Synthesis of High Valence Silver-Loaded Mesoporous Silica with Strong Antibacterial Properties
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
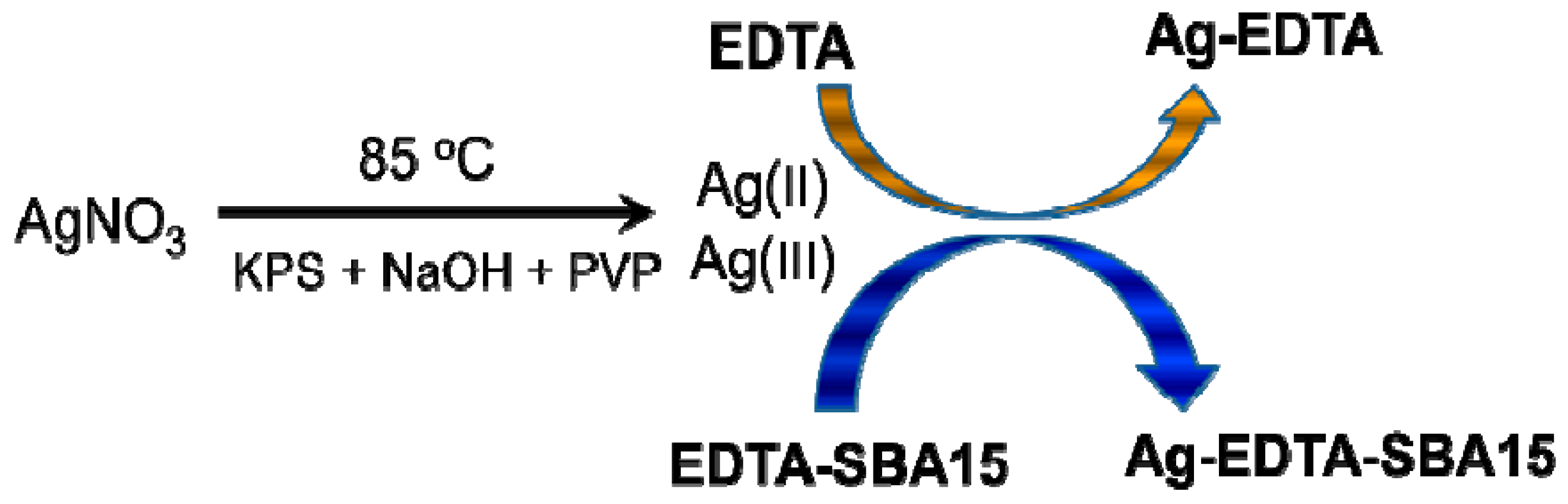
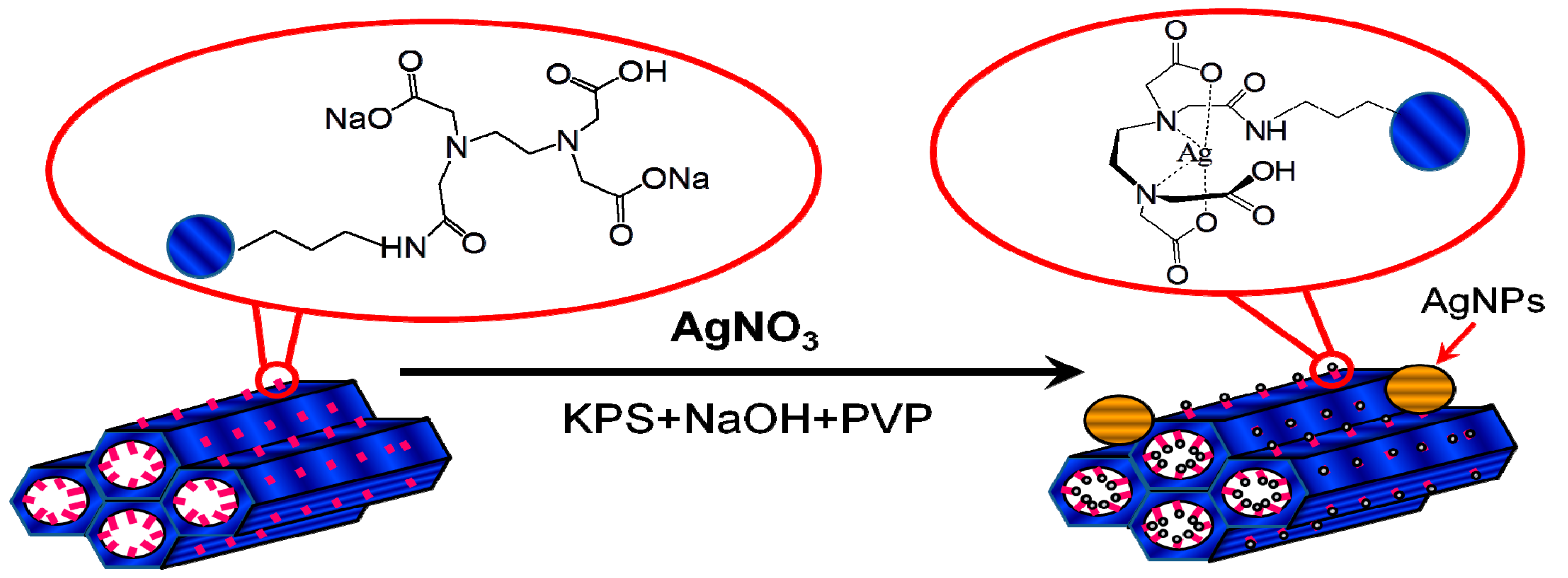
2.2. Synthesis of High Valence Ag-EDTA Complex
2.3. Synthesis of Functionalized Mesoporous Silica
2.4. Synthesis of High Valence Ag-Loaded Mesoporous Silica
2.5. Instruments and Characterization
2.6. Bacteria Culture for Antibacterial Activity
2.7. Disc Diffusion Assay
2.8. Microdilution Method for Minimum Inhibitory Concentration Assay
3. Results and Discussion
3.1. Formation of High Valence Ag Materials
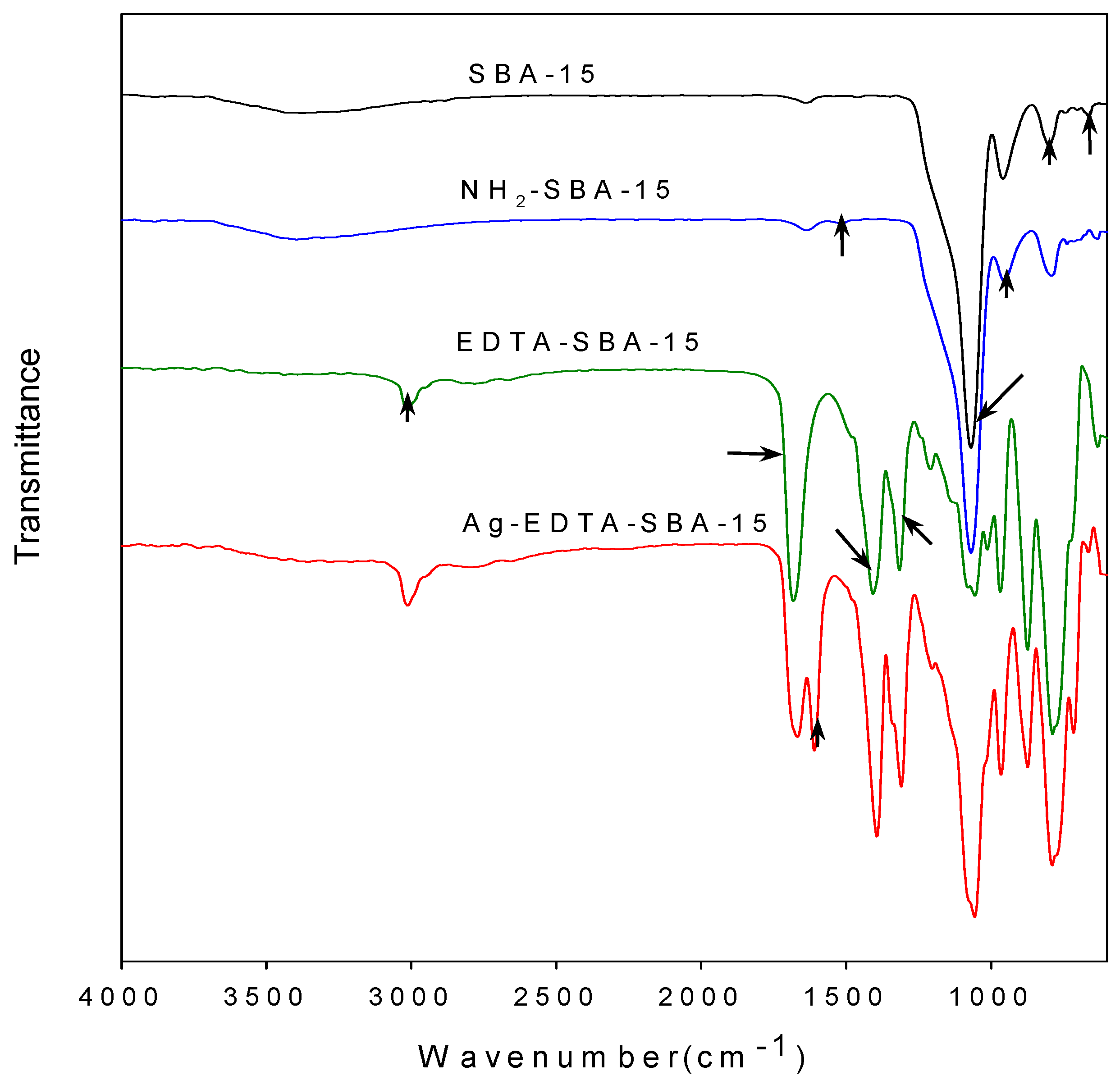
3.2. FTIR Analysis
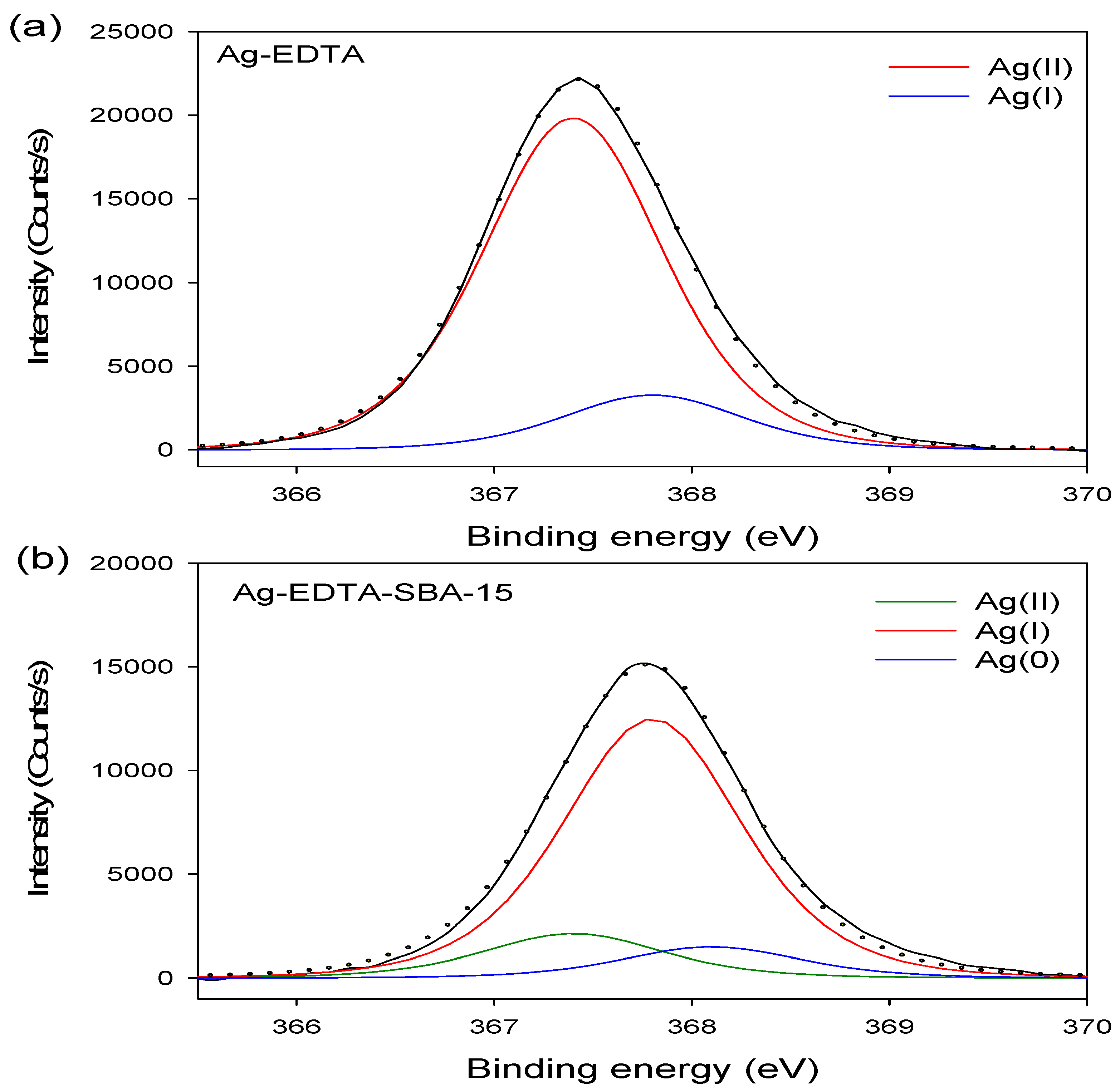
3.3. XPS Analysis
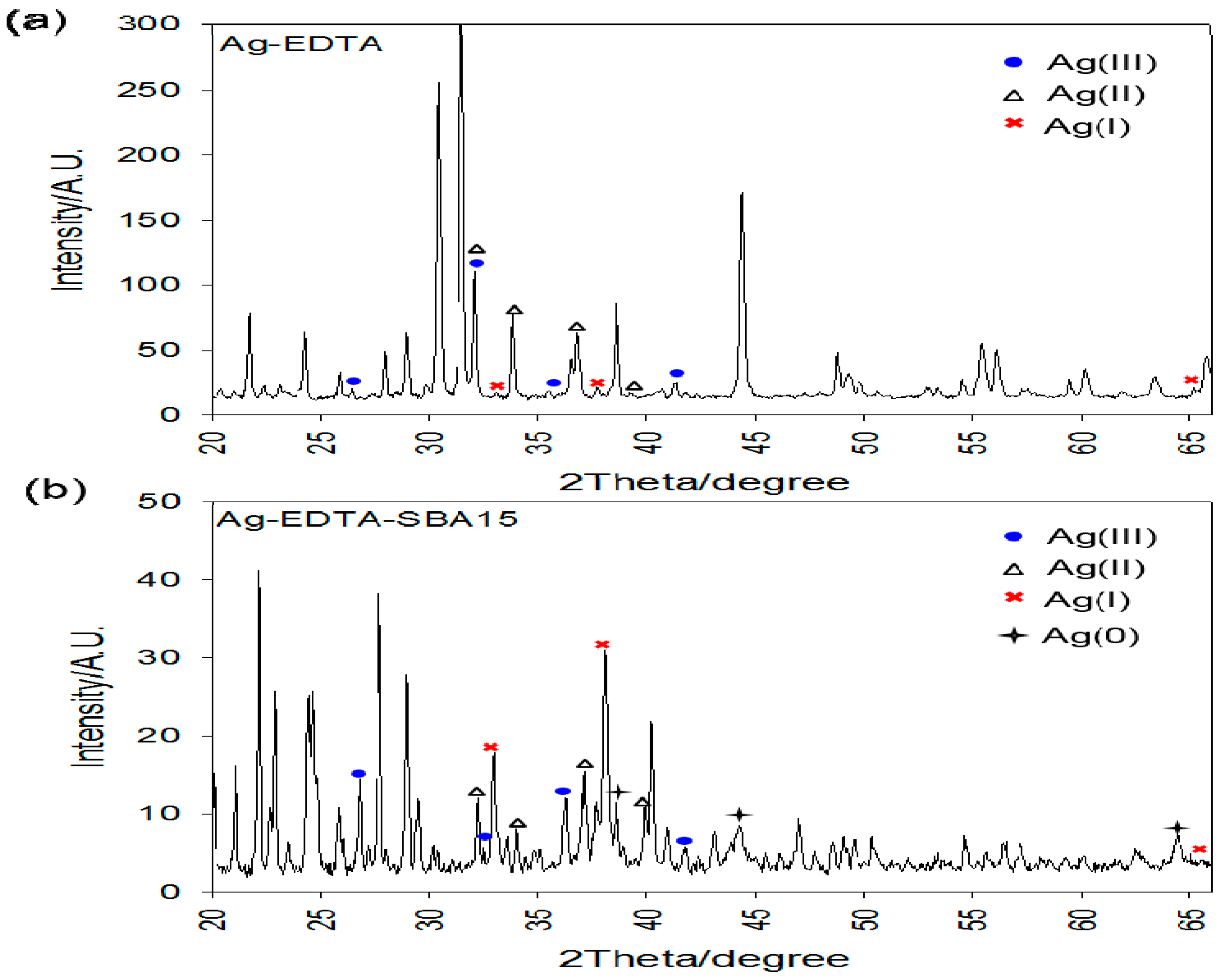
3.4. X-ray Diffraction Analysis
3.5. TEM Analysis

| Sample | SBET (m2/g) | Vp (cm3/g) |
|---|---|---|
| SBA-15 | 341 | 0.48 |
| Ag-EDTA-SBA-15 | 311 | 0.45 |
3.6. EDX Analysis

| Label | A | B | C | D |
|---|---|---|---|---|
| O | 10.6 | 10.9 | 10.5 | 0.94 |
| Si | 76.0 | 73.8 | 72.3 | 12.8 |
| Ag | 11.1 | 12.9 | 14.4 | 86.3 |
3.7. Antibacterial Activity
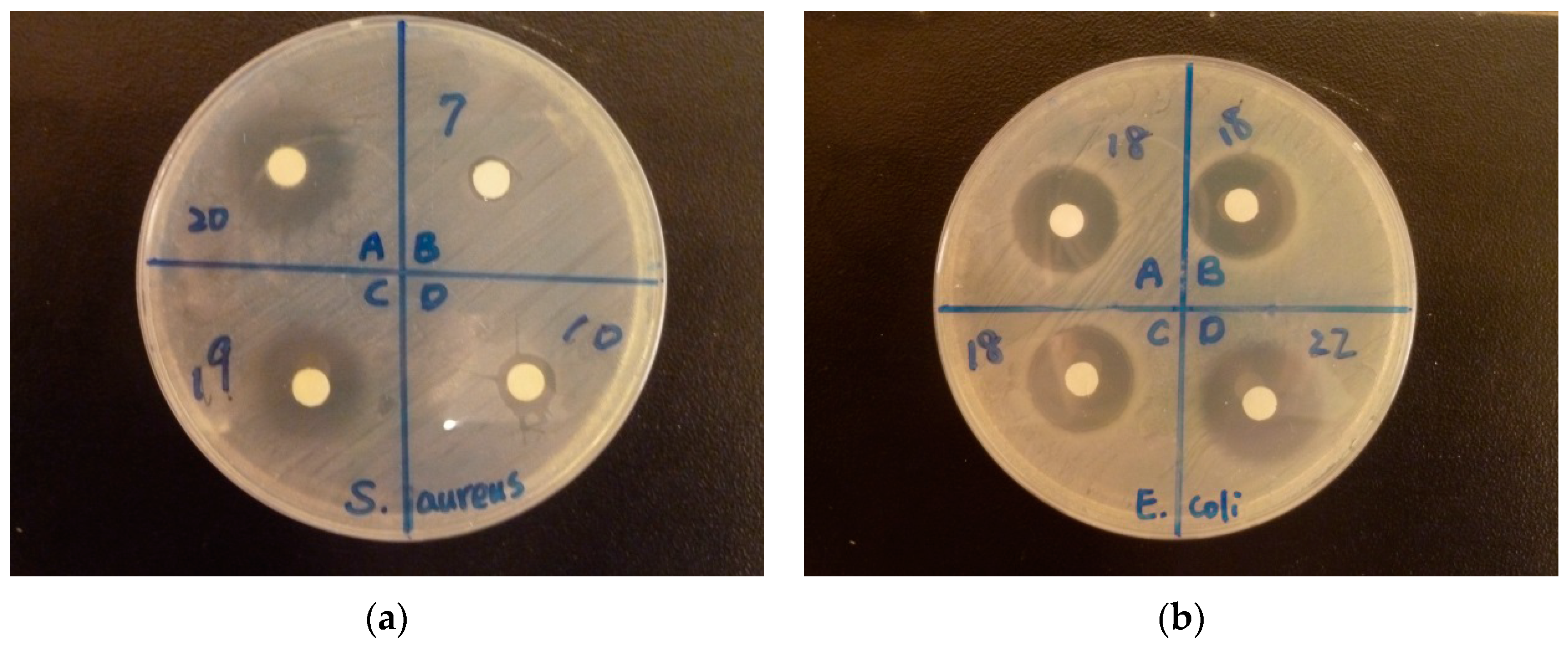
| Sample | Inhibitory Zone (mm) | |||||
| E. coli | S. aureus | |||||
| 100 mg/mL | 10 mg/mL | 1 mg/mL | 100 mg/mL | 10 mg/mL | 1 mg/mL | |
| EDTA | 9.2 ± 1.9 | 6 ± 0 | 6 ± 0 | 7.7 ± 0.5 | 6 ± 0 | 6 ± 0 |
| SBA-15 | 6 ± 0 | 6 ± 0 | 6 ± 0 | 6 ± 0 | 6 ± 0 | 6 ± 0 |
| EDTA-SBA-15 | 19.5 ± 2.8 | 6 ± 0 | 6 ± 0 | 20.5 ± 0.9 | 6 ± 0 | 6 ± 0 |
| Ag-EDTA | 23.3 ± 1.2 | 12.7 ± 2.1 | 10.5 ± 0.7 | 23 ± 0 | 11.3 ± 0.6 | 8.7 ± 0.6 |
| Ag-EDTA-SBA-15 | 20.3 ± 0.6 | 11.7 ± 1.2 | 9 ± 1 | 21.3 ± 0.6 | 11.5 ± 0.5 | 6.5 ± 0 |
| IONPURE | 9.8 ± 0.3 | 8 ± 0.5 | 6.5 ± 0 | 10.5 ± 0.9 | 6.5 ± 0 | 6 ± 0 |
| MNS-Ag-n30 | 7.5 ± 0 | 6 ± 0 | 6 ± 0 | 6.5 ± 0 | 6 ± 0 | 6 ± 0 |
| Minimum Inhibitory Concentrations (μg/mL) | ||||||
| E. coli | S. aureus | |||||
| MIC50 | MIC90 | MIC99 | MIC50 | MIC90 | MIC99 | |
| EDTA-SBA-15 | (NO Bacterial Inhibition) | (NO Bacterial Inhibition) | ||||
