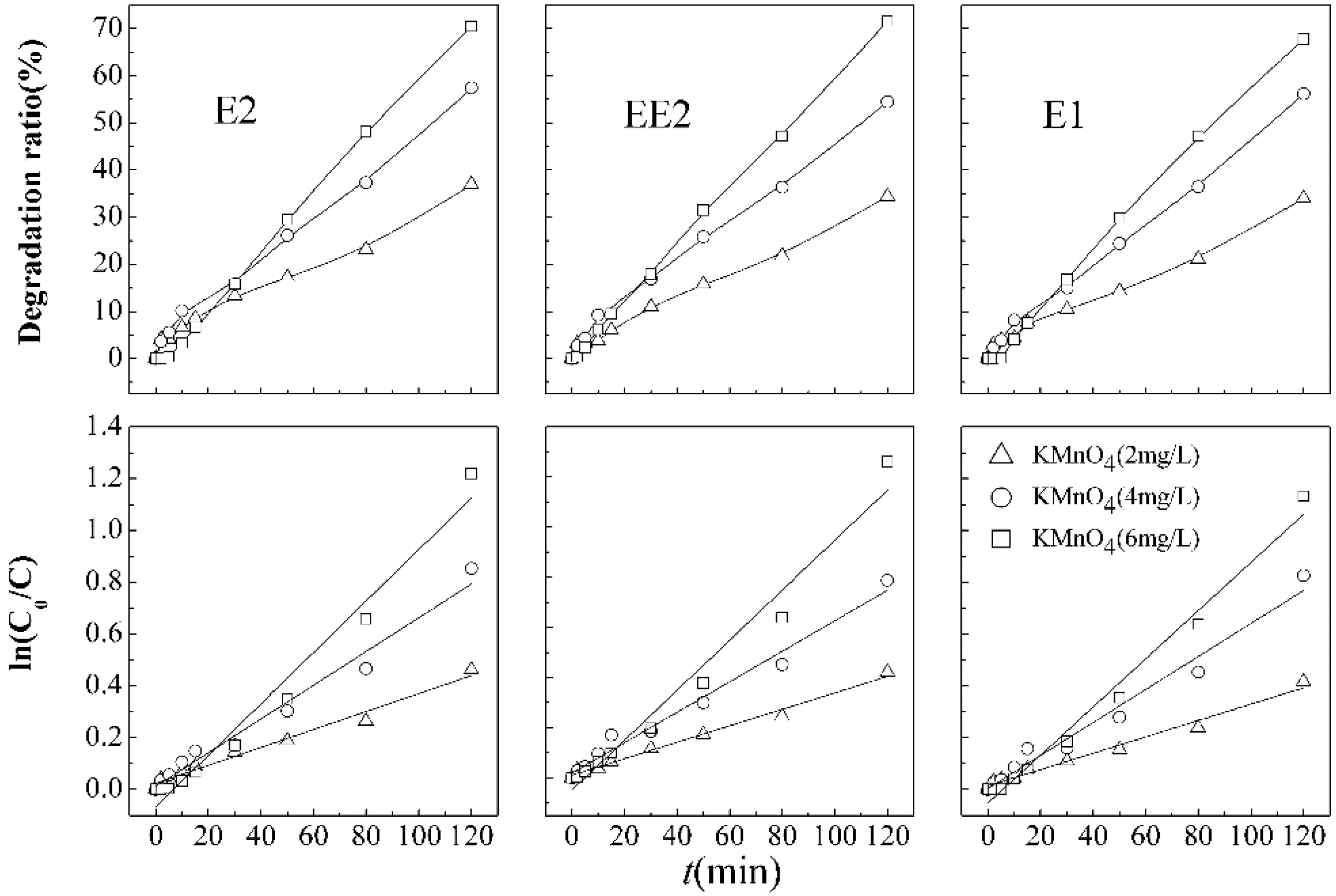
3.3.1. Degradation Efficiency of SEs in Different Treatment Process
Degradation of SEs fit pseudo-first-order kinetics in each treatment process. The removal curves for the different treatment processes are shown in
Figure 3. After a 120 min reaction, the degradation efficiencies of E2 (49.2%), EE2 (48.4%), and E1 (47.4%) by ultrasound/KMnO
4 were higher than those by single treatment processes, which reveal more effectiveness of combined ultrasound/KMnO
4 process.
where
k is an apparent rate constant, incorporating the constant concentration of oxidizing agents. The rate constants of oxidation (
kKMnO4), ultrasonic irradiation(
kUS), and their combination (
kUS/KMnO4) can be calculated according to Equation (4). The degree of synergy S can then be assessed as follows [
30]:
In this case, S < 0 indicates that there is not a good synergy between them, possibly due to the low frequencies of the ultrasonic generator in the experiments. It is generally known that radical generation by ultrasound is more effective at higher ultrasound frequencies. At lower frequencies, the cavitation effect is the dominant factor. Frontistis
et al. investigated the photocatalytic (UV-A/TiO
2) degradation of 17-ethynylestradiol (EE2) in environmental matrices, and showed that there is a two-way synergy between them (S = 44.3%), while the horn-type sonicator operating at 80 kHz was employed for sonophotocatalytic experiments [
42].
