Vitamin D Status and Its Consequences for Health in South Africa
Abstract
:1. Introduction
2. Vitamin D Status in South Africa
3. Sun Exposure and Photoprotection in Relation to Vitamin D Production in South Africa
4. Effect of Urbanisation and Diet on Vitamin D Status in South Africa
5. Association of Vitamin D Status with Diseases in South Africa
6. Vitamin D Receptor Gene (VDR) Polymorphisms and Ethnicity in South Africa
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holick, M.F. Sunlight, UV-radiation, vitamin D and skin cancer. Adv. Exp. Med. Biol. 2008, 624, 1–15. [Google Scholar] [PubMed]
- Wimalawansa, S.J. Non-musculoskeletal benefits of vitamin D. J. Steroid Biochem. Mol. Biol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Hossein-nezhad, A.; Holick, M.F. Vitamin D for health: A global perspective. Mayo Clin. Proc. 2013, 88, 720–755. [Google Scholar] [CrossRef] [PubMed]
- Wahl, D.A.; Cooper, C.; Ebeling, P.R.; Eggersdorfer, M.; Hilger, J.; Hoffmann, K.; Josse, R.; Kasis, J.A.; Mithal, A.; Pierroz, D.D.; et al. A global representation of vitamin D status in healthy populations. Arch. Osteoporos. 2012, 7, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Hilger, J.; Friedel, A.; Herr, R.; Rausch, T.; Roos, F.; Wahl, D.A.; Pierros, D.D.; Weber, P.; Hoffmann, K. A systematic review of vitamin D status in populations worldwide. Br. J. Nutr. 2014, 111, 23–45. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Sunlight “D”ilemma: Risk of skin cancer or bone disease and muscle weakness. Lancet 2001, 357, 4–6. [Google Scholar] [CrossRef]
- Hewison, M.; Zehnder, D.; Chakraverty, R.; Adams, J.S. Vitamin D and barrier function: A novel role for extra-renal 1 alpha-hydroxylase. Mol. Cell. Endocrinol. 2004, 215, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.T.; Tavera-Mendoza, L.C.; Laperriere, D.; Libby, E.; MacLeod, N.B.; Nagal, Y.; Bourdeau, V.; Konstorum, A.; Lallemant, B.; Zhang, R.; et al. Large-scale in silico and microarrany-based indentification of direct 1,25-dihydroxyvitamin D3 target genes. Mol. Endocrinol. 2005, 19, 2685–2695. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.B.; Lai, J.; Lizaola, B.; Kane, L.; Markova, S.; Weyland, P.; Terrault, N.A.; Stotland, N.; Bikle, D. A comparison of measured and calculated free 25(OH)D levels in clinical populations. J. Clin. Endocrinol. Metab. 2014, 99, 1631–1637. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium. Dietary Reference Intakes for Calcium and Vitamin D; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Bischoff-Ferrari, H.A.; Giovannucci, E.; Willett, W.C.; Dietrich, T.; Dawson-Hughes, B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am. J. Clin. Nutr. 2006, 84, 18–28. [Google Scholar] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, A.; Girdin, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Carter, G.D.; Carter, R.; Jones, J.; Berry, J. How accurate are assays for 25-hydroxyvitamin D? Data from the international vitamin D external quality assessment scheme. Clin. Chem. 2004, 50, 2195–2197. [Google Scholar] [CrossRef] [PubMed]
- Sempos, C.T.; Vesper, H.W.; Phinney, K.W.; Thienpont, L.M.; Coates, P.M. Vitamin D status as an international issue: National surveys and the problem of standardization. Scand. J. Lab. Investig. Suppl. 2012, 243, 32–40. [Google Scholar]
- Pettifor, J.M.; Moodley, G.P.; Hough, F.S.; Koch, H.; Chen, T.; Lu, Z.; Holick, M.F. The effect of season and latitude on in vitro vitamin D formation by sunlight in South Africa. S. Afr. Med. J. 1996, 86, 1270–1272. [Google Scholar] [PubMed]
- Weather 2 Travel. Available online: www.weather2travel.com (accessed on 14 October 2016).
- World Weather Online. Available online: www.worldweatheronline.com (accessed on 14 October 2016).
- Weather South African. Available online: www.weathersa.co.za (accessed on 14 October 2016).
- Global Security. Available online: www.globalsecurity.org (accessed on 5 August 2016).
- Wilkes, M.; Wright, C.Y.; du Plessis, A.; Reeder, A. Fitzpatrick skin type, individual topology angle, and melanin index in an African population: Steps towards universally applicable skin photosensitivity assessments. JAMA Dermatol. 2015, 151, 902–903. [Google Scholar] [CrossRef] [PubMed]
- Statistics South Africa. Available online: http://www.statssa.gov.za/?page_id=1854&PPN=P0302&SCH=6334 (accessed on 14 October 2016).
