Molecular Analysis of Bacterial Microbiota on Brazilian Currency Note Surfaces
Abstract
:1. Introduction
2. Experimental Section
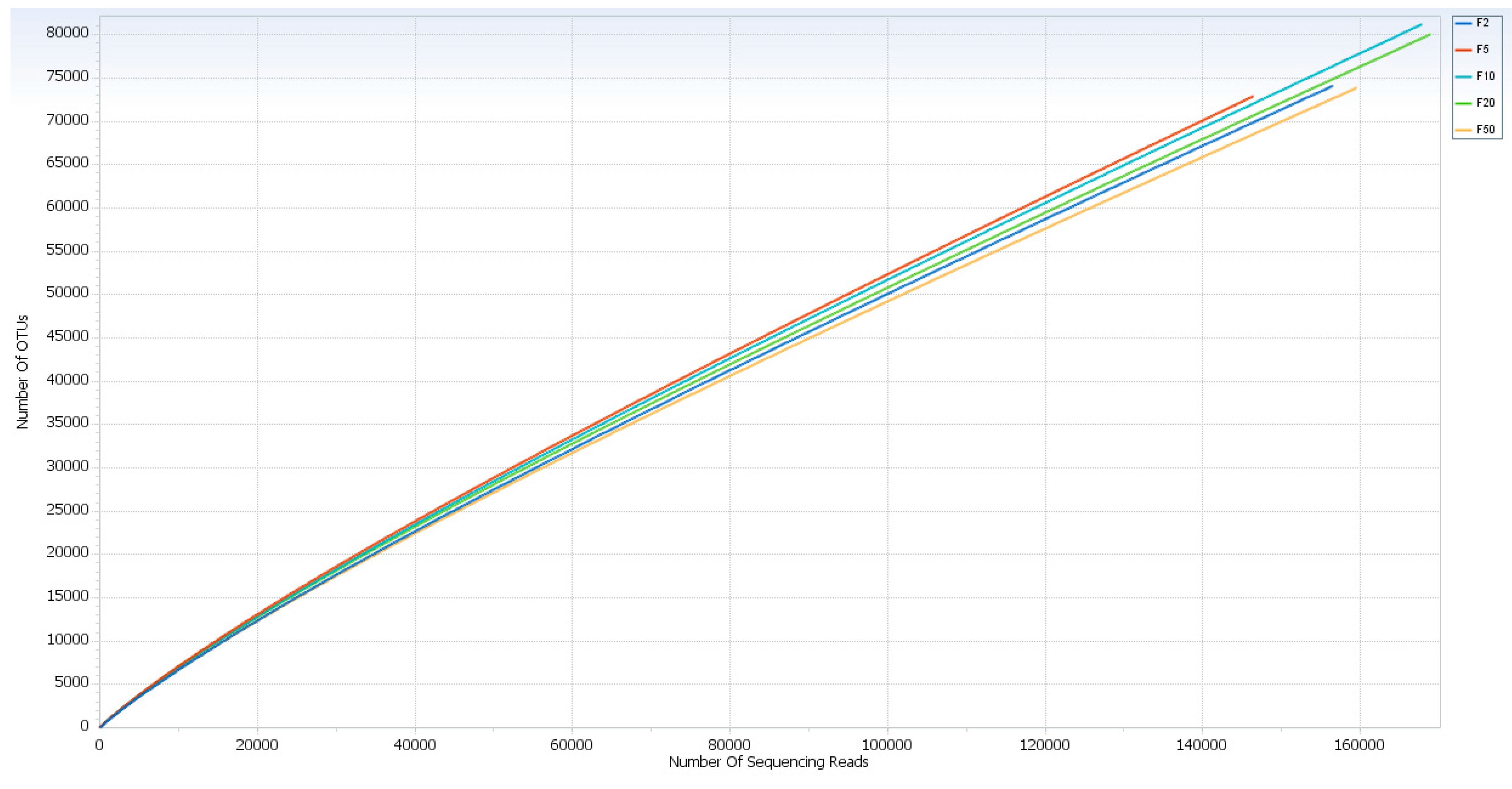
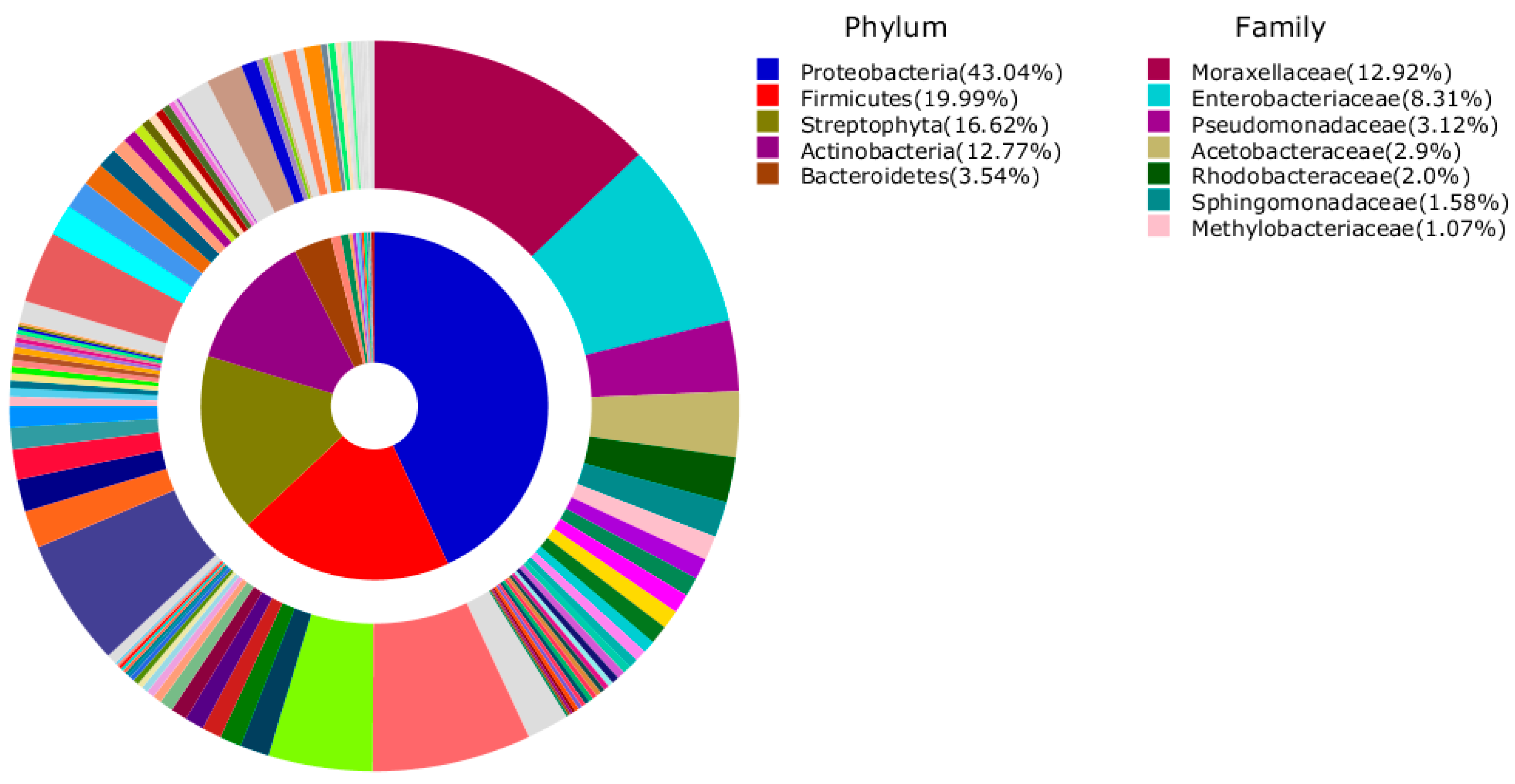
3. Results and Discussion
| Currency Note (R$) | Valid Reads | No. of OTU (>97% identity) | Chao1 Value | Shannon Index |
|---|---|---|---|---|
| F2 | 156,537 | 74,109 | 8,371,643,675 | 9,574,233 |
| F5 | 146,470 | 72,923 | 7,528,842,108 | 9,858,164 |
| F10 | 167,851 | 81,215 | 8,764,508,415 | 9,937,533 |
| F20 | 168,981 | 80,076 | 8,808,417,211 | 9,919,012 |
| F50 | 159,621 | 73,891 | 8,237,323,319 | 967,336 |
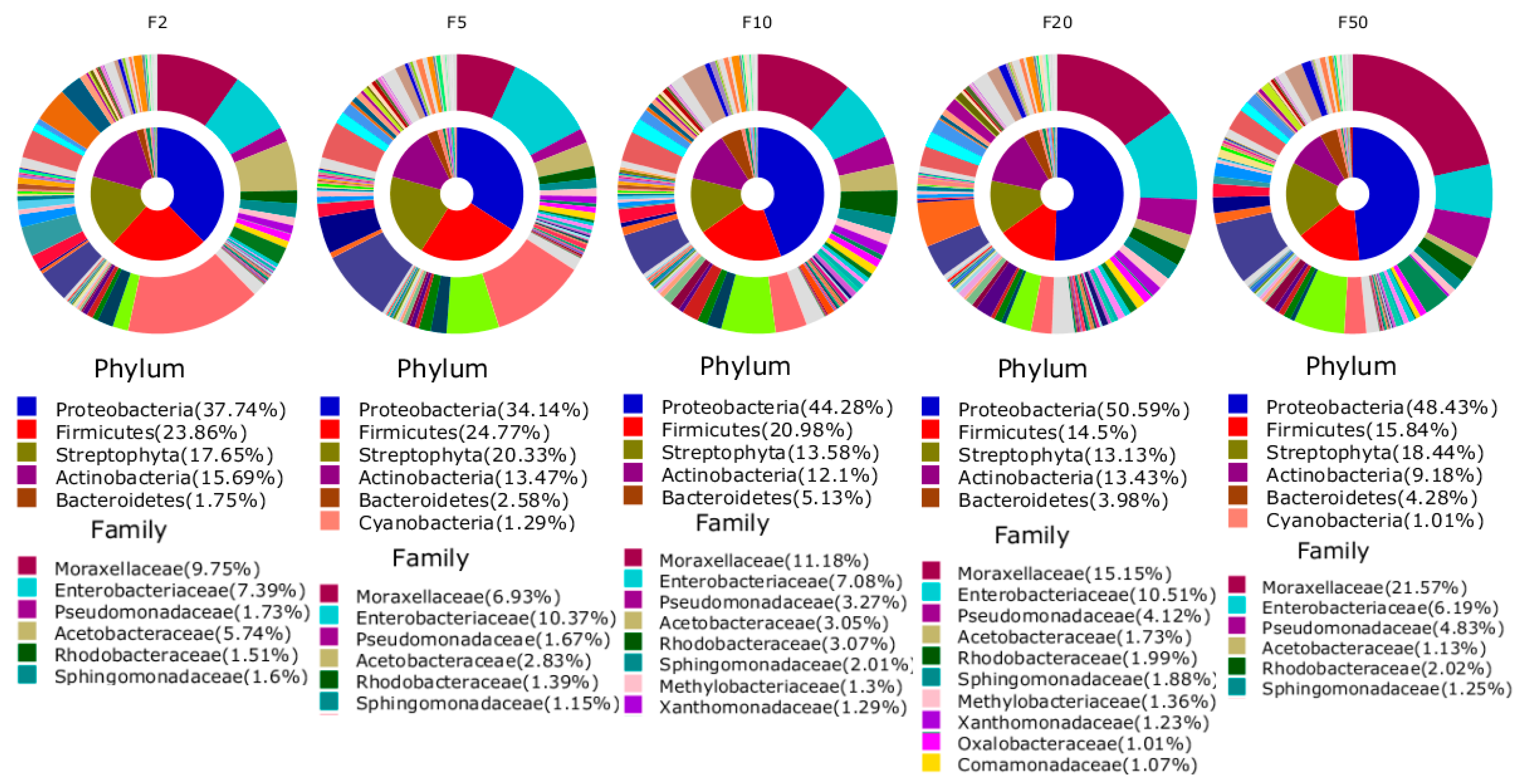
| F2 | F5 | F10 | F20 | F50 | |
|---|---|---|---|---|---|
| Abundance Order | Taxa | ||||
| (Abundance) | |||||
| 1 | Staphylococcus saprophyticus (2.8%) | Staphylococcus saprophyticus (2.7%) | Moraxellaceae_uc_s (1.6%) | Moraxellaceae_uc_s (1.5%) | Moraxellaceae_uc_s (1.7%) |
| 2 | Staphylococcaceae_uc_s (1.4%) | Enterobacteriaceae_uc_s (1.1%) | Acinetobacter_uc (0.8%) | Acinetobacter_uc (1.3%) | Acinetobacter_uc (1.1%) |
| 3 | Staphylococcus_uc (1.1%) | Staphylococcaceae_uc_s (0.9%) | Enterobacteriaceae_uc_s (0.8%) | Enterobacteriaceae_uc_s (1.0%) | Enterobacteriaceae_uc_s (1.1%) |
| 4 | Moraxellaceae_uc_s (1.1%) | Staphylococcus_uc (0.9%) | Staphylococcaceae_uc_s (0.7%) | Staphylococcaceae_uc_s (0.7%) | Staphylococcaceae_uc_s (0.6%) |
| 5 | Enterobacteriaceae_uc_s (0.9%) | Moraxellaceae_uc_s (0.6%) | Staphylococcus_uc (0.5%) | Staphylococcus_uc (0.5%) | Staphylococcus_uc (0.6%) |
| 6 | Acinetobacter_uc (0.9%) | Acinetobacter_uc (0.5%) | Staphylococcus saprophyticus (0.4%) | Staphylococcus saprophyticus (0.5%) | Staphylococcus saprophyticus (0.6%) |

4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Vriesekoop, F.; Russell, C.; Alvarez-Mayorga, B.; Aidoo, K.; Yuan, Q.; Scannell, A.; Beumer, R.R.; Jiang, X.; Barro, N.; Otokunefor, K.; et al. Dirty money: An investigation into the hygiene status of some of the world’s currencies as obtained from food outlets. Foodborne Pathog. Dis. 2010, 7, 1497–1502. [Google Scholar] [CrossRef] [PubMed]