| Ag-EDTA | 3.4 | 9.4 | 16.1 | 11.4 | 15.4 | 16.8 |
| Ag-EDTA-SBA-15 | 30 | 39 | 42 | 30 | 47 | 77 |
3.8. Antibacterial Durability
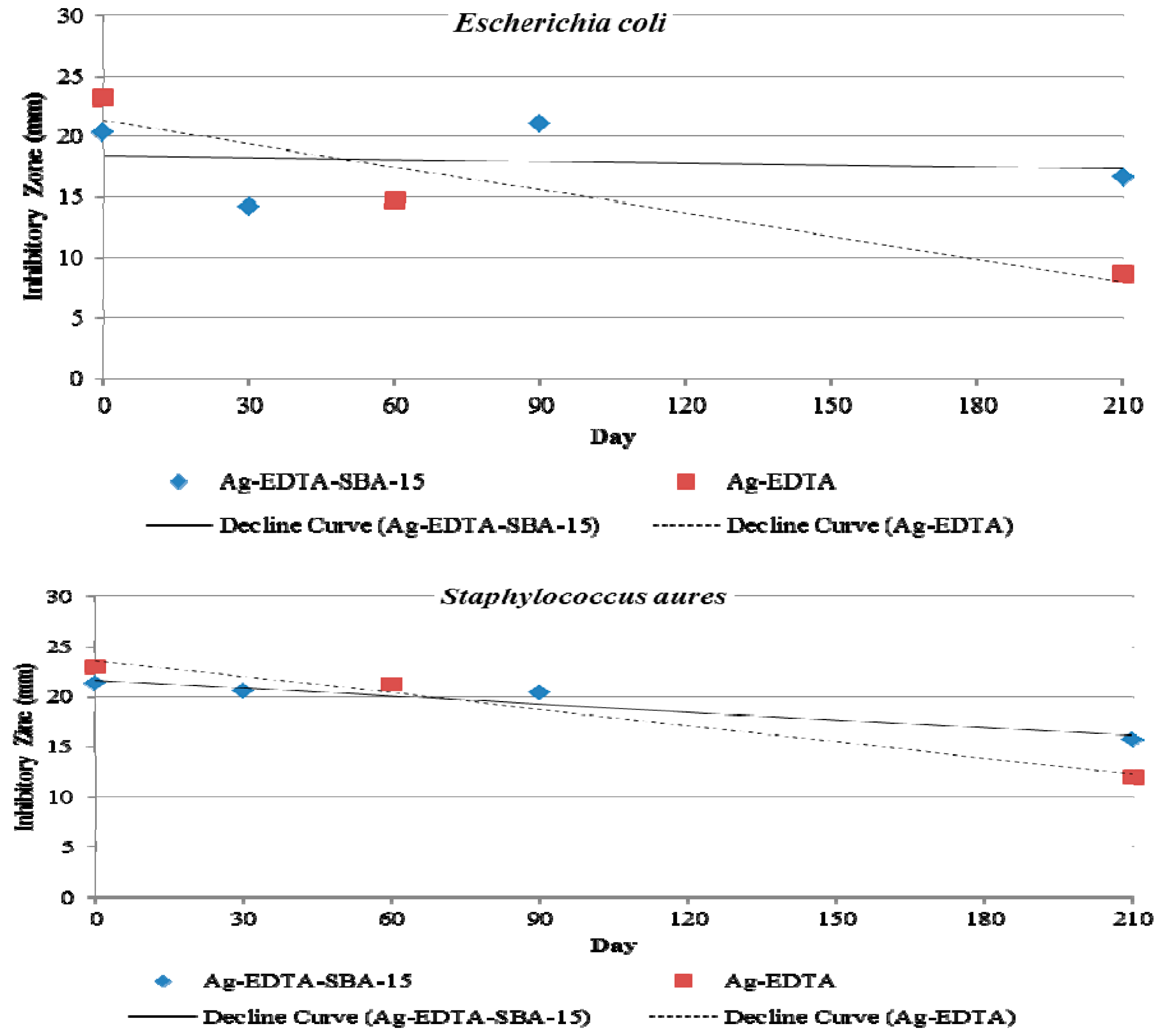
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cho, K.-H.; Park, J.-E.; Osaka, T.; Park, S.-G. The study of antimicrobial activity and preservative effects of nanosilver ingredient. Electrochim. Acta 2005, 51, 956–960. [Google Scholar] [CrossRef]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Wang, J.-X.; Kang, X.-L.; Chen, J.-F. Fabrication of antibacterial monodispersed Ag–SiO2 core–shell nanoparticles with high concentration. Mater. Lett. 2009, 63, 31–33. [Google Scholar] [CrossRef]
- Kokura, S.; Handa, O.; Takagi, T.; Ishikawa, T.; Naito, Y.; Yoshikawa, T. Silver nanoparticles as a safe preservative for use in cosmetics. Nanomed.: Nanotechnol. Biol. Med. 2010, 6, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Jain, J.; Arora, S.; Rajwade, J.M.; Omray, P.; Khandelwal, S.; Paknikar, K.M. Silver nanoparticles in therapeutics: Development of an antimicrobial gel formulation for topical use. Mol. Pharm. 2009, 6, 1388–1401. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.; Pradeep, T. Potential of silver nanoparticle-coated polyurethane foam as an antibacterial water filter. Biotechnol. Bioeng. 2005, 90, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Roe, D.; Karandikar, B.; Bonn-Savage, N.; Gibbins, B.; Roullet, J.B. Antimicrobial surface functionalization of plastic catheters by silver nanoparticles. J. Antimicrob. Chemother. 2008, 61, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Melaiye, A.; Sun, Z.; Hindi, K.; Milsted, A.; Ely, D.; Reneker, D.H.; Tessier, C.A.; Youngs, W.J. Silver(I)-Imidazole cyclophane gem-diol complexes encapsulated by electrospun tecophilic nanofibers: Formation of nanosilver particles and antimicrobial activity. J. Am. Chem. Soc. 2005, 127, 2285–2291. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Yoon, E.J.; Tak, Y.K.; Choi, E.C.; Song, J.M. Synthesis of highly antibacterial nanocrystalline trivalent silver polydiguanide. J. Am. Chem. Soc. 2009, 131, 16147–16155. [Google Scholar] [CrossRef] [PubMed]