- Palacios, C.; Gonzalez, L. Is vitamin D deficiency a major global public health problem? J. Steroid Biochem. Mol. Biol. 2014, 144, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Pettifor, J.M.; Ross, F.P.; Moodley, G.; Wang, J.; Margo, G.; Skjolde, C. Serum calcium, magnesium, phosphorus, alkaline phosphatase and 25-hydroxyvitamin D concentrations in children. S. Afr. Med. J. 1978, 53, 751–754. [Google Scholar] [PubMed]
- Pettifor, J.M.; Ross, P.; Moodley, G.; Shuenyane, E. Calcium deficiency in rural black children in South Africa—A comparison between rural and urban communities. Am. J. Clin. Nutr. 1979, 32, 2477–2483. [Google Scholar] [PubMed]
- Sochett, E.B.; Pettifor, J.M.; Moodley, G.; Pentopoulos, M. Vitamin D status in hospitalized black children under 2 years of age. S. Afr. Med. J. 1985, 67, 1041–1044. [Google Scholar] [PubMed]
- Van der Westhuyzen, J. Biochemical evaluation of black preschool children in the northern Transvaal. S. Afr. Med. J. 1986, 70, 146–148. [Google Scholar] [PubMed]
- Cornish, D.A.; Maluleke, V.; Mhlanga, T. An investigation into a possible relationship between vitamin D, parathyroid hormone, calcium and magnesium in normally pigmented and an albino rural black population in the Northern Province of South Africa. Biofactors 2000, 11, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Poopedi, M.A.; Norris, S.A.; Pettifor, J.M. Factors influencing the vitamin D status of 10-year-old urban South African children. Public Health Nutr. 2011, 14, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Fairney, A.; Sloan, M.A.; Patel, K.V.; Coumbe, A. Vitamin A and D status of black South African women and their babies. Hum. Nutr. Clin. Nutr. 1987, 41, 81–87. [Google Scholar] [PubMed]
- Daniels, E.D.; Pettifor, J.M.; Schnitzlet, C.M.; Moodley, G.P.; Zachen, D. Differences in mineral homeostasis, volumetric bone mass and femoral neck axis length in black and white South African women. Osteoporos. Int. 1997, 7, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Haarburger, D.; Hoffman, M.; Erasmus, R.T.; Pillay, T.S. Relationship between vitamin D, calcium and parathyroid hormone in Cape Town. J. Clin. Pathol. 2009, 26, 567–569. [Google Scholar] [CrossRef] [PubMed]
- Kruger, M.C.; Kruger, I.M.; Wentzel-Viljoen, E.; Kruger, A. Urbanization of black South African women may increase risk of low bone mass due to low vitamin D status, low calcium intake, and high bone turnover. Nutr. Res. 2011, 31, 748–758. [Google Scholar] [CrossRef] [PubMed]
- George, J.A.; Norris, S.A.; van Deventer, H.E.; Crowther, N.J. The association of 25 hydroxyvitamin D and parathyroid hormone with metabolic syndrome in two ethnic groups in South Africa. PLoS ONE 2013, 8, e61282. [Google Scholar] [CrossRef] [PubMed]
- Kruger, I.M.; Kruger, M.C.; Doak, C.M.; Schutte, A.E.; Huisman, H.W.; Van Rooyen, J.M.; Schutte, R.; Malan, L.; Malan, N.T.; Fourie, C.M.; et al. The association of 25(OH)D with blood pressure, pulse pressure and carotid-radial pulse wave velocity in African women. PLoS ONE 2013, 8, e54554. [Google Scholar] [CrossRef] [PubMed]
- George, J.A.; Micklesfield, L.K.; Norris, S.A.; Crowther, N.J. The association between body composition, 25(OH)D, and PTH and bone mineral density in black African and Asian Indian population groups. J. Clin. Endocrinol. Metab. 2014, 99, 2146–2154. [Google Scholar] [CrossRef] [PubMed]
- George, J.A.; Norris, S.A.; van Deventer, H.E.; Pettifor, J.M.; Crowther, N.J. Effect of adiposity, season, diet and calcium or vitamin D supplementation on the vitamin D status of healthy urban African and Asian-Indian adults. Br. J. Nutr. 2014, 112, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Durazo-Arvizu, R.A.; Camacho, P.; Bovet, P.; Forrester, T.; Lambert, E.V.; Plage-Rhule, J.; Hoofnagle, A.N.; Aloia, J.; Tayo, B.; Dugas, L.R.; et al. 25-Hydroxyvitamin D in African-origin populations at varying latitudes challenges the construct of a physiologic norm. Am. J. Clin. Nutr. 2014, 100, 908–914. [Google Scholar] [CrossRef] [PubMed]
- Coussens, A.K.; Naude, C.E.; Goloath, R.; Chaplin, G.; Wilkinson, R.J.; Jablonski, N.G. High-dose vitamin D3 reduces deficiency caused by low UVB exposure and limits HIV-1 replication in urban Southern Africans. Proc. Natl. Acad. Sci. USA 2015, 112, 8052–8057. [Google Scholar] [CrossRef] [PubMed]
- Pettifor, J.M.; Ross, F.P.; Solomon, L. Seasonal variation in serum 25-hydroxycholecalciferol concentrations in elderly South African patients with fractures of femoral neck. Br. Med. J. 1978, 1, 826–827. [Google Scholar] [CrossRef] [PubMed]
- Van Papendorp, D.H. The vitamin D status of South African women living in old-age homes. S. Afr. Med. J. 1990, 78, 556. [Google Scholar] [PubMed]
- Charlton, K.E.; Labadarios, D.; Lombard, C.J.; Louw, M.E. Vitamin D status of older South Africans. S. Afr. Med. J. 1996, 86, 1406–1410. [Google Scholar] [PubMed]
- Cancer Association of South Africa (CANSA). Fact Sheet on Vitamin D. Available online: http://www.cansa.org.za/files/2015/10/Fact-Sheet-Vitamin-D-October-2015.pdf (accessed on 14 October 2016).
