Flooding and Clostridium difficile Infection: A Case-Crossover Analysis
Abstract
:1. Introduction
1.1. Clostridium Difficile
1.2. Community Transmission
1.3. Transmission in the Environment
1.4. Flooding and Gastrointestinal Infections
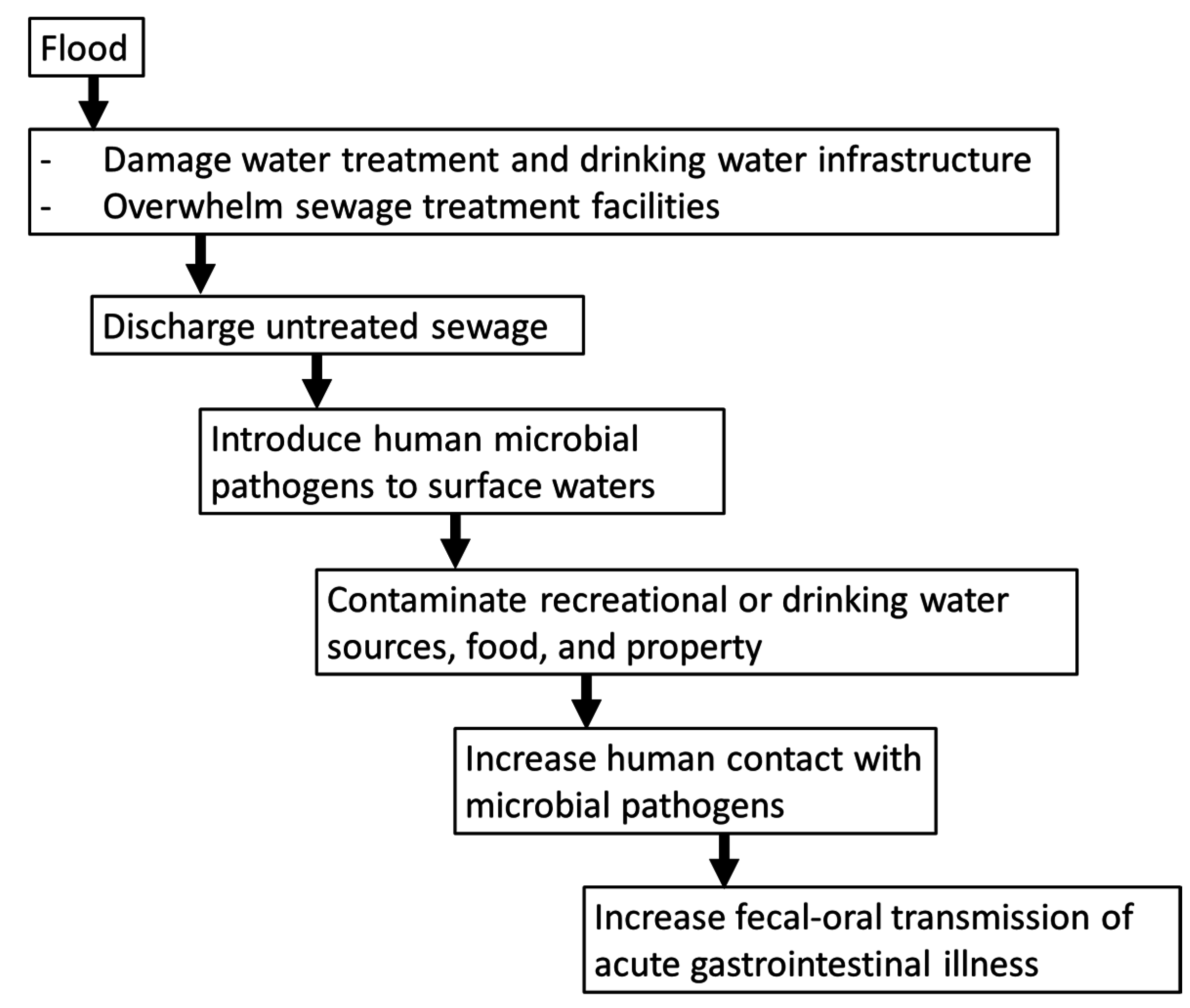
2. Methods
2.1. Heath Care Visits for Intestinal Infection due to C. Difficile
2.2. Flood Events
2.3. Study Design
2.4. Statistical Analysis
3. Results
3.1. Health Care Visits for C. Difficile
| Diagnosis Characteristic | Emergency Room (ER) | Outpatient | ER + Outpatient | |||
|---|---|---|---|---|---|---|
| N = 1023 | N = 552 | N = 1575 | ||||
| N | % | n | % | n | % | |
| Sex | ||||||
| Female | 708 | 69.2 | 341 | 61.8 | 1049 | 66.6 |
| Male | 315 | 30.8 | 211 | 38.2 | 526 | 33.4 |
| Age group | ||||||
| Children (≤5 years) | 21 | 2.1 | 21 | 3.8 | 42 | 2.7 |
| Youth (6–18 years) | 26 | 2.5 | 17 | 3.1 | 43 | 2.7 |
| Working Age Adults (19–64 years) | 470 | 45.9 | 256 | 46.4 | 726 | 46.1 |
| Elderly (≥65 years) | 506 | 49.5 | 258 | 46.7 | 764 | 48.5 |
| Race/ethnicity | ||||||
| Non-Hispanic White | 893 | 87.3 | 477 | 86.4 | 1370 | 87.0 |
| Non-Hispanic Black | 33 | 3.2 | 17 | 3.1 | 50 | 3.2 |
| Hispanic | 6 | 3.5 | 15 | 2.7 | 51 | 3.2 |
| Other | 7 | 1.7 | 10 | 1.8 | 7 | 1.7 |
| Missing/Unknown | 44 | 4.3 | 33 | 6.0 | 77 | 4.9 |
| Primary diagnosis | 597 | 58.4 | 338 | 61.2 | 935 | 59.4 |
| Weekend visit | 276 | 27.0 | 120 | 21.7 | 396 | 25.1 |
| Season | ||||||
| Fall | 237 | 23.2 | 130 | 23.6 | 367 | 23.3 |
| Winter | 218 | 21.3 | 137 | 24.8 | 355 | 22.5 |
| Spring | 277 | 27.1 | 149 | 27.0 | 426 | 27.1 |
| Summer | 291 | 28.5 | 136 | 24.6 | 427 | 27.1 |
| Year | ||||||
| 2003 | 174 | 17.0 | 86 | 15.6 | 260 | 16.5 |
| 2004 | 164 | 16.0 | 84 | 15.2 | 248 | 15.8 |
| 2005 | 190 | 18.6 | 126 | 22.8 | 316 | 20.1 |
| 2006 | 251 | 24.5 | 138 | 25.0 | 389 | 24.7 |
| 2007 | 244 | 23.9 | 118 | 21.4 | 362 | 23.0 |
3.2. Flood Events
| Number of Floods | % | |
|---|---|---|
| Season | ||
| Fall (September–November) | 32 | 24.8 |
