Treatment of Actual Chemical Wastewater by a Heterogeneous Fenton Process Using Natural Pyrite
Abstract
:1. Introduction
2. Experimental Section
2.1. Reagents and Wastewater
| Index | COD (mg/L) | BOD5/COD | TOC (mg/L) | Acute Biotoxicity (mg Zn2+/L) | Nitrobenzene (mg/L) | pH | Conductivity (μS/cm) |
|---|---|---|---|---|---|---|---|
| Values | 7500–8000 | 0.1 | 2000 | 471–490 | >300 | 1.8 | 38,000 |
2.2. Degradation of Chemical Wastewater by the Pyrite Fenton System and the Classic Fenton System
2.3. Effects of the Dosage of Pyrite and H2O2
2.4. Analytical Methods
3. Results and Discussion
3.1. Comparison of COD Removal in the Pyrite Fenton System and the Classic Fenton System
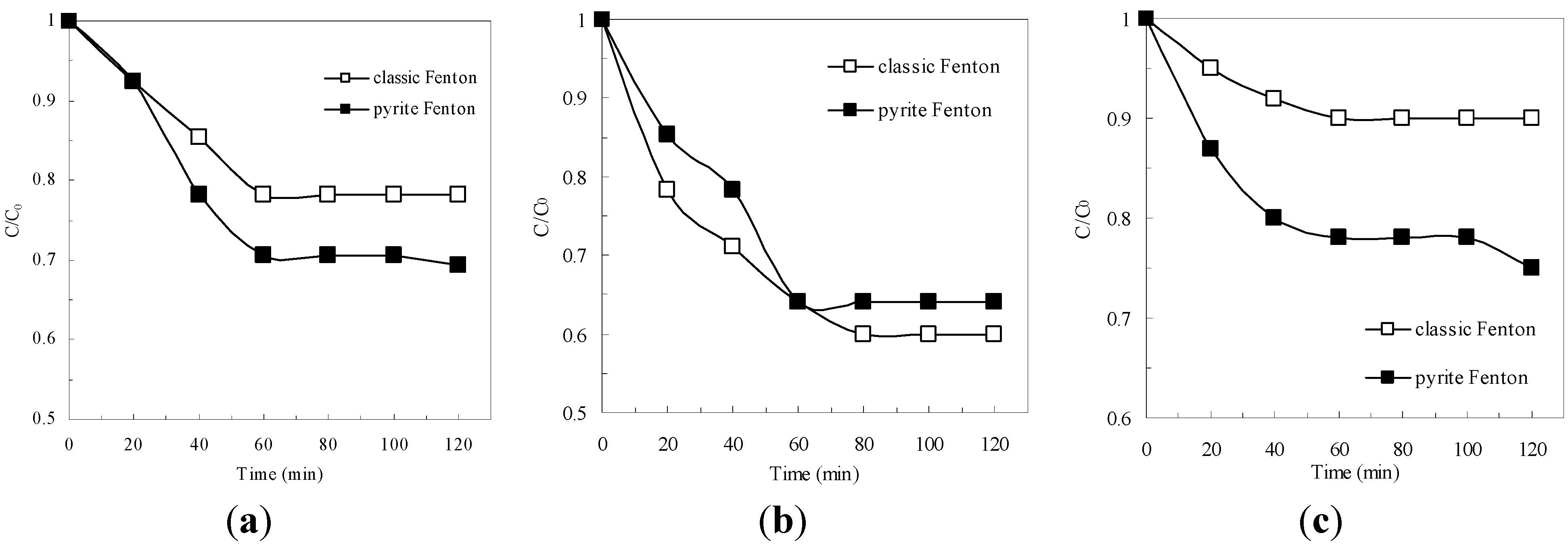
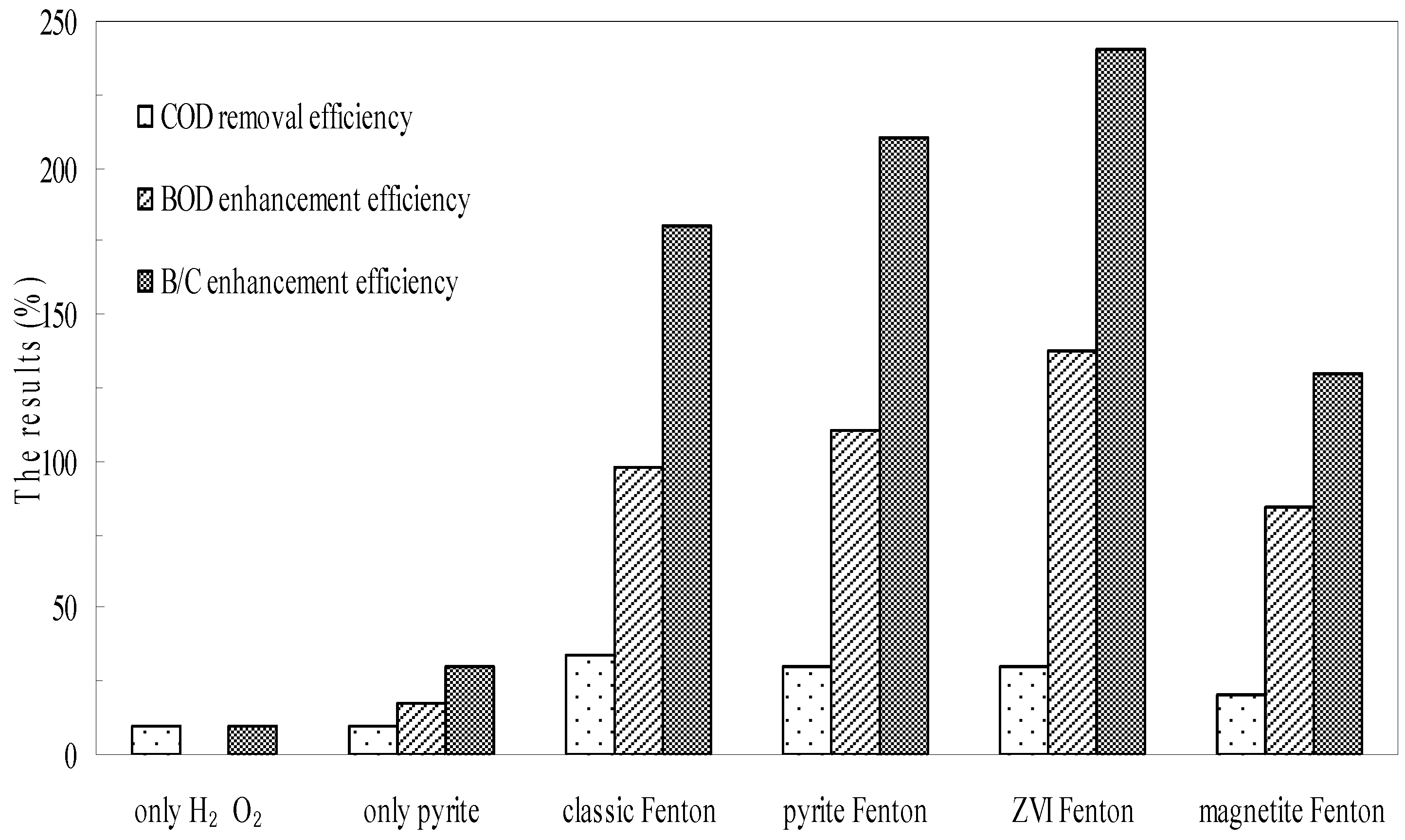
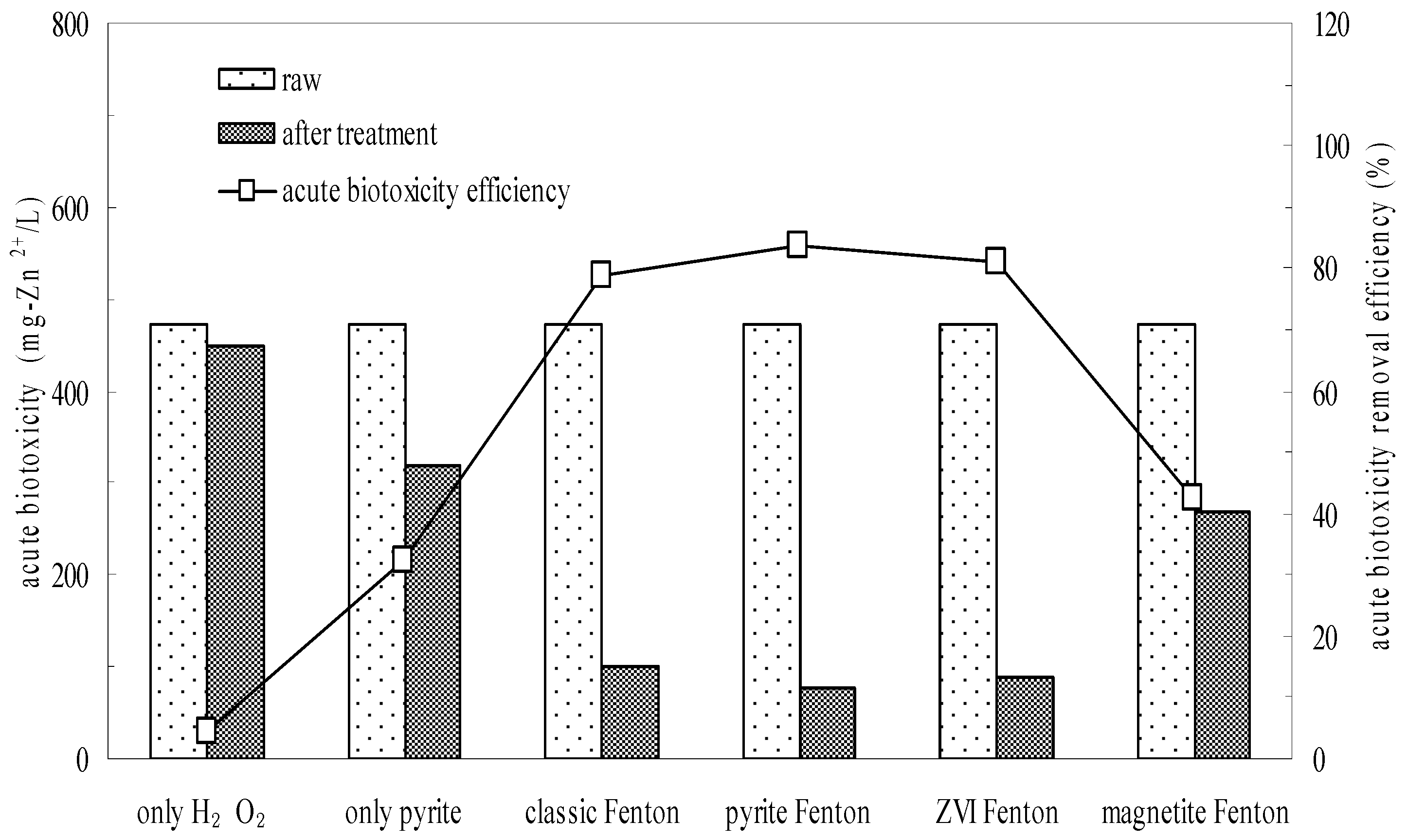
3.2. Biodegradability Enhancement and Acute Biotoxicity Reduction by the Pyrite Fenton System
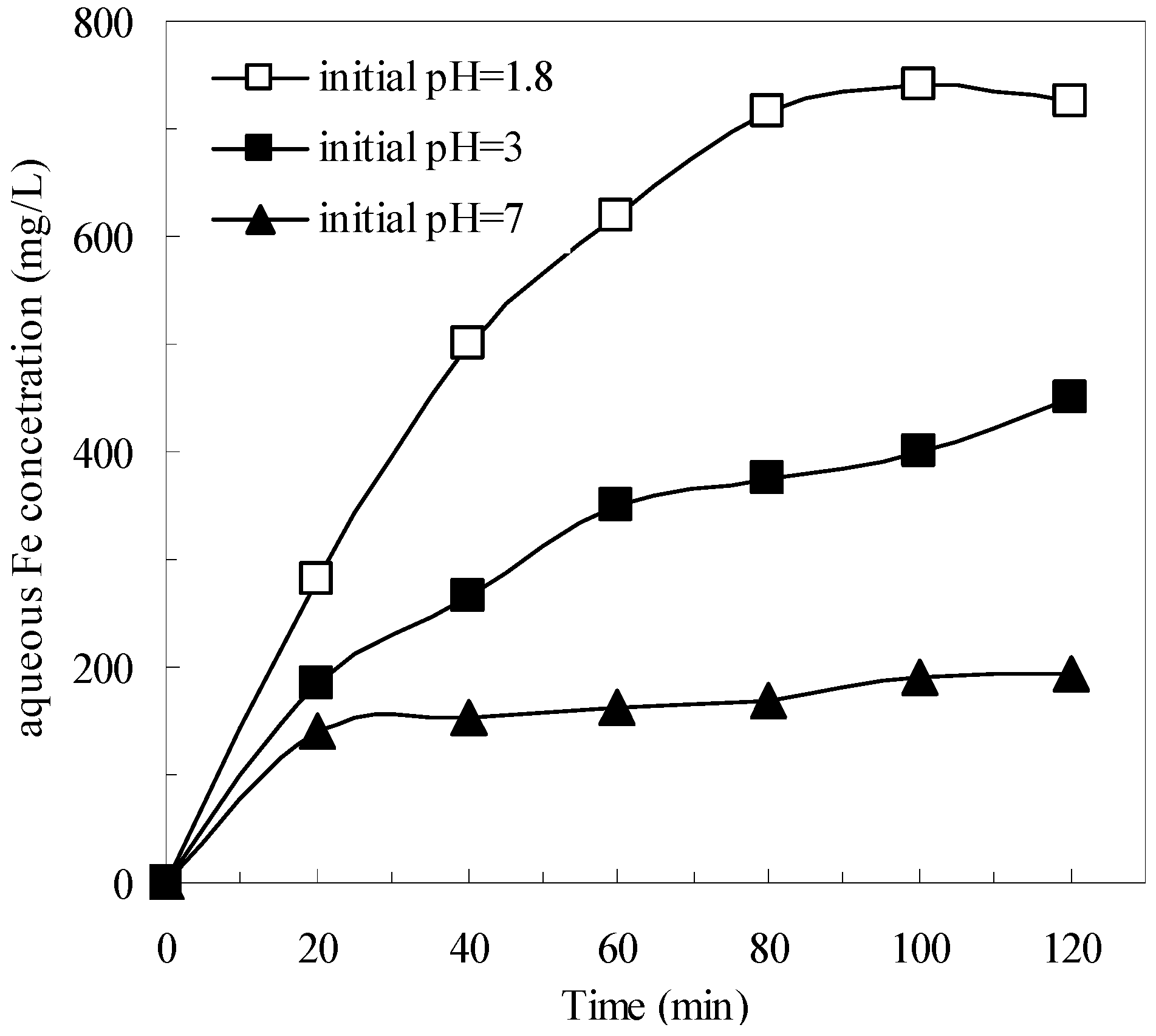
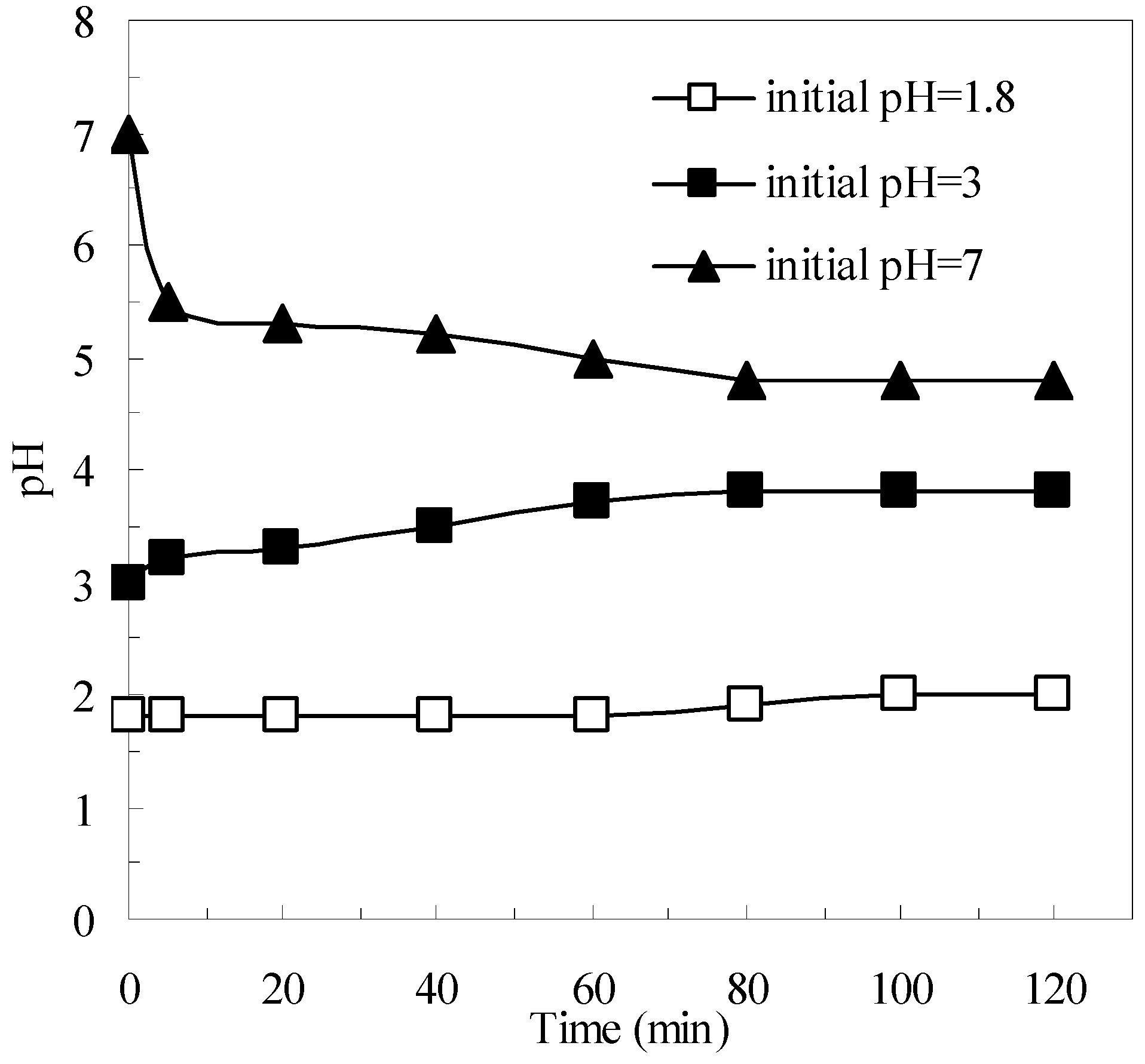
3.3. Effects of the Pyrite and H2O2 on the Removal of Organics in the Pyrite Fenton System


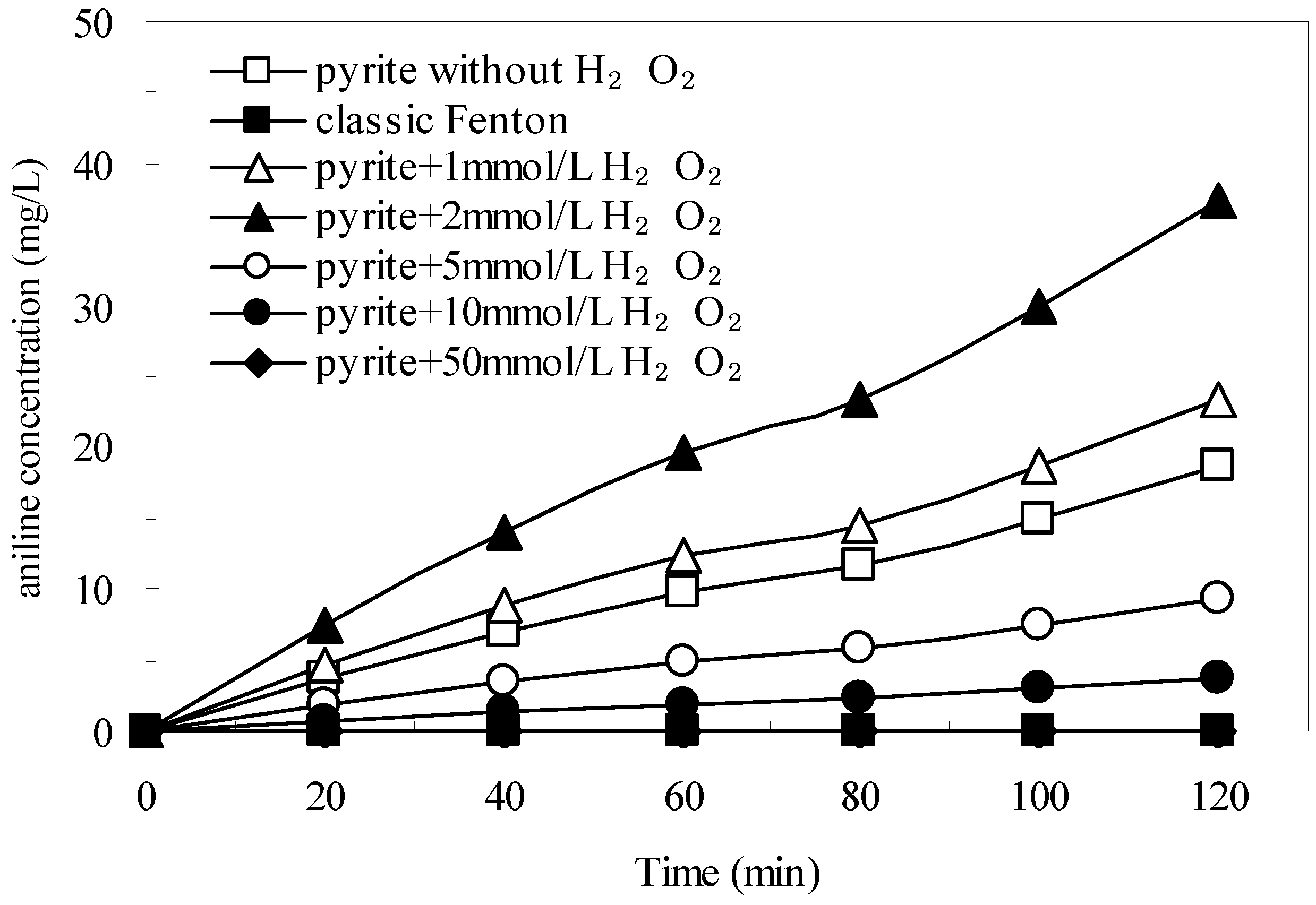
3.4. The Generation of Aniline in the Pyrite Fenton System

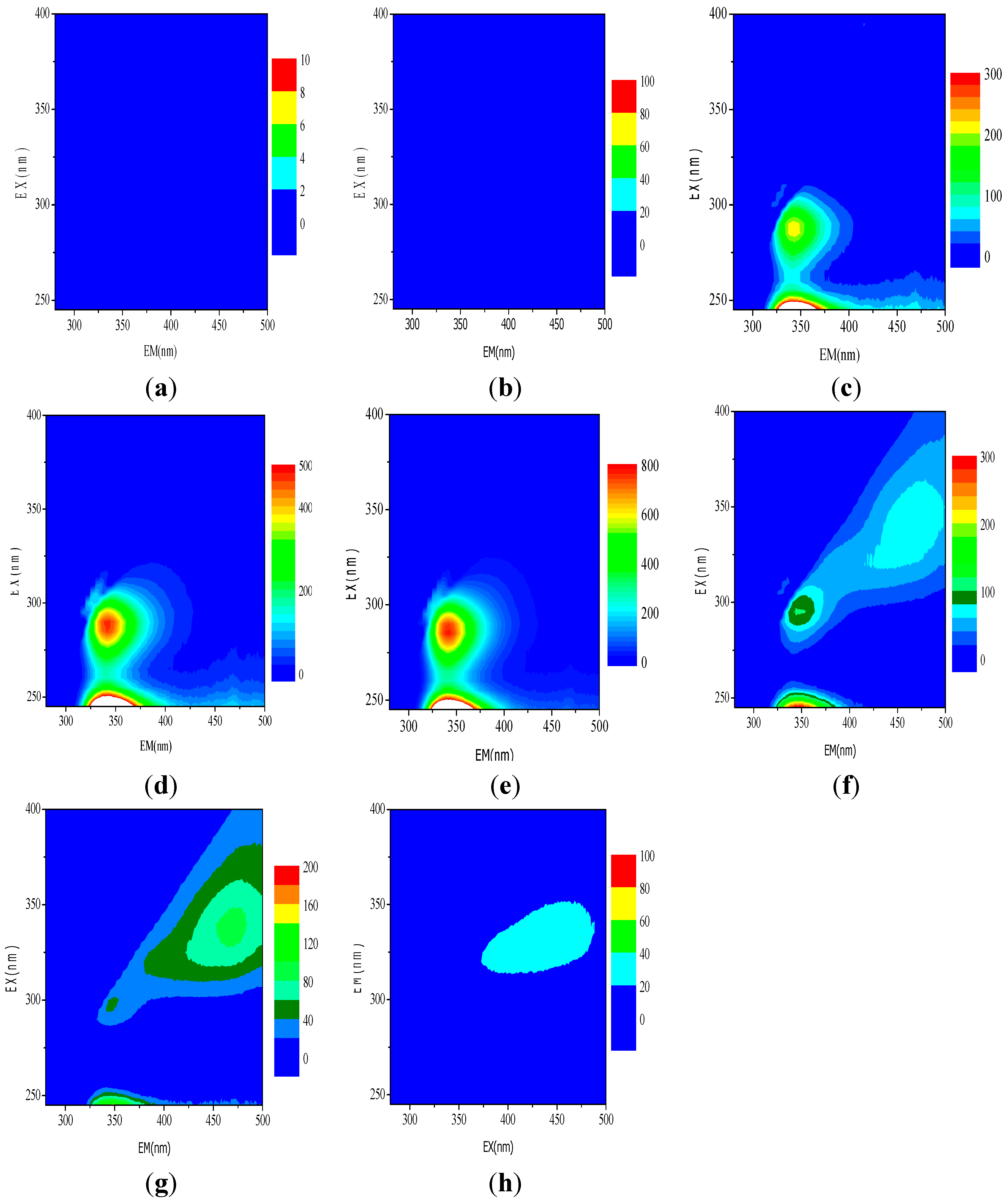
3.5. Evaluation of Organic Functional Groups by EEM in the Pyrite Fenton System
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fan, J.H.; Ma, L.M. The pretreatment by the Fe-Cu process for enhancing biological degradability of the mixed wastewater. J. Hazard. Mater. 2009, 164, 1392–1397. [Google Scholar] [CrossRef]
