Nitrogen Removal over Nitrite by Aeration Control in Aerobic Granular Sludge Sequencing Batch Reactors
Abstract
:1. Introduction
2. Materials and Methods
2.1. Technical Setup
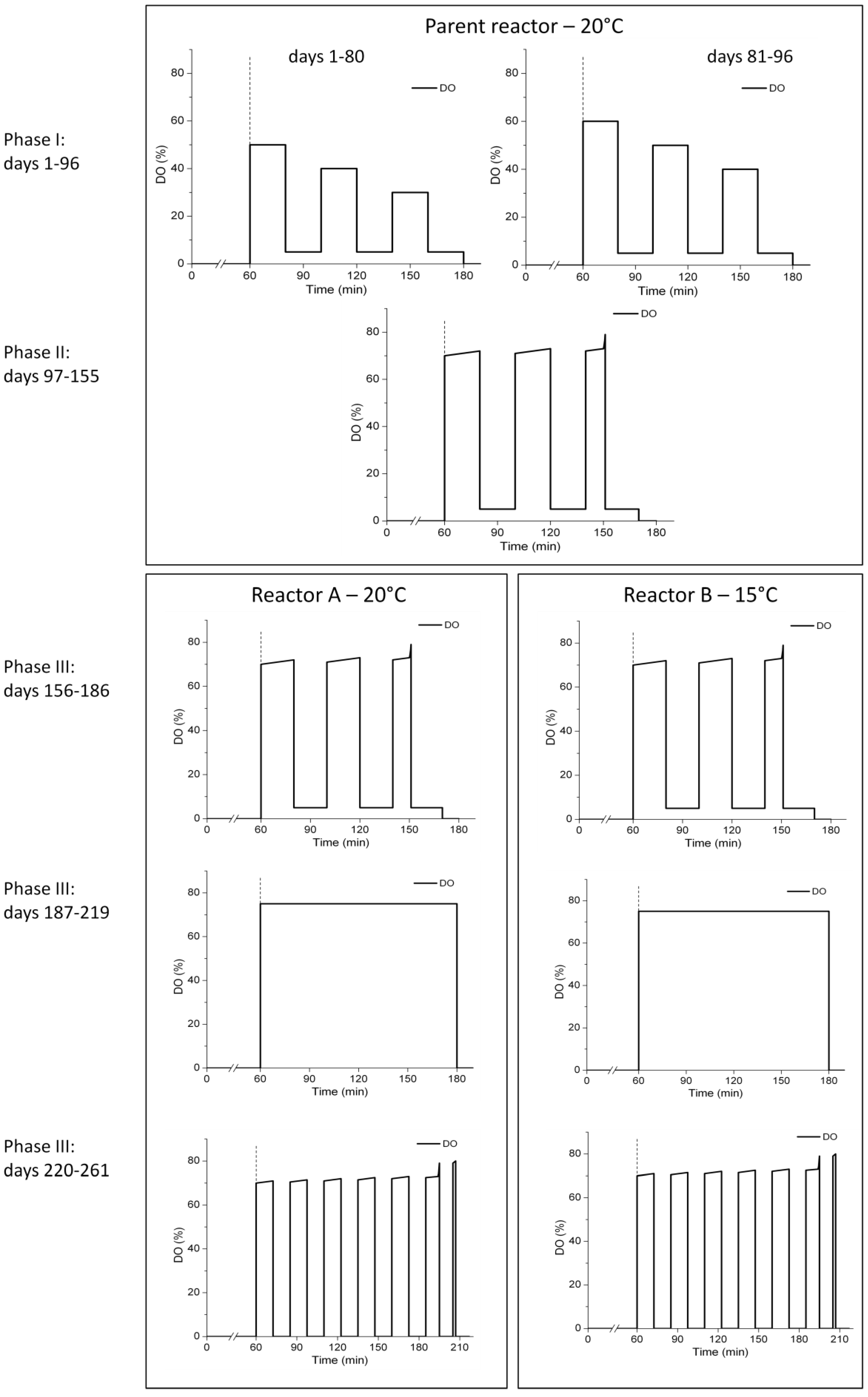
2.2. Experimental Schedule and Aeration Strategies
2.3. Analytical Methods
2.4. Nitrification Batch Tests
2.5. Quantification of Nitrite-Oxidizing Bacteria
2.6. Nitrous Oxide Tests
| Run | N-Removal over | Aeration Strategy | COD Concentration |
|---|---|---|---|
| No. | (mgCOD·L−1) | ||
| 1 | NO2− | Constant DO 30% | 400 |
| 2 | NO2− | Constant DO 30% | 600 |
| 3 | NO2− | Intermittent aeration | 400 |
| 4 | NO2− | Intermittent aeration | 600 |
| 5 | NO3− | Constant DO 30% | 400 |
| 6 | NO3− | Constant DO 30% | 600 |
| 7 | NO3− | Intermittent aeration | 400 |
| 8 | NO3− | Intermittent aeration | 600 |
3. Results
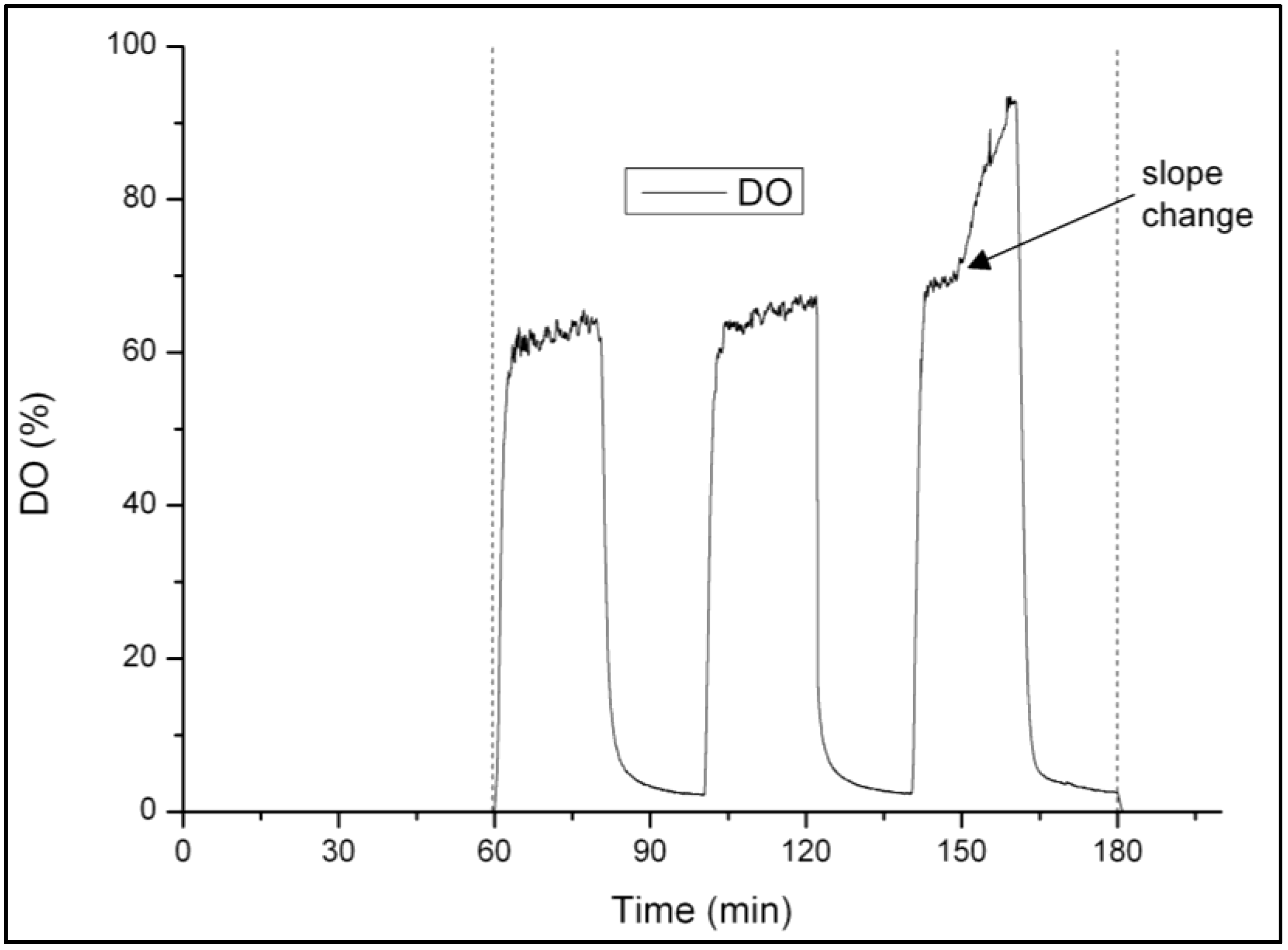
3.1. Strategy for Aeration Phase Length Control
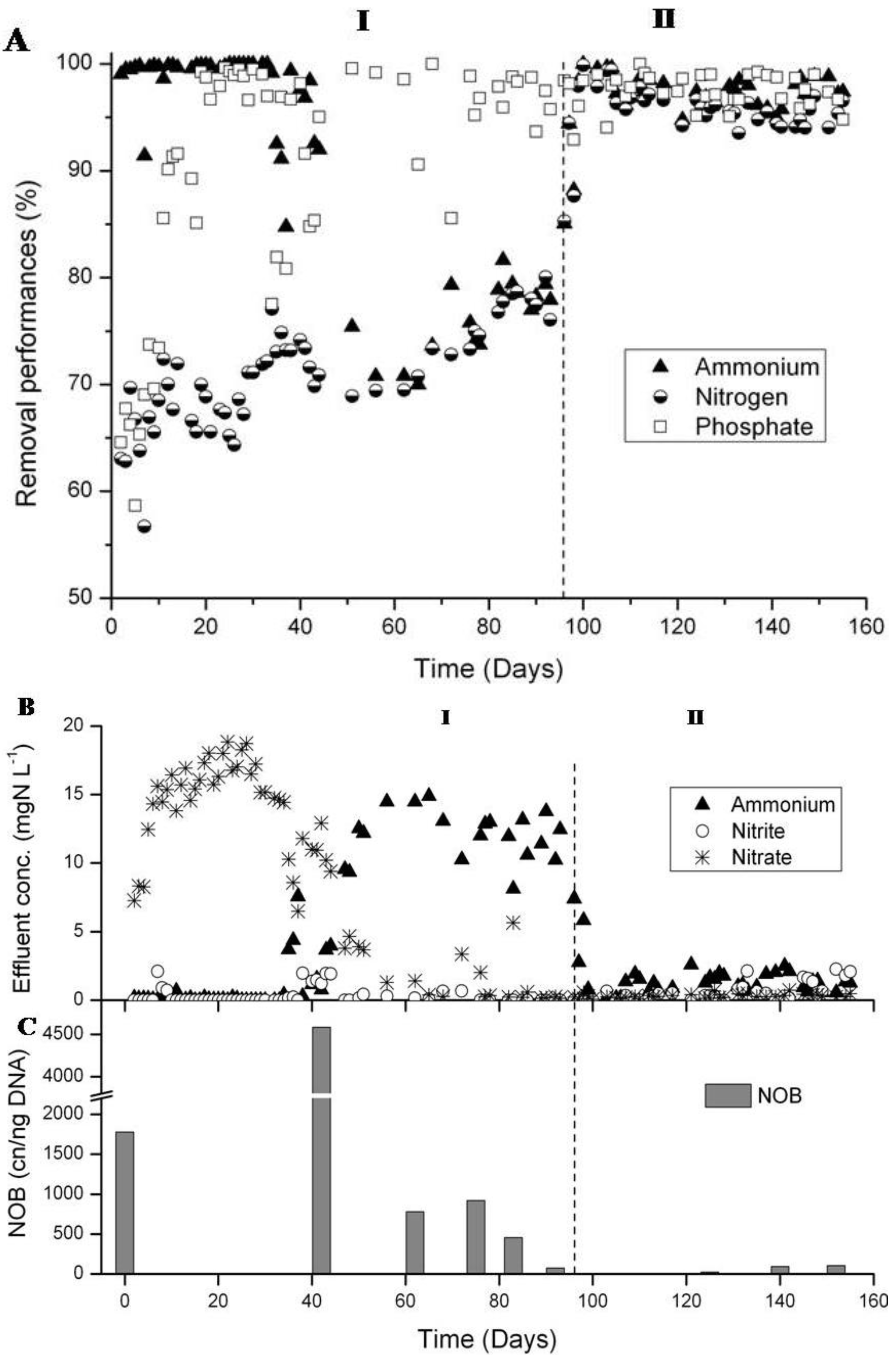
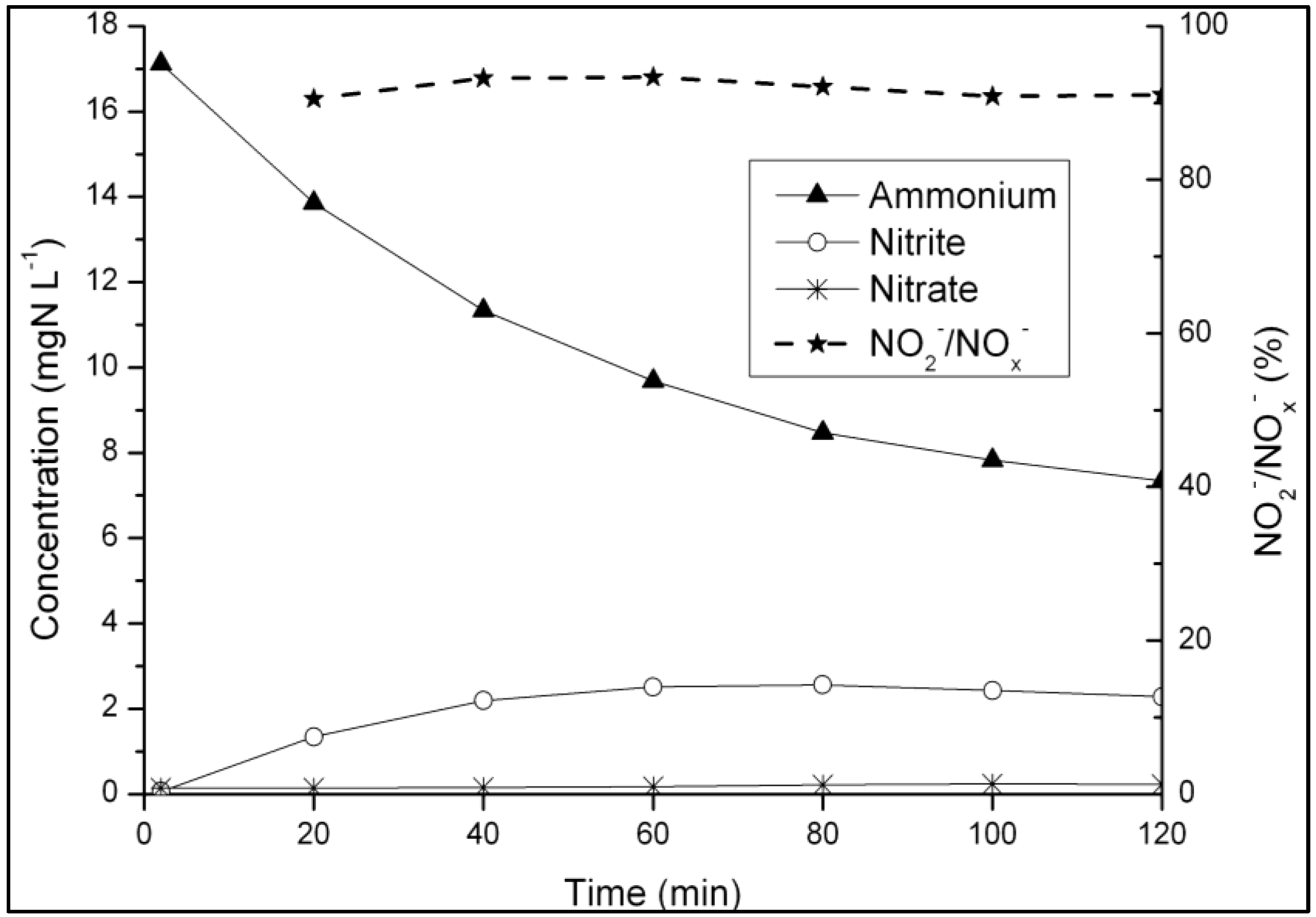
3.2. N-removal and NOB Abundance in the Parent Reactor
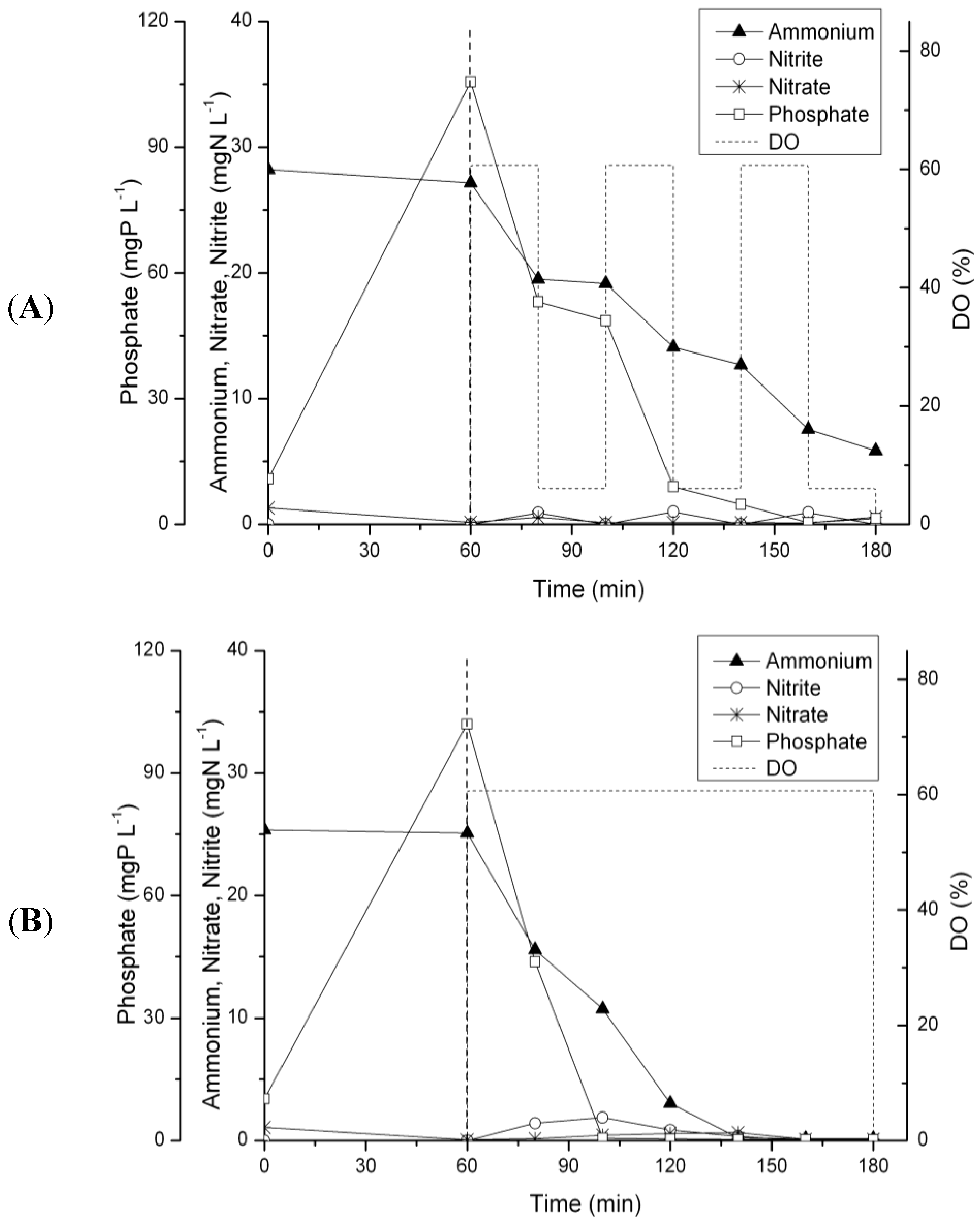
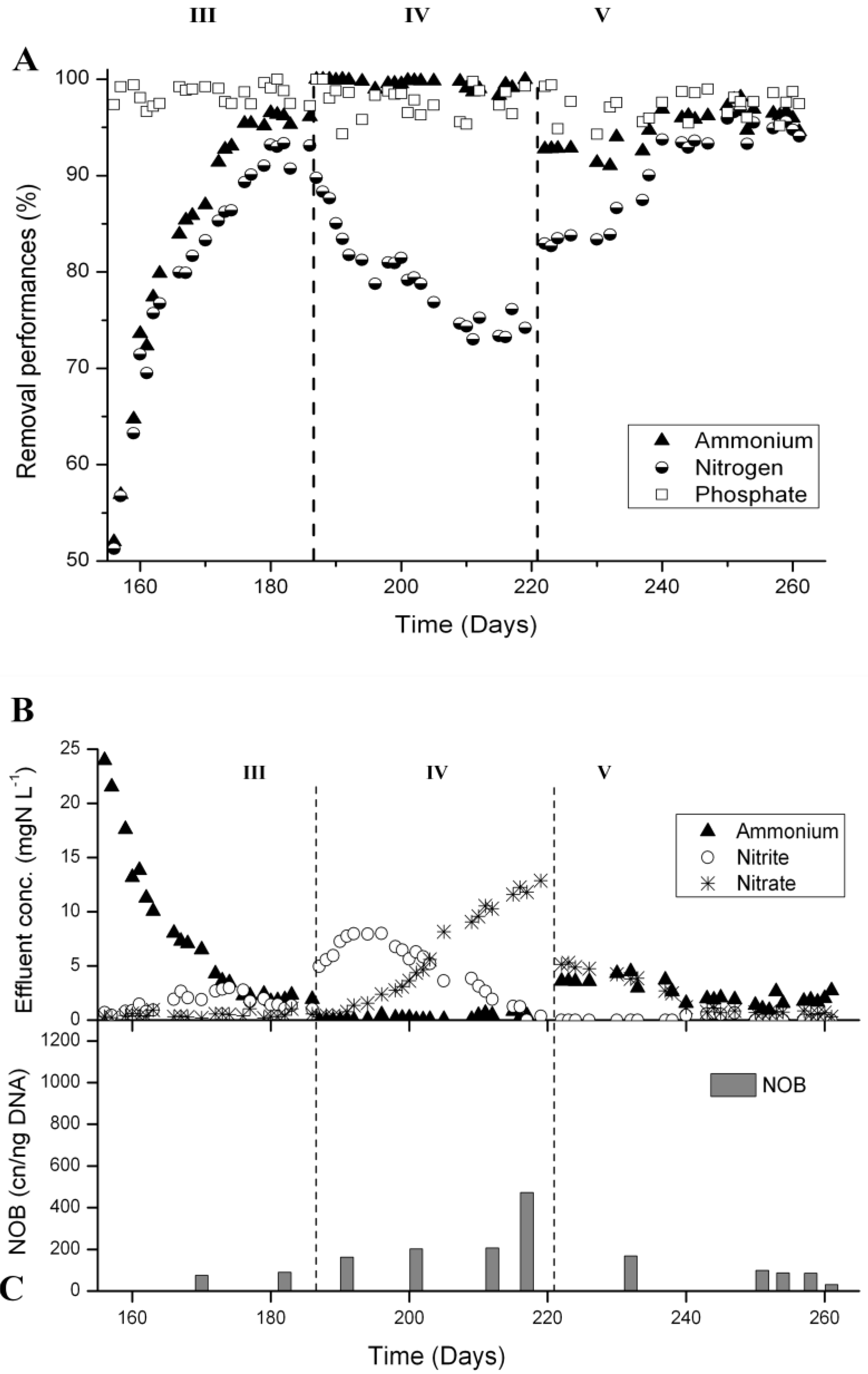
3.3. N-Removal and NOB Abundance at 20 °C
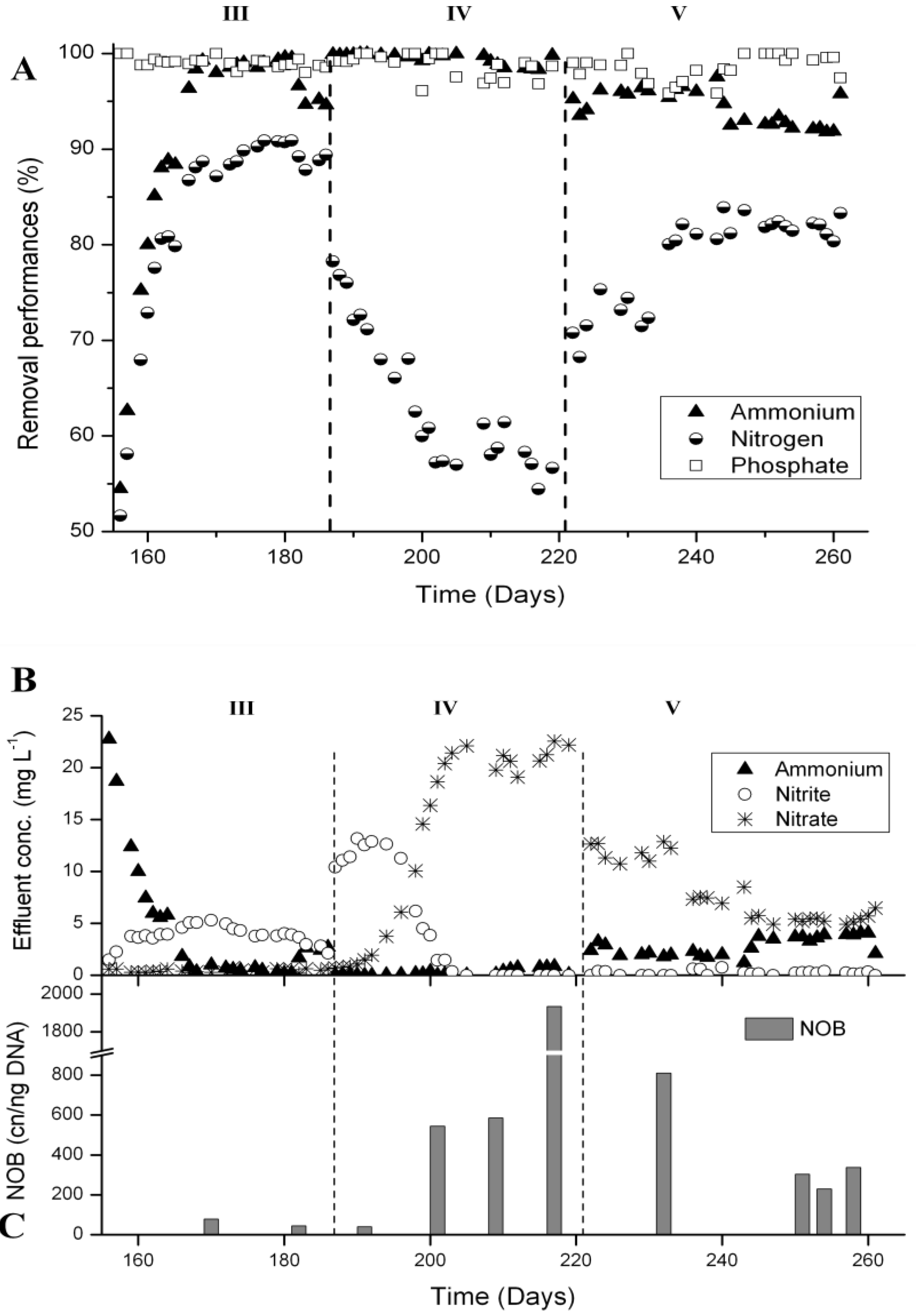
3.4. N-Removal and NOB Abundance at 15 °C
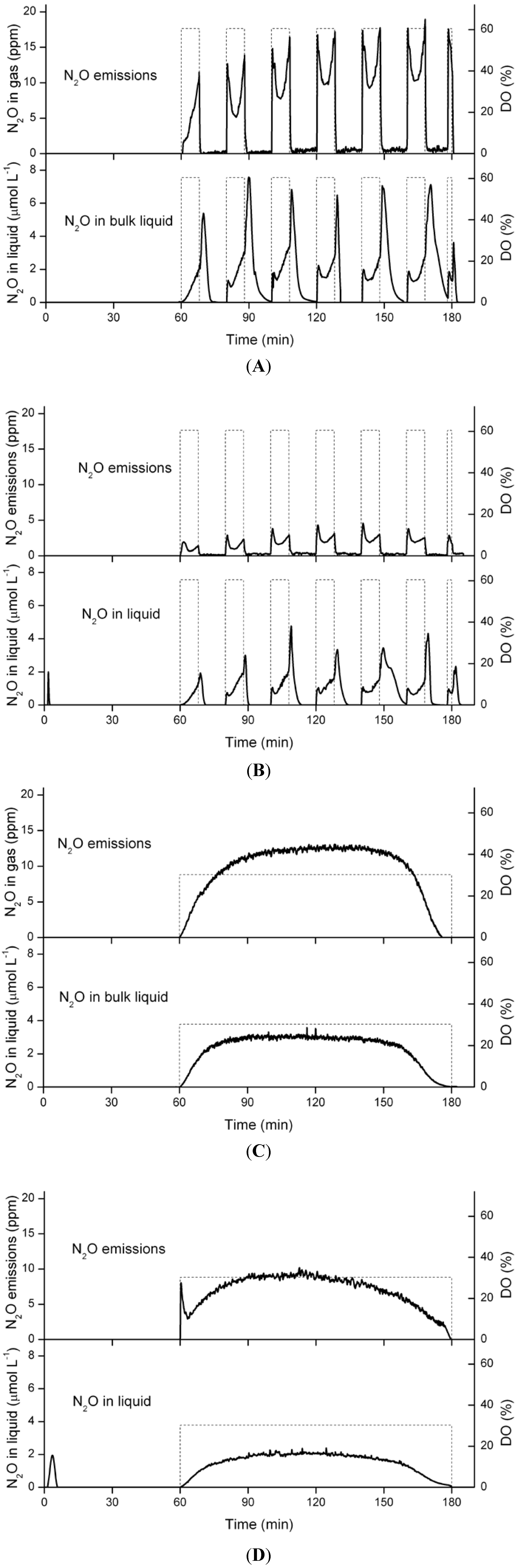
3.5. Nitrous Oxide Emissions and Concentrations in the Liquid Phase
| Aeration Strategy | COD Concentration (mgCOD·L−1) | Nitrite Pathway (% of N load) 1 | Nitrate Pathway (% of N load) 1 |
|---|---|---|---|
| Intermittent aeration | 400 | 5.2 ± 1.1 | 0.8 ± 0.2 |
| Intermittent aeration | 600 | 2.4 ± 0.8 | 0.7 ± 0.3 |
| Constant aeration | 400 | 12.9 ± 2.1 | 9.3 ± 2.4 |
| Constant aeration | 600 | 8.1 ± 1.7 | 5.9 ± 0.9 |
4. Discussion
5. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zeng, R.J.; Lemaire, R.; Yuan, Z.; Keller, J. Simultaneous nitrification, denitrification, and phosphorus removal in a lab-scale sequencing batch reactor. Biotechnol. Bioeng. 2003, 84, 170–178. [Google Scholar] [CrossRef]