Reaction rate constants in
Table 2 manifest that there are minor differences between the degradation rate of E1, E2, and slightly lower of EE2. Accordingly, the results elucidate that there is almost no obvious competitive degradation of target SEs under the condition of lower initial concentrations.
The mechanisms of the synergetic effect of ultrasounds and KMnO
4 could be explained as follows Ma
et al. [
41].
H
2O produces H
2O
2, which undergoes partial dissociation in the presence of ultrasound.
The H
3O
+ makes the solution acidic, which improves the generation of •OH decomposed by KMnO
4.
Furthermore, the redox reaction occurs between H
2O
2 and KMnO
4. Mn
2+ and MnO
2, produced in the reaction, have a strong catalytic effect on cavitations, thus promoting further degradation of the organic contaminants. Accordingly, KMnO
4 can improve the ultrasound to produce •OH in an aqueous environment.
Permanganate was usually applied as a pre-oxidant in the drinking water treatment processes. The MnO2 as a type of light yellow granule, can be removed easily in the following traditional treatment units, e.g., coagulation/sedimentation, filtration, etc. Considering the residual manganese and color problem, dosages of permanganate were always controlled to under a certain value.
Table 2.
Degradation kinetic parameters of SEs under different processes.
Table 2.
Degradation kinetic parameters of SEs under different processes.
| Steroid Estrogens | Treatment Technologies | Kinetic Equation | Reaction Rate Constant K/min−1 | R2 | Half-Life t1/2/min |
|---|
| E2 | KMnO4 (2mg·L−1) | ln(C0/C) = 0.0035t + 0.025 | 0.0035 | 0.978 | 191 |
| Pure ultrasound | ln(C0/C) = 0.0036t + 0.064 | 0.0036 | 0.888 | 175 |
| KMnO4/ultrasound | ln(C0/C) = 0.0056t + 0.003 | 0.0056 | 0.996 | 123 |
| EE2 | KMnO4 (2mg·L−1) | ln(C0/C) = 0.0033t + 0.011 | 0.0033 | 0.988 | 207 |
| Pure ultrasound | ln(C0/C) = 0.0032t + 0.024 | 0.0032 | 0.874 | 210 |
| KMnO4/ultrasound | ln(C0/C) = 0.0055t + 0.010 | 0.0055 | 0.994 | 125 |
| E1 | KMnO4 (2mg·L−1) | ln(C0/C) = 0.0032t + 0.015 | 0.0032 | 0.980 | 212 |
| Pure ultrasound | ln(C0/C) = 0.0032t + 0.023 | 0.0032 | 0.852 | 210 |
| KMnO4/ultrasound | ln(C0/C) = 0.0056t + 0.020 | 0.0056 | 0.993 | 120 |
3.3.2. Effects of Initial Concentration of SEs
Sample solutions of mixed SEs were prepared in concentrations of approximately 50, 100, and 500 μg·L
−1. The impact of the initial concentration on the SEs’ simultaneous degradation in a KMnO
