The Effects of Leptin on Breastfeeding Behaviour
Abstract
:1. Introduction
2. Experimental Section
2.1. Ethics Statement
2.2. Participants
| Mother and Infant Characteristics | Mean ± SD | Range |
|---|---|---|
| Mother | ||
| age (years) | 32 ± 3 | 27–37 |
| BMI * | 25 ± 4 | 18–33 |
| parity | NA | 1–3 |
| Infant | ||
| age (weeks) | 10 ± 5 | 3–21 |
| birth weight (g) | 3515 ± 416 | 2930–4325 |
| current weight (g) | 5912 ± 1296 | 4062–8990 |
2.3. Sample Collection
2.4. Biochemical Analyses
2.5. Leptin Dose and Intake
2.6. Statistical Analysis
3. Results and Discussion
3.1. Results
3.1.1. Participants
3.1.2. Variability of Skim Milk Leptin
| Breastmilk Constituents | Pre-Feed Value | Post-Feed Value | N Samples | p-Value |
|---|---|---|---|---|
| Protein (g/L) | 11.18 ± 1.67 | 11.33 ± 1.94 | 483 | 0.14 |
| Lactose (g/L) | 67.04 ± 6.03 | 66.04 ± 5.89 | 483 | 0.001 |
| Fat (g/L) | 31.37 ± 11.95 | 56.95 ± 20.19 | 479 | <0.0001 |
| Leptin (ng/mL) | 0.43 ± 0.10 | 0.42 ± 0.11 | 479 | 0.184 |
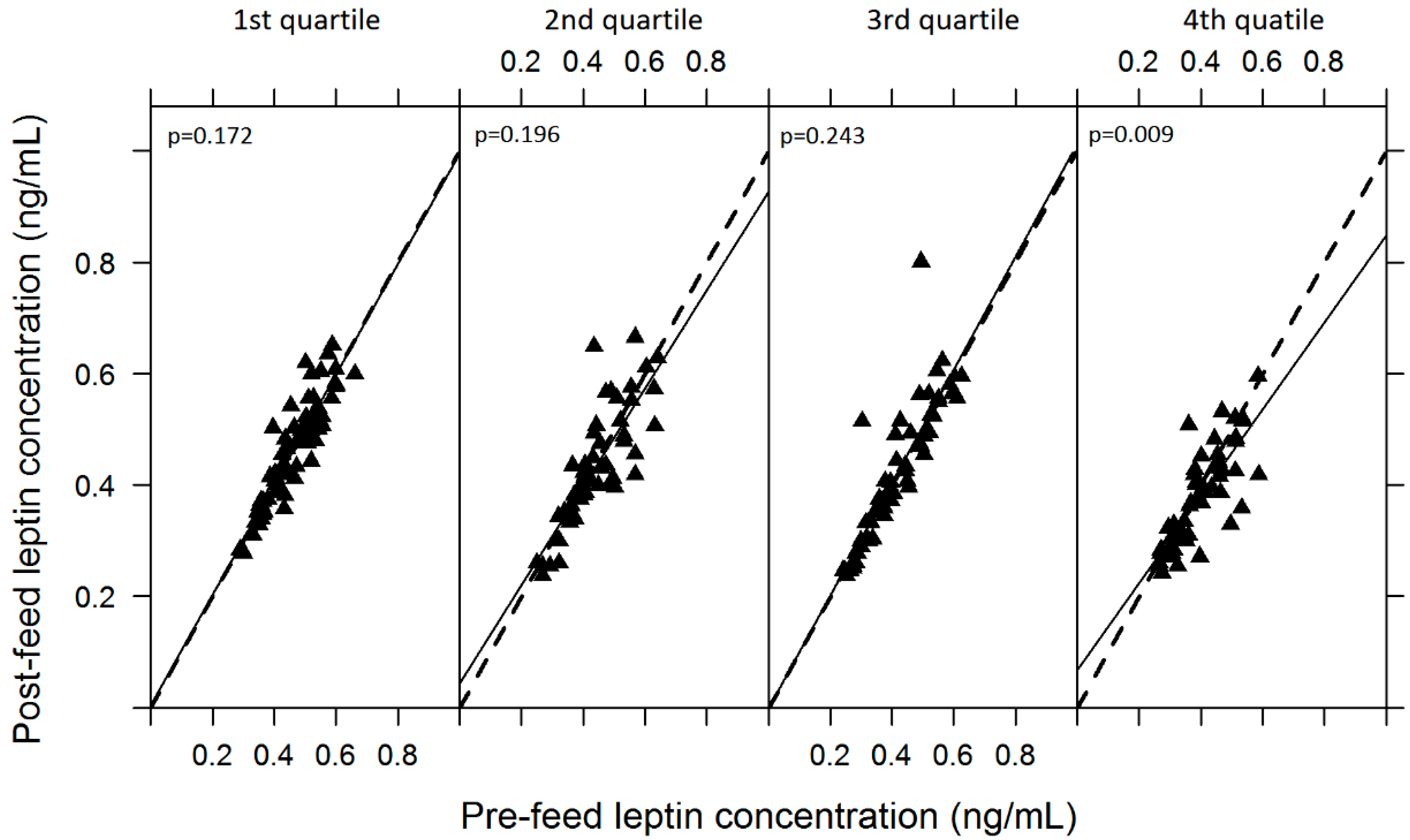
3.1.3. Relationship of Skim Milk Leptin with Macronutrients
3.1.4. Skim Milk Leptin and Breastfeeding Patterns
| Breastmilk and Feeding Characteristics | Mean ± SD | Range |
|---|---|---|
| Total production of both breasts (mL) | 822 ± 166 | 496–1232 |
| Left breast production (mL) | 373 ± 140 | 89–666 |
| Right breast production (mL) | 434 ± 119 | 259–676 |
| Number of feeds * | 9 ± 2 | 7–13 |
| Volume of feeds (mL) * | 108 ± 45 | 18–220 |
| Feed duration (min) * | 24 ± 13 | 4–105 |
| Feed interval (min) ** | 172 ± 102 | 23–590 |
| Skim milk leptin dose per feed (ng) * | 45.94 ± 21.25 | 9.62–104.84 |
| Skim milk leptin intake (ng) *** | 403.36 ± 80.08 | 239.64–501.40 |
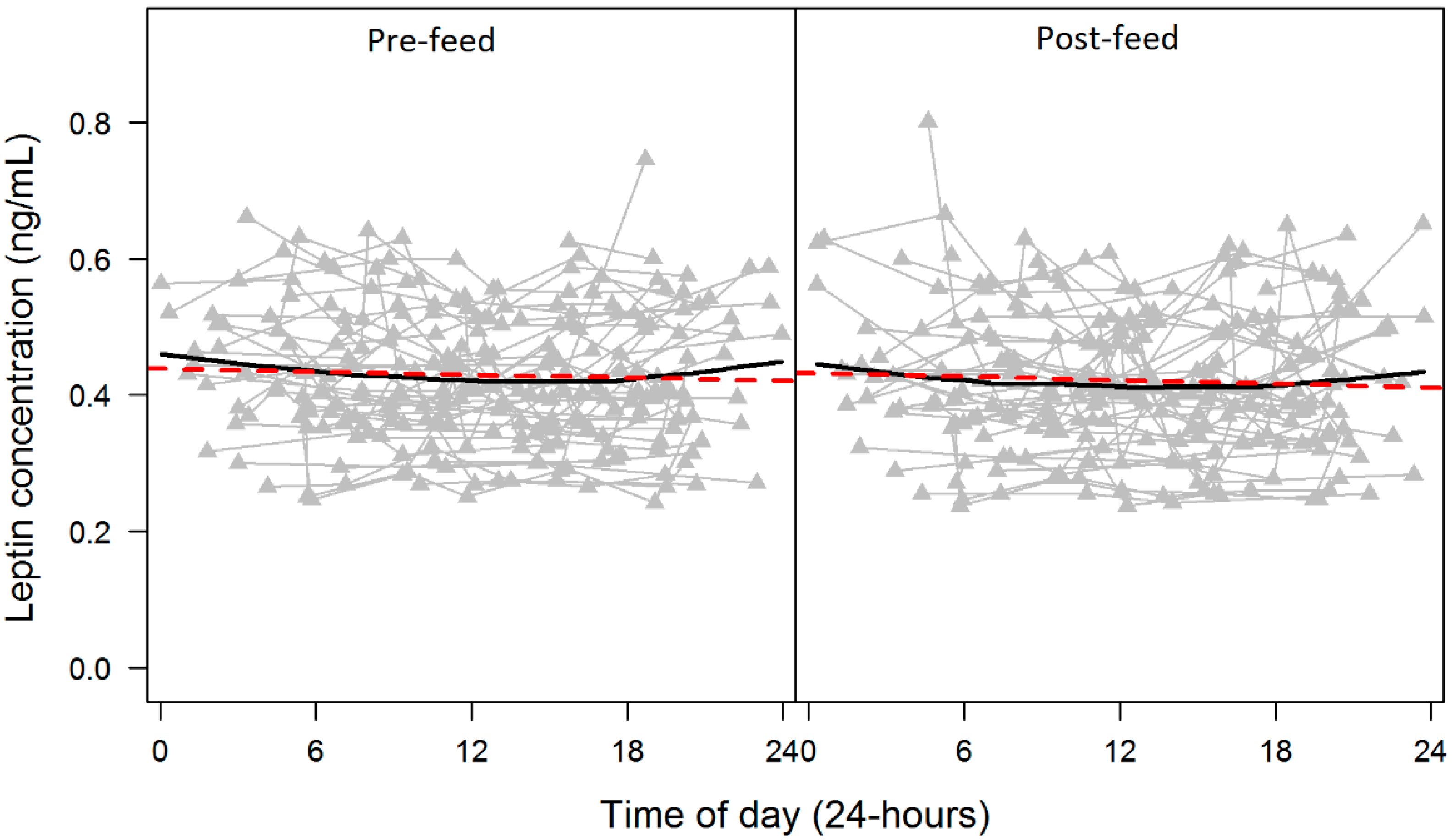
3.1.5. Twenty-Four Hour Pattern of Skim Milk Leptin and Macronutrients
3.1.6. Skim Milk Leptin and Demographics
3.2. Discussion
| Demographic Characteristics | Leptin Concentration (ng/mL) p-Value | Conditional R2 for Univariate 1 and Multivariate 2 Models | Total Leptin Intake (ng/24-h) p-Value | Conditional R2 for Univariate 1 and Multivariate 2 Models |