- Lucas, R.M.; Norval, M.; Neale, R.E.; Young, A.R.; de Gruijl, F.R.; Takizawa, Y.; van der Leun, J.C. The consequences for human health of stratospheric ozone depletion in association with other environmental factors. Photochem. Photobiol. Sci. 2015, 14, 53–87. [Google Scholar] [CrossRef] [PubMed]
- Ultraviolet Radiation and the INTERSUN Programme. Sun Protection; World Health Organization (WHO): Geneva, Switzerland, 2015. [Google Scholar]
- Norval, M.; Wulf, H.C. Does chronic sunscreen use reduce vitamin D production to insufficient levels? Br. J. Dermatol. 2009, 161, 732–736. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.; Thieden, E.; Philipsen, P.A.; Heydenreich, J.; Wulf, H.C.; Young, A.R. Determinants of personal ultraviolet-radiation exposure doses on a sun holiday. Br. J. Dermatol. 2013, 168, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Guy, C.Y.; Diab, R.; Martincigh, B. Ultraviolet radiation exposure of children and adolescents in Durban, South Africa. Photochem. Photobiol. 2003, 77, 265–270. [Google Scholar] [CrossRef]
- Gies, P.; Roy, C.; Toomey, S.; MacLennan, R.; Watson, M. Solar UVR exposures of primary school children at three locations in Queensland. Photochem. Photobiol. 1998, 68, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Diffey, B.L.; Gibson, C.J.; Haylock, R.; McKinlay, A.F. Outdoor ultraviolet exposure of children and adolescents. Br. J. Dermatol. 1996, 124, 1030–1034. [Google Scholar] [CrossRef]
- Wright, C.Y.; Albers, P.N.; Oosthuizen, M.A.; Phala, N. Self-reported sun-related knowledge, attitudes and behaviours among schoolchildren attending South African primary schools. Photodermatol. Photoimmunol. Photomed. 2014, 30, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Lund, P.M.; Taylor, J.S. Lack of adequate sun protection for children with oculocutaneous albinism in South Africa. BMC Public Health 2008, 8, 225. [Google Scholar] [CrossRef] [PubMed]
- Lund, P.M.; Gaigher, R. A health intervention programme for children with albinism at a special school in South Africa. Health Educ. Res. 2002, 17, 365–372. [Google Scholar] [CrossRef] [PubMed]
- United Nations Human Settlements Programme (UN-Habitat). The State of African Cities 2014; UN-Habitat: Nairobi, Kenya, 2014. [Google Scholar]
- Kolahdooz, F.; Spearing, K.; Sharma, S. Dietary adequacies among South African adults in rural KwaZulu-Natal. PLoS ONE 2013, 8, e67184. [Google Scholar] [CrossRef] [PubMed]
- Spearing, K.; Kolahdooz, F.; Lukasewich, M.; Mathe, N.; Khamis, T.; Sharma, S. Nutritional composition of commonly consumed composite dishes from rural villages in Empangeni, KwaZulu-Natal, South Africa. J. Hum. Nutr. Diet. 2013, 26, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Bourne, L.T.; Steyn, K.; Laubscher, J.A. Poor nutritional status in older black South Africans. Asia Pac. J. Clin. Nutr. 2001, 10, 31–38. [Google Scholar]
- Van Stuijvenberg, M.E.; Nel, J.; Schoeman, S.E.; Lombard, C.J.; du Plessis, L.M.; Dhansay, M.A. Low intake of calcium and vitamin D, but not zinc, iron or vitamin A, is associated with stunting in 2- to 5-year-old children. Nutrition 2015, 31, 841–846. [Google Scholar] [CrossRef] [PubMed]
- MacKeown, J.M.; Cleaton-Jones, P.E.; Edwards, A.W.; Turgeon-O’Brien, H. Energy, macro- and micronutrient intake of 5-year-old urban black South African children in 1984 and 1995. Paediatr. Perinat. Epidemiol. 1998, 12, 297–312. [Google Scholar] [CrossRef] [PubMed]
- Bischof, F.; Basu, D.; Pettifor, J.M. Pathological long-bone fractures in residents with cerebral palsy in a long-term care facility in South Africa. Dev. Med. Child Neurol. 2002, 44, 119–122. [Google Scholar] [CrossRef] [PubMed]
- Bhimma, R.; Pettifor, J.M.; Coovadia, H.M.; Moodley, M.; Adhikari, M. Rickets in black children beyond infancy in Natal. S. Afr. Med. J. 1995, 85, 668–672. [Google Scholar] [PubMed]
- Schnitzler, C.M.; Pettifor, J.M.; Patel, D.; Mesquita, J.M.; Moodley, G.P.; Zachen, D. Metabolic bone disease in black teenagers with genu valgum or varum without radiologic rickets: A bone histomorphometric study. J. Bone Miner. Res. 1994, 9, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Hamill, M.M.; Ward, K.A.; Pettifor, J.M.; Norris, S.A.; Prentice, A. Bone mass, body composition and vitamin D status of ARV-naive, urban, black South African women with HIV infection, stratified by CD4 count. Osteoporos. Int. 2013, 24, 2855–2861. [Google Scholar] [CrossRef] [PubMed]