- Atiyeh, B.S.; Costagliola, M.; Hayek, S.N.; Dibo, S.A. Effect of silver on burn wound infection control and healing: Review of the literature. Burns 2007, 33, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Dellasega, D.; Facibeni, A.; Fonzo, F.D.; Bogana, M.; Polissi, A.; Conti, C.; Ducati, C.; Casari, C.S.; Bassi, A.L.; Bottani, C.E. Nanostructured Ag4O4 films with enhanced antibacterial activity. Nanotechnology 2008, 19, 475602. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Feng, L.; Feng, H.; Kong, Z.; Guo, M. Ultrafine silver(II) oxide particles decorated porous ceramic composites for water treatment. Chem. Eng. J. 2011, 175, 592–599. [Google Scholar] [CrossRef]
- Lalueza, P.; Monzón, M.; Arruebo, M.; Santamaría, J. Bactericidal effects of different silver-containing materials. Mater. Res. Bull. 2011, 46, 2070–2076. [Google Scholar] [CrossRef]
- Levason, W.; Spicer, M.D. The chemistry of copper and silver in their higher oxidation states. Coord. Chem. Rev. 1987, 76, 45–120. [Google Scholar] [CrossRef]
- Liong, M.; France, B.; Bradley, K.A.; Zink, J.I. Antimicrobial activity of silver nanocrystals encapsulated in mesoporous silica nanoparticles. Adv. Mater. 2009, 21, 1684–1689. [Google Scholar] [CrossRef]
- Balogh, L.; Swanson, D.R.; Tomalia, D.A.; Hagnauer, G.L.; McManus, A.T. Dendrimer-silver complexes and nanocomposites as antimicrobial agents. Nano Lett. 2001, 1, 18–21. [Google Scholar] [CrossRef]
- Lee, H.Y.; Park, H.K.; Lee, Y.M.; Kim, K.; Park, S.B. A practical procedure for producing silver nanocoated fabric and its antibacterial evaluation for biomedical applications. Chem. Commun. 2007, 28, 2959–2961. [Google Scholar] [CrossRef] [PubMed]
- Oyanedel-Craver, V.A.; Smith, J.A. Sustainable colloidal-silver-impregnated ceramic filter for point-of-use water treatment. Environ. Sci. Technol. 2008, 42, 927–933. [Google Scholar] [CrossRef] [PubMed]