- De Kreuk, M.K.; Heijnen, J.J.; van Loosdrecht, M.C.M. Simultaneous COD, nitrogen, and phosphate removal by aerobic granular sludge. Biotechnol. Bioeng. 2005, 90, 761–769. [Google Scholar] [CrossRef]
- Yilmaz, G.; Lemaire, R.; Keller, J.; Yuan, Z. Simultaneous nitrification, denitrification, and phosphorus removal from nutrient-rich industrial wastewater using granular sludge. Biotechnol. Bioeng. 2008, 100, 529–541. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, J.; Wang, J.; Guo, H.; Tong, J. Integration of nitrification and denitrifying dephosphatation in airlift loop sequencing batch biofilm reactor. Process. Biochem. 2006, 41, 599–608. [Google Scholar] [CrossRef]
- Ekama, G.A.; Wentzel, M.C. Difficulties and developments in biological nutrient removal technology and modelling. Water Sci. Technol. 1999, 39, 1–11. [Google Scholar] [CrossRef]
- Kuba, T.; van Loosdrecht, M.C.M.; Heijnen, J.J. Phosphorus and nitrogen removal with minimal COD requirement by integration of denitrifying dephosphatation and nitrification in a two-sludge system. Water Res. 1996, 30, 1702–1710. [Google Scholar] [CrossRef]
- Keller, J.; Subramaniam, K.; Gösswein, J.; Greenfield, P.F. Nutrient removal from industrial wastewater using single tank sequencing batch reactors. Water Sci. Technol. 1997, 35, 137–144. [Google Scholar]
- Cassidy, D.P.; Belia, E. Nitrogen and phosphorus removal from an abattoir wastewater in a SBR with aerobic granular sludge. Water Res. 2005, 39, 4817–4823. [Google Scholar] [CrossRef]
- Lochmatter, S.; Gonzalez-Gil, G.; Holliger, C. Optimized aeration strategies for nitrogen and phosphorus removal with aerobic granular sludge. Water Res. 2013, 47, 6187–6197. [Google Scholar] [CrossRef]