4/ultrasound process were investigated, with KMnO
4 (2 mg·L
−1), ultrasonic power (210 W) and a frequency of (20 kHz). Degradation kinetic models of SEs under different initial concentrations were showed in
Table 3.
Table 3.
Parameters of the degradation kinetic model of SEs under different initial concentrations.
Table 3.
Parameters of the degradation kinetic model of SEs under different initial concentrations.
| Steroid Estrogens | Initial Concentration (μg·L−1) | Kinetic Equation | Reaction Rate Constant K/min−1 | R2 | Half-Lif t1/2/min |
|---|
| E2 | 50 | ln(C0/C) = 0.0056t + 0.003 | 0.0056 | 0.996 | 123 |
| 100 | ln(C0/C) = 0.0041t + 0.020 | 0.0041 | 0.984 | 163 |
| 500 | ln(C0/C) = 0.0029t + 0.019 | 0.0029 | 0.972 | 230 |
| EE2 | 50 | ln(C0/C) = 0.0055t + 0.010 | 0.0055 | 0.994 | 125 |
| 100 | ln(C0/C) = 0.0039t + 0.032 | 0.0039 | 0.964 | 169 |
| 500 | ln(C0/C) = 0.0024t + 0.022 | 0.0024 | 0.944 | 212 |
| E1 | 50 | ln(C0/C) = 0.0056t + 0.020 | 0.0056 | 0.993 | 120 |
| 100 | ln(C0/C) = 0.0038t + 0.042 | 0.0038 | 0.959 | 172 |
| 500 | ln(C0/C) = 0.0024t + 0.025 | 0.0024 | 0.965 | 282 |
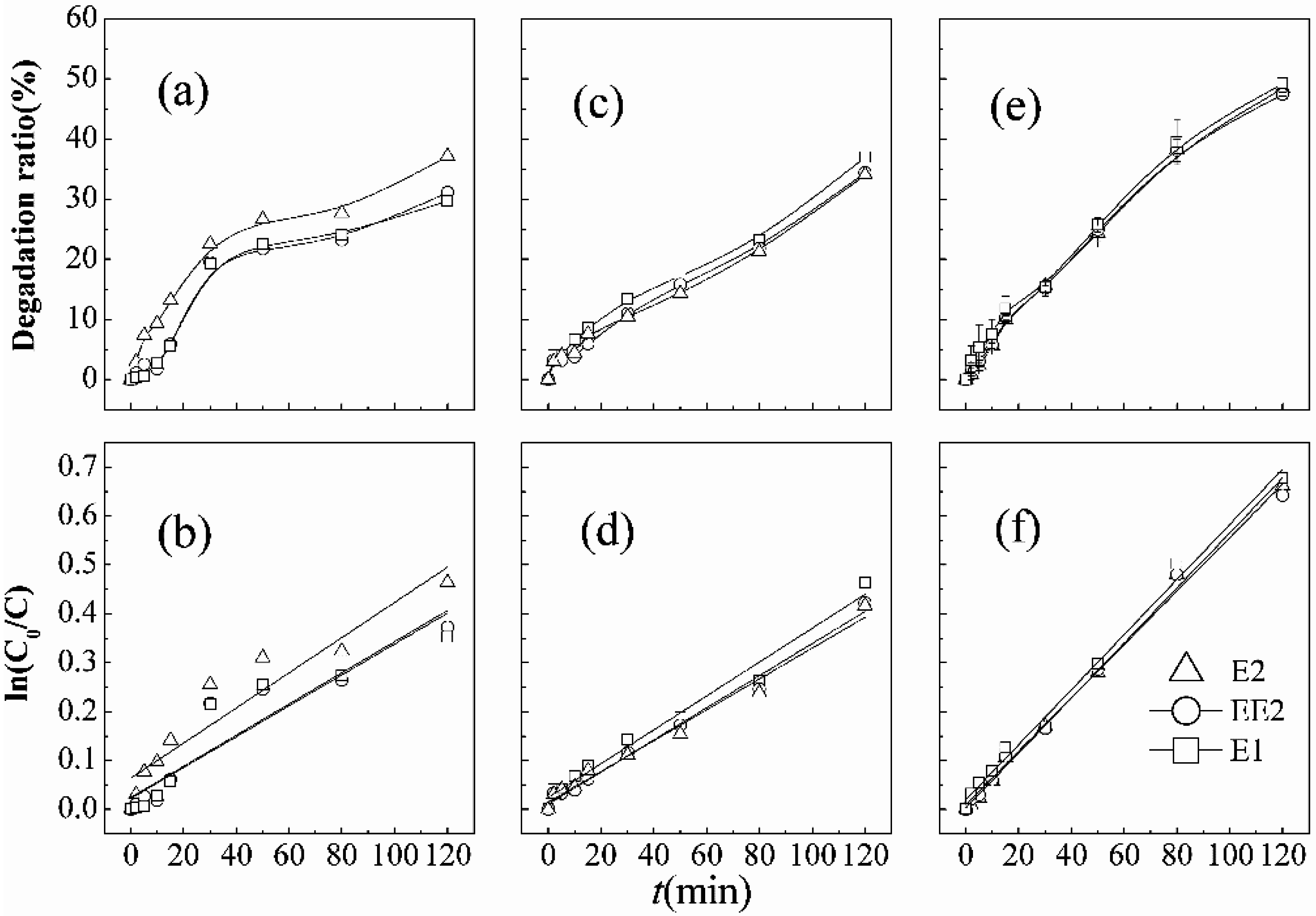
As shown in
Figure 4, when the initial concentration of SEs is 100 μg·L
−1, the degradation efficiencies of E2, EE2, and E1 are 38.3%, 36.6%, and 36.5%, respectively, with little difference in the coexisting system after 120-min-contact. However, it was observed that E2, EE2, and E1 were only removed by 28.8%, 24.4%, and 24.8%, respectively, when the initial concentration of SEs increases to about 500 μg·L
−1 with the same reaction time. Simultaneous degradation of the coexisting combinations of estrogens in KMnO
4/ultrasound system follows the apparent first-order kinetics shown in
Figure 4. The degradation ratio and removal rate decrease with increasing initial concentrations of estrogens, which is similar to the conclusions of He
et al. who used combined Fe(III)/H
2O
2 technology [
43]. Low concentration favors the degradation of SEs. The reason may be due to the low concentration accompanied by preferable contact with free hydroxyl radicals (•OH) and little oxidant demand.
Figure 4.
Degradation curves and first order kinetic models of SEs in combined processes under different initial concentrations.
Figure 4.
Degradation curves and first order kinetic models of SEs in combined processes under different initial concentrations.
