A Temporal Association between Accumulated Petrol (Gasoline) Lead Emissions and Motor Neuron Disease in Australia
Abstract
:1. Introduction
1.1. Trends of Increases in Rates of Motor Neuron Disease (MND)
1.2. Lead and MND
1.3. Lead Exposure in Australia
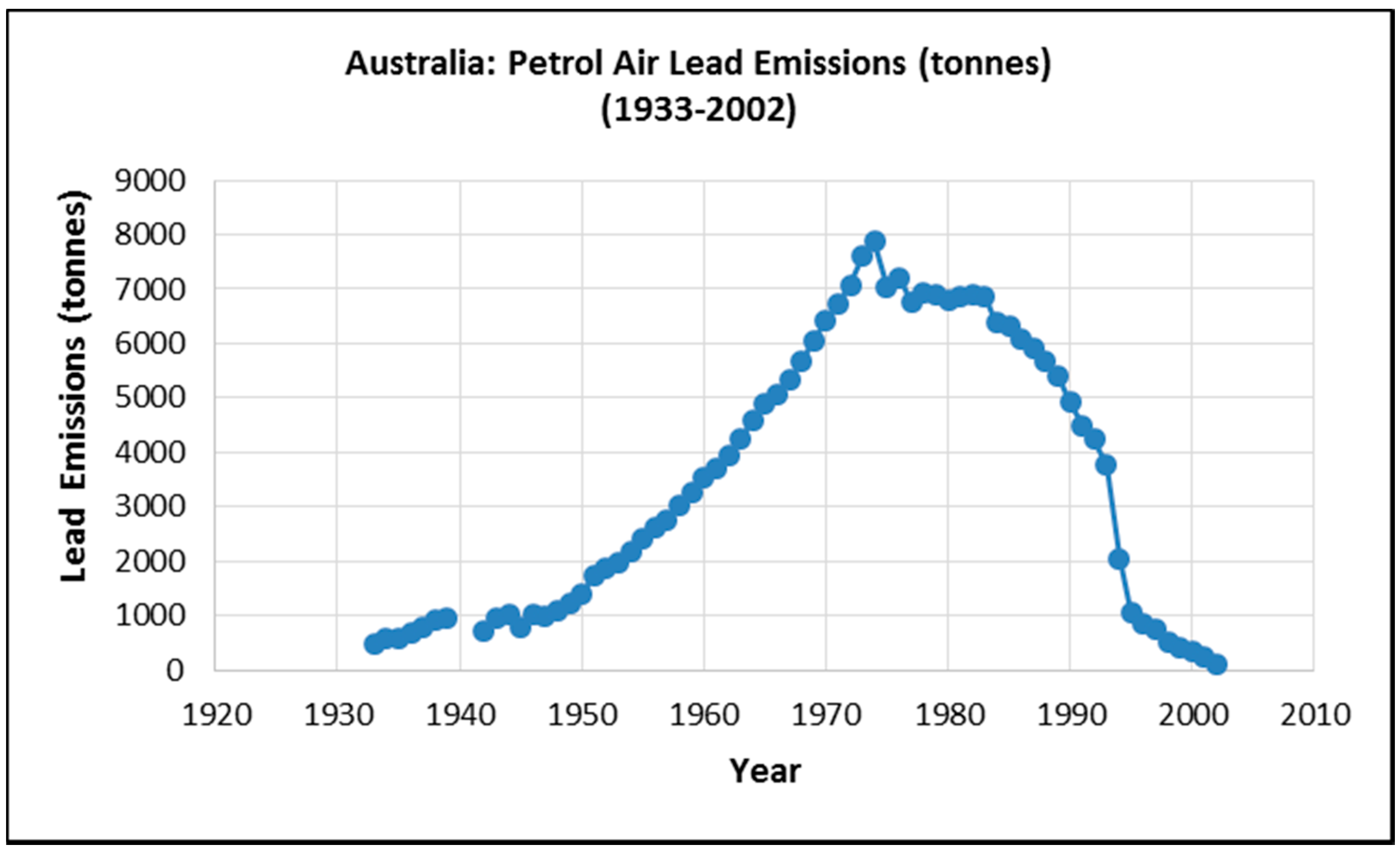
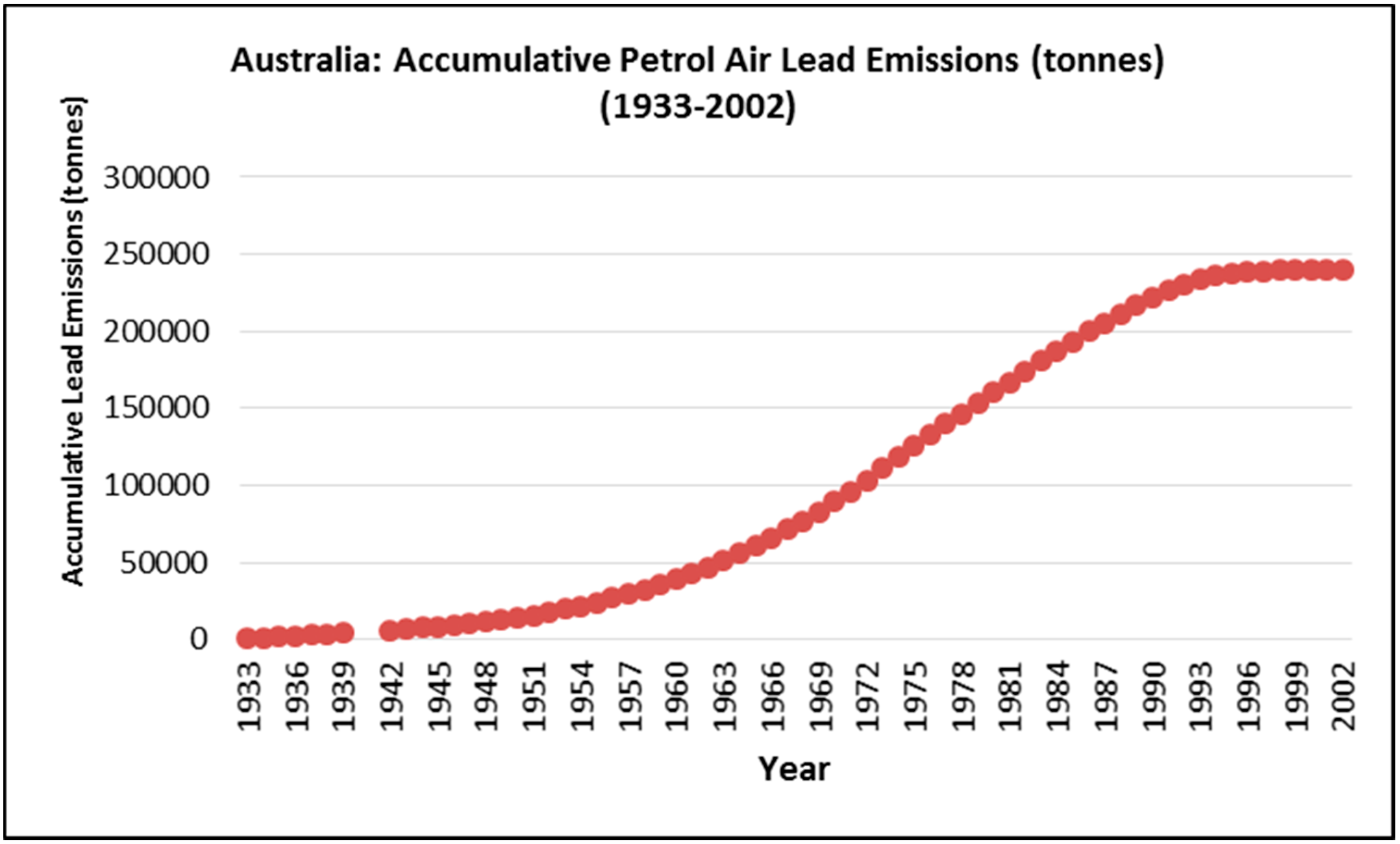
1.4. Accumulated Lead
2. Methods
3. Results
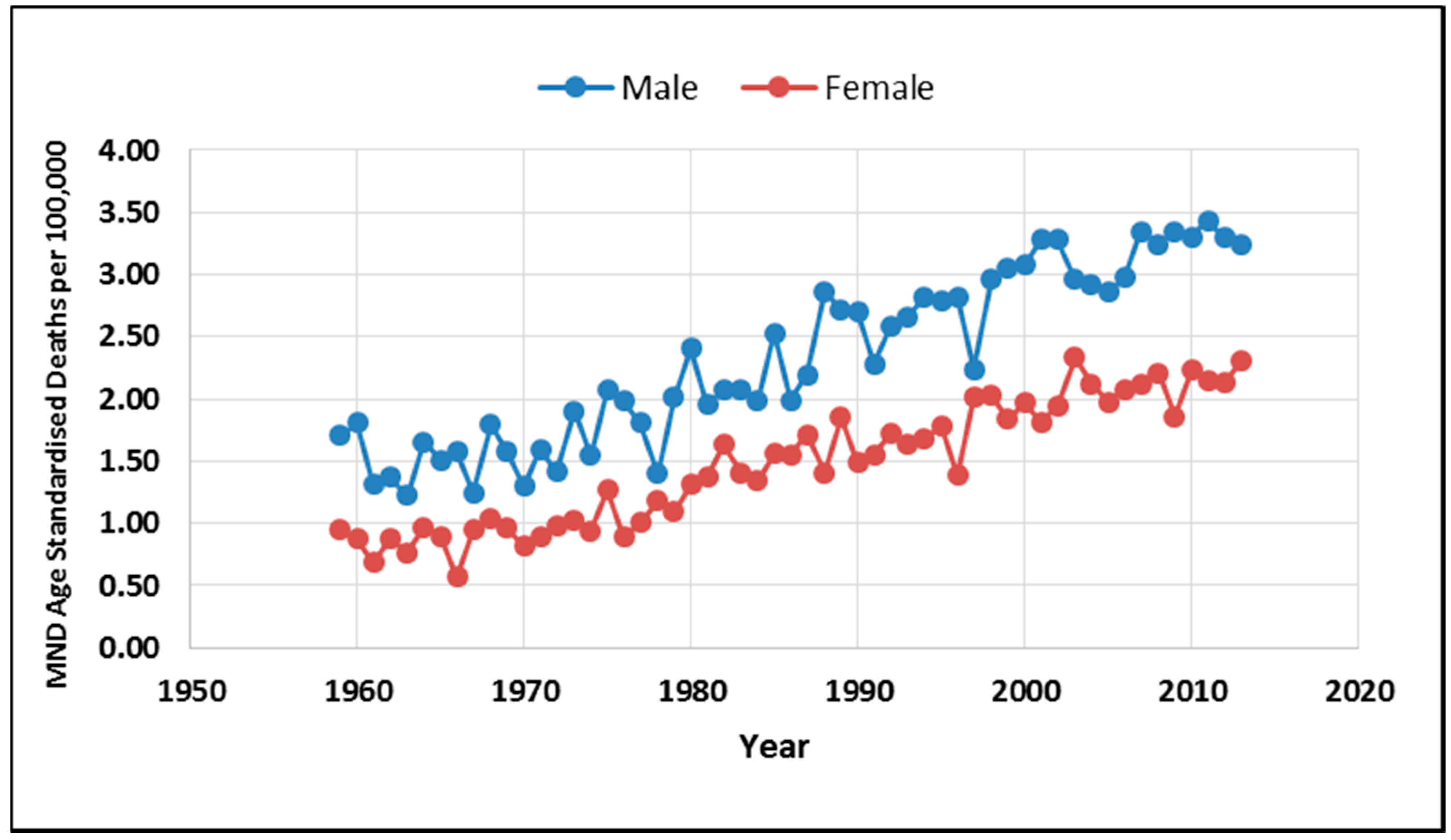

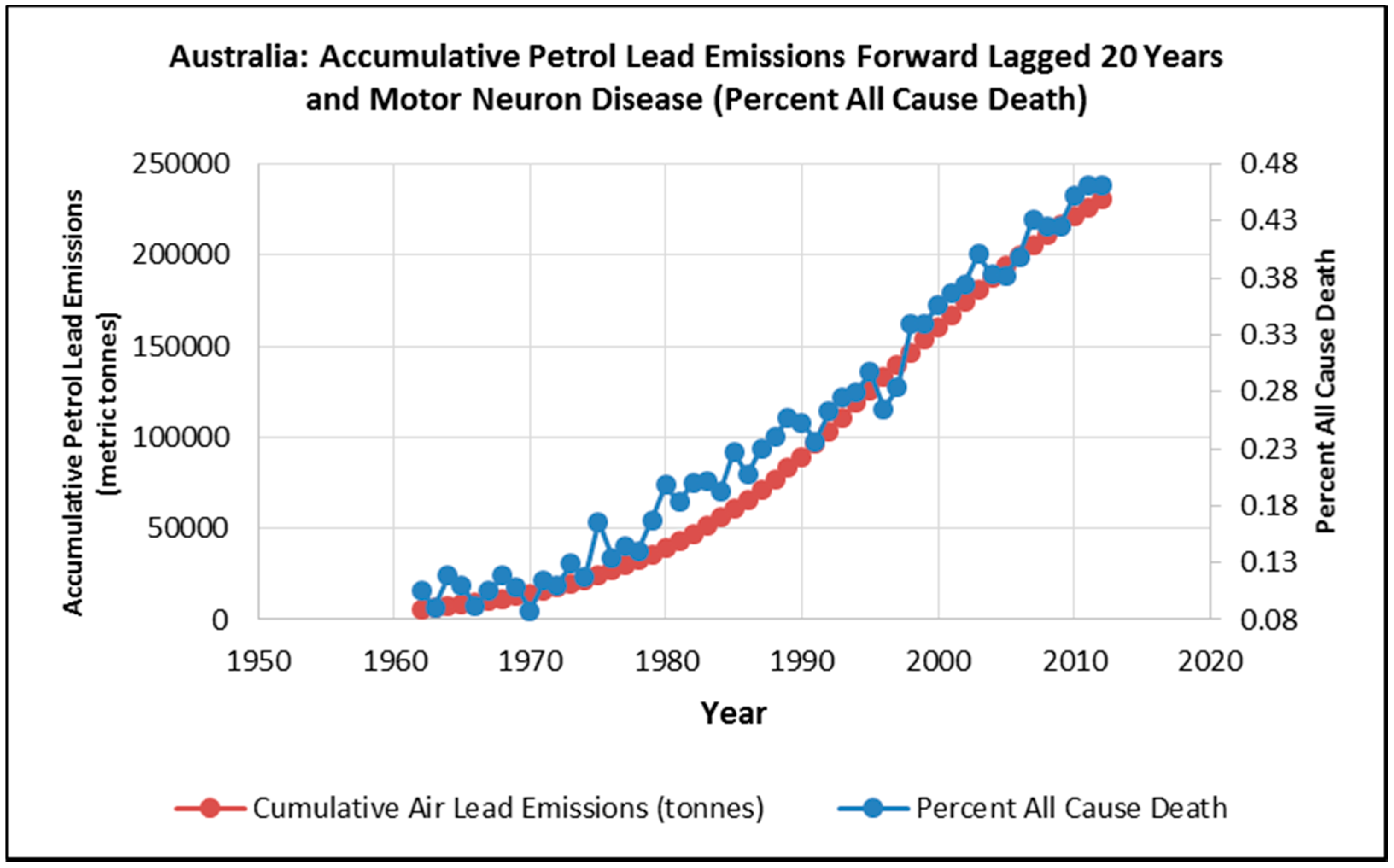
4. Discussion
4.1. Limitations and Additional Trend Rates
4.2. Possible Prediction of Future MND Death Trends in Australia
4.3. Need for Further Study
4.4. Need for Further Study
5. Conclusions
Supplementary Files
Supplementary File 1Supplementary File 2Acknowledgments
Author Contributions
Conflicts of Interest
References
- National Institute of Health (NIH). Motor Neuron Disease Fact Sheet. Available online: http://www.ninds.nih.gov/disorders/motor_neuron_diseases/detail_motor_neuron_diseases.htm (accessed on 24 November 2015).
- Veiga-Cabo, J.; Almazan-Isla, J.M.; Sendra-Gutiérrez, J.M.; de Pedro-Cuesta, J. Differential features of motor neuron disease mortality in Spain. Int. J. Epidemiol. 1997, 26, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Sylvain, D.; Alperovitch, A. Increasing trend of ALS in France and elsewhere: Are the changes real? Neurology 1989, 39, 768–768. [Google Scholar]
- Stuart, N.; Robinson, I.; Alperovitch, A. Rising amyotrophic lateral sclerosis mortality in France 1968–1990: Increased life expectancy and inter-disease competition as an explanation. J. Neurol. 1994, 241, 448–455. [Google Scholar]
