Methicillin-Resistant Staphylococcus aureus (MRSA) Contamination in Bedside Surfaces of a Hospital Ward and the Potential Effectiveness of Enhanced Disinfection with an Antimicrobial Polymer Surfactant
Abstract
:1. Introduction
2. Experimental Section
2.1. Setting of the Study Hospital Ward
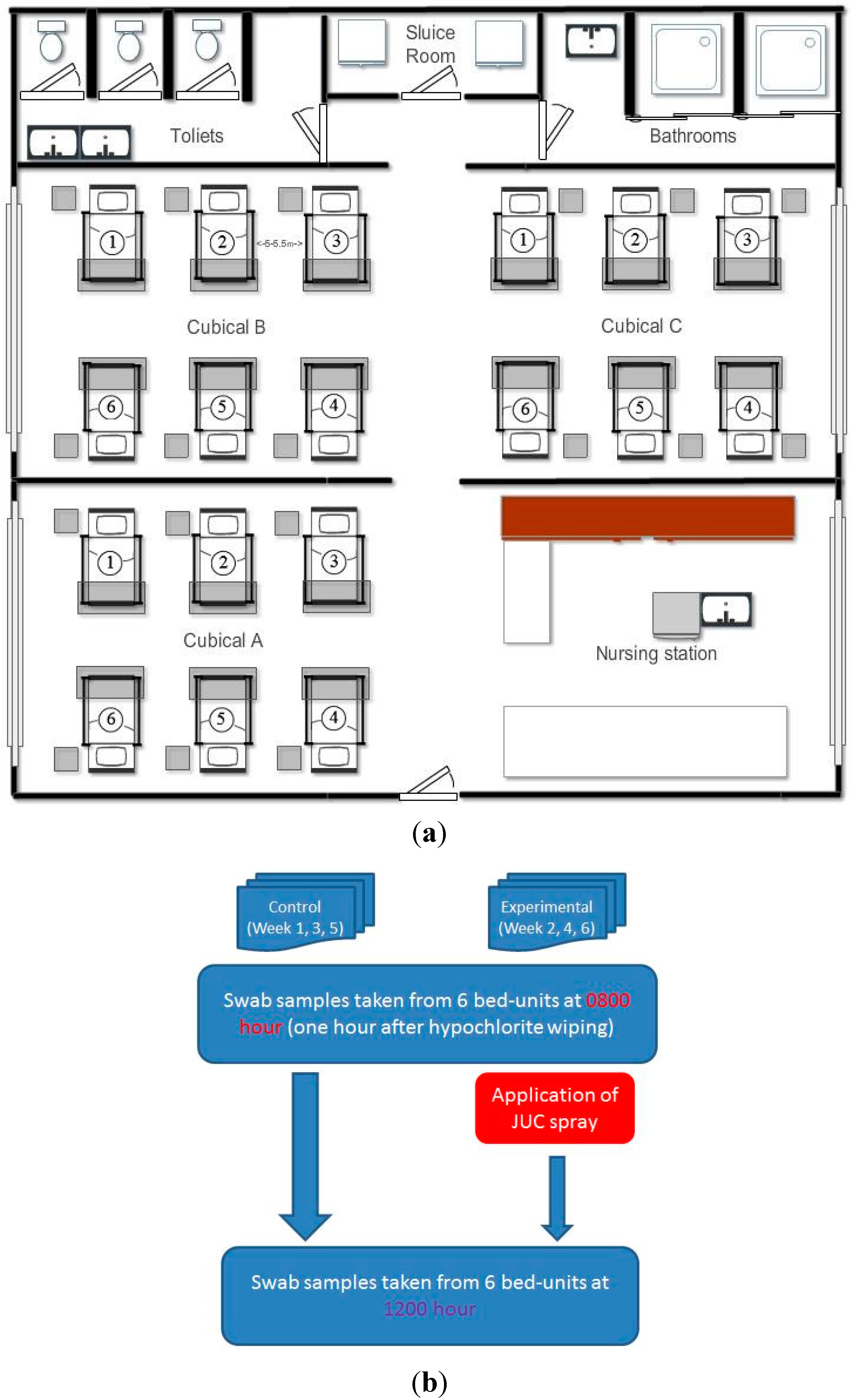
2.2. Assessing Staphylococcal Contamination on Bedside Surfaces
2.3. Experimental Design for Evaluating the Decontamination Effects of JUC Polymer
| Parameters | Experimental Group (n = 18) | Control Group (n = 18) | Paired t-Test | ||
|---|---|---|---|---|---|
| Mean Age ± SD (Range) | 81.13 ± 7.17 (67–91) | 73.44 ± 16.26 (38–92) | 0.1041 | ||
| Total number of patients | 18 | 18 | N.A. | ||
| Admitted from elderly homes (Number of patients) | Yes | 11 (61%) | 10 (63%) | N.A. | |
| No | 7 (39%) | 6 (37%) | N.A. | ||
| Positive culture for MRSA# | Wound swab | 1 | 1 | N.A. | |
| (Number of patients) | Nasal swab | 0 | 1 | ||
| Sputum | 2 | 1 | N.A. | ||
| None | 15 | 15 | N.A. | ||
| Mean hospitalization days ± SD (Range) | 7.81 ± 4.35 (2–16) | 6.75 ± 2.95 (1–14) | 0.4395 |
2.4. Bacterial Culture and Identification
2.5. Ethical Considerations
3. Results
| Types of Staphylococci and Sampling Sites | Mean cfu/cm2 ± Standard Error of Mean (SEM) | ||||
|---|---|---|---|---|---|
| MRSA-Positive | MRSA-Negative | p Value | |||
| Bed-Units (n = 6) | Bed-Units (n = 30) | ||||
| CNS | 0.1953 | ||||
| Bedside table | N.D. | 0.04 ± 0.03 | |||
| Left-side handrail | 0.39 ± 0.33 | 0.12 ± 0.09 | |||
| Right-side handrail | 1.94 ± 1.13 | 1.20 ± 1.13 | |||
| Overbed rolling table | N.D. | 0.29 ± 0.25 | |||
| MSSA | Undetermined | ||||
| Bedside table | N.D. | 0.48 ± 0.39 | |||
| Left-side handrail | N.D. | 1.94 ± 1.83 | |||
| Right-side handrail | N.D. | 3.08 ± 2.17 | |||
| Overbed rolling table | N.D. | 0.02 ± 0.01 | |||
| MRSA | 0.0392 | ||||
| Bedside table | N.D. | 0.68 ± 0.63 | |||
| Left-side handrail | 0.72 ± 0.72 | 6.37 ± 4.03 | |||
| Right-side handrail | 1.11 ± 0.98 | 0.57 ± 0.30 | |||
| Overbed rolling table | 2.11 ± 1.41 | 0.28 ± 0.14 | |||
| Types of Staphylococci and Parameters | Site for Surface Swabbing (n = 108 for Each Site) | ||||