- Dirty Paper Money. Available online: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1707435/?page=1 (accessed on 16 September 2015).
- Debajit, B.; Pratap, P.; Tarun, K. Paper currencies, a potential carrier of pathogenic microorganisms. Int. J. Appl. Biol. Pharm. Technol. 2012, 3, 23–25. [Google Scholar]
- Angelakis, E.; Azhar, E.I.; Bibi, F.; Yasir, M.; Al-Ghamdi, A.K.; Ashshi, A.M.; Elshemi, A.G.; Raoult, D. Paper money and coins as potential vectors of transmissible disease. Future Microbiol. 2014, 9, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Awodi, N.; Nock, I.; Aken’Ova, I. Prevalence and public health significance of parasite cysts and eggs on the Nigerian money. Nig. J. Parasitol. 2000, 9, 91–94. [Google Scholar]
- Prasai, T.; Yami, K.D.; Joshi, D.R. Microbial load on paper/polymer money and coins. Nepal J. Sci. Tech. 2008, 9, 105–109. [Google Scholar]
- Ayandele, A.A.; Adeniyi, S.A. Prevalence and antimicrobial resistance pattern of micro organism isolated from naria notes in Nigeria. J. Res. Biol. 2011, 587–593. [Google Scholar]
- Pradeep, N.V.; Marulasiddaiah, B.S.; Chetana, M. Microbial contamination of Indian currency notes in circulation. J. Res. Biol. 2012, 2, 377–382. [Google Scholar]
- Tagoe, D.N.A.; Baidoo, S.E.; Dadzie, I.; Ahator, D. A study of bacterial contamination of Ghanaian currency notes in circulation. Internet J. Microbiol. 2010, 8, 1–5. [Google Scholar]
- Bhat, N.; Bhat, S.; Asawa, K.; Agarwal, A. An assessment of oral health risk associated with handling of currency notes. Int. J. Dent. Clin. 2010, 2, 14–16. [Google Scholar]
- Ahmed, S.; Parveen, S.; Nasreen, T.; Feroza, B. Evaluation of the microbial contamination of Bangladesh paper currency notes (taka) in circulation. Adv. Biol. Res. 2010, 4, 266–271. [Google Scholar]
- Abid, H.S. Bacterial contamination of Iraqi paper currency notes in circulation & resistance of pathogenic bacteria to antibiotics. Iraqi J. Sci. 2012, 53, 245–248. [Google Scholar]
