Engineered Nanomaterials in Food: Implications for Food Safety and Consumer Health
Abstract
:1. Introduction
2. Applications of ENMs in the Food Sector
- (i)
- agriculture-pesticide, fertilizer or vaccine delivery; animal and plant pathogen detection; and targeted genetic engineering,
- (ii)
- food processing-encapsulation of flavor or odor enhancers; food textural or quality improvement; new gelation or viscosifying agents,
- (iii)
- food packaging-pathogen, gas or abuse sensors; anticounterfeiting devices, UV-protection, and stronger, more impermeable polymer films,
- (iv)
- nutrient supplements-nutraceuticals with higher stability and bioavailability.
| Area of application | Application | Reference |
|---|---|---|
| Agriculture (Nano-modification of seed and fertilisers/pesticides) | Pesticides | [27] |
| Targeted genetic engineering | [29] | |
| Preservation | [29,30] | |
| Agrichemical delivery | [29] | |
| Sensors to monitor soil conditions | [31] | |
| Processing (Interactive smart food) | Nanoencapsulation of flavors/aromas | [32,33,34,35,36,37] |
| Nanoemulsions | [38] | |
| Anti-caking agents | [39] | |
| Nutrition (Food fortification and modification) | Nutraceuticals | [40,41,42] |
| Nutrient delivery | [32,41] | |
| Mineral and vitamin fortification | [43,44] | |
| Drinking water purification | [45] | |
| Sensory characteristics of supplements | [46,47,48,49,50,51] | |
| Products (Smart packaging and food tracking) | UV protection | [52,53,54] |
| Antimicrobials | [55,56,57,58,59,60,61,62,63] | |
| Condition and abuse monitors | [46,47,64] | |
| High barrier plastics | [65,66,67,68] | |
| Security | [45,69,70] | |
| Contaminant sensors | [51,71,72,73,74,75] |
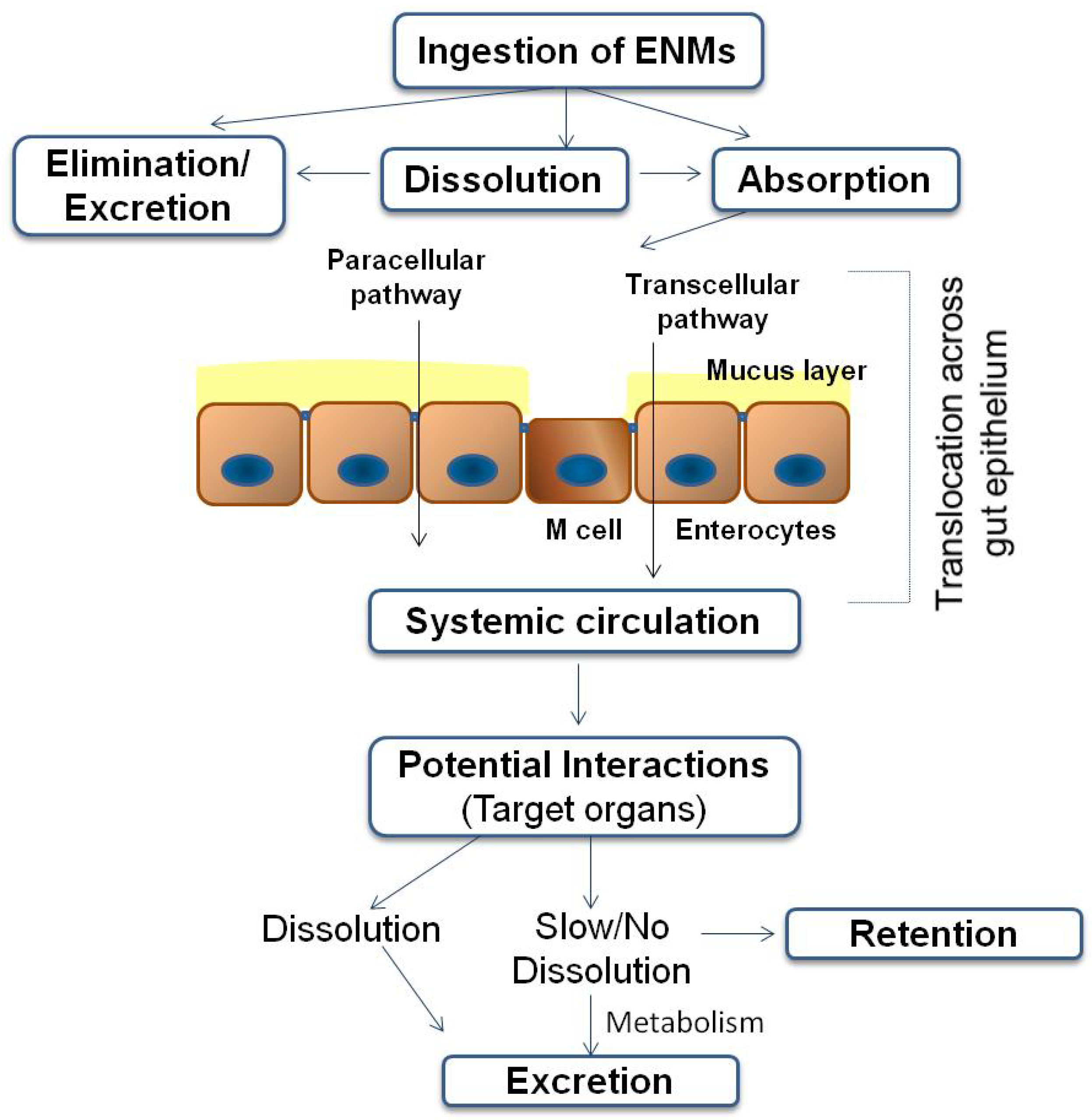
3. Fate of ENMs Following Ingestion
- As ENMs have been shown to have a greater ability to cross the gut wall, as compared to microparticles of the same kind, their enhanced absorption and bioavailability would result in higher internal exposure (higher plasma concentrations). The nanofood may also alter the way by which food ingredients are distributed or behave in the GIT [7].
- It is not only the bulk material of the ENM that may trigger biological effects. ENMs may form a biofunctional corona and thus act as carriers of these substances to different biological tissues, potentially influencing the absorption of molecules, e.g., by introducing unintended molecules across the GIT and lead to unpredictable effects, known as “Trojan horse” effect [83].
- In addition, it has been shown that the consumption of food containing NPs has the potential to alter body metabolism of experimental animals. For instance, oral administration of nanocalcium-enriched milk has been shown to alter the calcium metabolism in rats [84].