|---|---|---|---|---|
| Maternal | ||||
| BMI | 0.008 1 | 0.77 1 | 0.394 1 | 0.05 1 |
| breast left or right | 0.619 1 | 0.77 1 | N/A | N/A |
| pre-/post-feed sample | 0.317 2 | 0.77 2 | N/A | N/A |
| total milk intake | 0.595 2 | 0.78 2 | N/A | N/A |
| Infant age (weeks) | 0.638 1 | 0.78 1 | 0.21 1 | 0.09 1 |
| Infant current weight (g) | 0.769 1 | 0.78 1 | 0.067 1 | 0.19 1 |
| Infant gender | 0.220 1 | 0.78 1 | 0.616 1 | 0.02 1 |
| Total milk intake (mL/24 h) | 0.394 1 | 0.78 1 | 0.002 1 | 0.45 1 |
3.3. Limitations
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Miralles, O.; Sanchez, J.; Palou, A.; Pico, C. A physiological role of breast milk leptin in body weight control in developing infants. Obesity 2006, 14, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Palou, A.; Pico, C. Leptin intake during lactation prevents obesity and affects food intake and food preferences in later life. Appetite 2009, 52, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Geddes, D.T.; Prescott, S.L. Developmental origins of health and disease: The role of human milk in preventing disease in the 21st century. J. Hum. Lact. 2013, 29, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Von Kries, R.; Koletzko, B.; Sauerwald, T.; von Mutius, E.; Barnert, D.; Grunert, V.; von Voss, H. Breast feeding and obesity: Cross sectional study. BMJ 1999, 319, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Gillman, M.W.; Rifas-Shiman, S.L.; Camargo, C.A., Jr.; Berkey, C.S.; Frazier, A.L.; Rockett, H.R.; Field, A.E.; Colditz, G.A. Risk of overweight among adolescents who were breastfed as infants. JAMA 2001, 285, 2461–2467. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, K.E.; Bergmann, R.L.; von Kries, R.; Bohm, O.; Richter, R.; Dudenhausen, J.W.; Wahn, U. Early determinants of childhood overweight and adiposity in a birth cohort study: Role of breast-feeding. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 162–172. [Google Scholar] [CrossRef] [PubMed]