- Furumai, H.; Kazmi, A.A.; Fujita, M.; Furuya, Y.; Sasaki, K. Modeling long term nutrient removal in a sequencing batch reactor. Water Res. 1999, 33, 2708–2714. [Google Scholar] [CrossRef]
- Peng, Z.; Peng, Y.; Gui, L.; Liu, X. Competition for single carbon source between denitrification and phosphorus release in sludge under anoxic condition. Chin. J. Chem. Eng. 2010, 18, 472–477. [Google Scholar] [CrossRef]
- Turk, O.; Mavinic, D.S. Benefits of using selective inhibition to remove nitrogen from highly nitrogenous wastes. Environ. Technol. Lett. 1987, 8, 419–426. [Google Scholar] [CrossRef]
- Lackner, S.; Gilbert, E.M.; Vlaeminck, S.E.; Joss, A.; Horn, H.; van Loosdrecht, M.C.M. Full-scale partial nitritation/anammox experiences—An application survey. Water Res. 2014, 55, 292–303. [Google Scholar] [CrossRef]
- Hellinga, C.; Schellen, A.A.J.C.; Mulder, J.W.; van Loosdrecht, M.C.M.; Heijnen, J.J. The sharon process: An innovative method for nitrogen removal from ammonium-rich waste water. Water Sci. Technol. 1998, 37, 135–142. [Google Scholar]
- Munz, G.; Lubello, C.; Oleszkiewicz, J.A. Factors affecting the growth rates of ammonium and nitrite oxidizing bacteria. Chemosphere 2011, 83, 720–725. [Google Scholar] [CrossRef]
- Anthonisen, A.C.; Loehr, R.C.; Prakasam, T.B.S.; Srinath, E.G. Inhibition of nitrification by ammonia and nitrous acid. J. Water Pollut. Control. Federation 1976, 48, 835–852. [Google Scholar]
- Vadivelu, V.M.; Keller, J.; Yuan, Z. Free ammonia and free nitrous acid inhibition on the anabolic and catabolic processes of Nitrosomonas and Nitrobacter. Water Sci. Technol. 2007, 56, 89–97. [Google Scholar] [CrossRef]
- Xu, G.; Xu, X.; Yang, F.; Liu, S. Selective inhibition of nitrite oxidation by chlorate dosing in aerobic granules. J. Hazard. Mater. 2011, 185, 249–254. [Google Scholar] [CrossRef]
- Philips, S.; Laanbroek, H.; Verstraete, W. Origin, causes and effects of increased nitrite concentrations in aquatic environments. Rev. Environ. Sci. Biotechnol. 2002, 1, 115–141. [Google Scholar] [CrossRef]