|---|---|---|---|---|---|
| Bedside Table | Left-Side Handrail | Right-Side Handrail | Overbed Rolling Table | ||
| CNS | |||||
| Number (%) of positive culture | 5 (4.6) | 6 (5.6) | 12 (11.1) | 7 (6.5) | |
| Range of cfu/cm2 | 1–2 | 1–9 | 1–16 | 1–14 | |
| among positive culture | |||||
| MSSA | |||||
| Number (%) of positive culture | 7 (6.5) | 10 (9.3) | 4 (3.7) | 3 (2.8) | |
| Range of cfu/cm2 | 1–36 | 1–164 | 1–193 | 1–2 | |
| MRSA | among positive culture | ||||
| Number (%) of positive culture | 8 (7.4) | 19 (17.6) | 19 (17.6) | 12 (11.1) | |
| Range of cfu/cm2 | 1–23 | 1–276 | 1–75 | 1–26 | |
| among positive culture | |||||
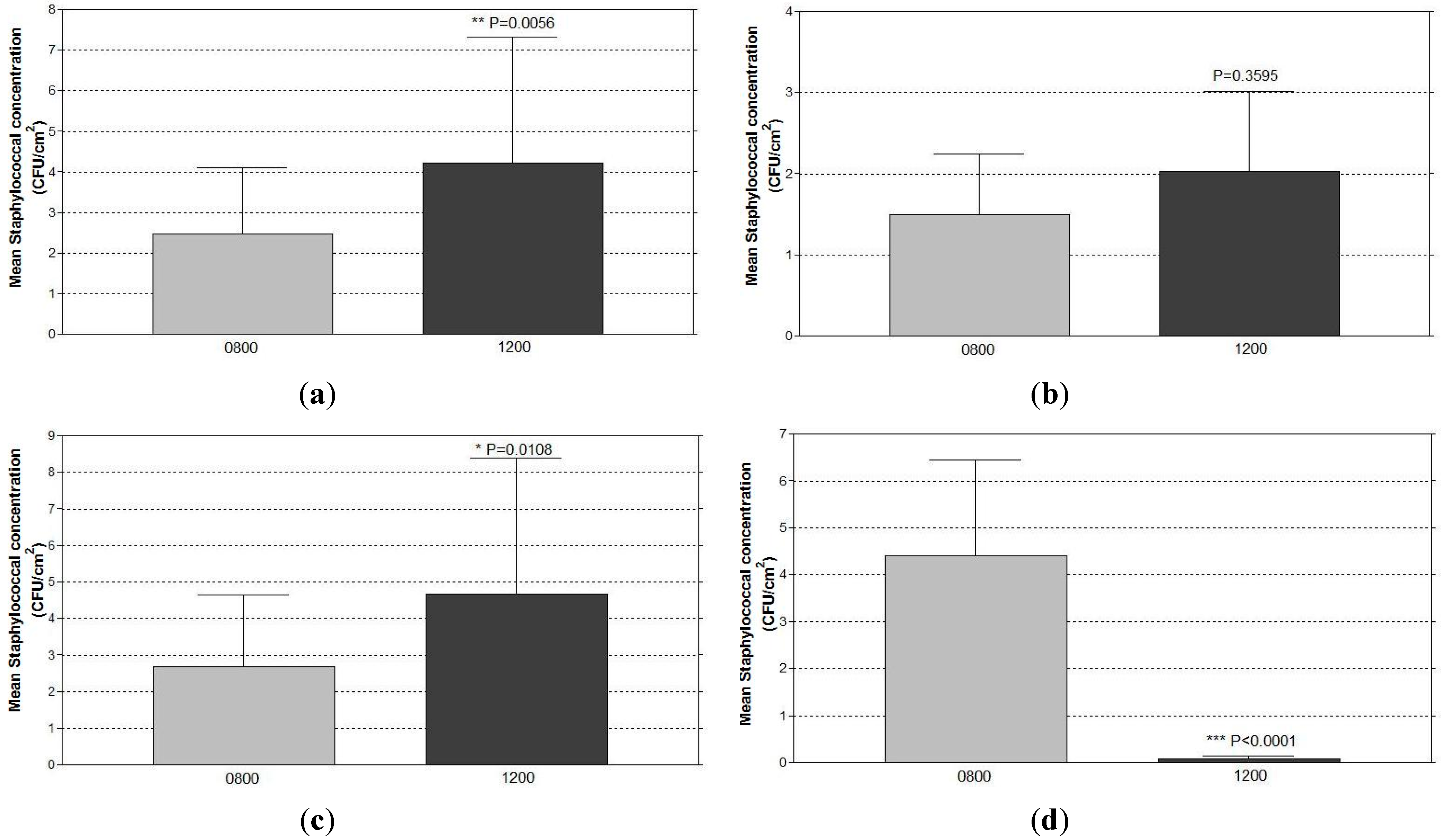
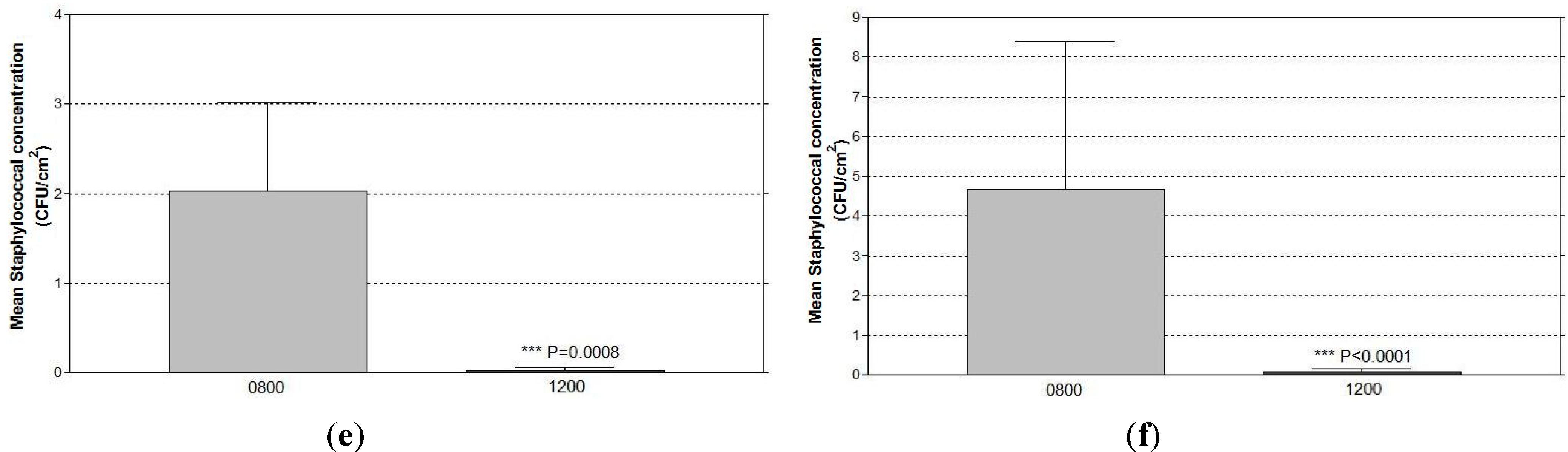
4. Discussion
| Bed-Units with Positive Culture and Types of Staphylococci | No. of Bed-Units (%) | ||
|---|---|---|---|
| Control | Experimental | ||
| (Hypochlorite Cleaning) | (JUC + Hypochlorite Cleaning) | ||
| Bed-units of MRSA carrier (n = 3) | |||
| Positive culture | 3 (100%) | 0 (0%) | |
| CNS | 1 (33%) | 0 (0%) | |
| MSSA | 2 (67%) | 0 (0%) | |
| MRSA | 1 (33%) | 0 (0%) | |
| Bed-units of non-MRSA carrier (n = 15) | |||
| Positive culture | 11 (73%) | 2 (13%) | |
| CNS | 2 (13%) | 2 (13%) | |
| MSSA | 8 (53%) | 0 (0%) | |
| MRSA | 3 (20%) | 0 (0%) | |
| All bed-units (n = 18) | |||
| Positive culture | 14 (78%) | 2 (11%) | |
| CNS | 3 (17%) | 2 (11%) | |
| MSSA | 10 (56%) | 0 (0%) | |
| MRSA | 4 (22%) | 0 (0%) | |
4.1. Staphylococcal Contamination on the Bedside Surfaces of Bed-Units in the Studied Ward
4.2. Significant Reduction in Surface Staphylococcal Burdens from Using the Antimicrobial Coating
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yuen, J.W.M.; Yung, J. Medical implications of antimicrobial coating polymers—Organosilicon quaternary ammonium chloride. Mod. Chem. Appl. 2013, 1. [Google Scholar] [CrossRef]
- Kenawy, E.R.; Worley, S.D.; Broughton, R. The chemistry and applications of antimicrobial polymers: A state-of-the-art review. Biomacromolecules 2007, 8, 1359–1384. [Google Scholar] [CrossRef] [PubMed]