- McCrory, C.; Layte, R. Breastfeeding and risk of overweight and obesity at nine-years of age. Soc. Sci. Med. 2012, 75, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Owen, C.G.; Martin, R.M.; Whincup, P.H.; Smith, G.D.; Cook, D.G. Effect of infant feeding on the risk of obesity across the life course: A quantitative review of published evidence. Pediatrics 2005, 115, 1367–1377. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, J.; Reilly, J.J.; Child Health Information Team. Breastfeeding and lowering the risk of childhood obesity. Lancet 2002, 359, 2003–2004. [Google Scholar] [CrossRef]
- Savino, F.; Liguori, S.A.; Fissore, M.F.; Oggero, R. Breast milk hormones and their protective effect on obesity. Int. J. Pediatr. Endocinol. 2009, 2009. [Google Scholar] [CrossRef]
- Stocker, C.J.; Cawthorne, M.A. The influence of leptin on early life programming of obesity. Trends Biotechnol. 2008, 26, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Hassiotou, F.; Geddes, D.T. Programming of appetite control during breastfeeding as a preventative strategy against the obesity epidemic. J. Hum. Lact. 2014, 30, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Magadia, J.; Fein, S.B.; Grummer-Strawn, L.M. Risk of bottle-feeding for rapid weight gain during the first year of life. Arch. Pediatr. Adolesc. Med. 2012, 166, 431–436. [Google Scholar] [PubMed]
- Bartok, C.J. Babies fed breastmilk by breast versus by bottle: A pilot study evaluationg early growth patterns. Breastfeed. Med. 2011, 6, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Costamagna, M.; Prino, A.; Oggero, R.; Silvestro, L. Leptin levels in breast-fed and formula-fed infants. Acta Paediatr. 2002, 91, 897–902. [Google Scholar] [CrossRef] [PubMed]
- Dewey, K.G.; Heinig, M.J.; Nommsen, L.A.; Lonnerdal, B. Maternal versus infant factors related to breast milk intake and residual milk volume: The darling study. Pediatrics 1991, 87, 829–837. [Google Scholar] [PubMed]