- Vittorio, G.; Granieri, E.; Capone, J.; Manconi, M.; Casetta, I. Incidence of amyotrophic lateral sclerosis in the local health district of Ferrara, Italy, 1964–1998. Neuroepidemiology 2003, 22, 229–234. [Google Scholar]
- Seljeseth, Y.M.; Vollset, S.E.; Tysnes, O.B. Increasing mortality from amyotrophic lateral sclerosis in Norway? Neurology 2000, 55, 1262–1266. [Google Scholar] [CrossRef] [PubMed]
- Seals, R.M.; Hansen, J.; Gredal, O.; Weisskopf, M.G. Age-Period-Cohort analysis of trends in amyotrophic lateral sclerosis in Denmark, 1970–2009. Am. J. Epidemiol. 2013, 178, 1265–1271. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kazushi, O.; Kobashi, G.; Washio, M.; Sasaki, S.; Yokoyama, T.; Miyake, Y.; Sakamoto, N.; Tanaka, H.; Inaba, Y. Descriptive epidemiology of amyotrophic lateral sclerosis in Japan, 1995–2001. J. Epidemiol. 2005, 15, 20–23. [Google Scholar]
- Noonan, C.W.; White, M.C.; Thurman, D.; Wong, L. Temporal and geographic variation in United States motor neuron disease mortality, 1969–1998. Neurology 2005, 64, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Jeans, A.F.; Ansorge, O. Recent developments in the pathology of motor neurone disease. ACNR 2009, 9, 25–26. [Google Scholar]
- Wang, M.D.; Gomes, J.; Cashman, N.R.; Little, J.; Krewski, D. A meta-analysis of observational studies of the association between chronic occupational exposure to lead and amyotrophic lateral sclerosis. J. Occup. Environ. Med. 2014, 56, 1235–1242. [Google Scholar] [CrossRef] [PubMed]
- Caller, T.; Henegan, P.; Andrew, A.; Stommel, E. Possible environmental causes of amyotrophic lateral sclerosis in Northern New England: A preliminary analysis (P4.142). Available online: http://www.neurology.org/content/84/14_Supplement/P4.142.short (accessed on 8 September 2015).