- Ciudad, G.; Werner, A.; Bornhardt, C.; Muñoz, C.; Antileo, C. Differential kinetics of ammonia- and nitrite-oxidizing bacteria: A simple kinetic study based on oxygen affinity and proton release during nitrification. Process. Biochem. 2006, 41, 1764–1772. [Google Scholar] [CrossRef]
- Guisasola, A.; Jubany, I.; Baeza, J.A.; Carrera, J.; Lafuente, J. Respirometric estimation of the oxygen affinity constants for biological ammonium and nitrite oxidation. J. Chem. Technol. Biotechnol. 2005, 80, 388–396. [Google Scholar] [CrossRef]
- Tokutomi, T. Operation of a nitrite-type airlift reactor at low DO concentration. Water Sci. Technol. 2004, 49, 81–88. [Google Scholar]
- Bernet, N.; Peng, D.; Delgenes, J.-P.; Moletta, R. Nitrification at low oxygen concentration in biofilm reactor. J. Environ. Eng. 2001, 127, 266–271. [Google Scholar] [CrossRef]
- Ruiz, G.; Jeison, D.; Chamy, R. Nitrification with high nitrite accumulation for the treatment of wastewater with high ammonia concentration. Water Res. 2003, 37, 1371–1377. [Google Scholar] [CrossRef]
- Blackburne, R.; Yuan, Z.; Keller, J. Demonstration of nitrogen removal via nitrite in a sequencing batch reactor treating domestic wastewater. Water Res. 2008, 42, 2166–2176. [Google Scholar] [CrossRef]
- Lemaire, R.; Marcelino, M.; Yuan, Z. Achieving the nitrite pathway using aeration phase length control and step-feed in an SBR removing nutrients from abattoir wastewater. Biotechnol. Bioeng. 2008, 100, 1228–1236. [Google Scholar] [CrossRef]
- Li, J.; Elliott, D.; Nielsen, M.; Healy, M.G.; Zhan, X. Long-term partial nitrification in an intermittently aerated sequencing batch reactor (SBR) treating ammonium-rich wastewater under controlled oxygen-limited conditions. Biochem. Eng. J. 2011, 55, 215–222. [Google Scholar] [CrossRef]