- Abrams, B.L.; Waterman, N.G. Dirty money. JAMA 1972, 219, 1202–1203. [Google Scholar] [CrossRef] [PubMed]
- Basavarajappa, K.G.; Rao, P.N.; Suresh, K. Study of bacterial, fungal, and parasitic contaminaiton of currency notes in circulation. Indian J. Pathol. Microbiol. 2005, 48, 278–279. [Google Scholar] [PubMed]
- Neel, R. Isolation of pathogenic microorganisms from contaminated paper currency notes in circulation from different market places in Korogwe and Mombo towns in Tanzania. J. Microbiol. Biotech. Res. 2012, 2, 470–474. [Google Scholar]
- Saeed, S.; Rasheed, H. Evaluation of bacterial contamination of Pakistani paper currency notes (rupee) in circulation in Karachi. J. Microbiol. Biotech. Res. 2011, 3, 94–98. [Google Scholar]
- Herlemann, D.P.; Labrenz, M.; Jurgens, K.; Bertilsson, S.; Waniek, J.J.; Andersson, A.F. Transitions in bacterial communities along the 2000 km salinity gradient of the baltic sea. ISME J. 2011, 5, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Masella, A.P.; Bartram, A.K.; Truszkowski, J.M.; Brown, D.G.; Neufeld, J.D. PANDAseq: Paired-end assembler for illumina sequences. BMC Bioinformatics 2012, 13, 31. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. Uchime improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Kim, O.S.; Cho, Y.J.; Lee, K.; Yoon, S.H.; Kim, M.; Na, H.; Park, S.C.; Jeon, Y.S.; Lee, J.H.; Yi, H.; et al. Introducing EzTaxon-e: A prokaryotic 16s rRNA gene sequence database with phylotypes that represent uncultured species. Int. J. Syst. Evol. Microbiol. 2012, 62, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Hamady, M.; Lozupone, C.; Knight, R. Fast unifrac: Facilitating high-throughput phylogenetic analyses of microbial communities including analysis of pyrosequencing and PhyloChip data. ISME J. 2010, 4, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Molecular Analysis of Bacterial Microbiota on Brazilian Currency Note Surfaces. Available online: http://zenodo.org/record/30279#.Vib1tYUXOLM (accessed on 2 September 2015).