| Winter (December–February) | 4 | 3.1 |
| Spring (March–May) | 37 | 28.7 |
| Summer (June–August) | 56 | 43.4 |
| Year | ||
| 2003 | 22 | 17.1 |
| 2004 | 10 | 7.8 |
| 2005 | 29 | 22.5 |
| 2006 | 42 | 32.6 |
| 2007 | 26 | 20.2 |
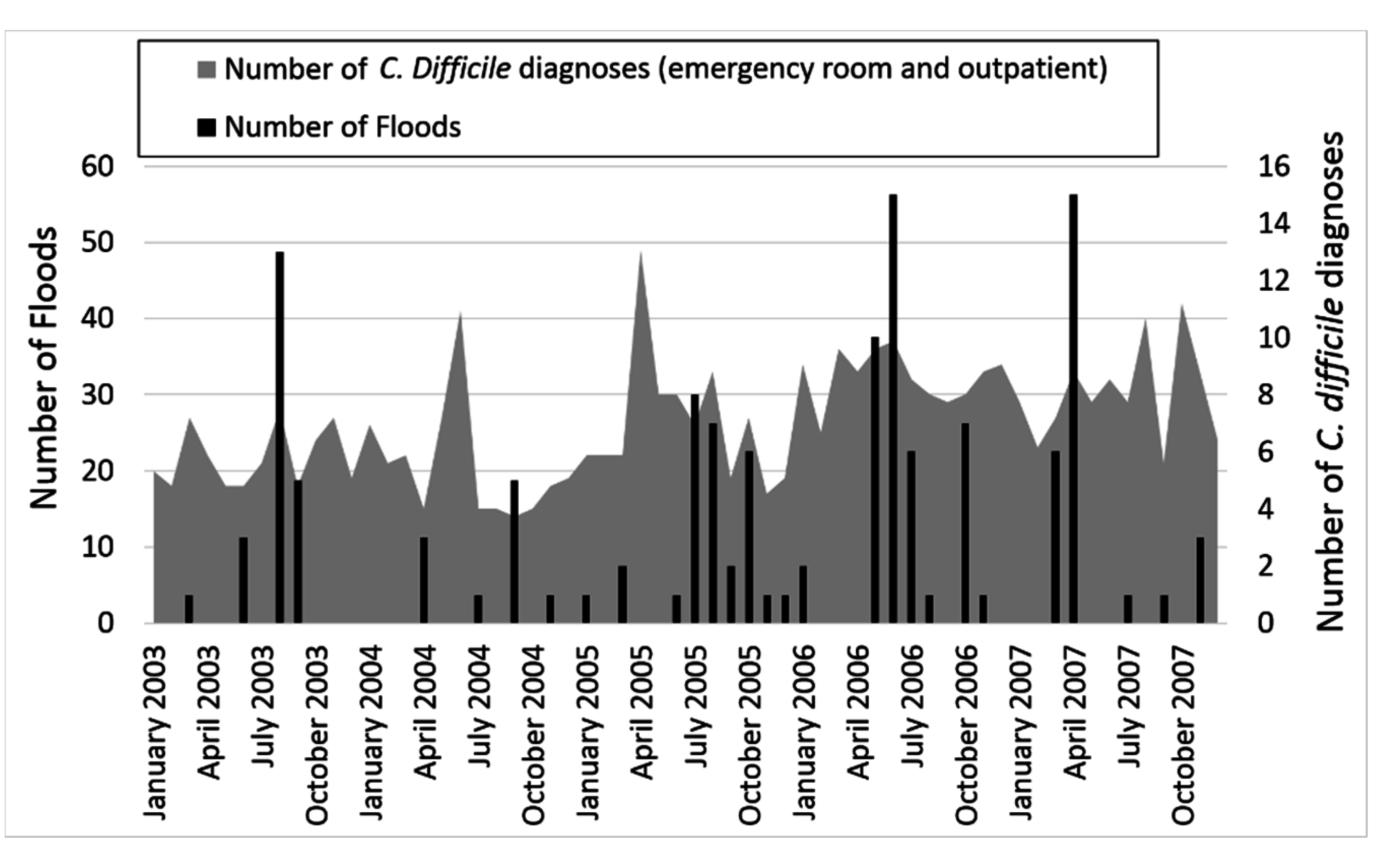
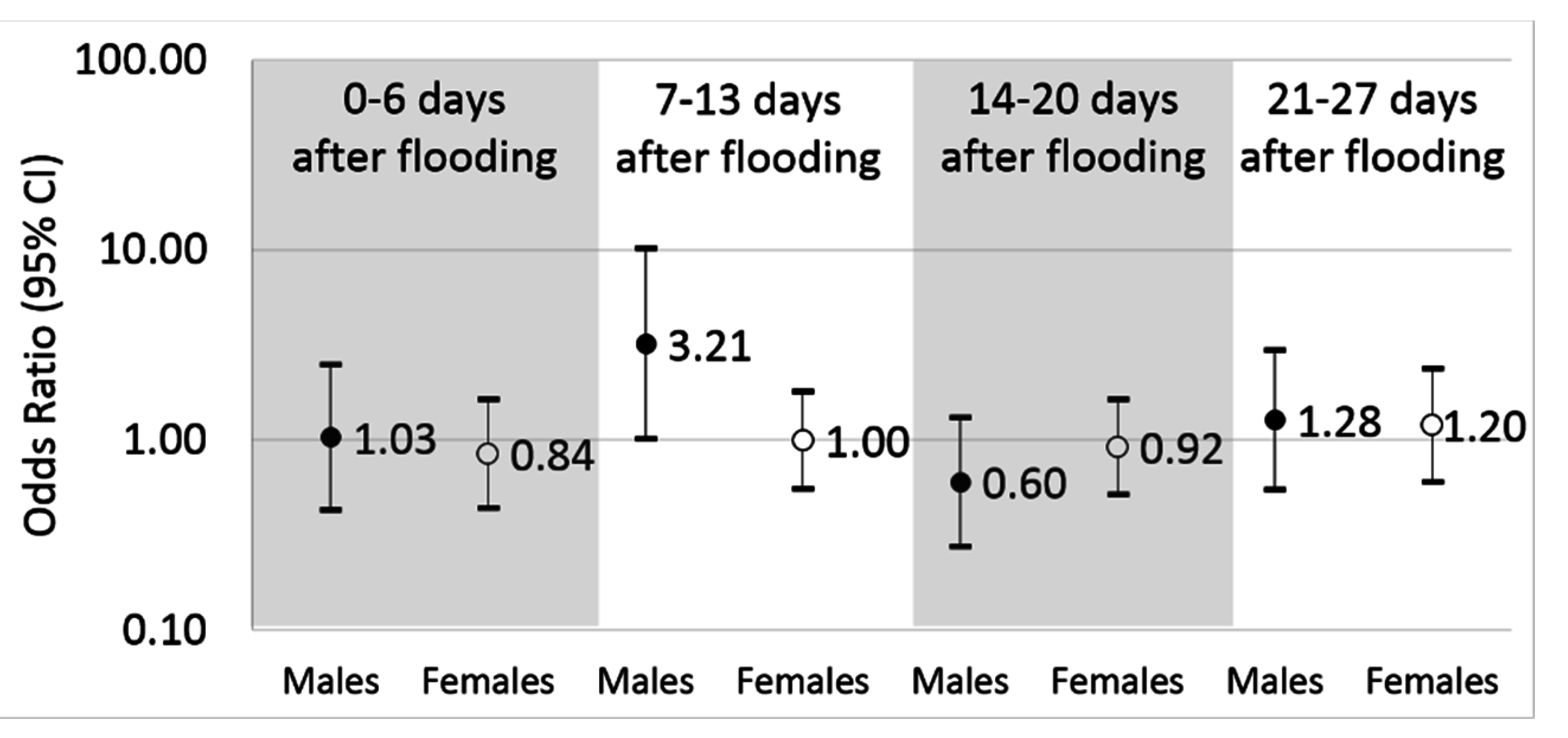
3.3. Association between Flooding and Diagnoses of Clostridium Difficile
| Emergency Room or Outpatient Diagnosis 1 | Number of Diagnoses | 0–6 Days after Flooding | 7–13 Days after Flooding | 14–20 Days after Flooding | 21–27 Days after Flooding |
|---|---|---|---|---|---|
| OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | ||
| Any C. difficile | 1575 | 0.91 (0.54, 1.54) | 1.32 (0.79, 2.20) | 0.79 (0.50, 1.25) | 1.23 (0.72, 2.09) |
| Primary diagnosis of C. difficile | 935 | 1.02 (0.50, 2.08) | 1.23 (0.65, 2.31) | 0.65 (0.35, 1.22) | 1.42 (0.71, 2.83) |
| Any C. difficile, Working Age Adults (19–64 years) | 726 | 1.22 (0.60, 2.52) | 1.69 (0.84, 3.37) | 0.76 (0.39, 1.49) | 1.12 (0.54, 2.33) |
| Any C. difficile, Elderly (≥65 years) | 764 | 0.66 (0.29, 1.53) | 0.86 (0.37, 1.99) | 0.76 (0.40, 1.46) | 1.30 (0.57, 2.96) |
| Any C. difficile, Males | 526 | 1.03 (0.43, 2.49) | 3.21 * (1.01, 10.19) | 0.60 (0.28, 1.32) | 1.28 (0.55, 2.97) |
| Any C. difficile, Females | 1049 | 0.84 (0.44, 1.63) | 1.00 (0.55, 1.81) | 0.92 (0.51, 1.63) | 1.20 (0.60, 2.38) |
4. Discussion
4.1. Differences by Sex
4.2. Age Group
4.3. Study Strengths and Limitations
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kachrimanidou, M.; Malisiovas, N. Clostridium difficile infection: A comprehensive review. Crit. Rev. Microbiol. 2011, 37, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Kyne, L.; Farrell, R.J.; Kelly, C.P. Clostridium difficile. Gastroenterol. Clin. N. Am. 2001, 30, 753–777. [Google Scholar] [CrossRef]
- Guerrant, R.L.; Hughes, J.M.; Lima, N.L.; Crane, J. Diarrhea in developed and developing-countries—Magnitude, special settings, and etiologies. Rev. Infect. Dis. 1990, 12, S41–S50. [Google Scholar] [CrossRef] [PubMed]