- Loher, S.; Schneider, O.D.; Maienfisch, T.; Bokorny, S.; Stark, W.J. Micro-organism-triggered release of silver nanoparticles from biodegradable oxide carriers allows preparation of self-sterilizing polymer surfaces. Small 2008, 4, 824–832. [Google Scholar] [CrossRef] [PubMed]
- Schneider, O.D.; Loher, S.; Brunner, T.J.; Schmidlin, P.; Stark, W.J. Flexible, silver containing nanocomposites for the repair of bone defects: Antimicrobial effect against E. coli infection and comparison to tetracycline containing scaffolds. J. Mater. Chem. 2008, 18, 2679–2684. [Google Scholar] [CrossRef]
- Gunawan, C.; Teoh, W.Y.; Marquis, C.P.; Lifia, J.; Amal, R. Reversible antimicrobial photoswitching in nanosilver. Small 2009, 5, 341–344. [Google Scholar] [CrossRef] [PubMed]
- Lok, C.-N.; Ho, C.-M.; Chen, R.; He, Q.-Y.; Yu, W.-Y.; Sun, H.; Tam, P.-H.; Chiu, J.-F.; Che, C.-M. Silver nanoparticles: Partial oxidation and antibacterial activities. J. Biol. Inorg. Chem. 2007, 12, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Tak, Y.K.; Song, J.M. Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the gram-negative bacterium escherichia coli. Appl. Environ. Microbiol. 2007, 73, 1712–1720. [Google Scholar] [CrossRef] [PubMed]
- Aruguete, D.M.; Kim, B.; Hochella, M.F.; Ma, Y.; Cheng, Y.; Hoegh, A.; Liu, J.; Pruden, A. Antimicrobial nanotechnology: Its potential for the effective management of microbial drug resistance and implications for research needs in microbial nanotoxicology. Environ. Sci.: Process. Impacts 2013, 15, 93–102. [Google Scholar] [CrossRef]
- Jin, R.; Cao, Y.; Mirkin, C.A.; Kelly, K.L.; Schatz, G.C.; Zheng, J.G. Photoinduced conversion of silver nanospheres to nanoprisms. Science 2001, 294, 1901–1903. [Google Scholar] [CrossRef] [PubMed]
- Sohn, B.H.; Cohen, R.E.; Papaefthymiou, G.C. Magnetic properties of iron oxide nanoclusters within microdomains of block copolymers. J. Magn. Magn. Mater. 1998, 182, 216–224. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Lu, Y.-C.; Chou, K.-S. Enhancement of photoluminescence of different quantum dots by Ag@SiO2 core-shell nanoparticles. Mater. Res. Bull. 2013, 48, 2076–2078. [Google Scholar] [CrossRef]
