UV radiation. J. Investig. Dermatol. 2009, 129, 834–842. [Google Scholar] [CrossRef] [PubMed]
- Juzeniene, A.; Moan, J. Beneficial effects of UV radiation other than vitamin D production. Dermatoendocrinol 2012, 4, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Hiramoto, K. Ultraviolet A irradiation of the eye activates a nitric oxide-dependent hypothalamo-pituitary pro-opiomelanocortin pathway and modulates the functions of langerhans cells. J. Dermatol. 2009, 36, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Hiramoto, K.; Yamate, Y.; Sato, E.F. The effects of ultraviolet eye irradiation on dextran sodium sulfate-induced ulcerative colitis in mice. Photochem. Photobiol. 2016, 92, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.D.; Ashmore, T.; Kotwica, A.O.; Murfitt, S.O.; Fernandez, B.O.; Feelisch, M.; Murray, A.J.; Griffin, J.L. Inorganic nitrate promotes the browning of white adipose tissue through the nitrate-nitrite-nitric oxide pathway. Diabetes 2015, 64, 471–484. [Google Scholar] [CrossRef] [PubMed]
- Carlstrom, M.; Larsen, F.J.; Nystrom, T.; Hezel, M.; Borniquel, S.; Weitzberg, E.; Lundberg, J.O. Dietary inorganic nitrate reverses features of metabolic syndrome in endothelial nitric oxide synthase-deficient mice. Proc. Natl. Acad. Sci. USA 2010, 107, 17716–17720. [Google Scholar] [CrossRef] [PubMed]
- Zand, J.; Lanza, F.; Garg, H.K.; Bryan, N.S. All-natural nitrite and nitrate containing dietary supplement promotes nitric oxide production and reduces triglycerides in humans. Nutr. Res. 2011, 31, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Gruber, H.J.; Mayer, C.; Mangge, H.; Fauler, G.; Grandits, N.; Wilders-Truschnig, M. Obesity reduces the bioavailability of nitric oxide in juveniles. Int. J. Obes. 2008, 32, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Brenner, C.; Galluzzi, L.; Kepp, O.; Kroemer, G. Decoding cell death signals in liver inflammation. J. Hepatol. 2013, 59, 583–594. [Google Scholar] [CrossRef] [PubMed]
- Skobwiat, C.; Slominski, A.T. Ultraviolet B stimulates proopiomelanocortin signalling in the arcuate nucleus of the hypothalamus in mice. Exp. Dermatol. 2016, 25, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Martin-Gronert, M.S.; Ozanne, S.E. Metabolic programming of insulin action and secretion. Diabetes Obes. Metab. 2012, 14 (Suppl. S3), 29–39. [Google Scholar] [CrossRef] [PubMed]
- Wardlaw, S.L. Hypothalamic proopiomelanocortin processing and the regulation of energy balance. Eur. J. Pharmacol. 2011, 660, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Fell, G.L.; Robinson, K.C.; Mao, J.; Woolf, C.J.; Fisher, D.E. Skin β-endorphin mediates addiction to UV light. Cell 2014, 157, 1527–1534. [Google Scholar] [CrossRef] [PubMed]
- Skobowiat, C.; Slominski, A.T. UVB activates hypothalamic-pituitary-adrenal axis in C57BL/6 mice. J. Investig. Dermatol. 2015, 135, 1638–1648. [Google Scholar] [CrossRef] [PubMed]
- Dutia, R.; Meece, K.; Dighe, S.; Kim, A.J.; Wardlaw, S.L. Beta-endorphin antagonizes the effects of alpha-MSH on food intake and body weight. Endocrinology 2012, 153, 4246–4255. [Google Scholar] [CrossRef] [PubMed]
- Grochot-Przeczek, A.; Dulak, J.; Jozkowicz, A. Haem oxygenase-1: Non-canonical roles in physiology and pathology. Clin. Sci. 2012, 122, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Gozzelino, R.; Jeney, V.; Soares, M.P. Mechanisms of cell protection by heme oxygenase-1. Ann. Rev. Pharmacol. Toxicol. 2010, 50, 323–354. [Google Scholar] [CrossRef] [PubMed]
- Otterbein, L.E.; Bach, F.H.; Alam, J.; Soares, M.; Lu, H.T.; Wysk, M.; Davis, R.J.; Flavell, R.; Choi, A.M.K. Carbon monoxide has anti-inflammatory effects involving the mitogen activated protein kinase pathway. Nat. Med. 2000, 6, 422–428. [Google Scholar] [PubMed]