- Sun, L.; Wang, C.; Ji, M.; Kong, X. Treatment of mixed chemical wastewater and the agglomeration mechanism via an internal electrolysis filter. Chem. Eng. J. 2013, 215–216, 50–56. [Google Scholar] [CrossRef]
- Wu, C.Y.; Zhou, Y.X.; Wang, P.C.; Guo, S.J. Improving hydrolysis acidification by limited aeration in the pretreatment of petrochemical wastewater. Bioresour. Technol. 2015, 194, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.X.; Li, Y.M.; Li, J.F.; Sheng, G.D.; Hu, L.J.; Zheng, X.M. Immobilization of nanoscale zero valent iron on organobentonite for accelerated reduction of nitrobenzene. J. Chem. Technol. Biotechnol. 2014, 89, 1961–1966. [Google Scholar] [CrossRef]
- Yin, W.Z.; Wu, J.H.; Huang, W.L.; Wei, C.H. Enhanced nitrobenzene removal and column longevity by coupled abiotic and biotic processes in zero-valent iron column. Chem. Eng. J. 2015, 259, 417–423. [Google Scholar] [CrossRef]
- Chamarro, E.; Marco, A.; Esplugas, E. Use of Fenton reagent to improve organic chemical biodegradability. Water Res. 2001, 35, 1047–1051. [Google Scholar] [CrossRef]
- Canizares, P.; Paz, R.; Saez, C.; Rodrigo, M.A. Costs of the electrochemical oxidation of wastewaters: A comparison with ozonation and Fenton oxidation processes. J. Environ. Manag. 2009, 90, 410–420. [Google Scholar] [CrossRef] [PubMed]