- Kim, Y.H.; Lee, D.K.; Cha, H.G.; Kim, C.W.; Kang, Y.S. Synthesis and characterization of antibacterial Ag−SiO2 nanocomposite. J. Phys. Chem. 2007, 111, 3629–3635. [Google Scholar]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.-H.; Park, S.J.; Lee, H.J.; Kim, S.H.; Park, Y.K.; Park, Y.H.; Hwang, C.-Y.; et al. Antimicrobial effects of silver nanoparticles. Nanomed.: Nanotechnol. Biol. Med. 2007, 3, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Min, S.-H.; Yang, J.-H.; Kim, J.Y.; Kwon, Y.-U. Development of white antibacterial pigment based on silver chloride nanoparticles and mesoporous silica and its polymer composite. Microporous Mesoporous Mater. 2010, 128, 19–25. [Google Scholar] [CrossRef]
- Ying, J.Y.; Mehnert, C.P.; Wong, M.S. Synthesis and applications of supramolecular-templated mesoporous materials. Angew. Chem. Int. Ed. 1999, 38, 56–77. [Google Scholar] [CrossRef]
- Wu, P.; Tatsumi, T.; Komatsu, T.; Yashima, T. Postsynthesis, Characterization, and catalytic properties in alkene epoxidation of hydrothermally stable mesoporous Ti-SBA-15. Chem. Mater. 2002, 14, 1657–1664. [Google Scholar] [CrossRef]
- Zhao, D.; Feng, J.; Huo, Q.; Melosh, N.; Fredrickson, G.H.; Chmelka, B.F.; Stucky, G.D. Triblock copolymer syntheses of mesoporous silica with periodic 50 to 300 angstrom pores. Science 1998, 279, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-C.; Bai, H.; Chiang, H.-S.; Karthik, M.; Li, S.-N.; Hsu, J.-N.; Shih, H.-Y. Development of regenerative dye impregnated mesoporous silica materials for assessing exposure to ammonia. J. Air Waste Manage. Assoc. 2012, 62, 838–845. [Google Scholar] [CrossRef]
- Vinu, A.; Hossain, K.Z.; Ariga, K. Recent advances in functionalization of mesoporous silica. J. Nanosci. Nanotechnol. 2005, 5, 347–371. [Google Scholar] [CrossRef] [PubMed]