4. Food-associated ENMs: Metal (Oxide) NPs and Their Toxicity Profiles
4.1. NanoSilver
4.2. NanoTitanium
4.3. NanoZinc
4.4. NanoSilica
5. Health Implications of Nanofood, Consumer Safety Issues and Regulatory Aspects
6. Discussion and Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Smita, S.; Gupta, S.K.; Bartonova, A.; Dusinska, M.; Gutleb, A.C.; Rahman, Q. Nanoparticles in the environment: Assessment using the causal diagram approach. Environ. Health 2012, 11, 1–13. [Google Scholar] [CrossRef]
- Hansen, S.F.; Michelson, E.S.; Kamper, A.; Borling, P.; Stuer-Lauridsen, F.; Baun, A. Categorization framework to aid exposure assessment of nanomaterials in consumer products. Ecotoxicology 2008, 17, 438–447. [Google Scholar] [CrossRef]
- Mueller, N.C.; Nowack, B. Exposure modeling of engineered nanoparticles in the environment. Environ. Sci. Technol. 2008, 42, 4447–4453. [Google Scholar] [CrossRef]
- Cassee, F.R.; van Balen, E.C.; Singh, C.; Green, D.; Muijser, H.; Weinstein, J.; Dreher, K. Exposure, health and ecological effects review of engineered nanoscale cerium and cerium oxide associated with its use as a fuel additive. Crit. Rev. Toxicol. 2011, 41, 213–219. [Google Scholar] [CrossRef]
- Klaine, S.J.; Alvarez, P.J.; Batley, G.E.; Fernandes, T.F.; Handy, R.D.; Lyon, D.Y.; Mahendra, S.; McLaughlin, M.J.; Lead, J.R. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects. Environ. Toxicol. Chem. 2008, 27, 1825–1851. [Google Scholar] [CrossRef]
- Buzea, C.; Pacheco, I.; Robbie, K. Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases 2007, 4, 17–71. [Google Scholar]
- Chaudhry, Q.; Scotter, M.; Blackburn, J.; Ross, B.; Boxall, A.; Castle, L.; Aitken, R.; Watkins, R. Applications and implications of nanotechnologies for the food sector. Food Addit. Contam. Pt A 2008, 25, 241–258. [Google Scholar] [CrossRef]
- Sanguansri, P.; Augustin, M.A. Nanoscale materials development—A food industry perspective. Trends Food Sci. Technol. 2006, 17, 547–556. [Google Scholar] [CrossRef]
- Lyon, D.Y.; Thill, A.; Rose, J.; Alvarez, P.J.J. Ecotoxicological Impacts of Nanomaterials. In Environmental Nanotechnology, Applications and Impacts of Nanomaterials; Wiesner, M.R., Bottero, J.Y., Eds.; McGraw Hill: New York, NY, USA, 2007; pp. 445–479. [Google Scholar]
- Lee, J.; Mahendra, S.; Alvarez, P.J. Nanomaterials in the construction industry: A review of their applications and environmental health and safety considerations. ACS Nano 2010, 4, 3580–3590. [Google Scholar]
- Stones, M. Nanoscience to Boost Food Safety, Quality and Shelf Life. Report from IFT 8 June 2009. Available online: http://www.foodproductiondaily.com/Safety-Regulation/Nanoscience-to-boost-food-safety-quality-and-shelf-life (accessed on 17 December 2013).
- Cho, Y.; Kim, C.; Kim, N.; Kim, C.; Park, B. Some cases in applications of nanotechnology to food and agricultural systems. Biochip J. 2008, 2, 183–185. [Google Scholar]
- Miller, G. Nanotechnology—The new threat to food. Friends of the Earth 30 October 2008. Available online: http://www.globalresearch.ca/PrintArticle.php?articleId=10755 (accessed on 17 December 2013).
- Project on Emerging Nanotechnologies: Woodrow Wilson International Center for Scholars. Available online: http://www.nanotechproject.org/cpi/ (accessed on 10 March 2013).
- Kah, M.; Tiede, K.; Beulke, S.; Hofmann, T. Nanopesticides: State of knowledge, environmental fate and exposure modeling. Crit. Rev. Environ. Sci. Technol. 2013, 43. [Google Scholar] [CrossRef]
- Takenaka, S.; Karg, E.; Roth, C.; Schulz, H.; Ziesenis, A.; Heinzmann, U.; Schramel, P.; Heyder, J. Pulmonary and systemic distribution of inhaled ultrafine silver particles in rats. Environ. Health Perspect. 2001, 109, 547–551. [Google Scholar] [CrossRef]
- Landsiedel, R.; Fabian, E.; Ma-Hock, L.; van Ravenzwaay, B.; Wohlleben, W.; Wiench, K.; Oesch, F. Toxico-/biokinetics of nanomaterials. Arch. Toxicol. 2012, 86, 1021–1060. [Google Scholar] [CrossRef]
- National Institute for Occupational Safety and Health (NIOSH). Filling the Knowledge Gaps for Safe Nanotechnology in the Workplace. In A Progress Report from the NIOSH Nanotechnology Research Center, 2004–2011; Centers for Disease Control and Prevention: Atlanta, Department of Health and Human Services, National Institute for Occupational Safety and Health, GA, USA, 2012. [Google Scholar]
- Donaldson, K.; Borm, P.J.; Castranova, V.; Gulumian, M. The limits of testing particle-mediated oxidative stress in vitro in predicting diverse pathologies; relevance for testing of nanoparticles. Part Fibre Toxicol. 2009, 6. [Google Scholar] [CrossRef]
- Brown, D.M.; Donaldson, K.; Borm, P.J.; Schins, R.P.; Dehnhardt, M.; Gilmour, P.; Jimenez, L.A.; Stone, V. Calcium and ROS-mediated activation of transcription factors and TNF-alpha cytokine gene expression in macrophages exposed to ultrafine particles. Amer. J. Physiol. Lung Cell Mol. Physiol. 2004, 286, 344–353. [Google Scholar] [CrossRef]
- Chen, Z.; Meng, H.; Xing, G.; Chen, C.; Zhao, Y. Toxicological and biological effects of nanomaterials. Int. J. Nanotechnol. 2007, 4, 179–196. [Google Scholar] [CrossRef]
- Cockburn, A.; Bradford, R.; Buck, N.; Constable, A.; Edwards, G.; Haber, B.; Hepburn, P.; Howlett, J.; Kampers, F.; Klein, C.; et al. Approaches to the safety assessment of engineered nanomaterials (ENM) in food. Food Chem. Toxicol. 2012, 50, 2224–2242. [Google Scholar] [CrossRef]
- Grobe, A.; Renn, O.; Jaeger, A. Risk Governance of Nanotechnology Applications in Food and Cosmetics. Available online: http://www.irgc.org/IMG/pdf/IRGC_Report_FINAL_For_Web.pdf (accessed on 19 May 2014).