- Dariush, S.; Moheb, A.; Abbas, R.; Larouk, S.; Roy, R.; Azzouz, A. Total mineralization of sulfamethoxazole and aromatic pollutants through Fe(2+)-montmorillonite catalyzed ozonation. J. Hazard. Mater. 2015, 298, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Nakai, S.; Okuda, T.; Okada, M. Production of mono-and di-carboxylated polyethylene glycols as a factor obstacle to the successful ozonation-assisted biodegradation of ethoxylated compounds. Chemosphere 2015, 136, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Verma, A.; Rajput, H. Potential use of foundry sand as heterogeneous catalyst in solar photo-fenton degradation of herbicide isoproturon. Int. J. Environ. Res. 2015, 9, 85–92. [Google Scholar]
- Doumic, L.I.; Soares, P.A.; Ayude, M.A.; Cassanello, M.; Boaventura, R.A.R.; Vilar, V.J.P. Enhancement of a solar photo-Fenton reaction by using ferrioxalate complexes for the treatment of a synthetic cotton-textile dyeing wastewater. Chem. Eng. J. 2015, 277, 86–96. [Google Scholar] [CrossRef]
- Che, H.; Lee, W. Selective redox degradation of chlorinated aliphatic compounds by Fenton reaction in pyrite suspension. Chemosphere 2011, 82, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Fang, Z.; Yan, X.; Cheng, W. Heterogeneous sono-Fenton catalytic degradation of bisphenol A by Fe3O4 magnetic nanoparticles under neutral condition. Chem. Eng. J. 2012, 197, 242–249. [Google Scholar] [CrossRef]
- Masomboon, N.; Ratanatamskul, C.; Lu, M.C. Chemical oxidation of 2,6-dimethylaniline in the Fenton process. Environ. Sci. Technol. 2009, 43, 8629–8634. [Google Scholar] [CrossRef] [PubMed]