- Rupnik, M.; Wilcox, M.H.; Gerding, D.N. Clostridium difficile infection: New developments in epidemiology and pathogenesis. Nat. Rev. Microbiol. 2009, 7, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Zilberberg, M.D.; Shorr, A.F.; Kollef, M.H. Increase in adult Clostridium difficile-related hospitalizations and case-fatality rate, united states, 2000–2005. Emerg. Infect. Dis. 2008, 14, 929–931. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.P.; LaMont, J.T. Clostridium difficile—More difficult than ever. N. Engl. J. Med. 2008, 359, 1932–1940. [Google Scholar] [CrossRef] [PubMed]
- Todd, B. Clostridium difficile: Familiar pathogen, changing epidemiology. Am. J. Nurs. 2006, 106, 33–36. [Google Scholar] [PubMed]
- O’Donoghue, C.; Kyne, L. Update on Clostridium difficile infection. Curr. Opin. Gastroenterol. 2011, 27, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Lucado, J.; Gould, C.; Elixhauser, A. Clostridium Difficile Infections (CDI) in Hospital Stays, 2009: Statistical Brief #124. Available online: http://www.hcup-us.ahrq.gov/reports/statbriefs/sb124.pdf (accessed on 17 April 2015).
- Centers for Disease Control and Prevention. Healthcare-Associated Infections (HAIS): Clostridium Difficile Infection. Available online: http://www.cdc.gov/hai/organisms/cdiff/cdiff_infect.html (accessed on 17 April 2015).
- Barbut, F.; Petit, J.C. Epidemiology of Clostridium difficile-associated infections. Clin. Microbiol. Infect. 2001, 7, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Severe Clostridium difficile-associated disease in populations previously at low risk—Four states, 2005. MMWR 2005, 54, 1201–1205. [Google Scholar]
- Pituch, H. Clostridium difficile is no longer just a nosocomial infection or an infection of adults. Int. J. Antimicrob. Agents 2009, 33, S42–S45. [Google Scholar] [CrossRef]
- Kuijper, E.J.; van Dissel, J.T. Spectrum of Clostridium difficile infections outside health care facilities. CMAJ Can. Med. Assoc. J. 2008, 179, 747–748. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Khanna, S. Community-acquired Clostridium difficile infection: An increasing public health threat. Infect. Drug Resist. 2014, 7, 63–72. [Google Scholar] [PubMed]