- Bouwmeester, H.; Dekkers, S.; Noordam, M.; Hagens, W.; Bulder, A.; de Heer, C.; ten Voorde, S.; Wijnhoven, S.; Sips, A. Health impact of nanotechnologies in food production. Report 2007.014 RIKILT (Institute of Food Safety, Wageningen UR) and RIVM (National Institute of Public Health and the Environment: Center for Substances and Integrated Risk Assessment). 2007, pp. 1–95. Available online: http://www.rivm.nl/bibliotheek/digitaaldepot/healthimpactnanotechnologies.pdf (accessed on 10 December 2013).
- FSAI (Food Safety Authority of Ireland). The Relevance for Food Safety of Applications of Nanotechnology in the Food and Feed Industries. 2008. Available online: http://www.fsai.ie/assets/0/86/204/b81b142b-9ef7–414c-9614–3a969835b392.pdf (accessed on 17 December 2013).
- Groves, K. Potential benefits of micro and nano technology for the food industry: Does size matter? New Food Mag. 2008, 4, 49–52. [Google Scholar]
- Kuzma, J.; VerHage, P. Nanotechnology in Agriculture and Food Production: Anticipated Applications. Available online: http://www.nanotechproject.org/file_download/94 (accessed on 16 December 2013).
- Morris, V.J. Nanotechnology in the food industry. New Food Mag. 2008, 4, 53–55. [Google Scholar]
- Kuzma, J. Nanotechnology in animal production—Upstream assessment of applications. Livestock Sci. 2010, 130, 14–24. [Google Scholar] [CrossRef]
- Chang, S.; Zhou, M.; Grover, C.P. Information coding and retrieving using fluorescent semiconductor nanocrystals for object identification. Opt. Express 2004, 12, 143–148. [Google Scholar] [CrossRef]
- Wang, L.; Maa, W.; Xub, L.; Chenb, W.; Zhub, Y.; Xua, C.; Kotov, N.A. Nanoparticle-based environmental sensors. Mater. Sci. Eng. 2010, 70, 265–274. [Google Scholar] [CrossRef]
- Huang, Q.R.; Yu, H.L.; Ru, Q.M. Bioavailability and delivery of nutraceuticals using nanotechnology. J. Food Sci. 2009, 75, 50–57. [Google Scholar] [CrossRef]
- Smith, A.; Giunta, B.; Bickford, P.C.; Fountain, M.; Tan, J.; Shytle, R.D. Nanolipidic particles improve the bioavailability and α-secretase inducing ability of epigallocatechin-3-gallate (EGCG) for the treatment of Alzheimer’s disease. Int. J. Pharm. 2010, 389, 207–212. [Google Scholar] [CrossRef]
- Barras, A.; Mezzetti, A.; Richard, A.; Lazzaroni, S.; Roux, S.; Melnyk, P.; Betbeder, D.; Monfilliette-Dupont, N. Formulation and characterization of polyphenol-loaded lipid nanocapsules. Int. J. Pharm. 2009, 379, 270–277. [Google Scholar] [CrossRef]
- Kumari, A.; Yadav, S.K.; Pakade, Y.B.; Kumar, V.; Singh, B.; Chaudhary, A.; Yadav, S.C. Nanoencapsulation and characterization of Albizia chinensis isolated antioxidant quercitrin on PLA nanoparticles. Colloid. Surface. B 2011, 82, 224–232. [Google Scholar] [CrossRef]
- Zheng, L.; Ding, Z.; Zhang, M.; Sun, J. Microencapsulation of bayberry polyphenols by ethyl cellulose: Preparation and characterization. J. Food Eng. 2011, 104, 89–95. [Google Scholar] [CrossRef]
- Luykx, D.; Peters, R.; van Ruth, S.M.; Bouwmeester, H. A review of analytical methods for the identification and characterisation of nano delivery systems in food. J. Agric. Food Chem. 2008, 56, 8231–8247. [Google Scholar] [CrossRef]
- Piorkowski, D.T.; McClements, D.J. Beverage emulsions: Recent developments in formulation, production, and applications. Food Hydrocolloids. Available online: http://www.sciencedirect.com/science/article/pii/S0268005X13002117 (accessed on 26 July 2013).
- Lomer, M.C.; Thompson, R.P.; Powell, J.J. Fine and ultrafine particles of the diet: Influence on the mucosal immune response and association with Crohn’s disease. Proc. Nutr. Soc. 2002, 61, 123–130. [Google Scholar] [CrossRef]
- Quintanilla-Carvajal, M.X.; Camacho-Díaz, B.H.; Meraz-Torres, L.S.; Chanona-Pérez, J.J.; Alamilla-Beltrán, L.; Jimenéz-Aparicio, A.; Gutiérrez-López, G.F. Nanoencapsulation: A new trend in food engineering processing. Food Eng. Rev. 2010, 2, 39–50. [Google Scholar] [CrossRef]
- Braithwaite, M.C.; Charu, T.; Tomar, L.K.; Kumar, P.; Choonara, Y.E.; Pillay, V. Nutraceutical-based therapeutics and formulation strategies augmenting their efficiency to complement modern medicine: An overview. J. Funct. Foods 2014, 6, 82–99. [Google Scholar] [CrossRef]
- Acosta, E. Bioavailability of nanoparticles in nutrient and nutraceutical delivery. Curr. Opin. Colloid Interface Sci. 2009, 14, 3–15. [Google Scholar] [CrossRef]
- Wiking, L.; Larsen, T.; Sehested, J. Transfer of dietary zinc and fat to milk—Evaluation of milk fat quality, milk fat precursors, and mastitis indicators. J. Dairy Sci. 2008, 91, 1544–1551. [Google Scholar] [CrossRef]
- Rincker, M.J.; Hill, G.M.; Link, J.E.; Meyer, A.M.; Rowntree, J.E. Effects of dietary zinc and iron supplementation on mineral excretion, body composition, and mineral status of nursery pigs. J. Anim. Sci. 2005, 83, 2762–2774. [Google Scholar]
- Li, Q.; Mahendra, S.; Lyon, D.Y.; Brunet, L.; Liga, M.V.; Li, D.; Alvarez, P.J. Antimicrobial nanomaterials for water disinfection and microbial control: Potential applications and implications. Water Res. 2008, 42, 4591–4602. [Google Scholar] [CrossRef]