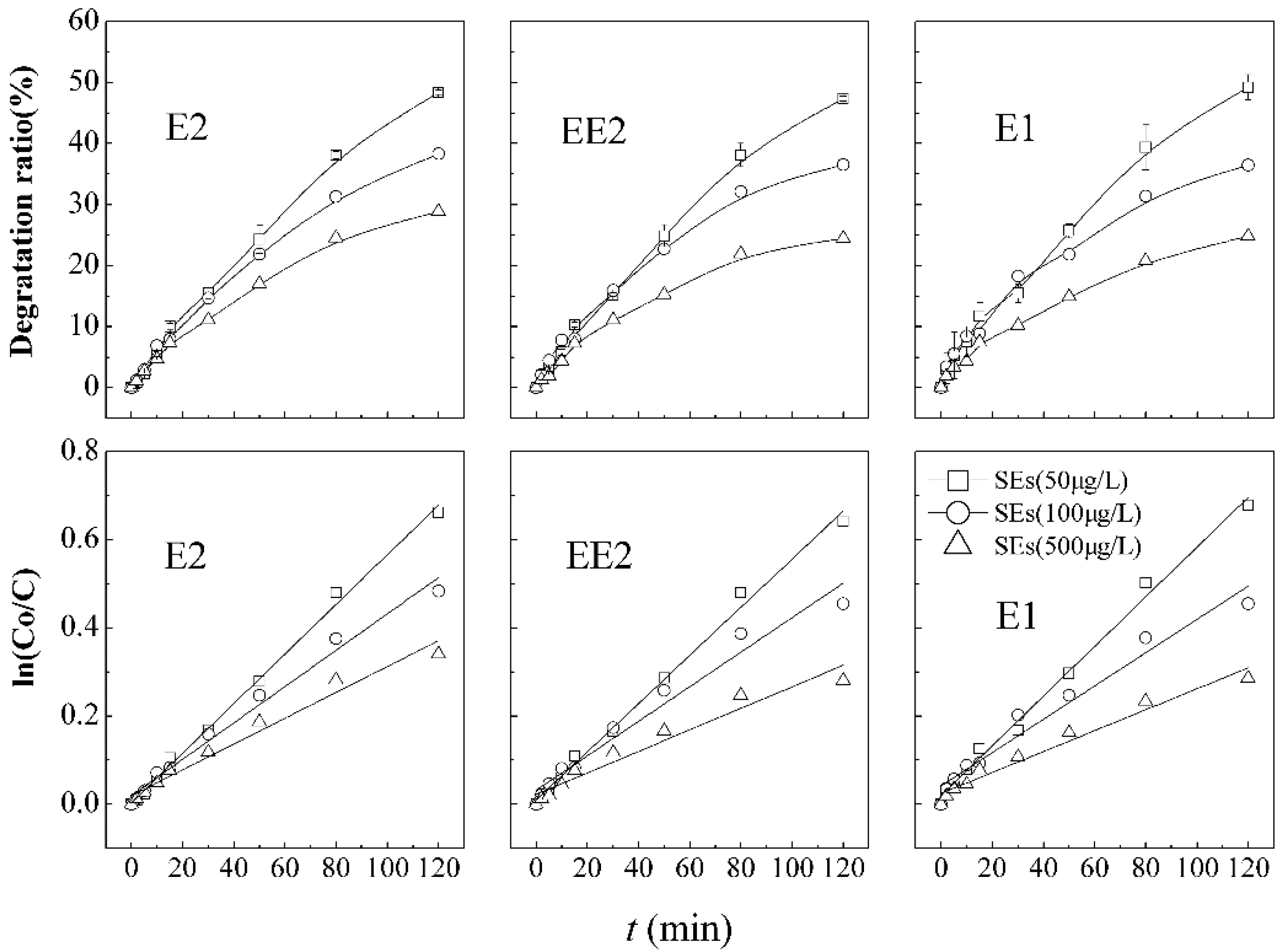
3.3.3. Analysis of Competitive Degradation of Coexisting Steroid Estrogens
The simultaneous and competitive degradation of SEs in the ultrasound/KMnO
4 process was further investigated by serial pair combinations of E2, EE2, and E1 and results were showed in
Figure 5 and
Table 4. Some researches pointed out that an estrogenic coexisting system had more adverse influences on the ecological environment [
7]. Additionally, it is supposed that coexisting SEs have mutual effects on each other in the decomposition process. Therefore, different coexisting combinations of estrogens (E2 and EE2; E2 and E1; EE2 and E1) were designed to summarize simultaneous or probable rules of competitive elimination and the impacts on each other. The concentration and percentage of each constituent were nearly identical to the experiments of the tri-coexisting SEs system.
Figure 5 shows that the initial concentrations of E2 and EE2 in dual-combination are 13.3 and 14.9 µg·L
−1, respectively, and the degradation ratios are 54.3% and 51.7% after 120 min. The initial concentrations of EE2 and E1 are 14.8 and 19.4 µg·L
−1, respectively, and the degradation ratios of them are 53.0% and 49.2%, respectively. The initial concentrations of E2 and E1 in dual-combination pattern are 10.9 and 16.1 µg·L
−1, respectively, and the degradation ratio of them are 55.6% and 50.7% after 120 min reaction.
Figure 5.
Degradation curves and first order kinetic models of dual- and tri-mixed SEs in the combination process.
Figure 5.
Degradation curves and first order kinetic models of dual- and tri-mixed SEs in the combination process.
Table 4.
Parameters of degradation kinetic model of dual-mixed SEs.
Table 4.