- Collins, C.E.; Ayturk, M.D.; Flahive, J.M.; Emhoff, T.A.; Anderson, F.A., Jr.; Santry, H.P. Epidemiology and outcomes of community-acquired Clostridium difficile infections in Medicare beneficiaries. J. Am. Coll. Surg. 2014, 218, 1141–1147. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, A.; Pant, C.; Pasupuleti, V.; Rolston, D.D.; Jain, A.; Deshpande, N.; Thota, P.; Sferra, T.J.; Hernandez, A.V. Association between proton pump inhibitor therapy and Clostridium difficile infection in a meta-analysis. Clin. Gastroenterol. Hepatol. 2012, 10, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Chitnis, A.S.; Holzbauer, S.M.; Belflower, R.M.; Winston, L.G.; Bamberg, W.M.; Lyons, C.; Farley, M.M.; Dumyati, G.K.; Wilson, L.E.; Beldavs, Z.G.; et al. Epidemiology of community-associated Clostridium difficile infection, 2009 through 2011. JAMA Intern. Med. 2013, 173, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- Naggie, S.; Miller, B.A.; Zuzak, K.B.; Pence, B.W.; Mayo, A.J.; Nicholson, B.P.; Kutty, P.K.; McDonald, L.C.; Woods, C.W. A case-control study of community-associated Clostridium difficile infection: No role for proton pump inhibitors. Am. J. Med. 2011, 124, 276:e1–276:e7. [Google Scholar] [CrossRef] [PubMed]
- Tleyjeh, I.M.; Bin Abdulhak, A.A.; Riaz, M.; Alasmari, F.A.; Garbati, M.A.; AlGhamdi, M.; Khan, A.R.; Al Tannir, M.; Erwin, P.J.; Ibrahim, T.; et al. Association between proton pump inhibitor therapy and Clostridium difficile infection: A contemporary systematic review and meta-analysis. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Freedberg, D.E.; Abrams, J.A. Clostridium difficile infection in the community: Are proton pump inhibitors to blame? World J. Gastroenterol. WJG 2013, 19, 6710–6713. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S.; Pardi, D.S.; Aronson, S.L.; Kammer, P.P.; Orenstein, R.; St Sauver, J.L.; Harmsen, W.S.; Zinsmeister, A.R. The epidemiology of community-acquired Clostridium difficile infection: A population-based study. Am. J. Gastroenterol. 2012, 107, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S.; Baddour, L.M.; Huskins, W.C.; Kammer, P.P.; Faubion, W.A.; Zinsmeister, A.R.; Harmsen, W.S.; Pardi, D.S. The epidemiology of Clostridium difficile infection in children: A population-based study. Clin. Infect. Dis. 2013, 56, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, M.H.; Mooney, L.; Bendall, R.; Settle, C.D.; Fawley, W.N. A case-control study of community-associated Clostridium difficile infection. J. Antimicrob. Chemother. 2008, 62, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Kutty, P.K.; Woods, C.W.; Sena, A.C.; Benoit, S.R.; Naggie, S.; Frederick, J.; Evans, S.; Engel, J.; McDonald, L.C. Risk factors for and estimated incidence of community-associated Clostridium difficile infection, North Carolina, USA. Emerg. Infect. Dis. 2010, 16, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Karlstrom, O.; Fryklund, B.; Tullus, K.; Burman, L.G. A prospective nationwide study of Clostridium difficile-associated diarrhea in Sweden. The Swedish C. difficile study group. Clin. Infect. Dis. 1998, 26, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Noren, T.; Akerlund, T.; Back, E.; Sjoberg, L.; Persson, I.; Alriksson, I.; Burman, L.G. Molecular epidemiology of hospital-associated and community-acquired Clostridium difficile infection in a Swedish county. J. Clin. Microbiol. 2004, 42, 3635–3643. [Google Scholar] [CrossRef] [PubMed]
- Lessa, F.C. Community-associated Clostridium difficile infection: How real is it? Anaerobe 2013, 24, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Al Saif, N.; Brazier, J.S. The distribution of Clostridium difficile in the environment of South Wales. J. Med. Microbiol. 1996, 45, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Resources for Patients: Clostridium Difficile Infection. Available online: http://www.cdc.gov/hai/organisms/cdiff/Cdiff-patient.html (accessed on 17 April 2015).