- Matta, R.; Hanna, K.; Chiron, S. Fenton-like oxidation of 2,4,6-trinitrotoluene using different iron minerals. Sci. Total. Environ. 2007, 385, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Maria, A.F.D.; Emilio, R.; María, F.F.; Marta, P.; Maria, Á.S. Degradation of organic pollutants by heterogeneous electro-Fenton process using Mn-alginate composite. J. Chem. Technol. Biotechnol. 2015, 90, 1439–1447. [Google Scholar] [CrossRef]
- Hu, X.B.; Deng, Y.H.; Gao, Z.Q.; Liu, B.Z.; Sun, C. Transformation and reduction of androgenic activity of 17 alpha-methyltestosterone in Fe3O4/MWCNTs-H2O2 system. Appl. Catal. B-Environ. 2012, 127, 167–174. [Google Scholar] [CrossRef]
- Todd, E. Surface oxidation of pyrite under ambient atmo-spheric and aqueous (pH = 2 to 10) conditions: Electronic structure and mineralogy from X-ray absorption spectroscopy. Geochim. Cosmochim. Acta 2003, 67, 881–893. [Google Scholar] [CrossRef]
- Arienzo, M. Oxidizing 2,4,6-trinitroluene with pyrite-H2O2 suspensions. Chemosphere 1999, 39, 1629–1638. [Google Scholar] [CrossRef]
- Che, H.; Bae, S.; Lee, W. Degradation of trichloroethylene by Fenton reaction in pyrite suspension. J. Hazard. Mater. 2011, 185, 1355–1361. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Kim, D.; Lee, W. Degradation of diclofenac by pyrite catalyzed Fenton oxidation. Appl. Catal. B-Environ. 2013, 134, 93–102. [Google Scholar] [CrossRef]
- Wu, D.L.; Feng, Y.; Ma, L.M. Oxidation of Azo Dyes by H2O2 in presence of natural pyrite. Water Air Soil. Pollut. 2013, 224. [Google Scholar] [CrossRef]