- Fux, C.; Velten, S.; Carozzi, V.; Solley, D.; Keller, J. Efficient and stable nitritation and denitritation of ammonium-rich sludge dewatering liquor using an SBR with continuous loading. Water Res. 2006, 40, 2765–2775. [Google Scholar] [CrossRef]
- Ge, S.; Peng, Y.; Qiu, S.; Zhu, A.; Ren, N. Complete nitrogen removal from municipal wastewater via partial nitrification by appropriately alternating anoxic/aerobic conditions in a continuous plug-flow step feed process. Water Res. 2014, 55, 95–105. [Google Scholar] [CrossRef]
- Regmi, P.; Miller, M.W.; Holgate, B.; Bunce, R.; Park, H.; Chandran, K.; Wett, B.; Murthy, S.; Bott, C.B. Control of aeration, aerobic SRT and COD input for mainstream nitritation/denitritation. Water Res. 2014, 57, 162–171. [Google Scholar] [CrossRef]
- Gao, D.W.; Lu, J.C.; Liang, H. Simultaneous energy recovery and autotrophic nitrogen removal from sewage at moderately low temperatures. Appl. Microbiol. Biotechnol. 2014, 98, 2637–2645. [Google Scholar] [CrossRef]
- Hu, Z.; Lotti, T.; de Kreuk, M.; Kleerebezem, R.; van Loosdrecht, M.; Kruit, J.; Jetten, M.S.M.; Kartal, B. Nitrogen removal by a nitritation-anammox bioreactor at low temperature. Appl. Environ. Microbiol. 2013, 79, 2807–2812. [Google Scholar] [CrossRef]
- Yang, Q.; Peng, Y.; Liu, X.; Zeng, W.; Mino, T.; Satoh, H. Nitrogen removal via nitrite from municipal wastewater at low temperatures using real-time control to optimize nitrifying communities. Environ. Sci. Technol. 2007, 41, 8159–8164. [Google Scholar] [CrossRef]
- Ebrahimi, S.; Gabus, S.; Rohrbach-Brandt, E.; Hosseini, M.; Rossi, P.; Maillard, J.; Holliger, C. Performance and microbial community composition dynamics of aerobic granular sludge from sequencing batch bubble column reactors operated at 20 degrees C, 30 degrees C, and 35 degrees C. Appl. Microbiol. Biotechnol. 2010, 87, 1555–1568. [Google Scholar] [CrossRef]