- Hosick, P.A.; AlAmodi, A.A.; Storm, M.V.; Gousset, M.U.; Pruett, B.E.; Gray, W., III; Stout, J.; Stec, D.E. Chronic carbon monoxide treatment attenuates development of obesity and remodels adipocytes in mice fed a high-fat diet. Int. J. Obes. 2014, 38, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Jansen, T.; Daiber, A. Direct antioxidant properties of bilirubin and biliverdin. Is there a role for biliverdin reductase? Front. Pharmacol. 2012, 3, 30. [Google Scholar] [CrossRef] [PubMed]
- McDonagh, A.F. The biliverdin-bilirubin antioxidant cycle of cellular protection: Missing a wheel? Free Radic. Biol. Med. 2010, 49, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Dong, H.; Zhang, Y.; Cao, M.; Song, L.; Pan, Q.; Bulmer, A.; Adams, D.B.; Dong, X.; Wang, H. Bilirubin increases insulin sensitivity by regulating cholesterol metabolism, adipokines and PPARγ levels. Sci. Rep. 2015, 5, 9886. [Google Scholar] [CrossRef] [PubMed]
- Gorman, S.; Kuritzky, L.A.; Judge, M.A.; Dixon, K.M.; McGlade, J.P.; Mason, R.S.; Finlay-Jones, J.J.; Hart, P.H. Topically applied 1,25-dihydroxyvitamin D3 enhances the suppressive activity of CD4+CD25+ cells in the draining lymph nodes. J. Immunol. 2007, 179, 6273–6283. [Google Scholar] [CrossRef] [PubMed]
- Ng, R.L.; Bisley, J.L.; Gorman, S.; Norval, M.; Hart, P.H. Ultraviolet irradiation of mice reduces the competency of bone marrow-derived CD11c+ cells via an indomethacin-inhibitable pathway. J. Immunol. 2010, 185, 7207–7215. [Google Scholar] [CrossRef] [PubMed]
- Byrne, S.N. How much sunlight is enough? Photochem. Photobiol. Sci. 2014, 13, 840–852. [Google Scholar] [CrossRef] [PubMed]
- Chatzigeorgiou, A.; Karalis, K.P.; Bornstein, S.R.; Chavakis, T. Lymphocytes in obesity-related adipose tissue inflammation. Diabetologia 2012, 55, 2583–2592. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; Mulder, I.E. Microbiome and immunological interactions. Nutr. Rev. 2012, 70 (Suppl. S1), S18–S30. [Google Scholar] [CrossRef] [PubMed]
- Gorman, S.; Geldenhuys, S.; Judge, M.A.; Weeden, C.E.; Waithman, J.; Hart, P.H. Dietary Vitamin D Increases Percentages and Function of Regulatory T Cells in the Skin-Draining Lymph Nodes and Suppresses Dermal Inflammation. Available online: https://www.hindawi.com/journals/jir/2016/1426503/ (accessed on 4 May 2016).
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fleury, N.; Geldenhuys, S.; Gorman, S. Sun Exposure and Its Effects on Human Health: Mechanisms through Which Sun Exposure Could Reduce the Risk of Developing Obesity and Cardiometabolic Dysfunction. Int. J. Environ. Res. Public Health 2016, 13, 999. https://doi.org/10.3390/ijerph13100999
Fleury N, Geldenhuys S, Gorman S. Sun Exposure and Its Effects on Human Health: Mechanisms through Which Sun Exposure Could Reduce the Risk of Developing Obesity and Cardiometabolic Dysfunction. International Journal of Environmental Research and Public Health. 2016; 13(10):999. https://doi.org/10.3390/ijerph13100999
Chicago/Turabian StyleFleury, Naomi, Sian Geldenhuys, and Shelley Gorman. 2016. "Sun Exposure and Its Effects on Human Health: Mechanisms through Which Sun Exposure Could Reduce the Risk of Developing Obesity and Cardiometabolic Dysfunction" International Journal of Environmental Research and Public Health 13, no. 10: 999. https://doi.org/10.3390/ijerph13100999
APA StyleFleury, N., Geldenhuys, S., & Gorman, S. (2016). Sun Exposure and Its Effects on Human Health: Mechanisms through Which Sun Exposure Could Reduce the Risk of Developing Obesity and Cardiometabolic Dysfunction. International Journal of Environmental Research and Public Health, 13(10), 999. https://doi.org/10.3390/ijerph13100999