- Wan, K.A.; Ng, M.Y.; Wong, Y.T. New horizon on community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA) skin and soft tissue infection: nanotechnology antimicrobial spray. H. K. J. Emerg. Med. 2011, 18, 432–436. [Google Scholar]
- Li, W.; Ma, X.; Peng, Y.; Cao, J.; Loo, W.T.Y.; Hao, L.; Cheung, M.N.B.; Chow, L.W.C.; Jin, L.J. Application of a nano-antimicrobial film to prevent ventilator-associated pneumonia: A pilot study. Afr. J. Biotechnol. 2011, 10, 1926–1931. [Google Scholar]
- Wu, L.; Dai, Y. T.; Wang, L.M.; Cheng, B.; Sun, Z.Y. Study on prevention of catheter associated urinary tract infection by using JUC physical antimicrobial spray dressing. Nat. J. Androl. 2005, 11, 581–583. [Google Scholar]
- Goodman, E.R.; Platt, R.; Bass, R.; Onderdonk, A.B.; Yokoe, D.S.; Huang, S.S. Impact of an environmental cleaning intervention on the presence of methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci on surfaces in intensive care unit rooms. Infect. Control Hosp. Epidemiol. 2008, 29, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Dancer, S.J.; Coyne, M.; Speekenbrink, A.; Samavedam, S.; Kennedy, J.; Wallace, P.G. MRSA acquisition in an intensive care unit. Am. J. Infect. Control 2006, 34, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Vickery, K.; Deva, A.; Jacombs, A.; Allan, J.; Valente, P.; Gosbell, I.B. Presence of biofilm containing viable multiresistant organisms despite terminal cleaning on clinical surfaces in an intensive care unit. J. Hosp. Infect. 2012, 80, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Oie, S.; Suenaga, S.; Sawa, A.; Kamiya, A. Association between isolation sites of methicillin-resistant Staphylococcus aureus (MRSA) in patients with MRSA-positive body sites and MRSA contamination in their surrounding environmental surfaces. Jpn. J. Infect. Dis. 2007, 60, 367–369. [Google Scholar] [PubMed]
- Rutala, W.A.; Katz, E.B.S.; Sherertz, R.J.; Sarubbi, F.A. Environmental-study of a methicillin-resistant Staphylococcus aureus epidemic in a burn unit. J. Clin. Microbiol. 1983, 18, 683–688. [Google Scholar] [PubMed]