- Kent, J.C.; Mitoulas, L.R.; Cregan, M.D.; Ramsay, D.T.; Doherty, D.A.; Hartmann, P.E. Volume and frequency of breastfeedings and fat content of breastmilk throughout the day. Pediatrics 2006, 117, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Karatas, Z.; Durmus Aydogdu, S.; Dinleyici, E.C.; Colak, O.; Dogruel, N. Breastmilk ghrelin, leptin, and fat levels changing foremilk to hindmilk: Is that important for self-control of feeding? Eur. J. Pediatr. 2011, 170, 1273–1280. [Google Scholar] [CrossRef] [PubMed]
- Bartok, C.J.; Ventura, A.K. Mechanisms underlying the association between breastfeeding and obesity. Int. J. Pediatr. Obes. 2009, 4, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Schuster, S.; Hechler, C.; Gebauer, C.; Kiess, W.; Kratzsch, J. Leptin in maternal serum and breast milk: Association with infants’ body weight gain in a longitudinal study over 6 months of lactation. Pediatr. Res. 2011, 70, 633–637. [Google Scholar] [CrossRef] [PubMed]
- Hale, T.W.; Hartmann, P.E. Hale and Hartmann’s Textbook of Human Lactation, 1st ed.; Hale Publishing, L.P.: Amarillo, TX, USA, 2007. [Google Scholar]
- Kent, J.C.; Prime, D.K.; Garbin, C.P. Principles for maintaining or increasing breast milk production. J. Obstet. Gynecol. Neonatal. Nurs. 2011, 41, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Arthur, P.G.; Hartmann, P.E.; Smith, M. Measurement of the milk intake of breast-fed infants. J. Pediatr. Gastroenterol. Nutr. 1987, 6, 758–763. [Google Scholar] [CrossRef] [PubMed]
- Fleet, I.R.; Linzell, J.L. A rapid method of estimating fat in very small quantities of milk. J. Physiol. 1964, 175, 15–17. [Google Scholar]
- Meier, P.P.; Engstrom, J.L.; Zuleger, J.L.; Motykowski, J.E.; Vasan, U.; Meier, W.A.; Hartmann, P.E.; Williams, T.M. Accuracy of a user-friendly centrifuge for measuring creamatocrits on mothers’ milk in the clinical setting. Breastfeed. Med. 2006, 1, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Mitoulas, L.R.; Kent, J.C.; Cox, D.B.; Owens, R.A.; Sherriff, J.L.; Hartmann, P.E. Variation