- Cohen, S.H.; Gerding, D.N.; Johnson, S.; Kelly, C.P.; Loo, V.G.; McDonald, L.C.; Pepin, J.; Wilcox, M.H.; Society for Healthcare Epidemiology of American; Infectious Diseases Society of American. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 Update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA). Infect. Control Hosp. Epidemiol. 2010, 31, 431–455. [Google Scholar] [PubMed]
- Wilcox, M.H.; Fawley, W.N. Hospital disinfectants and spore formation by Clostridium difficile. Lancet 2000, 356. [Google Scholar] [CrossRef]
- Kramer, A.; Schwebke, I.; Kampf, G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect. Dis. 2006, 6. [Google Scholar] [CrossRef] [PubMed]
- Nazarko, L. Clostridium difficile in the community: Time to clean up? Br. J. Community Nurs. 2014, 19, 512–513. [Google Scholar] [PubMed]
- Best, E.L.; Sandoe, J.A.; Wilcox, M.H. Potential for aerosolization of Clostridium difficile after flushing toilets: The role of toilet lids in reducing environmental contamination risk. J. Hosp. Infect. 2012, 80, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Hoover, D.G.; Rodriguez-Palacios, A. Transmission of Clostridium difficile in foods. Infect. Dis. Clin. N. Am. 2013, 27, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, K.R.; Colvin, H.V.; Patel, K.V.; Clokie, J.J.; Clokie, M.R. Genetically diverse Clostridium difficile strains harboring abundant prophages in an estuarine environment. Appl. Environ. Microbiol. 2013, 79, 6236–6243. [Google Scholar] [CrossRef] [PubMed]
- Kotila, S.M.; Pitkanen, T.; Brazier, J.; Eerola, E.; Jalava, J.; Kuusi, M.; Kononen, E.; Laine, J.; Miettinen, I.T.; Vuento, R.; et al. Clostridium difficil contamination of public tap water distribution system during a waterborne outbreak in Finland. Scand. J. Public Health 2013, 41, 541–545. [Google Scholar]
- U.S. Global Change Research Program. Global Climate Change Impacts in the United States. Available online: http://purl.access.gpo.gov/GPO/LPS116712 (accessed on 17 April 2015).