- Rohr, U.; Kaminski, A.; Wilhelm, M.; Jurzik, L.; Gatermann, S.; Muhr, G. Colonization of patients and contamination of the patients’ environment by MRSA under conditions of single-room isolation. Int. J. Hyg. Environ. Health 2009, 212, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Sexton, T.; Clarke, P.; O’Neill, E.; Dillane, T.; Humphreys, H. Environmental reservoirs of methicillin-resistant Staphylococcus aureus in isolation rooms: Correlation with patient isolates and implications for hospital hygiene. J. Hosp. Infect. 2006, 62, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Dancer, S.J.; White, L.; Robertson, C. Monitoring environmental cleanliness on two surgical wards. Int. J. Environ. Health Res. 2008, 18, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Boyce, J.M. Environmental contamination makes an important contribution to hospital infection. J. Hosp. Infect. 2007, 65 (Suppl. 2), 50–54. [Google Scholar] [CrossRef] [PubMed]
- Kibbler, C.C.; Quick, A.; O’Neill, A.M. The effect of increased bed numbers on MRSA transmission in acute medical wards. J. Hosp. Infect. 1998, 39, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Borg, M.A. Bed occupancy and overcrowding as determinant factors in the incidence of MRSA infections within general ward settings. J. Hosp. Infect. 2003, 54, 316–318. [Google Scholar] [CrossRef] [PubMed]
- Stefani, S.; Chung, D.R.; Lindsay, J.A.; Friedrich, A.W.; Kearns, A.M.; Westh, H.; Mackenzie, F.M. Meticillin-resistant Staphylococcus aureus (MRSA): Global epidemiology and harmonisation of typing methods. Int. J. Antimicrob. Agents 2012, 39, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H.; Hsueh, P.R.; Chung, D.R.; Ko, K.S.; Kang, C.I.; Peck, K.R.; Yeom, J.S.; Kim, S.W.; Chang, H.H.; Kim, Y.S.; et al. Spread of methicillin-resistant Staphylococcus aureus between the community and the hospitals in Asian countries: An ANSORP study. J. Antimicrob. Chemother. 2011, 66, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Huslage, K.; Rutala, W.A.; Sickbert-Bennett, E.; Weber, D.J. A quantitative approach to defining “high-touch” surfaces in hospitals. Infect. Control Hosp. Epidemiol. 2010, 31, 850–853. [Google Scholar] [CrossRef] [PubMed]