- Fang, F.; Kwee, L.C.; Allen, K.D.; Umbach, D.M.; Ye, W.; Watson, M.; Keller, J.; Oddone, E.Z.; Sandler, D.P.; Schmidt, S.; et al. Association between blood lead and the risk of amyotrophic lateral sclerosis. Am. J. Epidemiol. 2010, 171, 1126–1133. [Google Scholar] [CrossRef] [PubMed]
- Afridi, H.I.; Talpur, F.N.; Kazi, T.G.; Kazi, N.; Arain, S.S.; Shah, F. Estimation of calcium, magnesium, cadmium, and lead in biological samples from paralyzed quality control and production steel mill workers. Environ. Monit. Assess. 2015, 187. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, M.; Brown, R.H., Jr.; Rogers, J.T.; Cudkowicz, M.E. Serum ferritin and metal levels as risk factors for amyotrophic lateral sclerosis. Open Neurol. J. 2008, 2. [Google Scholar] [CrossRef] [PubMed]
- Kurlander, H.M.; Patten, B.M. Metals in spinal cord tissue of patients dying of motor neuron disease. Ann. Neurol. 1979, 6, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Rabinowitz, M.B. Toxicokinetics of bone lead. Environ. Health Perspect. 1991, 91, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Silbergeld, E.K.; Sauk, J.; Somerman, M.; Todd, A.; McNeill, F.; Fowler, B.; Fontaine, A.; Van Buren, J. Lead in bone: Storage site, exposure source, and target organ. Neurotoxicology 1992, 14, 225–236. [Google Scholar]
- Gulson, B.L.; Mizon, K.J.; Korsch, M.J.; Palmer, J.M.; Donnelly, J.B. Mobilization of lead from human bone tissue during pregnancy and lactation: A summary of long-term research. Sci. Total. Env. 2003, 303, 79–104. [Google Scholar] [CrossRef]
- Pamphlett, R. Exposure to environmental toxins and the risk of sporadic motor neuron disease: An expanded Australian case—Control study. Eur. J. Neurol. 2012, 19, 1343–1348. [Google Scholar] [CrossRef] [PubMed]