Parameters of degradation kinetic model of dual-mixed SEs.
| Combinations | Constituent | Kinetic Equation | Reaction Rate Constant K/min−1 | R2 | Half-Life t1/2/min |
|---|
| E2+EE2 | E2 | ln(C0/C) = 0.0063t + 0.071 | 0.0063 | 0.967 | 99 |
| EE2 | ln(C0/C) = 0.0059t + 0.058 | 0.0059 | 0.974 | 108 |
| EE2+E1 | EE2 | ln(C0/C) = 0.0061t + 0.011 | 0.0061 | 0.990 | 112 |
| E1 | ln(C0/C) = 0.0058t + 0.008 | 0.0058 | 0.992 | 118 |
| E2+E1 | E2 | ln(C0/C) = 0.0070t + 0.035 | 0.0070 | 0.997 | 94 |
| E1 | ln(C0/C) = 0.0061t + 0.038 | 0.0061 | 0.996 | 107 |
The results indicated that dual-combinations of E2, EE2, and E1 have better removal ratios than those in a tri-estrogens coexisting system, the degradation efficiency and reaction rate constant decrease with increasing numbers of constitute species and increasing total initial concentrations. The low concentration led to more effective degradation. In combined KMnO4/ultrasounds, the generation of the free hydroxyl radicals (•OH) is accompanied with the consumption. When the parameters of the KMnO4/ultrasound process were fixed, the yield of •OH was constant, and the collision probability between the molecules and •OH increases when a lower amount of target compounds joined the competition.
3.3.4. Steroid Estrogens Removal by KMnO4/Ultrasound in Natural Water Background
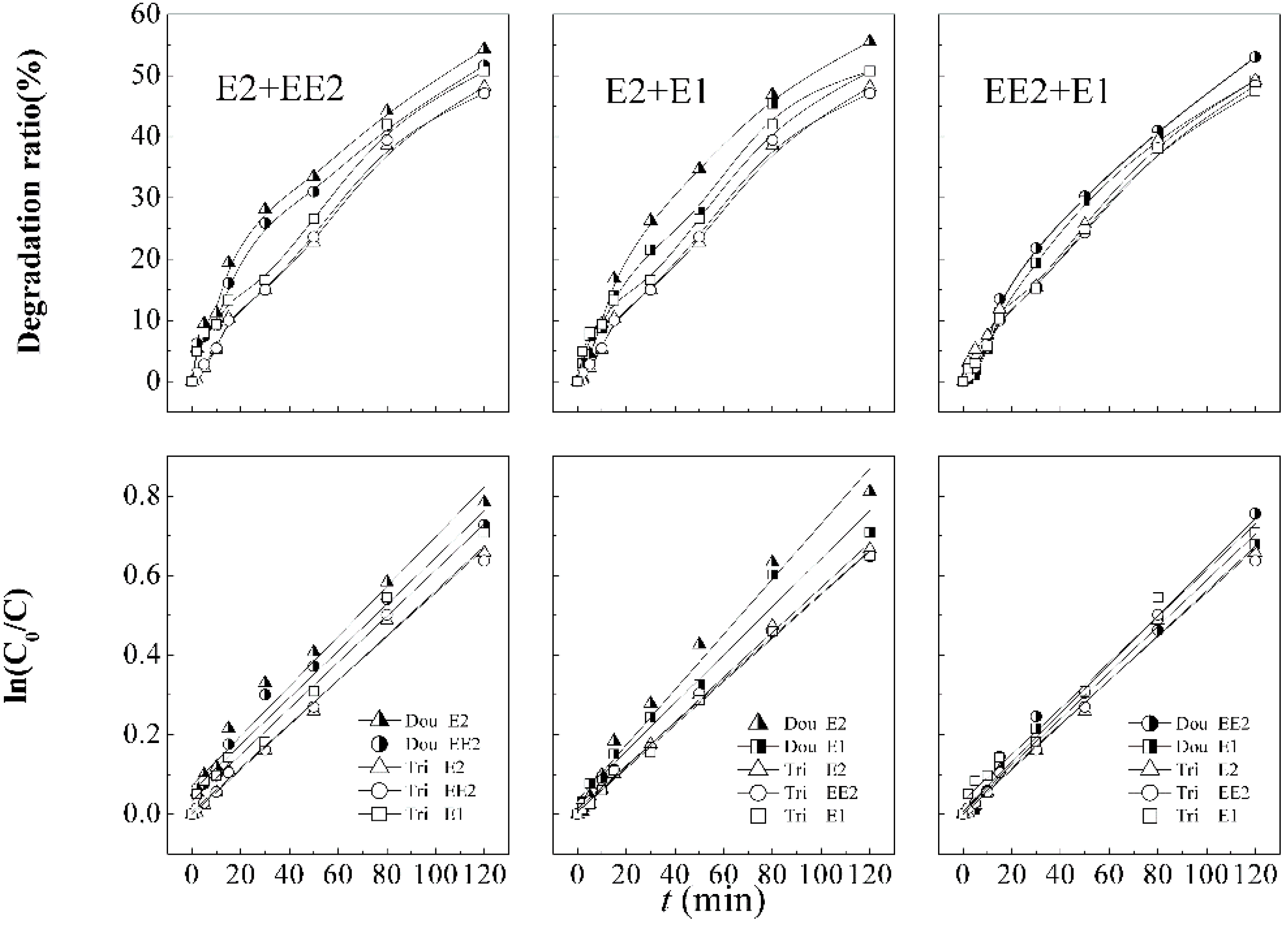
The competitive degradation for the tri-coexisting system occurring in raw surface water was studied, and the results were showed in
Figure 6 and
Table 6. The surface water was drawn from Shangtang River on campus, which had been pretreated by coagulation with PAC and sedimentation. The desired total concentration for target pollution is 50 µg·L
−1. The characteristic of the pretreated natural water is shown in
Table 5.