- Dolan, A.; Bartlett, M.; McEntee, B.; Creamer, E.; Humphreys, H. Evaluation of different methods to recover meticillin-resistant Staphylococcus aureus from hospital environmental surfaces. J. Hosp. Infect. 2011, 79, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Creamer, E.; Shore, A.C.; Deasy, E.C.; Galvin, S.; Dolan, A.; Walley, N.; McHugh, S.; Fitzgerald-Hughes, D.; Sullivan, D.J.; Cunney, R.; et al. Air and surface contamination patterns of meticillin-resistant Staphylococcus aureus on eight acute hospital wards. J. Hosp. Infect. 2014, 86, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Cimolai, N. The role of healthcare personnel in the maintenance and spread of methicillin-resistant Staphylococcus aureus. J. Infect. Public Health 2008, 1, 78–100. [Google Scholar] [CrossRef] [PubMed]
- Pittet, D.; Dharan, S.; Touveneau, S.; Sauvan, V.; Perneger, T.V. Bacterial contamination of the hands of hospital staff during routine patient care. Arch. Intern. Med. 1999, 159, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Ziebuhr, W.; Hennig, S.; Eckart, M.; Kranzler, H.; Batzilla, C.; Kozitskaya, S. Nosocomial infections by Staphylococcus epidermidis: How a commensal bacterium turns into a pathogen. Int. J. Antimicrob. Agents 2006, 28 (Suppl. 1), S14–S20. [Google Scholar] [CrossRef] [PubMed]
- Kundrapu, S.; Sunkesula, V.; Jury, L.A.; Sitzlar, B.M.; Donskey, C.J. Daily disinfection of high-touch surfaces in isolation rooms to reduce contamination of healthcare workers’ hands. Infect. Control Hosp. Epidemiol. 2012, 33, 1039–1042. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.L.; Boost, M.V.; Chung, J.W. Study on the effectiveness of disinfection with wipes against methicillin-resistant Staphylococcus aureus and implications for hospital hygiene. Am. J. Infect. Control 2011, 39, 577–580. [Google Scholar] [CrossRef]
- Chan, C.S. The use of JUC physical antimicrobial spray dressing in managing radiation-induced acute skin reaction, pain and pruritus in cancer patient receiving radiation therapy. Master’s Thesis, The Hong kong Polytechnic University, Hong kong, China, 2012. [Google Scholar]