- Campbell, A.M.G.; Williams, E.R.; Barltrop, D. Motor neurone disease and exposure to lead. J. Neurol. Neurosurg. Psychiatry 1970, 33, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Chancellor, A.M.; Slattery, J.M.; Fraser, H.; Warlow, C.P. Risk factors for motor neuron disease: A case-control study based on patients from the Scottish Motor Neuron Disease Register. J. Neurol. Neurosurg. Psychiatry 1993, 56, 1200–1206. [Google Scholar] [CrossRef] [PubMed]
- Kamel, F.; Umbach, D.M.; Munsat, T.L.; Shefner, J.M.; Hu, H.; Sandler, D.P. Lead exposure and amyotrophic lateral sclerosis. Epidemiology 2002, 13, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Kamel, F.; Umbach, D.M.; Lehman, T.A.; Park, L.P.; Munsat, T.L.; Shefner, J.M.; Sandler, D.P.; Hu, H.; Taylor, J.A. Amyotrophic lateral sclerosis, lead, and genetic susceptibility: Polymorphisms in the deltaaminolevulinic acid dehydratase and vitamin D receptor genes. Environ. Health Perspect. 2003, 111, 1335–1339. [Google Scholar] [CrossRef] [PubMed]
- Kamel, F.; Umbach, D.M.; Hu, H.; Munsat, T.L.; Shefner, J.M.; Taylor, J.A.; Sandler, D.P. Lead exposure as a risk factor for amyotrophic lateral sclerosis. Neurodegener. Dis. 2005, 2, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Eum, K.D.; Seals, R.M.; Taylor, K.M.; Grespin, M.; Umbach, D.M.; Hu, H.; Sandler, D.P.; Kamel, F.; Weisskopf, M.G. Modification of the association between lead exposure and amyotrophic lateral sclerosis by iron and oxidative stress related gene polymorphisms. Amyotroph. Lateral Scler. 2015, 16, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.P.; Hudson-Edwards, K.A.; Mackay, A.K.; Holz, E. Soil Cd, Cu, Pb and Zn contaminants around Mount Isa city, Queensland, Australia: Potential sources and risks to human health. Appl. Geochem. 2010, 25, 841–855. [Google Scholar] [CrossRef]