- Quang, D.V.; Sarawade, P.B.; Hilonga, A.; Park, S.D.; Kim, J.-K.; Kim, H.T. Facile route for preparation of silver nanoparticle-coated precipitated silica. Appl. Surf. Sci. 2011, 257, 4250–4256. [Google Scholar] [CrossRef]
- Lee, J.-M.; Kim, D.-W.; Kim, T.-H.; Oh, S.-G. Facile route for preparation of silica-silver heterogeneous nanocomposite particles using alcohol reduction method. Mater. Lett. 2007, 61, 1558–1562. [Google Scholar] [CrossRef]
- Lv, Y.; Liu, H.; Wang, Z.; Liu, S.; Hao, L.; Sang, Y.; Liu, D.; Wang, J.; Boughton, R.I. Silver nanoparticle-decorated porous ceramic composite for water treatment. J. Membr. Sci. 2009, 331, 50–56. [Google Scholar] [CrossRef]
- Pallavicini, P.; Taglietti, A.; Dacarro, G.; Antonio Diaz-Fernandez, Y.; Galli, M.; Grisoli, P.; Patrini, M.; Santucci de Magistris, G.; Zanoni, R. Self-assembled monolayers of silver nanoparticles firmly grafted on glass surfaces: Low Ag+ release for an efficient antibacterial activity. J. Colloid Interface Sci. 2010, 350, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Repo, E.; Kurniawan, T.A.; Warchol, J.K.; Sillanpää, M.E.T. Removal of Co (II) and Ni (II) ions from contaminated water using silica gel functionalized with EDTA and/or DTPA as chelating agents. J. Hazard. Mater. 2009, 171, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Repo, E.; Warchol, J.K.; Kurniawan, T.A.; Sillanpää, M.E.T. Adsorption of Co (II) and Ni (II) by EDTA- and/or DTPA-modified chitosan: Kinetic and equilibrium modeling. Chem. Eng. J. 2010, 161, 73–82. [Google Scholar] [CrossRef]
- Pereira, F.V.; Gurgel, L.V.A.; Gil, L.F. Removal of Zn2+ from aqueous single metal solutions and electroplating wastewater with wood sawdust and sugarcane bagasse modified with EDTA dianhydride (EDTAD). J. Hazard. Mater. 2010, 176, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Tong, M.; Sun, X.; Li, B. Enhanced and selective adsorption of Pb2+ and Cu2+ by EDTAD-modified biomass of baker’s yeast. Bioresource Technol. 2008, 99, 2588–2593. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Ye, M.; Qu, Y.; Chu, L.; Chen, R.; He, Q.; Xu, D. Pb (II) removal from aqueous media by EDTA-modified mesoporous silica SBA-15. J. Colloid Interface Sci. 2012, 385, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Rajesh, M.; Sen, J.; Srujan, M.; Mukherjee, K.; Sreedhar, B.; Chaudhuri, A. Dramatic influence of the orientation of linker between hydrophilic and hydrophobic lipid moiety in liposomal gene delivery. J. Am. Chem. Soc. 2007, 129, 11408–11420. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-J.; Zeng, F.-X.; Jiang, H.; Zhang, X.-S. Adsorption of lead (Pb) from aqueous solution with Typha angustifolia biomass modified by SOCl2 activated EDTA. Chem. Eng. J. 2011, 170, 21–28. [Google Scholar] [CrossRef]
- Li, C.-S.; Tsai, Y.-H.; Lee, W.-C.; Kuo, W.-J. Synthesis and photophysical properties of Pyrrole/Polycyclic aromatic units hybrid fluorophores. J. Org. Chem. 2010, 75, 4004–4013. [Google Scholar] [CrossRef] [PubMed]
- Chicharro, R.; Alonso, M.; Arán, V.J.; Herradón, B. Studies on calpain inhibitors. Synthesis of partially reduced isoquinoline-1-thione derivatives and conversion to functionalized 1-chloroisoquinolines. Tetrahedron Lett. 2008, 49, 2275–2279. [Google Scholar] [CrossRef]
- Alba, M.D.; Luan, Z.; Klinowski, J. Titanosilicate mesoporous molecular sieve MCM-41: Synthesis and characterization. J. Phys. Chem. 1996, 100, 2178–2182. [Google Scholar] [CrossRef]
- Maria Chong, A.S.; Zhao, X.S. Functionalization of SBA-15 with APTES and characterization of functionalized materials. J. Phys. Chem. 2003, 107, 12650–12657. [Google Scholar] [CrossRef]
- Schon, G. ESCA Studies of Ag, Ag2O and AgO. Acta Chem. Scand. 1973, 27, 2623–2633. [Google Scholar] [CrossRef]
- Wang, X.; Li, S.; Yu, H.; Yu, J.; Liu, S. Ag2O as a new visible-light photocatalyst: Self-stability and high photocatalytic activity. Chem. A Eur. J. 2011, 17, 7777–7780. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, B.; Zhang, S.W.; Ogita, T.; Nishimoto, S.; Kagiya, T. Photoactivation of silver loaded on titanium(IV) oxide for room-temperature decomposition of ozone. J. Photochem. Photobiol. A: Chem. 1993, 71, 195–198. [Google Scholar] [CrossRef]