in fat, lactose and protein in human milk over 24 h and throughout the first year of lactation. Br. J. Nutr. 2002, 88, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, N.J.; Lowenstein, J.M. Lactogenesis in the rat. Changes in metabolic parameters at parturition. Biochem. J. 1967, 105, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2009. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. Nlme: Linear and Nonlinear Mix Effects Models, R package version 3.1–113; R Foundation for Statistical Computing: Vienna, Austria, 2009. [Google Scholar]
- Sarkar, D. Lattice: Multivariate Data Visualization with R; Springer: New York, NY, USA, 2008. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach; Springer-Verlag: New York, NY, USA, 2002. [Google Scholar]
- Sinha, M.K.; Ohannesian, J.P.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Magosin, S.; Marco, C.; Caro, J.F. Nocturnal rise of leptin in lean, obese, and non-insulin-dependent diabetes mellitus subjects. J. Clin. Investig. 1996, 97, 1344–1347. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Hepworth, A.R.; Prime, D.K.; Lai, C.T.; Trengove, N.J.; Hartmann, P.E. Variation in fat, lactose, and protein composition in breast milk over 24 h: Associations with infant feeding patterns. J. Hum. Lact. 2013, 29, 81–89. [Google Scholar] [CrossRef] [PubMed]
- WHO. The Who Child Growth Standards. Available online: http://www.who.int/childgrowth/standards/en/ (accessed on 31 July 2015).
- Australian Institute of Health and Walfare. 2010 Australian National Infant Feeding Survey: Indicator Results; Australian Institute of Health and Walfare: Canberra, Australia, 2011.
- Forde, K.A.; Miller, L.J. 2007–2009 North metropolitan perth breastfeeding cohort study: How long are mothers breastfeeding? Breastfeed. Rev. 2010, 10, 14–24. [Google Scholar]
- Centers for Disease Control and Prevention. Breastfeeding Report Card; Centers for Disease Control and Prevention: DeKalb County, GA, USA, 2014.