Table 5.
The water quality parameters of the pretreated natural water.
Table 5.
The water quality parameters of the pretreated natural water.
| Turbidity (NTU) | Color (CU) | Temperature (°C) | pH | TOC (mg·L−1) | UV254 (cm−1) |
|---|
| 1.18 | 8 | 20 | 6.8 | 5.387 | 0.0396 |
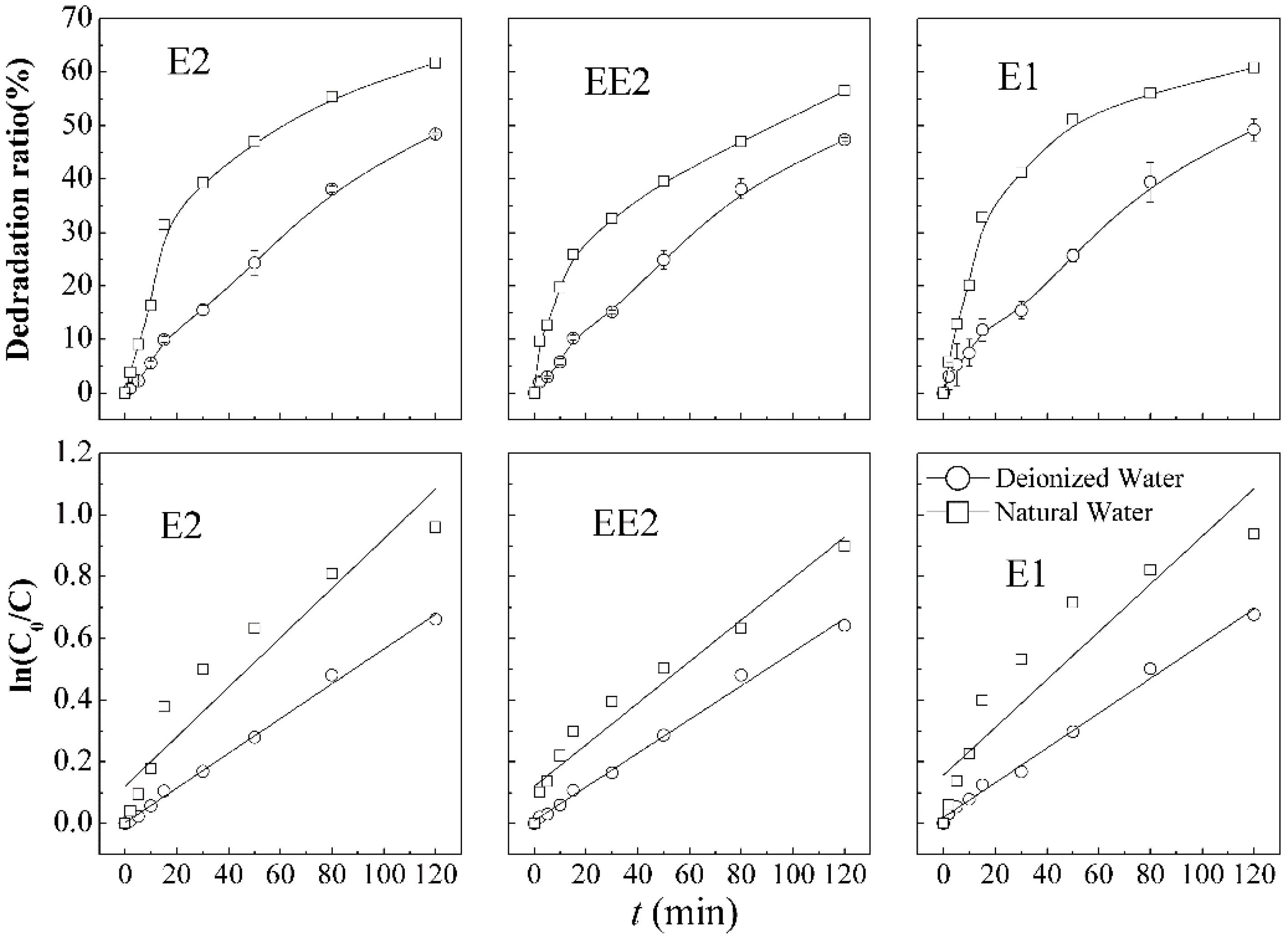
The comparison of SEs removal efficiency in different backgrounds is shown in
Figure 6. The degradation of E2 (49.2%), EE2 (48.4%), and E1 (47.4%) in pure water is lower than of E2 (61.8%), EE2 (56.5%), and E1 (60.8%) in natural water. The advantage was also reflected by the first order kinetic constants, with 0.00562 min
−1 (E2), 0.00546 min
−1 (EE2), 0.00563 min
−1 (E1) of pure water and 0.00814 min
−1 (E2), 0.00671 min
−1 (EE2), and 0.00774 min
−1 (E1) of natural water, respectively. The results demonstrate significant superior degradation in natural water background compared with those in pure water systems under the same process conditions.