- Zhang, Y.L.; Zhang, K.; Dai, C.M.; Zhou, X.F.; Si, H.P. An enhanced Fenton reaction catalyzed by natural heterogeneous pyrite for nitrobenzene degradation in an aqueous solution. Chem. Eng. J. 2014, 244, 438–445. [Google Scholar] [CrossRef]
- Wang, C.; Xi, J.Y.; Hu, H.Y. Chemical identification and acute biotoxicity assessment of gaseous chlorobenzene photodegradation products. Chemosphere 2008, 73, 1167–1171. [Google Scholar] [CrossRef] [PubMed]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA/AWWA/WEF: Washington, DC, USA, 1998. [Google Scholar]
- Wang, L.S.; Wei, D.B.; Wei, J.; Hu, H.Y. Screening and estimating of toxicity formation with photobacterium bioassay during chlorine disinfection of wastewater. J. Hazard. Mater. 2007, 141, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Stookey, L.L. Ferrozine—A new spectrophotometric reagent for iron. Anal. Chem. 1970, 42, 779–781. [Google Scholar] [CrossRef]
- Norwitz, G.; Keliher, P.N. Spectrophotometric determination of aniline by the diazotization-coupling method with N-(1-naphthyl) ethylenediamine as the coupling agent. Anal. Chem. 1981, 53, 1238–1240. [Google Scholar] [CrossRef]
- Stedmon, C.A.; Bro, R. Characterizing dissolved organic matter fluorescence with parallel factor analysis: A tutorial. Limnol. Oceanogr. Methods 2008, 6, 572–579. [Google Scholar] [CrossRef]
- Baghoth, S.A.; Sharma, S.K.; Amy, G.L. Tracking natural organic matter (NOM) in a drinking water treatment plant using fluorescence excitation—Emission matrices and PARAFAC. Water Res. 2011, 45, 797–809. [Google Scholar] [CrossRef] [PubMed]