- Kirshen, P.; Watson, C.; Douglas, E.; Gontz, A.; Lee, J.; Tian, Y. Coastal flooding in the Northeastern United States due to climate change. Mitig. Adapt. Strateg. Glob. Chang. 2008, 13, 437–451. [Google Scholar] [CrossRef]
- Knox, J.C. Large increases in flood magnitude in response to modest changes in climate. Nature 1993, 361, 430–432. [Google Scholar] [CrossRef]
- Milly, P.C.D.; Wetherald, R.T.; Dunne, K.A.; Delworth, T.L. Increasing risk of great floods in a changing climate. Nature 2002, 415, 514–517. [Google Scholar] [CrossRef] [PubMed]
- McMichael, A.J.; Woodruff, R.E.; Hales, S. Climate change and human health: Present and future risks. Lancet 2006, 367, 859–869. [Google Scholar] [CrossRef]
- Lane, K.; Charles-Guzman, K.; Wheeler, K.; Abid, Z.; Graber, N.; Matte, T. Health effects of coastal storms and flooding in urban areas: A review and vulnerability assessment. J. Environ. Public Health 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Wade, T.J.; Sandhu, S.K.; Levy, D.; Lee, S.; LeChevallier, M.W.; Katz, L.; Colford, J.M., Jr. Did a severe flood in the Midwest cause an increase in the incidence of gastrointestinal symptoms? Am. J. Epidemiol. 2004, 159, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Wade, T.J.; Lin, C.J.; Jagai, J.S.; Hilborn, E.D. Flooding and emergency room visits for gastrointestinal illness in Massachusetts: A case-crossover study. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, O.P.; Holt, J.; Kjerulf, A.; Muller, L.; Ethelberg, S.; Molbak, K. Personal protective equipment, hygiene behaviours and occupational risk of illness after July 2011 flood in Copenhagen, Denmark. Epidemiol. Infect. 2013, 141, 1756–1763. [Google Scholar] [CrossRef] [PubMed]
- Schnitzler, J.; Benzler, J.; Altmann, D.; Mucke, I.; Krause, G. Survey on the population’s needs and the public health response during floods in Germany 2002. J. Public Health Manag. Pract. JPHMP 2007, 13, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Alderman, K.; Turner, L.R.; Tong, S. Floods and human health: A systematic review. Environ. Int. 2012, 47, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Maclure, M. The case-crossover design: A method for studying transient effects on the risk of acute events. Am. J. Epidemiol. 1991, 133, 144–153. [Google Scholar] [PubMed]
- Janes, H.; Sheppard, L.; Lumley, T. Case-crossover analyses of air pollution exposure data: Referent selection strategies and their implications for bias. Epidemiology 2005, 16, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Maclure, M.; Mittleman, M.A. Should we use a case-crossover design? Ann. Rev. Public Health 2000, 21, 193–221. [Google Scholar] [CrossRef] [PubMed]
- Mittleman, M.A.; Maclure, M.; Robins, J.M. Control sampling strategies for case-crossover studies: An assessment of relative efficiency. Am. J. Epidemiol. 1995, 142, 91–98. [Google Scholar] [PubMed]
- Janes, H.; Sheppard, L.; Lumley, T. Overlap bias in the case-crossover design, with application to air pollution exposures. Stat. Med. 2005, 24, 285–300. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, J.L.; Chrischilles, E.A.; Pendergast, J.F.; Herwaldt, L.A.; Polgreen, P.M. Incidence of and risk factors for community-associated Clostridium difficile infection: A nested case-control study. BMC Infect. Dis. 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- StataCorp LP. Stata Statistical Software, Version SE12, StataCorp LP: College Station, TX, USA, 2011.