- Lochmatter, S.; Holliger, C. Optimization of operation conditions for the startup of aerobic granular sludge reactors biologically removing carbon, nitrogen, and phosphorous. Water Res. 2014, 59, 58–70. [Google Scholar] [CrossRef]
- Mosquera-Corral, A.; de Kreuk, M.K.; Heijnen, J.J.; van Loosdrecht, M.C.M. Effects of oxygen concentration on N-removal in an aerobic granular sludge reactor. Water Res. 2005, 39, 2676–2686. [Google Scholar] [CrossRef]
- Vishniac, W.; Santer, M. The thiobacilli. Bacteriol. Rev. 1957, 21, 195–213. [Google Scholar]
- Gonzalez-Gil, G.; Holliger, C. Dynamics of microbial community structure and enhanced biological phosphorus removal of propionate- and acetate-cultivated aerobic granules. Appl. Environ. Microbiol. 2011, 77, 8041–8051. [Google Scholar] [CrossRef]
- Weissbrodt, D.G.; Neu, T.R.; Kuhlicke, U.; Rappaz, Y.; Holliger, C. Assessment of bacterial and structural dynamics in aerobic granular biofilms. Front. Microbiol. 2013, 4, 175. [Google Scholar]
- Dionisi, H.M.; Layton, A.C.; Harms, G.; Gregory, I.R.; Robinson, K.G.; Sayler, G.S. Quantification of Nitrosomonas oligotropha-like ammonia-oxidizing bacteria and Nitrospira spp. from full-scale wastewater treatment plants by competitive PCR. Appl. Environ. Microbiol. 2002, 68, 245–253. [Google Scholar] [CrossRef]
- Rupakula, A.; Kruse, T.; Boeren, S.; Holliger, C.; Smidt, H.; Maillard, J. The restricted metabolism of the obligate organohalide respiring bacterium Dehalobacter restrictus: Lessons from tiered functional genomics. Philosophical Trans. R. Soc. Lond. Ser. B Biol. Sci. 2013, 368. [Google Scholar] [CrossRef]
- Lemaire, R.; Yuan, Z.; Blackall, L.L.; Crocetti, G.R. Microbial distribution of Accumulibacter spp. and Competibacter spp. in aerobic granules from a lab-scale biological nutrient removal system. Environ. Microbiol. 2008, 10, 354–363. [Google Scholar] [CrossRef]