- Li, W.T.; Chen, S.Y.; Xu, Z.X.; Li, Y.; Shuang, C.D.; Li, A.M. Characterization of dissolved organic matter in municipal wastewater using fluorescence PARAFAC analysis and chromatography multi-excitation/emission scan: A comparative study. Environ. Sci. Technol. 2014, 48, 2603–2609. [Google Scholar] [CrossRef] [PubMed]
- Pignatello, J.J.; Oliveros, E.; MacKay, A. Advanced oxidation processes for organic contaminant destruction based on the Fenton reaction and related chemistry. Crit. Rev. Environ. Sci. Technol. 2006, 36, 1–84. [Google Scholar] [CrossRef]
- Hashemian, S.; Tabatabaee, M.; Gafari, M. Fenton oxidation of methyl violet in aqueous solution. J. Chem. 2013, 2013. [Google Scholar] [CrossRef]
- Zhang, Y.L.; Zhang, K.; Dai, C.M.; Zhou, X.F. Performance and mechanism of pyrite for nitrobenzene removal in aqueous solution. Chem. Eng. Sci. 2014, 111, 135–141. [Google Scholar] [CrossRef]
- Segura, Y.; Martínez, F.; Melero, J.A.; Fierro, J.L.G. Zero valent iron (ZVI) mediated Fenton degradation of industrial wastewater: Treatment performance and characterization of final composites. Chem. Eng. J. 2015, 269, 298–305. [Google Scholar] [CrossRef]
- Moffett, J.W.; Zika, R.G. Reaction kinetics of hydrogen peroxide with copper and iron in seawater. Environ. Sci. Technol. 1987, 21, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Stumm, W.; James, J. Aquatic Chemistry. An Introduction Empha—Sizing Chemical Equilibria in Natural Waters; John Wiley & Sons: Michigan, MI, USA, 1981. [Google Scholar]