- Kuang, H.; Chen, W.; Yan, W.; Xu, L.; Zhu, Y.; Liu, L.; Chu, H.; Peng, C.; Wang, L.; Kotov, N.A.; et al. Crown ether assembly of gold nanoparticles: Melamine sensor. Biosens. Bioelectron. 2011, 26, 2032–2037. [Google Scholar] [CrossRef]
- Staiano, M.; Matveeva, E.G.; Rossi, M.; Crescenzo, R.; Gryczynski, Z.; Gryczynski, I.; Iozzino, L.; Akopova, I.; D’Auria, S. Nanostructured silver-based surfaces: New emergent methodologies for an easy detection of analytes. Appl. Mater. Interface. 2009, 1, 2909–2916. [Google Scholar] [CrossRef]
- Liu, Y.; Ai, K.; Cheng, X.; Huo, L.; Lu, L. Gold-nanocluster-based fluorescent sensors for highly sensitive and selective detection of cyanide in water. Adv. Funct. Mater. 2010, 20, 951–956. [Google Scholar] [CrossRef]
- Hong, S.; Choi, I.; Lee, S.; Yang, Y.I.; Kang, T.; Yi, J. Sensitive and colorimetric detection of the structural evolution of superoxide dismutase with gold nanoparticles. Anal. Chem. 2009, 81, 1378–1382. [Google Scholar] [CrossRef]
- Wang, L.; Chen, W.; Xu, D.; Shim, B.S.; Zhu, Y.; Sun, F.; Liu, L.; Peng, C.; Jin, Z.; Xu, C.; et al. Simple, rapid, sensitive, and versatile SWNT-paper sensor for environmental toxin detection competitive with ELISA. Nano Lett. 2009, 9, 4147–4152. [Google Scholar] [CrossRef]
- Ozdemir, C.; Yeni, F.; Odaci, D.; Timur, S. Electrochemical glucose biosensing by pyranose oxidase immobilized in gold nanoparticle-polyaniline/AgCl/gelatin nanocomposite matrix. Food Chem. 2010, 119, 380–385. [Google Scholar] [CrossRef]
- Barnard, A.S. One-to-one comparison of sunscreen efficacy, aesthetics and potential nanotoxicity. Nat. Nanotechnol. 2010, 5, 271–274. [Google Scholar]
- Zhang, H.; Millington, K.R.; Wang, X. The photostability of wool doped with photocatalytic titanium dioxide nanoparticles. Polym. Degrad. Stabil. 2009, 94, 278–283. [Google Scholar] [CrossRef]
- Li, G.; Liu, H.; Zhao, H.; Gao, Y.; Wang, J.; Jiang, H.; Boughton, R.I. Chemical assembly of TiO2 and TiO2@ Ag nanoparticles on silk fiber to produce multifunctional fabrics. J. Colloid Interface Sci. 2011, 358, 307–315. [Google Scholar] [CrossRef]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramírez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346–2353. [Google Scholar] [CrossRef]
- Yoon, K.Y.; Hoon, B.J.; Park, J.H.; Hwang, J. Susceptibility constants of Escherichia coli and Bacillus subtilis to silver and copper nanoparticles. Sci. Total Environ. 2007, 373, 572–575. [Google Scholar] [CrossRef]
- Eby, D.M.; Schaeublin, N.M.; Farrington, K.E.; Hussain, S.M.; Johnson, G.R. Lysozyme catalyzes the formation of antimicrobial silver nanoparticles. ACS Nano 2009, 3, 984–994. [Google Scholar] [CrossRef]
- Egger, S.; Lehmann, R.P.; Height, M.J.; Loessner, M.J.; Schuppler, M. Antimicrobial properties of a novel silver-silica nanocomposite material. Appl. Environ. Microbiol. 2009, 75, 2973–2976. [Google Scholar]
- Huang, L.; Li, DQ.; Lin, Y.J.; Wei, M.; Evans, D.G.; Duan, X. Controllable preparation of Nano-MgO and investigation of its bactericidal properties. J. Inorg. Biochem. 2005, 99, 986–993. [Google Scholar] [CrossRef]
- Lin, Y.J.; Li, D.Q.; Wang, G.; Huang, L.; Duan, X. Preparation and bactericidal property of MgO nanoparticles on gamma-Al2O3. J. Mater. Sci. Mater. Med. 2005, 16, 53–56. [Google Scholar] [CrossRef]
- Esteban-Tejeda, L.; Malpartida, F.; Esteban-Cubillo, A.; Pecharromán, C.; Moya, J.S. The antibacterial and antifungal activity of a soda-lime glass containing silver nanoparticles. Nanotechnology 2009, 20. [Google Scholar] [CrossRef]
- Ren, G.; Hu, D.; Cheng, E.W.C.; Vargas-Reus, M.A.; Reip, P.; Allaker, R.P. Characterisation of copper oxide nanoparticles for antimicrobial applications. Int. J. Antimicrob. Agents 2009, 33, 587–590. [Google Scholar] [CrossRef]
- Lu, Y.; Chen, Y.; Lin, H.; Wang, C.; Yang, Z. Preparation of chitosan nanoparticles and their application to Antheraea pernyi silk. J. Appl. Polym. Sci. 2010, 117, 3362–3369. [Google Scholar]
- Cao, Q.A.; Zhao, H.; He, Y.J.; Li, X.J.; Zeng, L.X.; Ding, N.; Wang, J.A.; Yang, J.; Wang, G.W. Hydrogen-bonding-induced colorimetric detection of melamine by nonaggregation-based Au-NPs as a probe. Biosens. Bioelectron. 2010, 25, 2680–2685. [Google Scholar] [CrossRef]
- Robertson, G.L. Food Packaging: Principles and Practice, 2nd ed.; CRC Press, Taylor & Francis Group: New York, NY, USA, 2006. [Google Scholar]
- Sanchez-Garcia, M.D.; Gimenez, E.; Lagaron, J.M. Novel PET nanocomposites of interest in food packaging applications and comparative barrier performance with biopolyester nanocomposites. J. Plast. Film Sheet. 2007, 23, 133–148. [Google Scholar] [CrossRef]
- Quintavalla, S.; Vicini, L. Antimicrobial food packaging in meat industry. Meat Sci. 2002, 62, 373–380. [Google Scholar] [CrossRef]
- Appendini, P.; Hotchkiss, J.H. Review of antimicrobial food packaging. Innov. Food Sci. Emerg. Technol. 2002, 3, 113–126. [Google Scholar] [CrossRef]