- Mackay, A.K.; Taylor, M.P.; Munksgaard, N.C.; Hudson-Edwards, K.A.; Burn-Nunes, L. Identification of environmental lead sources, pathways and forms in a mining and smelting town: Mount Isa, Australia. Environ. Pollut. 2013, 180, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Laidlaw, M.A.; Taylor, M.P. Potential for childhood lead poisoning in the inner cities of Australia due to exposure to lead in soil dust. Environ. Pollut. 2011, 159, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Laidlaw, M.A.S.; Zahran, S.; Pingitore, N.; Clague, J.; Devlin, G.; Taylor, M.P. Identification of lead sources in residential environments: Sydney, Australia. Environmental Pollut. 2014, 184, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, D.A.; Genuis, S.J.; Frye, R.E. Environmental toxicants and autism spectrum disorders: A systematic review. Transl. Psychiatry 2014, 11. [Google Scholar] [CrossRef] [PubMed]
- Gorini, F.; Muratori, F.; Morales, M.A. The role of heavy metal pollution in neurobehavioral disorders: A focus on autism. Rev. J. Autism Deve. Disord. 2014, 1, 354–372. [Google Scholar] [CrossRef]
- Kennedy, D.A.; Woodland, C.; Koren, G. Lead exposure, gestational hypertension and pre-eclampsia: A systematic review of cause and effect. J. Obstet. Gynaecol. 2012, 32, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Earl, R.; Burns, N.; Nettlebeck, T.; Baghurst, P. Low-level environmental lead exposure still negatively associated with children’s cognitive abilities. Aust. J. Psychol. 2015, 6. [Google Scholar] [CrossRef]