- Snyman, J.R.; de Sommers, K.; Steinmann, M.A.; Lizamore, D.J. Effects of calcitriol on eosinophil activity and antibody responses in patients with schistosomiasis. Eur. J. Clin. Pharmacol. 1997, 52, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Visser, D.H.; Schoeman, J.F.; Van Furth, A.M. Seasonal variation in the incidence rate of tuberculous meningitis is associated with sunshine hours. Epidemiol. Infect. 2013, 141, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Nhamoyebonde, S.; Oni, T.; Rangaka, M.X.; Marais, S.; Bangani, N.; Tsekela, R.; Bash, L.; de Azevedo, V.; Caldwell, J.; et al. Reciprocal seasonal variation in vitamin D status and tuberculosis notifications in Cape Town, South Africa. Proc. Natl. Acad. Sci. USA 2011, 108, 19013–19017. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, J.N.; Bicanic, T.; Loyse, A.; Meintjes, G.; Hogan, L.; Roberts, C.H.; Shoham, S.; Perfect, J.R.; Govender, N.P.; Harrison, T.S. Very low levels of 25-hydroxyvitamin D are not associated with immunologic changes or clinical outcome in South African patients with HIV-associated cryptococcal meningitis. Clin. Infect. Dis. 2014, 59, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Shivakoti, R.; Christian, P.; Yang, W.T.; Gupte, N.; Mwelase, N.; Kanyama, C.; Pillay, S.; Samaneka, W.; Santos, B.; Poongulali, S.; et al. Prevalence and risk factors of micronutrient deficiencies pre- and post-antiretroviral therapy (ART) among a diverse multicountry cohort of HIV-infected adults. Clin. Nutr. 2015, 61, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Havers, F.P.; Detrick, B.; Cardoso, S.W.; Bwewbdes, S.; Lama, J.R.; Sugandhavesa, P.; Mwelase, N.H.; Campbell, T.B.; Gupta, A. Change in vitamin d levels occurs early after antiretroviral therapy initiation and depends on treatment regimen in resource-limited settings. PLoS ONE 2014, 9, e95164. [Google Scholar] [CrossRef] [PubMed]
- Price, P.; Haddow, L.J.; Affandi, J.; Agerwal, U.; Easterbrook, P.J.; Elliott, J.; French, M.; Kumar, M.; Moosa, M.Y.; Oliver, B.; et al. Short communication: Plasma levels of vitamin D in HIV patients initiating antiretroviral therapy do not predict immune restoration disease associated with Mycobacterium tuberculosis. AIDS Res. Hum. Retrovir. 2012, 28, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Potocnik, F.C.; van Rensburg, S.J.; Hon, D.; Emsley, R.A.; Moodie, I.M.; Erasmus, R.T. Oral zinc augmentation with vitamins A and D increases plasma zinc concentration: Implications for burden of disease. Metab. Brain Dis. 2006, 21, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Naude, C.E.; Carey, P.D.; Laubscher, R.; Fein, G.; Senekal, M. Vitamin D and calcium status in South African adolescents with alcohol use disorders. Nutrients 2012, 4, 1076–1094. [Google Scholar] [CrossRef] [PubMed]
- Coussens, A.K.; Martineau, A.R.; Wilkinson, R.J. Anti-inflammatory and antimicrobial actions of vitamin D in combating TB/HIV. Scientifica 2014. [Google Scholar] [CrossRef] [PubMed]
- Pinzone, M.R.; di Rosa, M.; Celesia, B.M.; Condorelli, F.; Malaguarnera, M.; Madeddu, G. LPS and HIV gp120 modulate monocyte/macrophage CYP27B1 and CYP24A1 expression leading to vitamin D consumption and hypovitaminosis in HIV-infected individuals. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 1938–1950. [Google Scholar] [PubMed]
- Campbell, G.R.; Spector, S.A. Hormonally active vitamin D3 (1alpha,25-dihydroxycholecalciferol) triggers autophage in human macrophages that inhibits HIV-1 infection. J. Biol. Chem. 2011, 286, 18890–18902. [Google Scholar] [CrossRef] [PubMed]
- Quinones-Mateu, M.E.; Lederman, M.M.; Feng, Z.; Chakraborty, B.; Weber, J.; Rangel, H.R. Human epithelial beta-defensins 2 and 3 inhibit HIV-1 replication. AIDS 2003, 17, 39–48. [Google Scholar] [CrossRef]
- Feng, Z.; Dubyak, G.R.; Lederman, M.M.; Weinberg, A. Cutting edge: Human beta-defensin 3—A novel antagonist of the HIV-coreceptor CXCR4. J. Immunol. 2006, 177, 782–786. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Diaz, S.; Valle, N.; Ferrer-Mayorga, G.; Lombardia, L.; Herrara, M.; Dominguez, O. MicroRNA-22 is induced by vitamin D and contributes to its antiproliferative, antimigratory and gene regulatory effects in colon cancer cells. Hum. Mol. Genet. 2012, 21, 2157–2165. [Google Scholar] [CrossRef] [PubMed]