- Chu, C.S.; Matylevitch, N.P.; McManus, A.T.; Goodwin, C.W.; Pruitt, B.A., Jr. Accelerated healing with a mesh autograft/allodermal composite skin graft treated with silver nylon dressings with and without direct current in rats. J. Trauma 2000, 49, 115–125. [Google Scholar] [CrossRef]
- Blaker, J.J.; Nazhat, S.N.; Boccaccini, A.R. Development and characterisation of silver-doped bioactive glass-coated sutures for tissue engineering and wound healing applications. Biomaterials 2004, 25, 1319–1329. [Google Scholar] [CrossRef]
- Warner, M.G.; Grate, J.W.; Tyler, A.; Ozanich, R.M.; Miller, K.D.; Lou, J.; Marks, J.D.; Bruckner-Lea, C.J. Quantum dot immunoassays in renewable surface column and 96-well plate formats for the fluorescence detection of botulinum neurotoxin using high-affinity antibodies. Biosens. Bioelectron. 2009, 25, 179–184. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, L.; Pan, D.; Song, S.; Boey, F.Y.; Zhang, H.; Fan, C. Visual cocaine detection with gold nanoparticles and rationally engineered aptamer structures. Small 2008, 4, 1196–1200. [Google Scholar]
- Sozer, N.; Kokini, J.L. Nanotechnology and its applications in the food sector. Trends Biotechnol. 2009, 27, 82–89. [Google Scholar]
- Graveland-Bikkerand, J.F.; de Kruif, C.G. Food Nanotechnology. Trends Food Sci. Technol. 2006, 17, 196–203. [Google Scholar] [CrossRef]
- Augustin, M.A.; Sanguansri, P. Nanostructured materials in the food industry. Adv. Food Nutr. Res. 2009, 58, 183–213. [Google Scholar] [CrossRef]
- Li, F.; Zhang, J.; Cao, X.; Wang, L.; Li, D.; Song, S.; Ye, B.; Fan, C. Adenosine detection by using gold nanoparticles and designed aptamer sequences. Analyst 2009, 134, 1355–1360. [Google Scholar] [CrossRef]
- Jin, X.; Jin, X.; Chen, L.; Jiang, J.; Shen, G.; Yu, R. Piezoelectric immunosensor with gold nanoparticles enhanced competitive immunoreaction technique for quantification of aflatoxin B1. Biosens. Bioelectron. 2009, 24, 2580–2585. [Google Scholar]
- Kaushik, A.; Solanki, P.R.; Pandey, M.K.; Ahmad, S.; Malhotra, B.D. Cerium oxide-chitosan based nanobiocomposite for food borne mycotoxin detection. Appl. Phys. Lett. 2009, 95, 1–3. [Google Scholar]
- Weiss, J.; Takhistov, P.; McClements, J. Functional materials in food nanotechnology. J. Food Sci. 2006, 71, 107–116. [Google Scholar] [CrossRef]
- International Union of Food Science & Technology (IUFoST). Nanotechnology and Food. IUFoST Scientific Information Bulletin. Available online: http://www.iufost.org/iufost-scientific-information-bulletins-sib (accessed on 20 December 2013).
- Hsieh, Y.H.P.; Ofori, J.A. Innovation in food technology for health. Asia Pac. J. Clin. Nutr. 2007, 16, 65–73. [Google Scholar]
- McClements, D.J.; Weiss, J.; Decker, E.A. Emulsion-based delivery systems for lipoliphic bioactive components. J. Food Sci. 2007, 72, 109–124. [Google Scholar]
- Report of Joint FAO/WHO Expert Meeting on the Application of Nanotechnologies in the Food and Agriculture Sectors: Potential Food Safety Implications. Available online: http://www.who.int/foodsafety/fs_management/meetings/nano_june09/en/ (accessed on 16 December 2013).
- Park, H.S.; Ahn, J.; Kwak, H.S. Effect of nano-calcium-enriched milk on calcium metabolism in ovariectomised rats. J. Med. Food 2008, 11, 454–459. [Google Scholar] [CrossRef]
- Martirosyan, A.; Polet, M.; Bazes, A.; Sergent, T.; Schneider, Y.-J. Food Nanoparticles and Intestinal Inflammation: A Real Risk? In Inflammatory Bowel Disease; Szabo, I., Ed.; InTech: Rijeka, Croatia, 2012; Chapter 8; pp. 259–282. [Google Scholar]
- Lai, S.K.; Wang, Y.-Y.; Hanes, J. Mucus-penetrating nanoparticles for drug and gene delivery to mucosal tissues. Adv. Drug Deliv. 2009, 61, 158–171. [Google Scholar] [CrossRef]
- Crater, J.S.; Carrier, R.L. Barrier properties of gastrointestinal mucus to nanoparticle transport. Macromol. Biosci. 2010, 10, 1473–1483. [Google Scholar] [CrossRef]
- Powell, J.J.; Faria, N.; Thomas-McKay, E.; Pele, L.C. Origin and fate of dietary nanoparticles and microparticles in the gastrointestinal tract. J. Autoimmun. 2010, 34, 226–233. [Google Scholar] [CrossRef]
- OECD Guidance Manual for the Testing of Manufactured Nanomaterials: OECD’s Sponsorship Programme. Available online: http://www.oecd.org/science/nanosafety/publicationsintheseriesonthesafetyofmanufacturednanomaterials.htm (accessed on 20 December 2013).
- Bar-Ilan, O.; Albrecht, R.M.; Fako, V.E.; Furgeson, D.Y. Toxicity assessments of multisized gold and silver nanoparticles in zebrafish embryos. Small 2009, 5, 1897–1910. [Google Scholar] [CrossRef]
- Scientific Committee on Emerging and Newly Identified Health Risks (SCENIHR) Opinion on: Risk Assessment of Products of nanotechnologies. Available online: http://ec.europa.eu/health/archive/ph_risk/committees/04_scenihr/docs/scenihr_o_023.pdf (accessed on 20 December 2013).
- FDA Draft Guidance for Industry: Safety of Nanomaterials in Cosmetic Products. Available online: http://www.fda.gov/Cosmetics/GuidanceComplianceRegulatoryInformation/GuidanceDocuments/ucm300886.htm (accessed on 20 December 2013).