- Navas-Acien, A.; Guallar, E.; Silbergeld, E.K.; Rothenberg, S.J. Lead exposure and cardiovascular disease—A systematic review. Environ. Health. Perspect. 2007, 115, 472–482. [Google Scholar]
- Goodlad, J.K.; Marcus, D.K.; Jessica, J. Fulton lead and attention-deficit/hyperactivity disorder (ADHD) symptoms: A meta-analysis. Clin. Psych. Rev. 2013, 33, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Genuis, S.J.; Kelln, K.L. Toxicant exposure and bioaccumulation: A common and potentially reversible cause of cognitive dysfunction and dementia. Behav. Neurol. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Tomás, R.; Guilarte, T.R.; Opler, M.; Pletnikov, M. Is lead exposure in early life an environmental risk factor for Schizophrenia? Neurobiological connections and testable hypotheses. NeuroToxicology 2012, 33, 560–574. [Google Scholar]
- Bellinger, D.C. The protean toxicities of lead: New chapters in a familiar story. Int. J. Environ. Res. Public Health 2011, 8, 2593–2628. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, L.J. Quantification of atmospheric lead emissions from 70 years of leaded petrol consumption in Australia. Atmos. Environ. 2015, 111, 195–201. [Google Scholar] [CrossRef]
- Semlali, R.M.; Dessogne, J.B.; Monna, F.; Bolte, J.; Azimi, S.; Denaix, L.; Loubet, M.; Chateau, C.; van Oort, F. Modeling lead input and output in soils using lead isotopic geochemistry. Environ. Sci. Tech. 2004, 38, 1513–1521. [Google Scholar] [CrossRef]
- MND Australia. Facts and Figures. Available online: https://www.mndaust.asn.au/Discover-our-research/grants-and-research-meetings/Statistics.aspx (accessed on 24 November 2015).