- Tien, D.-C.; Chen, L.-C.; Thai, N.V.; Ashraf, S. Study of Ag and au nanoparticles synthesized by arc discharge in deionized water. J. Nanomater. 2010, 2010, 1–9. [Google Scholar] [CrossRef]
- Zhou, Y.-T.; Nie, H.-L.; Branford-White, C.; He, Z.-Y.; Zhu, L.-M. Removal of Cu2+ from aqueous solution by chitosan-coated magnetic nanoparticles modified with α-ketoglutaric acid. J. Colloid Interface Sci. 2009, 330, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Mancic, L.; Marinkovic, K.; Marinkovic, B.A.; Dramicanin, M.; Milosevic, O. YAG: Ce3+ nanostructured particles obtained via spray pyrolysis of polymeric precursor solution. J. Eur. Ceram. Soc. 2010, 30, 577–582. [Google Scholar] [CrossRef]
- Riviere, J.C.; Myhra, S. Handbook of Surface and Interface Analysis: Methods for Problem-Solving; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-C.; Wu, H.-H.; Huang, H.-Y.; Liu, C.-W.; Chen, Y.-N. Synthesis of High Valence Silver-Loaded Mesoporous Silica with Strong Antibacterial Properties. Int. J. Environ. Res. Public Health 2016, 13, 99. https://doi.org/10.3390/ijerph13010099
Chen C-C, Wu H-H, Huang H-Y, Liu C-W, Chen Y-N. Synthesis of High Valence Silver-Loaded Mesoporous Silica with Strong Antibacterial Properties. International Journal of Environmental Research and Public Health. 2016; 13(1):99. https://doi.org/10.3390/ijerph13010099
Chicago/Turabian StyleChen, Chun-Chi, Hsin-Hsien Wu, Hsin-Yi Huang, Chen-Wei Liu, and Yi-Ning Chen. 2016. "Synthesis of High Valence Silver-Loaded Mesoporous Silica with Strong Antibacterial Properties" International Journal of Environmental Research and Public Health 13, no. 1: 99. https://doi.org/10.3390/ijerph13010099
APA StyleChen, C.-C., Wu, H.-H., Huang, H.-Y., Liu, C.-W., & Chen, Y.-N. (2016). Synthesis of High Valence Silver-Loaded Mesoporous Silica with Strong Antibacterial Properties. International Journal of Environmental Research and Public Health, 13(1), 99. https://doi.org/10.3390/ijerph13010099