- Girma, G.; Ketema, T.; Bacha, K. Microbial load and safety of paper currencies from some food vendors in Jimma town, southwest Ethiopia. BMC Res. Notes 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Pope, T.W.; Ender, P.T.; Woelk, W.K.; Koroscil, M.A.; Koroscil, T.M. Bacterial contamination of paper currency. South Med. J. 2002, 95, 1408–1410. [Google Scholar] [CrossRef] [PubMed]
- El-Dars, F.M.; Hassan, W.M. A preliminary bacterial study of Egyptian paper money. Int. J. Environ. Health Res. 2005, 15, 235–239. [Google Scholar] [CrossRef] [PubMed]
- De Bentzmann, S.; Plesiat, P. The Pseudomonas aeruginosa opportunistic pathogen and human infections. Environ. Microbiol. 2011, 13, 1655–1665. [Google Scholar] [CrossRef] [PubMed]
- Murphy, T.F.; Parameswaran, G.I. Moraxella catarrhalis, a human respiratory tract pathogen. Clin. Infect. Dis. 2009, 49, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Berrocal, A.M.; Scott, I.U.; Miller, D.; Flynn, H.W., Jr. Endophthalmitis caused by Moraxella osloensis. Graef. Arch. Clin. Exp. 2002, 240, 329–330. [Google Scholar] [CrossRef] [PubMed]
- Sifri, C.D.; Brassinga, A.K.; Flohr, T.; Kinchen, J.M.; Hazen, K.C.; Sawyer, R.G.; Pruett, T.L.; Bonatti, H. Moraxella osloensis bacteremia in a kidney transplant recipient. Transpl. Int. 2008, 21, 1011–1013. [Google Scholar] [CrossRef] [PubMed]
- Kuria, J.K.; Wahome, R.G.; Jobalamin, M.; Kariuki, S.M. Profile of bacteria and fungi on money coins. East Afr. Med. J. 2009, 86, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Fournier, P.E.; Richet, H. The epidemiology and control of Acinetobacter baumannii in health care facilities. Clin. Infect. Dis. 2006, 42, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Peleg, A.Y.; de Breij, A.; Adams, M.D.; Cerqueira, G.M.; Mocali, S.; Galardini, M.; Nibbering, P.H.; Earl, A.M.; Ward, D.V.; Paterson, D.L.; et al. The success of Acinetobacter species; genetic, metabolic and virulence attributes. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Touchon, M.; Cury, J.; Yoon, E.J.; Krizova, L.; Cerqueira, G.C.; Murphy, C.; Feldgarden, M.; Wortman, J.; Clermont, D.; Lambert, T.; et al. The genomic diversification of the whole Acinetobacter genus: Origins, mechanisms, and consequences. Genome Biol. Evol. 2014, 6, 2866–2882. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Price, L.S.; Weinstein, R.A. Acinetobacter infection. N. Engl. J. Med. 2008, 358, 1271–1281. [Google Scholar] [CrossRef] [PubMed]
- Turton, J.F.; Shah, J.; Ozongwu, C.; Pike, R. Incidence of Acinetobacter species other than A. baumannii among clinical isolates of Acinetobacter: Evidence for emerging species. J. Clin. Microbiol. 2010, 48, 1445–1449. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Karveli, E.A.; Kelesidis, I.; Kelesidis, T. Community-acquired Acinetobacter infections. Eur. J. Clin. Microbiol. 2007, 26, 857–868. [Google Scholar] [CrossRef] [PubMed]
- Eveillard, M.; Kempf, M.; Belmonte, O.; Pailhories, H.; Joly-Guillou, M.L. Reservoirs of Acinetobacter baumannii outside the hospital and potential involvement in emerging human community-acquired infections. Int. J. Infect. Dis. 2013, 17, e802–e805. [Google Scholar] [CrossRef] [PubMed]
- Gayoso, C.M.; Mateos, J.; Mendez, J.A.; Fernandez-Puente, P.; Rumbo, C.; Tomas, M.; Martinez de Ilarduya, O.; Bou, G. Molecular mechanisms involved in the response to desiccation stress and persistence in Acinetobacter baumannii. J. Proteome Res. 2014, 13, 460–476. [Google Scholar] [CrossRef] [PubMed]
- Dijkshoorn, L.; Nemec, A.; Seifert, H. An increasing threat in hospitals: Multidrug-resistant Acinetobacter baumannii. Nat. Rev. Microbiol. 2007, 5, 939–951. [Google Scholar] [CrossRef] [PubMed]
- Yoon, E.J.; Chabane, Y.N.; Goussard, S.; Snesrud, E.; Courvalin, P.; De, E.; Grillot-Courvalin, C. Contribution of resistance-nodulation-cell division efflux systems to antibiotic resistance and biofilm formation in Acinetobacter baumannii. MBio 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Podschun, R.; Ullmann, U. Klebsiella spp. as nosocomial pathogens: Epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin. Microbiol. Rev. 1998, 11, 589–603. [Google Scholar] [PubMed]