- Schueler, J.; Alexander, B.; Hart, A.M.; Austin, K.; Larson-Meyer, D.E. Presence and dynamics of leptin, glp-1, and pyy in human breast milk at early postpartum. Obesity 2013, 21, 1451–1458. [Google Scholar] [CrossRef] [PubMed]
- Eilers, E.; Ziska, T.; Harder, T.; Plagemann, A.; Obladen, M.; Loui, A. Leptin determination in colostrum and early human milk from mothers of preterm and term infants. Early Hum. Dev. 2011, 87, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Uysal, F.K.; Onal, E.E.; Aral, Y.Z.; Adam, B.; Dilmen, U.; Ardicolu, Y. Breast milk leptin: Its relationship to maternal and infant adiposity. Clin. Nutr. 2002, 21, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Bronsky, J.; Mitrova, K.; Karpisek, M.; Mazoch, J.; Durilova, M.; Fisarkova, B.; Stechova, K.; Prusa, R.; Nevoral, J. Adiponectin, AFABP, and leptin in human breast milk during 12 months of lactation. J. Pediatr. Gastroenterol. Nutr. 2011, 52, 474–477. [Google Scholar] [CrossRef] [PubMed]
- Weyermann, M.; Beermann, C.; Brenner, H.; Rothenbacher, D. Adiponectin and leptin in maternal serum, cord blood, and breast milk. Clin. Chem. 2006, 52, 2095–2102. [Google Scholar] [CrossRef] [PubMed]
- Houseknecht, K.L.; McGuire, M.K.; Potrocarrero, C.P.; McGuire, M.A.; Beerman, K. Leptin is present in human milk and is related to maternal plasma leptin concentration and adiposity. Biochem. Biophys. Res. Commun. 1997, 240, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Smith-Kirwin, S.M.; O’Connor, D.M.; De Johnston, J.; Lancey, E.D.; Hassink, S.G.; Funanage, V.L. Leptin expression in human mammary epithelial cells and breast milk. J. Clin. Endocrinol. Metab. 1998, 83, 1810–1813. [Google Scholar] [CrossRef] [PubMed]
- Ucar, B.; Kirel, B.; Bor, O.; Kilic, F.S.; Dogruel, N.; Aydogdu, S.D.; Tekin, N. Breast milk leptin concentrations in initial and terminal milk samples: Relationships to maternal and infant plasma leptin concentrations, adiposity, serum glucose, insulin, lipid and lipoprotein levels. J. Pediatr. Endocrinol. Metab. 2000, 13, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Lonnerdal, B.; Havel, P.J. Serum leptin concentrations in infants: Effects of diet, sex, and adiposity. Am. J. Clin. Nutr. 2000, 72, 484–489. [Google Scholar] [PubMed]
- Bielicki, J.; Huch, R.; von Mandach, U. Time-course of leptin levels in term and preterm human milk. Eur. J. Endocrinol. 2004, 151, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Sakalidis, V.S.; Kent, J.C.; Garbin, C.P.; Hepworth, A.R.; Hartmann, P.E.; Geddes, D.T. Longitudinal changes in suck-swallow-breathe, oxygen saturation, and heart rate patterns in term breastfeeding infants. J. Hum. Lact. 2013, 29, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Daly, S.E.; di Rosso, A.; Owens, R.A.; Hartmann, P.E. Degree of breast emptying explains changes in the fat content, but not fatty acid composition, of human milk. Exp. Physiol. 1993, 78, 741–755. [Google Scholar] [CrossRef] [PubMed]
- Hassiotou, F.; Hepworth, A.R.; Williams, T.M.; Twigger, A.J.; Perrella, S.; Lai, C.T.; Filgueira, L.; Geddes, D.T.; Hartmann, P.E. Breastmilk cell and fat contents respond similarly to removal of breastmilk by the infant. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Ilcol, Y.O.; Hizli, Z.B.; Ozkan, T. Leptin concentration in breast milk and its relationship to duration of lactation and hormonal status. Int. Breastfeed. J. 2006, 1. [Google Scholar] [CrossRef] [PubMed]
- Langendonk, J.G.; Pijl, H.; Toornvliet, A.C.; Burggraaf, J.; Frolich, M.; Schoemaker, R.C.; Doornbos, J.; Cohen, A.F.; Meinders, A.E. Circadian rhythm of plasma leptin levels in upper and lower body obese women: Influence of body fat distribution and weight loss. J. Clin. Endocrinol. Metab. 1998, 83, 1706–1712. [Google Scholar] [CrossRef] [PubMed]