- Oberdörster, G.; Maynard, A.; Donaldson, K.; Castranova, V.; Fitzpatrick, J.; Ausman, K.; Carter, J.; Karn, B.; Kreyling, W.; Lai, D.; et al. Principles for characterizing the potential human health effects from exposure to nanomaterials: Elements of a screening strategy. Part. Fibre Toxicol. 2005, 2. [Google Scholar] [CrossRef]
- Yin, H.; Too, H.P.; Chow, G.M. The effect of particle size and surface coating on the cytotoxicity of nickel ferrite. Biomaterial 2005, 26, 5818–5826. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Cytotoxicity suppression and cellular uptake enhancement of surface modified magnetic nanoparticles. Biomaterial 2005, 26, 1565–1573. [Google Scholar] [CrossRef]
- Araujo, L.; Lobenberg, R.; Kreuter, J. Influence of the surfactant concentration on the body distribution of nanoparticles. J. Drug Target. 1999, 6, 373–385. [Google Scholar] [CrossRef]
- Kennedy, D.C.; Tay, L.L.; Lyn, R.K.; Rouleau, Y.; Hulse, J.; Pezacki, J.P. Nanoscale aggregation of cellular beta2-adrenergic receptors measured by plasmonic interactions of functionalized nanoparticles. ACS Nano 2009, 3, 2329–2339. [Google Scholar] [CrossRef]
- Radomski, A.; Jurasz, P.; Alonso-Escolano, D.; Drews, M.; Morandi, M.; Malinski, T.; Radomski, M.W. Nanoparticle-induced platelet aggregation and vascular thrombosis. Brit. J. Pharmacol. 2005, 146, 882–893. [Google Scholar] [CrossRef]
- Chen, X.; Schluesener, H.J. Nanosilver: A nanoproduct in medical application. Toxicol. Lett. 2008, 176, 1–12. [Google Scholar] [CrossRef]
- Lundqvist, M.; Stigler, J.; Elia, G.; Lynch, I.; Cedervall, T.; Dawson, K.A. Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. Proc. Natl. Acad. Sci. USA 2008, 105, 14265–14270. [Google Scholar] [CrossRef]
- Leszczynski, J. Bionanoscience: Nano meets Bio at the interface. Nat. Nanotechnol. 2010, 5, 633–634. [Google Scholar] [CrossRef]
- Monopoli, M.P.; Walczyk, D.; Campbell, A.; Elia, G.; Lynch, I.; Bombelli, F.B.; Dawson, K.A. Physical-chemical aspects of protein corona: Relevance to in vitro and in vivo biological impacts of nanoparticles. J. Am. Chem. Soc. 2011, 133, 2525–2534. [Google Scholar] [CrossRef]
- Adiseshaiah, P.P.; Hall, J.B.; McNeil, S.E. Nanomaterial standards for efficacy and toxicity assessment. Interdiscip. Rev. Nanomed. Nanobiotechnol. 2010, 2, 99–112. [Google Scholar] [CrossRef]
- Ge, C.; Du, J.; Zhao, L.; Wang, L.; Liu, Y.; Li, D.; Yang, Y.; Zhou, R.; Zhao, Y.; Chai, Z.; et al. Binding of blood proteins to carbon nanotubes reduces cytotoxicity. Proc. Natl. Acad. Sci. USA 2011, 108, 16968–16973. [Google Scholar]
- Safi, M.; Courtois, J.; Seigneuret, M.; Conjeaud, H.; Berret, J.F. The effects of aggregation and protein corona on the cellular internalization of iron oxide nanoparticles. Biomaterials 2011, 32, 9353–9363. [Google Scholar] [CrossRef]
- Amiri, H.; Bordonali, L.; Lascialfari, A.; Wan, S.; Monopoli, M.P.; Lynch, I.; Laurent, S.; Mahmoudi, M. Protein corona affects the relaxivity and MRI contrast efficiency of magnetic nanoparticles. Nanoscale 2013. [Google Scholar] [CrossRef]
- Wang, F.; Yu, L.; Monopoli, M.P.; Sandin, P.; Mahon, E.; Salvati, A.; Dawson, K.A. The biomolecular corona is retained during nanoparticle uptake and protects the cells from the damage induced by cationic nanoparticles until degraded in the lysosomes. Nanomedicine 2013, 9, 1159–1168. [Google Scholar] [CrossRef]
- Powell, J.J.; Thoree, V.; Pele, L.C. Dietary microparticles and their impact on tolerance and immune responsiveness of the gastrointestinal tract. Brit. J. Nutr. 2007, 98, S59–S63. [Google Scholar]
- Van der Zande, M.; Vandebriel, R.J.; van Doren, E.; Kramer, E.; Rivera, Z.H.; Serrano-Rojero, C.S.; Gremmer, E.R.; Mast, J.; Peters, R.J.; Hollman, P.C.; et al. Distribution, elimination, and toxicity of silver nanoparticles and silver ions in rats after 28-day oral exposure. ACS Nano 2012, 6, 7427–7442. [Google Scholar] [CrossRef]
- Dekkers, S.; Krystek, P.; Peters, R.J.; Lankveld, D.X.; Bokkers, B.G.; van Hoeven-Arentzen, P.H.; Bouwmeester, H.; Oomen, A.G. Presence and risks of nanosilica in food products. Nanotoxicology 2011, 5, 393–405. [Google Scholar] [CrossRef]
- Frohlich, E.; Roblegg, E. Models for oral uptake of nanoparticles in consumer products. Toxicology 2012, 291, 10–17. [Google Scholar] [CrossRef]
- Luoma, S.N. Silver Nanotechnologies and the Environment: Old Problems or New Challenges? Woodrow Wilson International Center for Scholars: The Project of Emerging Nanotechnologies. 9 September 2008. Available online: http://www.nanotechproject.org/publications/archive/silver/ (accessed on 17 December 2013).
- Woodrow Wilson Institute’s Project of Emerging Nanotechnologies: Analysis of Consumer Products. Available online: http://www.nanotechproject.org/cpi/browse/nanomaterials/silver-nanoparticle/ (accessed on 12 November 2013).