- Pham, A.L.T.; Lee, C.; Doyle, F.M.; Sedlak, D.L. A silica-supported iron oxide catalyst capable of activating hydrogen peroxide at neutral pH values. Environ. Sci. Technol. 2009, 43, 8930–8935. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.X.; Hanna, K.; Abdelmoula, M.; Deng, N. Adsorption and oxidation of PCP on the surface of magnetite: Kinetic experiments and spectroscopic investigations. Appl. Catal. B-Environ. 2009, 89, 432–440. [Google Scholar] [CrossRef]
- Kong, L.H.; Hu, X.Y.; He, M.C. Mechanisms of Sb(III) oxidation by pyrite-induced hydroxyl radicals and hydrogen peroxide. Environ. Sci. Technol. 2015, 49, 3499–3505. [Google Scholar] [CrossRef] [PubMed]
- Kwan, W.P.; Voelker, B.M. Decomposition of hydrogen peroxide and organic compounds in the presence of dissolved iron and ferrihydrite. Environ. Sci. Technol. 2002, 36, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Westerhoff, P.; Chen, W.; Esparza, M. Fluorescence analysis of a standard fulvic acid and tertiary treated wastewater. J. Environ. Qual. 2001, 30, 2037–2046. [Google Scholar] [CrossRef] [PubMed]
- Goldman, J.H.; Rounds, S.A.; Needoba, J.A. Applications of fluorescence spectroscopy for predicting percent wastewater in an urban stream. Environ. Sci. Technol. 2012, 46, 4374–4381. [Google Scholar] [CrossRef] [PubMed]
- Stedmon, C.A.; Markager, S.; Bro, R. Tracing dissolved organic matter in aquatic environments using a new approach to fluorescence spectroscopy. Mar. Chem. 2003, 82, 239–254. [Google Scholar] [CrossRef]