- Weissbrodt, D.G.; Shani, N.; Holliger, C. Linking bacterial population dynamics and nutrient removal in the granular sludge biofilm ecosystem engineered for wastewater treatment. FEMS Microbiol. Ecol. 2014, 88, 579–595. [Google Scholar] [CrossRef]
- Chandran, K.; Smets, B.F. Single-step nitrification models erroneously describe batch ammonia oxidation profiles when nitrite oxidation becomes rate limiting. Biotechnol. Bioeng. 2000, 68, 396–406. [Google Scholar] [CrossRef]
- Knowles, G.; Downing, A.L.; Barrett, M.J. Determination of kinetic constants for nitrifying bacteria in mixed culture, with the aid of an electronic computer. J. Gen. Microbiol. 1965, 38, 263–278. [Google Scholar] [CrossRef]
- Chung, J.; Shim, H.; Park, S.-J.; Kim, S.J.; Bae, W. Optimization of free ammonia concentration for nitrite accumulation in shortcut biological nitrogen removal process. Bioprocess. Biosyst. Eng. 2006, 28, 275–282. [Google Scholar] [CrossRef]
- Bae, W.; Baek, S.; Chung, J.; Lee, Y. Optimal operational factors for nitrite accumulation in batch reactors. Biodegradation 2001, 12, 359–366. [Google Scholar] [CrossRef]
- Kornaros, M.; Dokianakis, S.N.; Lyberatos, G. Partial nitrification/denitrification can be attributed to the slow response of nitrite oxidizing bacteria to periodic anoxic disturbances. Environ. Sci. Technol. 2010, 44, 7245–7253. [Google Scholar] [CrossRef]
- Bassin, J.P.; Kleerebezem, R.; Dezotti, M.; van Loosdrecht, M.C.M. Simultaneous nitrogen and phosphate removal in aerobic granular sludge reactors operated at different temperatures. Water Res. 2012, 46, 3805–3816. [Google Scholar] [CrossRef]
- Saito, T.; Brdjanovic, D.; van Loosdrecht, M.C.M. Effect of nitrite on phosphate uptake by phosphate accumulating organisms. Water Res. 2004, 38, 3760–3768. [Google Scholar] [CrossRef]
- Ahn, J.H.; Kwan, T.; Chandran, K. Comparison of partial and full nitrification processes applied for treating high-strength nitrogen wastewaters: microbial ecology through nitrous oxide production. Environ. Sci. Technol. 2011, 45, 2734–2740. [Google Scholar] [CrossRef]