- Microsoft. Microsoft Office 2013, Microsoft: Redmond, WA, USA, 2013.
- ESRI. Arcgis for Desktop, Version 10.2.1, Environmental Systems Research Institute: Redlands, CA, USA, 2014.
- Martinez, F.J.; Leffler, D.A.; Kelly, C.P. Clostridium difficile outbreaks: Prevention and treatment strategies. Risk Manag. Healthc. Policy 2012, 5, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Paredes-Sabja, D.; Shen, A.; Sorg, J.A. Clostridium difficile spore biology: Sporulation, germination, and spore structural proteins. Trends Microbiol. 2014, 22, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.A.; Lamont, J.T. Editorial: Not so nosocomial anymore: The growing threat of community-acquired Clostridium difficile. Am. J. Gastroenterol. 2012, 107, 96–98. [Google Scholar] [CrossRef] [PubMed]
- Fothergill, A. Women’s roles in a disaster. Appl. Behav. Sci. Rev. 1999, 7, 125–143. [Google Scholar] [CrossRef]
- Riggs, M.A.; Rao, C.Y.; Brown, C.M.; Van Sickle, D.; Cummings, K.J.; Dunn, K.H.; Deddens, J.A.; Ferdinands, J.; Callahan, D.; Moolenaar, R.L.; et al. Resident cleanup activities, characteristics of flood-damaged homes and airborne microbial concentrations in New Orleans, Louisiana, October 2005. Environ. Res. 2008, 106, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, D. Reinforcing vulnerability? Disaster relief, recovery, and response to the 2001 flood in Rawalpindi, Pakistan. Glob. Environ. Chang. Part B Environ. Hazards 2003, 5, 71–82. [Google Scholar] [CrossRef]
- Sullivent, E.E., 3rd; West, C.A.; Noe, R.S.; Thomas, K.E.; Wallace, L.J.; Leeb, R.T. Nonfatal injuries following Hurricane Katrina—New Orleans, Louisiana, 2005. J. Saf. Res. 2006, 37, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Fayard, G.M. Fatal work injuries involving natural disasters, 1992–2006. Disaster Med. Public Health Prep. 2009, 3, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Nufer, K.E.; Wilson-Ramirez, G.; Shah, M.B.; Hughes, C.E.; Crandall, C.S. Analysis of patients treated during four disaster medical assistance team deployments. J. Emerg. Med. 2006, 30, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Dubberke, E.R.; Reske, K.A.; McDonald, L.C.; Fraser, V.J. Icd-9 Codes and surveillance for Clostridium difficile-associated disease. Emerg. Infect. Dis. 2006, 12, 1576–1579. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.H.; Liu, J.; Tzeng, Y.H.; Wu, J. Impact of visitors and hospital staff on nosocomial transmission and spread to community. J. Theor. Biol. 2014, 356, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.J.; Anu, A.; Walk, S.T.; Garey, K.W. Investigation of potentially pathogenic Clostridium difficile contamination in household environs. Anaerobe 2014, 27, 31–33. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, C.J.; Wade, T.J.; Hilborn, E.D. Flooding and Clostridium difficile Infection: A Case-Crossover Analysis. Int. J. Environ. Res. Public Health 2015, 12, 6948-6964. https://doi.org/10.3390/ijerph120606948
Lin CJ, Wade TJ, Hilborn ED. Flooding and Clostridium difficile Infection: A Case-Crossover Analysis. International Journal of Environmental Research and Public Health. 2015; 12(6):6948-6964. https://doi.org/10.3390/ijerph120606948
Chicago/Turabian StyleLin, Cynthia J., Timothy J. Wade, and Elizabeth D. Hilborn. 2015. "Flooding and Clostridium difficile Infection: A Case-Crossover Analysis" International Journal of Environmental Research and Public Health 12, no. 6: 6948-6964. https://doi.org/10.3390/ijerph120606948
APA StyleLin, C. J., Wade, T. J., & Hilborn, E. D. (2015). Flooding and Clostridium difficile Infection: A Case-Crossover Analysis. International Journal of Environmental Research and Public Health, 12(6), 6948-6964. https://doi.org/10.3390/ijerph120606948