- Talon, D. The role of the hospital environment in the epidemiology of multi-resistant bacteria. J. Hosp. Infect. 1999, 43, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Rutala, W.A.; Weber, D.J. Surface disinfection: Should we do it? J. Hosp. Infect. 2001, 48 (Suppl. A), S64–S68. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; O’Donoghue, M.M.; Ito, T.; Hiramatsu, K.; Boost, M.V. Prevalence of antiseptic-resistance genes in Staphylococcus aureus and coagulase-negative staphylococci colonising nurses and the general population in Hong Kong. J. Hosp. Infect. 2011, 78, 113–117. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuen, J.W.M.; Chung, T.W.K.; Loke, A.Y. Methicillin-Resistant Staphylococcus aureus (MRSA) Contamination in Bedside Surfaces of a Hospital Ward and the Potential Effectiveness of Enhanced Disinfection with an Antimicrobial Polymer Surfactant. Int. J. Environ. Res. Public Health 2015, 12, 3026-3041. https://doi.org/10.3390/ijerph120303026
Yuen JWM, Chung TWK, Loke AY. Methicillin-Resistant Staphylococcus aureus (MRSA) Contamination in Bedside Surfaces of a Hospital Ward and the Potential Effectiveness of Enhanced Disinfection with an Antimicrobial Polymer Surfactant. International Journal of Environmental Research and Public Health. 2015; 12(3):3026-3041. https://doi.org/10.3390/ijerph120303026
Chicago/Turabian StyleYuen, John W. M., Terence W. K. Chung, and Alice Y. Loke. 2015. "Methicillin-Resistant Staphylococcus aureus (MRSA) Contamination in Bedside Surfaces of a Hospital Ward and the Potential Effectiveness of Enhanced Disinfection with an Antimicrobial Polymer Surfactant" International Journal of Environmental Research and Public Health 12, no. 3: 3026-3041. https://doi.org/10.3390/ijerph120303026
APA StyleYuen, J. W. M., Chung, T. W. K., & Loke, A. Y. (2015). Methicillin-Resistant Staphylococcus aureus (MRSA) Contamination in Bedside Surfaces of a Hospital Ward and the Potential Effectiveness of Enhanced Disinfection with an Antimicrobial Polymer Surfactant. International Journal of Environmental Research and Public Health, 12(3), 3026-3041. https://doi.org/10.3390/ijerph120303026