- Matkovic, V.; Ilich, J.Z.; Badenhop, N.E.; Skugor, M.; Clairmont, A.; Klisovic, D.; Landoll, J.D. Gain in body fat is inversely related to the nocturnal rise in serum leptin level in young females. J. Clin. Endocrinol. Metab. 1997, 82, 1368–1372. [Google Scholar] [CrossRef] [PubMed]
- Laughlin, G.A.; Yen, S.S. Hypoleptinemia in women athletes: Absence of a diurnal rhythm with amenorrhea. J. Clin. Endocrinol. Metab. 1997, 82, 318–321. [Google Scholar] [CrossRef] [PubMed]
- Ando, H.; Yanagihara, H.; Hayashi, Y.; Obi, Y.; Tsuruoka, S.; Takamura, T.; Kaneko, S.; Fujimura, A. Rhythmic messenger ribonucleic acid expression of clock genes and adipocytokines in mouse visceral adipose tissue. Endocrinology 2005, 146, 5631–5636. [Google Scholar] [CrossRef] [PubMed]
- Bodosi, B.; Gardi, J.; Hajdu, I.; Szentirmai, E.; Obal, F., Jr.; Krueger, J.M. Rhythms of ghrelin, leptin, and sleep in rats: Effects of the normal diurnal cycle, restricted feeding, and sleep deprivation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 287, R1071–R1079. [Google Scholar] [CrossRef] [PubMed]
- Pico, C.; Oliver, P.; Sanchez, J.; Palou, A. Gastric leptin: A putative role in the short-term regulation of food intake. Br. J. Nutr. 2003, 90, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Ramsey, K.M.; Marcheva, B.; Bass, J. Circadian rhythms, sleep, and metabolism. J. Clin. Investig. 2011, 121, 2133–2141. [Google Scholar] [CrossRef] [PubMed]
- Bouret, S.G.; Simerly, R.B. Development of leptin-sensitive circuits. J. Neuroendocrinol. 2007, 19, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Prime, D.K.; Hepworth, A.R.; Lai, C.T.; Trengove, N.J.; Hartmann, P.E. Investigation of short-term variations in term breast milk composition during repeated breast expression sessions. J. Hum. Lact. 2013, 29, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Bouret, S.G.; Simerly, R.B. Minireview: Leptin and development of hypothalamic feeding circuits. Endocrinology 2004, 145, 2621–2626. [Google Scholar] [CrossRef] [PubMed]
- Vickers, M.H.; Sloboda, D.M. Leptin as mediator of the effects of developmental programming. Best Pract. Res. Clin. Endocrinol. Metab. 2012, 26, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Benetti, S.; Liguori, S.A.; Sorrenti, M.; Cordero, D.I.M.L. Advances on human milk hormones and protection against obesity. Cell Mol. Biol. 2013, 59, 89–98. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cannon, A.M.; Kakulas, F.; Hepworth, A.R.; Lai, C.T.; Hartmann, P.E.; Geddes, D.T. The Effects of Leptin on Breastfeeding Behaviour. Int. J. Environ. Res. Public Health 2015, 12, 12340-12355. https://doi.org/10.3390/ijerph121012340
Cannon AM, Kakulas F, Hepworth AR, Lai CT, Hartmann PE, Geddes DT. The Effects of Leptin on Breastfeeding Behaviour. International Journal of Environmental Research and Public Health. 2015; 12(10):12340-12355. https://doi.org/10.3390/ijerph121012340
Chicago/Turabian StyleCannon, Anna M., Foteini Kakulas, Anna R. Hepworth, Ching Tat Lai, Peter E. Hartmann, and Donna T. Geddes. 2015. "The Effects of Leptin on Breastfeeding Behaviour" International Journal of Environmental Research and Public Health 12, no. 10: 12340-12355. https://doi.org/10.3390/ijerph121012340
APA StyleCannon, A. M., Kakulas, F., Hepworth, A. R., Lai, C. T., Hartmann, P. E., & Geddes, D. T. (2015). The Effects of Leptin on Breastfeeding Behaviour. International Journal of Environmental Research and Public Health, 12(10), 12340-12355. https://doi.org/10.3390/ijerph121012340