- Rodriguez-Caballero, A.; Ribera, A.; Balcázar, J.L.; Pijuan, M. Nitritation versus full nitrification of ammonium-rich wastewater: comparison in terms of nitrous and nitric oxides emissions. Bioresour. Technol. 2013, 139, 195–202. [Google Scholar]
- Wunderlin, P.; Mohn, J.; Joss, A.; Emmenegger, L.; Siegrist, H. Mechanisms of N2O production in biological wastewater treatment under nitrifying and denitrifying conditions. Water Res. 2012, 46, 1027–1037. [Google Scholar]
- Kampschreur, M.J.; Temmink, H.; Kleerebezem, R.; Jetten, M.S.M.; van Loosdrecht, M.C.M. Nitrous oxide emission during wastewater treatment. Water Res. 2009, 43, 4093–4103. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lochmatter, S.; Maillard, J.; Holliger, C. Nitrogen Removal over Nitrite by Aeration Control in Aerobic Granular Sludge Sequencing Batch Reactors. Int. J. Environ. Res. Public Health 2014, 11, 6955-6978. https://doi.org/10.3390/ijerph110706955
Lochmatter S, Maillard J, Holliger C. Nitrogen Removal over Nitrite by Aeration Control in Aerobic Granular Sludge Sequencing Batch Reactors. International Journal of Environmental Research and Public Health. 2014; 11(7):6955-6978. https://doi.org/10.3390/ijerph110706955
Chicago/Turabian StyleLochmatter, Samuel, Julien Maillard, and Christof Holliger. 2014. "Nitrogen Removal over Nitrite by Aeration Control in Aerobic Granular Sludge Sequencing Batch Reactors" International Journal of Environmental Research and Public Health 11, no. 7: 6955-6978. https://doi.org/10.3390/ijerph110706955
APA StyleLochmatter, S., Maillard, J., & Holliger, C. (2014). Nitrogen Removal over Nitrite by Aeration Control in Aerobic Granular Sludge Sequencing Batch Reactors. International Journal of Environmental Research and Public Health, 11(7), 6955-6978. https://doi.org/10.3390/ijerph110706955