- Emamifar, A.; Kadivar, M.; Shahedi, M.; Solaimanianzad, S. Effect of nanocomposite packaging containing Ag and ZnO on inactivation of Lactobacillus plantarum in orange juice. Food Control 2011, 22, 408–413. [Google Scholar] [CrossRef]
- Fernández, A.; Picouet, P.; Lloret, E. Reduction of the spoilage-related microflora in absorbent pads by silver nanotechnology during modified atmosphere packaging of beef meat. J. Food Prot. 2010, 73, 2263–2269. [Google Scholar]
- Braydich-Stolle, L.; Hussain, S.; Schlager, J.J.; Hofmann, M.C. In vitro cytotoxicity of nanoparticles in mammalian germline stem cells. Toxicol. Sci. 2005, 88, 412–419. [Google Scholar] [CrossRef]
- Yang, L.; Watts, D.J. Particle surface characteristics may play an important role in phytotoxicity of alumina nanoparticles. Toxicol. Lett. 2005, 158, 122–132. [Google Scholar] [CrossRef]
- Hussain, S.M.; Javorina, A.K.; Schrand, A.M.; Duhart, H.M.H.M.; Ali, S.F.; Schlager, J.J. The interaction of manganese nanoparticles with PC-12 cells induces dopamine depletion. Toxicol. Sci. 2006, 92, 456–463. [Google Scholar] [CrossRef]
- Hussain, S.M.; Hess, K.L.; Gearhart, J.M.; Geiss, K.T.; Schlager, J.J. In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol. Vitro 2005, 19, 975–983. [Google Scholar] [CrossRef]
- Martirosyan, A.; Bazes, A.; Schneider, Y.-J. In vitro toxicity assessment of silver nanoparticles in the presence of phenolic compounds—preventive agents against the harmful effect? Nanotoxicology 2014, 8, 573–582. [Google Scholar] [CrossRef]
- Ahamed, M.; Alsalhi, M.S.; Siddiqui, M.K. Silver nanoparticle applications and human health. Clin. Chim. Acta 2010, 411, 1841–1848. [Google Scholar] [CrossRef]
- Trickler, W.J. Silver nanoparticle induced blood–brain barrier inflammation and increased permeability in primary rat brain microvessel endothelial cells. Toxicol. Sci. 2010, 118, 160–170. [Google Scholar] [CrossRef]
- Sharma, H.S.; Hussain, S.; Schlager, J.; Ali, S.F.; Sharma, A. Influence of nanoparticles on blood—brain barrier permeability and brain edema formation in rats. Acta Neurochir. Suppl. 2010, 106, 359–364. [Google Scholar] [CrossRef]
- Sharma, H.S.; Ali, S.F.; Hussain, S.M.; Schlager, J.J.; Sharma, A. Influence of engineered nanoparticles from metals on the blood—brain barrier permeability, cerebral blood flow, brain edema and neurotoxicity. An experimental study in the rat and mice using biochemical and morphological approaches. J. Nanosci. Nanotechnol. 2009, 9, 5055–5072. [Google Scholar] [CrossRef]
- Kawata, K.; Osawa, M.; Okabe, S. In vitro toxicity of silver nanoparticles at noncytotoxic doses to HepG2 human hepatoma cells. Environ. Sci. Technol. 2009, 43, 6046–6051. [Google Scholar] [CrossRef]
- Kim, W.Y.; Kim, J.; Park, J.D.; Ryu, H.Y.; Yu, I.J. Histological study of gender differences in accumulation of silver nanoparticles in kidneys of Fischer 344 rats. J. Toxicol. Environ. Health A 2009, 72, 1279–1284. [Google Scholar] [CrossRef]
- Kim, Y.S.; Kim, J.S.; Cho, H.S.; Rha, D.S.; Kim, J.M.; Park, J.D.; Choi, B.S.; Lim, R.; Chang, H.K.; Chung, Y.H.; et al. Twenty-eight-day oral toxicity, genotoxicity, and gender-related tissue distribution of silver nanoparticles in Sprague-Dawley rats. Inhal. Toxicol. 2008, 20, 575–583. [Google Scholar] [CrossRef]
- Fondevila, M.; Herrer, R.; Casallas, M.C.; Abecia, L.; Ducha, J.J. Silver nanoparticles as potential antimicrobial additive for weaned pigs. Anim. Feed Sci. Technol. 2009, 150, 259–269. [Google Scholar] [CrossRef]
- Cha, K.; Hong, H.W.; Choi, Y.G.; Lee, M.J.; Park, J.H.; Chae, H.K.; Ryu, G.; Myung, H. Comparison of acute responses of mice livers to short-term exposure to nano-sized or micro-sized silver particles. Biotechnol. Lett. 2008, 30, 1893–1899. [Google Scholar] [CrossRef]
- Shukla, R.K.; Sharma, V.; Pandey, A.K.; Singh, S.; Sultana, S.; Dhawan, A. ROS-mediated genotoxicity induced by titanium dioxide nanoparticles in human epidermal cells. Toxicol. Vitro 2011, 25, 231–241. [Google Scholar] [CrossRef]
- Baan, R.; Straif, K.; Grosse, Y.; Secretan, B.; El Ghissassi, F.; Cogliano, V. Carcinogenicity of carbon black, titanium dioxide, and talc. Lancet Oncol. 2006, 7, 295–296. [Google Scholar] [CrossRef]
- Ortlieb, M. White giant or white dwarf? Particle size distribution measurements of TiO2. GIT Lab J. Eur. 2010, 14, 42–43. [Google Scholar]
- Kaida, T.; Kobayashi, K.; Adachi, M.; Suzuki, F. Optical characteristics of titanium oxide interference film and the film laminated with oxides and their applications for cosmetics. J. Cosmet. Sci. 2004, 55, 219–220. [Google Scholar]
- Wang, J.; Zhou, G.; Chen, C.; Yu, H.; Wang, T.; Ma, Y.; Jia, G.; Gao, Y.; Li, B.; Sun, J.; et al. Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration. Toxicol. Lett. 2007, 168, 176–185. [Google Scholar] [CrossRef]
- Wang, J.J.; Sanderson, B.J.; Wang, H. Cyto- and genotoxicity of ultrafine TiO2 particles in cultured human lymphoblastoid cells. Mutat. Res. 2007, 628, 99–106. [Google Scholar] [CrossRef]
- Trouiller, B.; Reliene, R.; Westbrook, A.; Solaimani, P.; Schiestl, R.H. Titanium dioxide nanoparticles induce DNA damage and genetic instability in vivo in mice. Cancer Res. 2009, 69, 8784–8789. [Google Scholar] [CrossRef]
- Wolf, R.; Matz, H.; Orion, E.; Lipozencic, J. Sunscreens–The ultimate cosmetic. Acta Dermatovenerol. Croat 2003, 11, 158–162. [Google Scholar]
- Hagens, W.I.; Oomen, A.G.; de Jong, W.H.; Cassee, F.R.; Sips, A.J. What do we (need to) know about the kinetic properties of nanoparticles in the body? Regul. Toxicol. Pharmacol. 2007, 49, 217–229. [Google Scholar] [CrossRef]
- Hillyer, J.F.; Albrecht, R.M. Gastrointestinal persorption and tissue distribution of differently sized colloidal gold nanoparticles. J. Pharm. Sci. 2001, 90, 1927–1936. [Google Scholar] [CrossRef]
- Lin, C.C.; Lin, W.J. Sun protection factor analysis of sunscreens containing titanium dioxide nanoparticles. J. Food Drug Anal. 2011, 19, 1–8. [Google Scholar]
- Ge, Y.; Schimel, J.P.; Holden, P.A. Identification of soil bacteria susceptible to TiO2 and ZnO nanoparticles. Appl. Environ. Microbiol. 2012, 78, 6749–6758. [Google Scholar] [CrossRef]
- Chawengkijwanich, C.; Hayata, Y. Development of TiO2 powder-coated food packaging film and its ability to inactivate Escherichia coli in vitro and in actual tests. Int. J. Food Microbiol. 2008, 123, 288–292. [Google Scholar] [CrossRef]
- Gelover, S.; Gómez, L.A.; Reyes, K.; Leal, M.T. A practical demonstration ofwater disinfection using TiO2 films and sunlight. Water Res. 2006, 40, 3274–3280. [Google Scholar] [CrossRef]
- Weir, A.; Westerhoff, P.; Fabricius, L.; Hristovski, K.; von Goetz, N. Titanium dioxide nanoparticles in food and personal care products. Environ. Sci. Technol. 2012, 46, 2242–2250. [Google Scholar] [CrossRef]
- Lomer, M.C.; Harvey, R.S.; Evans, S.M.; Thompson, R.P.; Powell, J.J. Efficacy tolerability of a low microparticle diet in a double blind randomized pilot study in Crohn’s disease. Eur. J. Gastroenterol. Hepatol. 2001, 13, 101–106. [Google Scholar] [CrossRef]
- Shi, H.; Magaye, R.; Castranova, V.; Zhao, J. Titanium dioxide nanoparticles: A review of current toxicological data. Part Fibre Toxicol. 2013, 10. [Google Scholar] [CrossRef]
- Dankovic, D.; Kuempel, E.; Wheeler, M. An approach to risk assessment for TiO2. Inhal. Toxicol. 2007, 19, 205–212. [Google Scholar] [CrossRef]
- FDA: Listing of Color Additives Exempt from Certification. In Code of Federal Regulations Title 21—Food and Drugs; U.S. Government Printing Office: Washington, DC, USA, 2002.