- Bro, R. The N-Way On-Line Course on PARAFAC and PLS. Available online: http://www.models.life.ku.dk/~pih/parafac/chap0contents.htm (accessed on 21 June 2010).
- Lei, Y.Q.; Wang, G.H.; Guo, P.R.; Li, G.B.; Cai, D.C. The application of three-dimensional fluorescence spectroscopy in the electrochemical degradation of organic pollutions. Chin. J. Anal. Lab. 2014, 33, 373–376. (In Chinese) [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, L.; Li, Y.; Li, A. Treatment of Actual Chemical Wastewater by a Heterogeneous Fenton Process Using Natural Pyrite. Int. J. Environ. Res. Public Health 2015, 12, 13762-13778. https://doi.org/10.3390/ijerph121113762
Sun L, Li Y, Li A. Treatment of Actual Chemical Wastewater by a Heterogeneous Fenton Process Using Natural Pyrite. International Journal of Environmental Research and Public Health. 2015; 12(11):13762-13778. https://doi.org/10.3390/ijerph121113762
Chicago/Turabian StyleSun, Liang, Yan Li, and Aimin Li. 2015. "Treatment of Actual Chemical Wastewater by a Heterogeneous Fenton Process Using Natural Pyrite" International Journal of Environmental Research and Public Health 12, no. 11: 13762-13778. https://doi.org/10.3390/ijerph121113762
APA StyleSun, L., Li, Y., & Li, A. (2015). Treatment of Actual Chemical Wastewater by a Heterogeneous Fenton Process Using Natural Pyrite. International Journal of Environmental Research and Public Health, 12(11), 13762-13778. https://doi.org/10.3390/ijerph121113762