Figure 6.
Steroid estrogens degradation curves and first order kinetic models in combination process under different aqueous background.
Figure 6.
Steroid estrogens degradation curves and first order kinetic models in combination process under different aqueous background.
Table 6.
Parameters of kinetic model of degradation of steroid estrogens under different aqueous background.
Table 6.
Parameters of kinetic model of degradation of steroid estrogens under different aqueous background.
| Steroid Estrogens | Backgrounds | Kinetic Equation | Reaction Rate Constant K/min−1 | R2 | Half-Life t1/2/min |
|---|
| E2 | Pure water | ln(C0/C) = 0.0056t + 0.003 | 0.0056 | 0.996 | 123 |
| Natural water | ln(C0/C) = 0.0080t + 0.122 | 0.0080 | 0.888 | 71 |
| EE2 | Pure water | ln(C0/C) = 0.0055t + 0.010 | 0.0055 | 0.994 | 124 |
| Natural water | ln(C0/C) = 0.0067t + 0.121 | 0.0067 | 0.944 | 100 |
| E1 | Pure water | ln(C0/C) = 0.0056t + 0.020 | 0.0056 | 0.993 | 120 |
| Natural water | ln(C0/C) = 0.0077t + 0.157 | 0.0077 | 0.867 | 70 |
In the natural water simulation system, the removal of estrogens increased significantly. The reason might be the presence of background components, including dissolved organic matter (DOM), cations, anions, organics
etc., enhancing the oxidation of estrogens by permanganate [
44]. DOM was reported to generate photo-oxidants that can accelerate organic contaminants when irradiated at a solar wavelength of 254 nm [
45]. The common bicarbonate ions were reported to be favorable in the elimination of micro-pollutants through carbonate radical producing, which can migrate towards the bulk of the solution and therefore induce the degradation, unlike the HO·radicals [
46].
In a natural aquatic environment, the degradation rates of these three types of SEs increase prominently in 40 min compared with that of SEs in pure water, as illustrated in
Figure 6, which also showed the removal rates decrease after that time point. Koyuncu
et al. pointed out that when humic acid (10 mg·L
−1) was added to the feed solution, the removal of the hormones increased to approximately 95% or greater with slightly higher values observed for the hormones alone [
47]. These observations of enhanced removal of hormones in raw water can be explained by the influence of natural organic matter. Shao
et al. reported the degradation of E1 by permanganate in different backgrounds and found that the removal efficiency of E1 in natural water is significantly better than that in an ultra-pure water system. Pétrier
et al. [
46] found that bicarbonate ion present in natural waters was favorable in the elimination of micro-pollutants through carbonate radical producing. Therefore, humic acid, reducing substances (SO
32−, NO
2− et al.), complexes (EDTA, citrate, oxalate), HCO
3−, phosphate