- Ranijbar, S.; Jasenosky, L.D.; Chow, N.; Goldfield, A.E. Regulation of Mycobacterium tuberculosis-dependent HIV-1 transcription reveals a new role for NFAT5 in the toll-like receptor pathway. PLoS Pathogens 2012, 8, e1002620. [Google Scholar] [CrossRef] [PubMed]
- Nunnari, G.; Fagone, P.; Lazzara, F.; Longo, A.; Cambria, D.; Di Stefano, G. Vitamin D inhibitis TNFalpha-induced latent HIV reactivation in J-LAT cells. Mol. Cell. Biochem. 2016, 418, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Statistics South Africa. Mortality and Causes of Death in South Africa, 2011: Findings from Death Notification; Statistics South Africa: Pretoria, South Africa, 2014. [Google Scholar]
- Feldman, D.; Krishnan, A.V.; Swami, S.; Giovannucci, E.; Feldman, B.J. The role of vitamin D in reducing cancer risk and progression. Nat. Rev. Cancer 2014, 14, 342–357. [Google Scholar] [CrossRef] [PubMed]
- Saccone, D.; Asani, F.; Bornman, L. Regulation of the vitamin D receptor gene by environment, genetics and epigenetics. Gene 2015, 561, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Creemers, P.C.; Du Toit, E.D.; Kriel, J. DBP (vitamin D binding protein) and BF (properdin factor B) allele distribution in Namibian San and Khoi and in other South African populations. Gene Geogr. 1995, 9, 185–189. [Google Scholar] [PubMed]
- Luxwolda, M.F.; Kuipers, R.S.; Kema, I.P.; van der Veer, E.; Dijck-Brouwer, D.A.; Muskiet, F.A. Vitamin D status indicators in indigenous populations in East Africa. Eur. J. Nutr. 2013, 52, 1115–1125. [Google Scholar] [CrossRef] [PubMed]
- Ojwang, P.J.; Pegoraro, R.J.; Rom, L.; Lanning, P. Collagen Ialpha1 and vitamin D receptor gene polymorphisms in South African whites, blacks and Indians. East Afr. Med. J. 2001, 78, 604–607. [Google Scholar] [CrossRef] [PubMed]
- Lombard, Z.; Dalton, D.-L.; Venter, P.A.; Williams, R.C.; Bornman, L. Association of HLA-DR, -DQ, and vitamin D receptor alleles and haplotypes with tuberculosis in the Venda of South Africa. Hum. Immunol. 2006, 67, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Babb, C.; van der Merwe, L.; Beyers, N.; Pheiffer, C.; Walzl, G.; Duncan, K.; van Helden, P.; Hoal, E.G. Vitamin D receptor gene polymorphisms and sputum conversion time in pulmonary tuberculosis patients. Tuberculosis 2007, 87, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Andraos, C.; Koorsen, G.; Knight, J.C.; Bornman, L. Vitamin D receptor gene methylation is associated with ethnicity, tuberculosis, and TaqI polymorphism. Hum. Immunol. 2011, 72, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Kresfelder, T.L.; Janssen, R.; Bont, L.; Pretorius, M.; Venter, M. Confirmation of an association between single nucleotide polymorphisms in the VDR gene with respiratory syncytial virus related disease in South African children. J. Med. Virol. 2011, 83, 1834–1840. [Google Scholar] [CrossRef] [PubMed]
- Neill, V.; Asani, F.F.; Jeffery, T.J.; Saccone, D.S.; Bornman, L. Vitamin D receptor gene expression and function in a South African population: Ethnicity, vitamin D and FokI. PLoS ONE 2013, 8, e67663. [Google Scholar]
- Janssen, R.; Bont, L.; Siezen, C.L.; Hodemaekers, H.M.; Ermers, M.J.; Doornbos, G.; van’t Slot, R.; Wijmenga, C.; Goeman, J.J.; Kimpen, J.L.; et al. Genetic susceptibility to respiratory syncytial virus bronchiolitis is predominantly associated with innate immune genes. J. Infect. Dis. 2007, 196, 826–834. [Google Scholar] [CrossRef] [PubMed]
- Hart, P.H.; Gorman, S. Exposure to UV wavelengths in sunlight suppresses immunity. To what extent is UV-induced vitamin D3 the mediator responsible? Clin. Biochem. Rev. 2013, 34, 3–13. [Google Scholar] [PubMed]
| Parameter | Cape Town Latitude 35° S, Altitude 0–300 m | Pretoria Latitude 26° S, Altitude 1339 m | ||
|---|---|---|---|---|
| Summer | Winter | Summer | Winter | |
| Average hours of sunshine per day | 11 | 5 | 10 | 8 |
| Ultraviolet index | 9–10 | 2–3 | 11+ | 4–6 |
| Average temperature in °C, minimum/maximum | 16/26 | 7/18 | 18/28 | 5/20 |
| Study Population, Sampling Frame | Age (Years) | Location (Latitude), Season of Sampling when Specified | Assay | Serum or Plasma 25(OH)D Concentration Means (Unless Specified), nmol/L | Reference (Year) |
|---|---|---|---|---|---|
| Children | |||||