- Laupland, K.B.; Ross, T.; Pitout, J.D.; Church, D.L.; Gregson, D.B. Community-onset urinary tract infections: A population-based assessment. Infection 2007, 35, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Shon, A.S.; Bajwa, R.P.; Russo, T.A. Hypervirulent (hypermucoviscous) klebsiella pneumoniae: A new and dangerous breed. Virulence 2013, 4, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Lowy, F.D. Staphylococcus aureus infections. N. Engl. J. Med. 1998, 339, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Becker, R.E.; Bubeck Wardenburg, J. Staphylococcus aureus and the skin: A longstanding and complex interaction. Skinmed 2015, 13, 111–119. [Google Scholar] [PubMed]
- Moosavy, M.H.; Shavisi, N.; Warriner, K.; Mostafavi, E. Bacterial contamination of Iranian paper currency. Iran J. Public Health 2013, 42, 1067–1070. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira da Fonseca, T.A.; Pessôa, R.; Sanabani, S.S. Molecular Analysis of Bacterial Microbiota on Brazilian Currency Note Surfaces. Int. J. Environ. Res. Public Health 2015, 12, 13276-13288. https://doi.org/10.3390/ijerph121013276
Pereira da Fonseca TA, Pessôa R, Sanabani SS. Molecular Analysis of Bacterial Microbiota on Brazilian Currency Note Surfaces. International Journal of Environmental Research and Public Health. 2015; 12(10):13276-13288. https://doi.org/10.3390/ijerph121013276
Chicago/Turabian StylePereira da Fonseca, Tairacan Augusto, Rodrigo Pessôa, and Sabri Saeed Sanabani. 2015. "Molecular Analysis of Bacterial Microbiota on Brazilian Currency Note Surfaces" International Journal of Environmental Research and Public Health 12, no. 10: 13276-13288. https://doi.org/10.3390/ijerph121013276
APA StylePereira da Fonseca, T. A., Pessôa, R., & Sanabani, S. S. (2015). Molecular Analysis of Bacterial Microbiota on Brazilian Currency Note Surfaces. International Journal of Environmental Research and Public Health, 12(10), 13276-13288. https://doi.org/10.3390/ijerph121013276