et al. obviously can promote the oxidation of steroid estrogens; appropriate common ions in natural water such as Mn
2+, Fe
2+ and Ca
2+ can enhance the removal of E1, slightly; however, Al
3+, Fe
3+ and Mg
2+ inhibit the degradation of estrogens during the permanganate oxidation process [
28,
44].
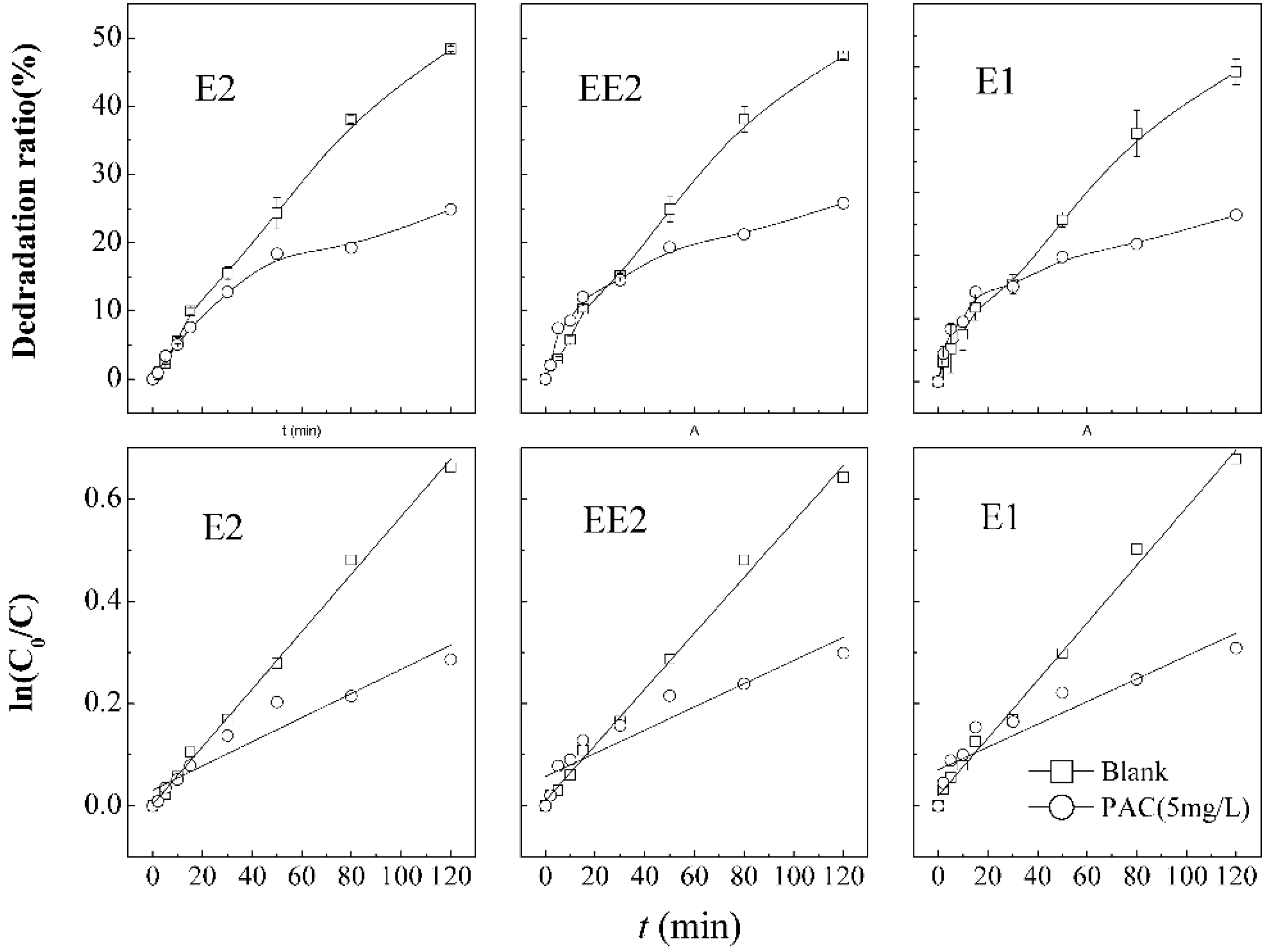
In this experiment, coagulation with PAC (5 mg·L
−1) was used to pre-treat the raw water, so it is unavoidable that Al
3+ ions will be introduced into the system. Investigation of the impact of Al
3+ ions on SEs degradation in the combined process was conducted and the results are shown in
Figure 7. The degradation of E2, EE2, and E1 decreases sharply to 24.9%, 25.8%, and 26.5%, respectively, after adding the coagulant PAC. The results indicated that coagulation of PAC exerts an inhibitory effect on the degradation, because the excess of Al
3+ ions produced in coagulation by the hydrolysis of PAC (Al
2(OH)
nCl
6−n, enhanced by ultrasound) had an adverse effect on the oxidation by the combined KMnO
4/Ultrasound process, which is similar to the conclusion of Shao
et al. [
28].
The TOC values of the samples were collected at different times to measure the steroid estrogens’ mineralizing degree by combined KMnO4/ultrasound process.
The results (presented in
Figure 8 and
Table 7) indicate that direct mineralizing degrees are unsatisfactory, with the TOC removal rate of 20.2% and 16.6% in natural water and pure water, respectively. In the combined simultaneous degradation system, the KMnO
4/ultrasound process has effects on the removal of SEs, but still a large proportion of SEs remains. Instead, some intermediate products may be generated which still devote themselves to TOC. The oxidant capacity of •OH and KMnO
4 is insufficient to completely decompose the steroid estrogens.
Figure 7.
Degradation curves and first order kinetic models of SEs in the presence of Al3+.
Figure 7.
Degradation curves and first order kinetic models of SEs in the presence of Al3+.
Figure 8.
Mineralization of SEs indicated by TOC in KMnO4 /ultrasound process.
Figure 8.
Mineralization of SEs indicated by TOC in KMnO4 /ultrasound process.
Table 7.
Parameters of degradation kinetic model of SEs in the presence of Al3+.
Table 7.
Parameters of degradation kinetic model of SEs in the presence of Al3+.
| Steroid Estrogens | Comparison of Condition | Kinetic Equation | Reaction Rate Constant K/min−1 | R2 | Half-Lifet1/2/min |
|---|
| E2 | Blank sample | ln(C0/C) = 0.0056t + 0.003 | 0.0056 | 0.9962 | 123 |
| Addition to PAC | ln(C0/C) = 0.0024t + 0.031 | 0.0024 | 0.9057 | 281 |
| EE2 | Blank sample | ln(C0/C) = 0.0055t + 0.010 | 0.0055 | 0.9939 | 125 |
| Addition to PAC | ln(C0/C) = 0.0023t + 0.057 | 0.0023 | 0.8526 | 281 |
| E1 | Blank sample | ln(C0/C) = 0.0056t + 0.020 | 0.0056 | 0.9931 | 120 |
| Addition to PAC | ln(C0/C) = 0.0022t + 0.070 | 0.0022 | 0.8337 | 280 |