| 285 Coloured (mixed race) children; community based | 1–17 | Western Township, Johannesburg (26° S), winter | Competitive protein binding | 78.0 in age 1–12 y, 58.5 in age 13–14 y, 56.8 in age 15–16 y; none <25; no difference between boys and girls | [23] (1978) |
| 60 Black African children; school sample | 7–12 | Rural, small urban and large urban communities near Johannesburg (26° S) | Competitive protein binding | 72.3 in rural, 77.3 in small urban, 82.8 in large urban; none <25 | [24] (1979) |
| 114 hospitalised Black African infants; random hospital admissions | 0–2 | Witwatersrand (26° S), throughout year | Competitive protein binding | 37.3 aged 1–24 months, no correlation with age or season; <12.5 in 7%. 19.8 aged 0–1 month, probably reflecting vitamin D status of mother | [25] (1985) |
| 20 Black African pre-school children; cluster sample of villages | 3–5 | Villages in Northern Transvaal, now Limpopo (24° S), end of summer | Competitive protein binding | 85.5, no difference between underweight and normal weight children | [26] (1986) |
| 82 Black Africans with OCA, 58 Black Africans; school sample | 6–18 | Pietersburg, Northern Province (24° S) | Radioimmunoassay | 125 in OCA Blacks, 103 in Blacks, 6–9 y; 116 in OCA Blacks, 86.3 in Blacks, 10–13 y; 90.3 in OCA Blacks, 90.8 in Blacks,14–18 y | [27] (2000) |
| 295 Black African children, 90 White children; bone health subcohort of Birth-to-Twenty longitudinal cohort | 10 | Johannesburg (26° S), all seasons | Chemiluminescence (DiaSorin Liaison) | 100 in Black boys, 129 in White boys, 86 in Black girls, 112 in White girls; <50 in 8% Blacks and 1% Whites; higher values in White children in summer/autumn than in winter/spring, no seasonal variation in Blacks | [28] (2011) |
| Adults | |||||
| 43 healthy Black African women and cord blood of their babies, shortly after delivery; hospital based | 16–40 | Transkei (31° S) | Competitive protein binding | 81.8 in mothers; 171 in cord blood | [29] (1987) |
| 105 healthy White and 74 Black African premenopausal nurses; 50 healthy White and 65 Black African postmenopausal nurses; hospital based | 20–64 | Witwatersrand (26° S) | Competitive protein binding | Medians–65.8 in premenopausal White, 48.3 Black; 64.5 in postmenopausal White, 67.5 Black | [30] (1997) |
| 216 requests for vitamin D testing (39% with suspected osteoporosis); hospital based | All ages, peaks at 2 and 64 | Western Cape (32° S), all seasons | Competitive protein binding | Medians–48.3 (range 5.5–106); <45 in 41%; no seasonal effect on level | [31] (2009) |
| 658 rural healthy Black African women, 603 urban healthy Black African women; random selection from Prospective Urban and Rural Epidemiology Study or community based | >35 | North West Province (27° S), rural and urban seasonally matched | Roche Eledsys 2010 COBAS system | Levels decreased with age in both rural and urban women from about 78 at <50 y to about 65 at >70 y; levels lower in urban than in rural women, aged 50–70 y | [32] (2011) |
| 373 Black Africans, 344 Asian/Indians; cohort from Birth-to-Twenty longitudinal study | Mean 42 | Johannesburg-Soweto (26° S), all seasons | HPLC | 70.9 Blacks, 41.8 Asian/Indian; <30 3% Blacks and 13% Asian/Indians; females lower than males in both groups; highest level in autumn | [33] (2013) |
| 291 healthy urban Black African women; random selection from Prospective Urban and Rural Epidemiology Study or community based | >47 (mean 57.6) | North West Province (27° S) | Roche Elecsys 2010 COBAS system | 65; those with levels <75 two-times more likely to have higher systolic blood pressure than those with >75 (151 vs. 146 mmHg) | [34] (2013) |
| 368 healthy Black Africans and 347 healthy Asian/Indians; random selection from Birth-to-Twenty longitudinal study | 18–65 | Johannesburg (26° S) | HPLC | 58.3 in Black females, 72.7 in Black males, 35.7 in Asian/Indian females, 45.4 in Asian/Indian males | [35] (2014) |
| 371 healthy Black Africans and 343 healthy Asian/Indians; random selection from Birth-to-Twenty longitudinal study | 18–65 | Johannesburg (26° S) | HPLC | 56.8 in Black females, 72.4 in Black males, 32.4 in Asian/Indian females, 43.9 in Asian/Indian males; <30 in 5% Blacks and 28.6% Asian/Indians; levels 40%–60% higher in autumn than in winter/spring; little 25(OH)D2 | [36] (2014) |