- General Record of Incidence of Mortality (GRIM) Books. Available online: http://www.aihw.gov.au/deaths/grim-books/ (accessed on 8 September 2015).
- Morgenstern, H. Ecological studies. In Modern Epidemiology, 2nd ed.; Rothman, K.J., Greenland, S., Eds.; Philadelphia: Lippincott-Raven, Philadelphia, USA, 1998; pp. 459–480. [Google Scholar]
- Chio, A. Risk factors in the early diagnosis of ALS: European epidemiological studies. Amyotroph. Lateral Scler. 2000, 1, S13–S18. [Google Scholar] [CrossRef]
- Agosta, F.; Al-Chalabi, A.; Filippi, M.; Hardiman, O.; Kaji, R.; Meininger, V.; Nakano, I.; et al. The El Escorial criteria: Strengths and weaknesses. Amyotroph. Lateral Scler. 2015, 16, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Valdmanis, P.N.; Rouleau, G. Genetics of familial amyotrophic lateral sclerosis. Neurology 2008, 70, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Quinlan, P.; Ye, W.; Barber, M.K.; Umbach, D.M.; Sandler, D.P.; Kamel, F. Workplace exposures and the risk of amyotrophic lateral sclerosis. Environ Health Perspect. 2009, 117, 1387–1392. [Google Scholar] [CrossRef] [PubMed]
- Morahan, J.; Pamphlett, R. Amyotrophic lateral sclerosis and exposure to environmental toxins: An Australian case-control study. Neuroepidemiology 2006, 27, 130–135. [Google Scholar] [CrossRef] [PubMed]
- McGuire, V.; Longstreth, W.T.; Nelson, L.M.; Koepsell, T.D.; Checkoway, H.; Morgan, M.S.; van Belle, G. Occupational exposures and amyotrophic lateral sclerosis. A population-based case-control study. Am. J. Epidemiol. 1997, 145, 1076–1088. [Google Scholar] [CrossRef] [PubMed]
- Francesca, B.; Marcello, N.; Mandrioli, J.; Pietrini, V.; Vinceti, M. Exposure to pesticides and risk of amyotrophic lateral sclerosis: A population-based case-control study. Ann. Ist. Super. Sanita 2010, 46, 284–287. [Google Scholar]
- Pablo, J.; Banack, S.A.; Cox, P.A.; Johnson, T.E.; Papapetropoulos, S.; Bradley, W.G.; Buck, A.; Mash, D.C. Cyanobacterial neurotoxin BMAA in ALS and Alzheimer’s disease. Acta Neurol. Scand. 2009, 120, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Mehal, J.M.; Holman, R.C.; Schonberger, L.B.; Sejvar, J.J. Amyotrophic lateral sclerosis/motor neuron disease deaths in the United States, 1999–2009. Amyotroph. Lateral Scler. 2013, 14, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Sorenson, E.J.; Stalker, A.P.; Kurland, L.T.; Windebank, A.J. Amyotrophic lateral sclerosis in Olmsted County, Minnesota, 1925 to 1998. Neurology 2002, 59, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Swingler, R.J.; Compston, D.A.S. The morbidity of multiple sclerosis. QJM 1992, 1, 325–337. [Google Scholar]
- Forbes, R.B.; Colville, S.; Cran, G.W.; Swingler, R.J. Unexpected decline in survival from amyotrophic lateral sclerosis/motor neurone disease. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1753–1755. [Google Scholar] [CrossRef] [PubMed]
- Alvaro, A.; Logroscino, G.; Jick, S.S.; Hernán, M.A. Incidence and lifetime risk of motor neuron disease in the United Kingdom: A population-based study. Eur. J. Neurol. 2009, 16, 745–751. [Google Scholar]
- Lovei, M. Phasing out Lead from Gasoline: Worldwide Experience and Policy Implications; World Bank Publications: Washington, DC, USA, 1998. [Google Scholar]
- Phasing Lead Out of Gasoline: An Examination of Policy Approaches in Different Countries. Available online: http://www.grida.no/news/press/2056.aspx (accessed on 8 September 2015).
- EPA Takes Final Step in Phaseout of Leaded Gasoline. Available online: http://www2.epa.gov/aboutepa/epa-takes-final-step-phaseout-leaded-gasoline (accessed on 9 November 2015).
- Laidlaw, M.A.; Filippelli, G.M. Resuspension of urban soils as a persistent source of lead poisoning in children: A review and new directions. Appl. Geochem. 2008, 23, 2021–2039. [Google Scholar] [CrossRef]
- Mielke, H.W. Nature and extent of metal contaminated soils in urban environments. Environ. Geochem. Health 2016, in press. [Google Scholar]
- Laidlaw, M.A.S.; Zahran, S.; Mielke, H.W.; Taylor, M.P.; Filippelli, G.M. Re-suspension of lead contaminated urban soil as a dominant source of atmospheric lead in Birmingham, Chicago, Detroit and Pittsburgh, USA. Atmos. Environ. 2012, 49, 302–310. [Google Scholar] [CrossRef]
- Gulson, B.; Taylor, A. Relationship of Pb in house dust and ambient. Air. Mod. Chem. Appl. 2014, 2. [Google Scholar] [CrossRef]
- Hunt, A.; Johnson, D.L. Suspension and re-suspension of dry soil indoors following track-in on footwear. Environ. Geochem. Health 2012, 34, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Hunt, A.; Johnson, D.L.; Griffith, D.A. Mass transfer of soil indoors by track-in on footwear. Sci. Total Environ. 2006, 370, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Zahran, S.; Mielke, H.W.; McElmurry, S.P.; Filippelli, G.M.; Laidlaw, M.A.S.; Taylor, M.P. Determining the relative importance of soil sample locations to predict risk of children’s lead (Pb) exposure. Environ. Int. 2013, 60, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Zahran, S.; Laidlaw, M.; McElmurry, S.; Filippelli, G.M.; Taylor, M. Linking source and effect: Resuspended soil lead, air lead, and children’s blood lead levels in Detroit, Michigan. Environ. Sci. Tech. 2013, 47, 2839–2845. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, M.; Siddiqui, M.K.J. Low level lead exposure and oxidative stress: Current opinions. Clin. Chim. Acta 2007, 383, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Lopes, A.; Almeida, C.B.; Peixe, T.S.; Mesas, A.E.; Paoliello, M. Lead exposure and oxidative stress: A systematic review. Rev. Environ. Contam. Toxicol. 2016, 236, 193–238. [Google Scholar] [PubMed]
- Hiroshi, M.; Santella, R.M.; Liu, X.; Bogdanov, M.; Zipprich, J.; Wu, H.; Mahata, J.; Kilty, M.; Bednarz, K.; Bell, D.; et al. Oxidative stress biomarkers in sporadic ALS. Amyotroph. Lateral Scler. 2008, 9, 177–183. [Google Scholar]
- Barber, S.C.; Mead, R.J.; Shaw, P.J. Oxidative stress in ALS: A mechanism of neurodegeneration and a therapeutic target. Biochim. Biophys. Acta 2006, 1762, 1051–1067. [Google Scholar] [CrossRef] [PubMed]
- The Precautionary Principle. World Commission on the Ethics of Scientific Knowledge and Technology. Available online: http://unesdoc.unesco.org/images/0013/001395/139578e.pdf (accessed on 27 August 2015).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laidlaw, M.A.S.; Rowe, D.B.; Ball, A.S.; Mielke, H.W. A Temporal Association between Accumulated Petrol (Gasoline) Lead Emissions and Motor Neuron Disease in Australia. Int. J. Environ. Res. Public Health 2015, 12, 16124-16135. https://doi.org/10.3390/ijerph121215047
Laidlaw MAS, Rowe DB, Ball AS, Mielke HW. A Temporal Association between Accumulated Petrol (Gasoline) Lead Emissions and Motor Neuron Disease in Australia. International Journal of Environmental Research and Public Health. 2015; 12(12):16124-16135. https://doi.org/10.3390/ijerph121215047
Chicago/Turabian StyleLaidlaw, Mark A. S., Dominic B. Rowe, Andrew S. Ball, and Howard W. Mielke. 2015. "A Temporal Association between Accumulated Petrol (Gasoline) Lead Emissions and Motor Neuron Disease in Australia" International Journal of Environmental Research and Public Health 12, no. 12: 16124-16135. https://doi.org/10.3390/ijerph121215047
APA StyleLaidlaw, M. A. S., Rowe, D. B., Ball, A. S., & Mielke, H. W. (2015). A Temporal Association between Accumulated Petrol (Gasoline) Lead Emissions and Motor Neuron Disease in Australia. International Journal of Environmental Research and Public Health, 12(12), 16124-16135. https://doi.org/10.3390/ijerph121215047