| 502 Black Africans; population sample from Modelling of the Epidemiologic Transition Study | 25–45 (mean 33.4) | Cape Town (34° S), winter and summer months | LC-MS/MS | 59.3; <30 in 6.6%, >50 in 65.9%; negative correlation of 25(OH)D level with distance from the equator (by comparing levels in Blacks living at latitudes 41° N, 17° N, 6° N, 4° S and Cape Town) | [37] (2014) |
| 50 healthy Black Africans, 50 healthy Coloured (Cape mixed); community based longitudinal study | 18–24 | Cape Town (34° S), winter and summer months | Chemiluminescence (DiaSorin, Liaison) | Medians: 72.6 Black, 65.5 Coloured in summer; 45.5 Black, 43.8 Coloured in winter | [38] (2015) |
| Elderly | |||||
| 232 patients with femoral neck fractures, ethnicity not specified; hospital admissions | Mean 72.7 | Johannesburg (26° S), throughout year | Competitive protein binding | 44.3 throughout year; 51 in summer/autumn, 38.1 in winter and spring; <25 in 17% subjects in winter/spring | [39] (1978) |
| 60 females living in old-age homes, ethnicity not specified | Mean 80 | Pretoria (26° S), winter | Not specified | 32 | [40] (1991) |
| 173 non-institutionalised Coloured (mixed race), 52% women; population sample | 65–92 (mean 73.7) | Cape Town (34° S), late winter | Not specified | 37; <25 in 17% | [41] (1996) |
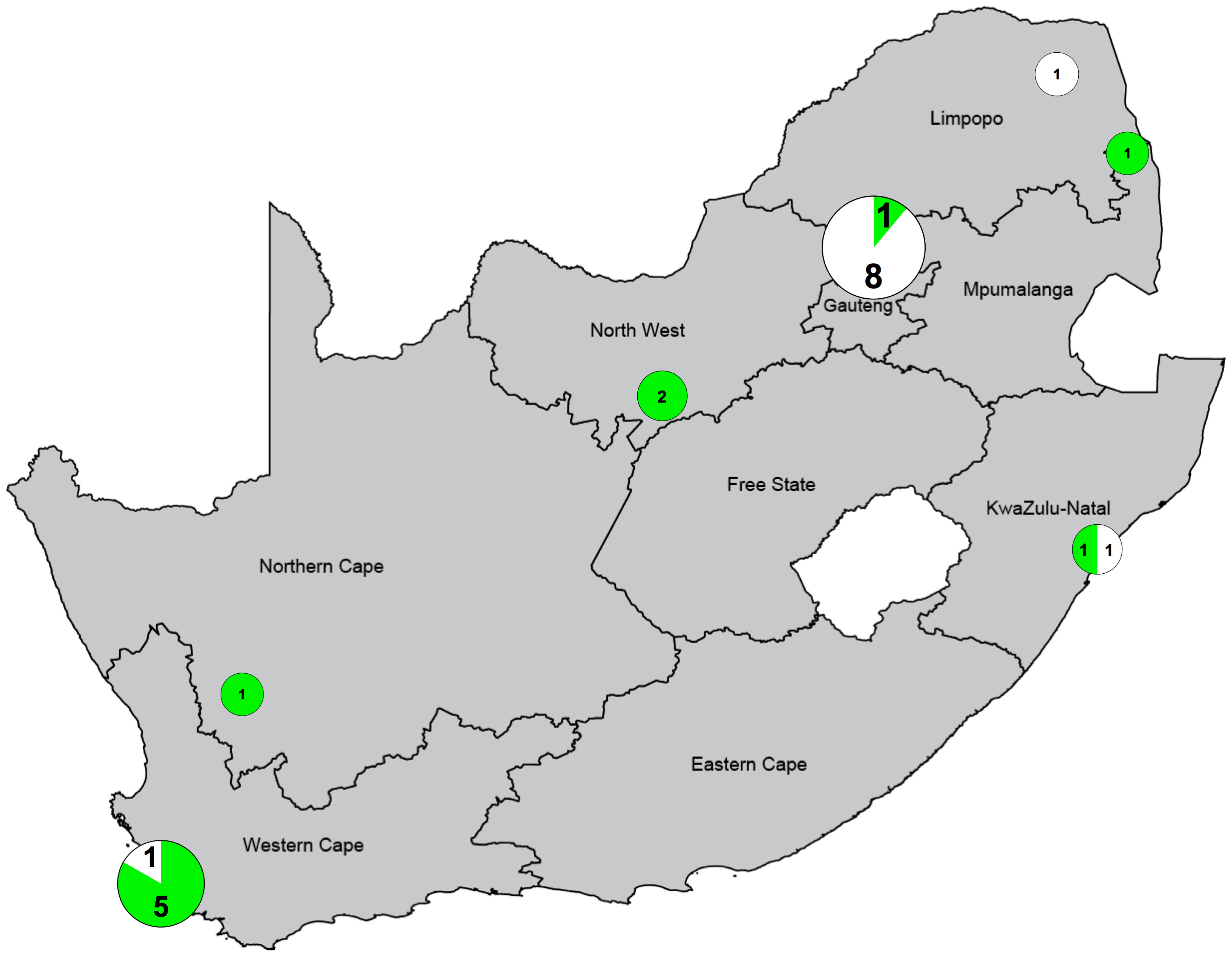
| Category | Disease/Study Groups | Assoc 1 | Study Type | Location | Population | Sample Size | Age Group | Gender | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Bone | Fractures | Yes | longitudinal | Gauteng | NA | 20, 20 | 6–29 y | M & F | [59] |
| Bone | Rickets | Yes | cross-sectional | KwaZulu-Natal | Black African | 37 | 1–12 y | M & F | [60] |
| Bone | Bone mineral density | Yes | cross-sectional | North-West | Black African | 658 | >45 y | F | [32] |
| Bone | Growth stunting | Yes | cross-sectional | Northern Cape | NA | 150 | 2–5 y | M & F | [57] |
| Bone | Rickets | No | cross-sectional | Gauteng | Black African | 114 | <2 y | M & F | [25] |
| Bone | Metabolic bone disease | No | cross-sectional | Gauteng | Black African | 26 | 16–19 y | M | [61] |
| Bone | Bone mineral density | No | cross-sectional | Gauteng | Black & Asian-Indian | 371, 343 | 18–65 y | M & F | [35] |
| Bone | Under weight vs. normal weight | No | case-control | Limpopo | Black African | 145 | 3–5 y | M & F | [22] |
| Bone/ID | BMD in HIV uninfected vs. HIV high CD4 vs. HIV low CD4 count | No | case-control | Gauteng | Black African | 98, 74, 75 | ≥18 y | F | [62] |
| ID | Schistosomiasis (PZQ vs. PZQ + vitamin D vs. vitamin D vs. placebo) | Yes 2 | RCT | Mozambique border | NA | 14, 16, 14, 15 | 14–18 y | M | [63] |
| ID | TB-Meningitis | Yes 3 | case-control | Western Cape | Black & Coloured | 42, 147 | 0–13 y | M & F | [64] |
| ID | HIV replication (Summer vs. winter vs. winter + vitamin D) | Yes | longitudinal | Western Cape | Black African | 30 | 18–24 y | M & F | [38] |
| ID | TB HIV (TB vs. HIV vs. TB-HIV vs. OD) | Yes | case-control | Western Cape | Black African | 93, 75, 99, 103 | ≥18 y | M & F | [65] |
| ID | HIV-Cryptococcal Meningitis vs. HIV | No 4 | case-control | Western Cape | NA | 150, 150 | ≥21 y | M & F | [66] |
| ID | HIV ART initiation | NA 5 | cross-sectional | Gauteng/KWN | NA | 270 | ≥18 y | M & F | [67] |
| ID | HIV ART initiation | NA 5 | cross-sectional | Gauteng/KWN | NA | 270 | ≥18 y | M & F | [68] |
| ID | Paradoxical TB-HIV IRIS vs TB-HIV no IRIS | No | case-control | KwaZulu-Natal | Black African | 11, 11 | 24–50 y | M & F | [69] |
| ID | ART-associated TB vs HIV+TB- | No | case-control | KwaZulu-Natal | Black African | 18, 38 | 23–57 y | M & F | [69] |
| NCD | Cardiovascular disease (blood pressure and pulse) | Yes | cross-sectional | North-West | Black African | 291 | >47 y | F | [34] |
| NCD | Metabolic syndrome | No | cross-sectional | Gauteng | Black & Asian-Indian | 374, 350 | 18–65 y | M & F | [33] |
| NCD | Obesity (total body fat, fat distribution) | No | cross-sectional | Gauteng | Black & Asian-Indian | 371, 343 | 18–65 y | M & F | [36] |
| Nutrition | Alzheimer’s zinc deficiency (Zn vs. Zn ± vitamin A ± vitamin D) | Yes 6 | RCT | Western Cape | NA | 70/group | 55 y | M | [70] |
| Nutrition | Alcohol use disorders vs. matched controls | Yes | cross-sectional | Western Cape | Mixed ancestry | 81, 81 | 12–16 y | M & F | [71] |
| SNP (rs) | Study Population; Gender when Specified | Factor Investigated | Study Design | Median/Mean Age, Years (Range) | Association | Reference |
|---|---|---|---|---|---|---|
| FokI (rs2544037) (rs10783219) (rs10735810) TaqI (rs731236) | 22 male, 28 female Cape mixed; 26 male, 24 female Black Africans (Zhosa) | Determinants of 25(OH)D status, before and after vitamin D supplementation | Longitudinal | (18–24) | FokI (rs10735810) “AG” contributed to lower 25(OH)D, and lower 25(OH)D in winter and post-supplementation, while “AA” contributed to higher 25(OH)D, in a linear model incorporating UVB exposure, skin pigmentation, ethnicity, age and sex | [38] |
| ApaI (rs7975232) TaqI (rs731236) | 264 Black Africans, 247 Whites, 194 Asian-Indians | Ethnicity | Retrospective cross-sectional cohort of healthy male and female blood donors | NA | Higher ApaI “AA” and TaqI “TT” in Blacks than Whites or Asian-Indians; no difference between Whites and Asian-Indians. Higher frequency of TaqI “T” may contribute to lower incidence of osteoporosis in Blacks | [85] |
| FokI (rs2228570) BsmI (rs1544410) ApaI (rs7975232) TaqI (rs731236) | 95 Black Africans (Venda) with TB, 117 ethnicity matched controls | TB | Case-control | NA | No independent SNP association; FokI-BsmI-ApaI-TaqI haplotype “F-b-A-T” (C-G-T-T) protects | [86] |
| FokI (rs2228570) ApaI (rs7975232) TaqI (rs731236) | All Colored: 249 TB cases, 352 ethnicity matched controls, 220 TB cases converting from positive to negative | TB and chemotherapy for TB | Case-control for TB; longitudinal for TB conversion | (18–65) | No association of VDR allele, genotype, haplotype or diplotype with TB. Quicker response to TB chemotherapy in ApaI “AA” (TT) vs. “aa” (GG), in TaqI “Tt” (TC) vs. “tt” (CC) and in TaqI “TT” (TT) vs. “tt” (CC) | [87] |
| ApaI (rs7975232) TaqI (rs731236) | 15 male, 15 female CAU (North America); 15 male, 15 female YRI (Nigeria); 16 male, 16 female Black Africans (Venda) with TB together with 12 male and 17 female healthy Black African controls | Ethnicity, TB, and DNA methylation of the VDR 3’ region CGI 1060 | Ethnicity; case-control for TB | CAU 32 (22–44), YRI unknown, TB cases 38 (18–62) TB controls 34 (21–62) | Methylation variable positions in 3’ end of VDR distinguish ethnicity and TB status. Higher regional methylation in TaqI “TC/CC” than “TT” in YRI and Venda, but not in CAU | [88] |
| FokI (rs10735810 merged into rs2228570) | 296 Black Africans with RSV, 113 Black African controls | RSV-related disease | Case-control | Cases 3.0 months, controls 3.5 months | FokI “C” frequency higher in Black South Africans than European, Asian and Japanese populations. “CT” and “T” predispose to RSV disease, while “CC” protects | [89] |
| FokI (rs2228570) | 40 healthy Black Africans, 20 healthy Whites | VDR expression and VDR function, considering ethnicity and 25(OH)D levels | Cross-sectional cohort of male and female blood donors | 35 (17–65) | Higher frequency FokI “CC” genotype and VDR protein in Blacks than Whites, but less VDR mRNA and CAMP mRNA. CAMP mRNA higher in FokI “CT/TT” genotypes than “CC”. Circulating 25(OH)D levels ≥50 nmol/L, and comparable between Blacks and Whites. Different effects on CAMP and CYP24A1 expression in Whites and Blacks on supplementation with 1,25(OH)2D | [90] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Norval, M.; Coussens, A.K.; Wilkinson, R.J.; Bornman, L.; Lucas, R.M.; Wright, C.Y. Vitamin D Status and Its Consequences for Health in South Africa. Int. J. Environ. Res. Public Health 2016, 13, 1019. https://doi.org/10.3390/ijerph13101019
Norval M, Coussens AK, Wilkinson RJ, Bornman L, Lucas RM, Wright CY. Vitamin D Status and Its Consequences for Health in South Africa. International Journal of Environmental Research and Public Health. 2016; 13(10):1019. https://doi.org/10.3390/ijerph13101019
Chicago/Turabian StyleNorval, Mary, Anna K. Coussens, Robert J. Wilkinson, Liza Bornman, Robyn M. Lucas, and Caradee Y. Wright. 2016. "Vitamin D Status and Its Consequences for Health in South Africa" International Journal of Environmental Research and Public Health 13, no. 10: 1019. https://doi.org/10.3390/ijerph13101019
APA StyleNorval, M., Coussens, A. K., Wilkinson, R. J., Bornman, L., Lucas, R. M., & Wright, C. Y. (2016). Vitamin D Status and Its Consequences for Health in South Africa. International Journal of Environmental Research and Public Health, 13(10), 1019. https://doi.org/10.3390/ijerph13101019