- Morimoto, Y.; Kobayashi, N.; Shinohara, N.; Myojo, T.; Tanaka, I.; Nakanishi, J. Hazard assessments of manufactured nanomaterials. J. Occup. Health 2010, 52, 325–324. [Google Scholar] [CrossRef]
- SCCS (Scientific Committee on Consumer Safety), Opinion on Titanium Dioxide (Nano Form). SCCS/1516/13 from 22 July 2013, revised on 22 April 2014. Available online: http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_136.pdf (accessed on 19 May 2014).
- Powell, J.; Harvey, R.; Ashwood, P.; Wolstencroft, R.; Gershwin, M.; Thompson, R. Immune potentiation of ultrafine dietary particles in normal subjects and patients with inflammatory bowel disease. J. Autoimmun. 2000, 14, 99–105. [Google Scholar] [CrossRef]
- Valant, J.; Drobne, D. Biological reactivity of TiO2 nanoparticles assessed by ex vivo testing. Protoplasma 2011, 249, 835–842. [Google Scholar] [CrossRef]
- Dufour, E.K.; Kumaravel, T.; Nohynek, G.J.; Kirkland, D.; Toutain, H. Clastogenicity, photo-clastogenicity or pseudo-photo-clastogenicity: Genotoxic effects of zinc oxide in the dark, in pre-irradiated or simultaneously irradiated Chinese hamster ovary cells. Mutat. Res. 2006, 607, 215–224. [Google Scholar] [CrossRef]
- Yammamoto, O. Influence of particle size on the antibacterial activity of zinc oxide. Int. J. Inorg. Mater. 2001, 3, 643–646. [Google Scholar] [CrossRef]
- Jones, N.; Ray, B.; Ranjit, K.T.; Manna, A.C. Antibacterial activity of ZnO nanoparticle suspensions on a broad spectrum of microorganisms. FEMS Microbiol. Lett. 2008, 279, 71–76. [Google Scholar] [CrossRef]
- Chandramouleeswaran, S.; Mhaske, S.T.; Kathe, A.A.; Varadarajan, P.V.; Prasad, V.; Vigneshwaran, N. Functional behaviour of polypropylene/ZnO-soluble starch nanocomposites. Nanotechnology 2007, 18. [Google Scholar] [CrossRef]
- Moos, P.J; Olszewski, K.; Honeggar, M.; Cassidy, P.; Leachman, S.; Woessner, D.; Cutler, N.S.; Veranth, J.M. Responses of human cells to ZnO nanoparticles: A gene transcription study. Metallomics 2011, 3, 1199–1211. [Google Scholar] [CrossRef]
- Moos, P.J.; Chung, K.; Woessner, D.; Honeggar, M.; Cutler, N.S.; Veranth, J.M. ZnO particulate matter requires cell contact for toxicity in human colon cancer cells. Chem. Res. Toxicol. 2010, 23, 733–739. [Google Scholar] [CrossRef]
- De Berardis, B.; Civitelli, G.; Condello, M.; Lista, P.; Pozzi, R.; Arancia, G.; Meschini, S. Exposure to ZnO nanoparticles induces oxidative stress and cytotoxicity in human colon carcinoma cells. Toxicol. Appl. Pharmacol. 2010, 246, 116–127. [Google Scholar] [CrossRef]
- Kang, T.; Guan, R.; Chen, X.; Song, Y.; Jiang, H.; Zhao, J. In vitro toxicity of different-sized ZnO nanoparticles in Caco-2 cells. Nanosc. Res Lett. 2013, 8. [Google Scholar] [CrossRef]
- Esmaeillou, M.; Moharamnejad, M.; Hsankhani, R.; Tehrani, A.A.; Maadi, H. Toxicity of ZnO nanoparticles in healthy adult mice. Environ. Toxicol. Pharmacol. 2013, 35, 67–71. [Google Scholar] [CrossRef]
- Pasupuleti, S.; Alapati, S.; Ganapathy, S.; Anumolu, G.; Pully, N.R.; Prakhya, B.M. Toxicity of zinc oxide nanoparticles through oral route. Toxicol. Ind. Health 2012, 28, 675–686. [Google Scholar] [CrossRef]
- Vandebriel, R.J.; de Jong, W.H. A review of mammalian toxicity of ZnO nanoparticles. Nanotechnol. Sci. Appl. 2012, 15, 61–71. [Google Scholar] [CrossRef]
- Wang, T.T.; Chai, F.; Fu, Q.; Zhang, L.Y.; Liu, H.Y.; Li, L.; Liao, Y.; Su, Z.; Wang, C.; Duanc, B.; et al. Iniform hollow mesoporous silica nanocages for drug delivery in vitro and in vivo for liver cancer therapy. J. Mater. Chem. 2011, 21, 5299–5306. [Google Scholar] [CrossRef]
- Yuan, L.; Tang, Q.Q.; Yang, D.; Zhang, J.Z.; Zhang, F.Y.; Hu, J.H. Preparation of pH-responsive mesoporous silica nanoparticles and their application in controlled drug delivery. J. Phys. Chem. C 2011, 115, 9926–9932. [Google Scholar] [CrossRef]
- Gao, F.; Li, L.; Liu, T.; Hao, N.; Liu, H.; Tan, L.; Li, H.; Huang, X.; Peng, B.; Yan, C.; et al. Doxorubicin loaded silica nanorattles actively seek tumors with improved anti-tumor effects. Nanoscale 2012, 4, 3365–3372. [Google Scholar] [CrossRef]
- Zhu, M.; Wang, H.X.; Liu, J.Y.; He, H.L.; Hua, X.G.; He, Q.J.; Zhang, L.; Ye, X.; Shi, J. A mesoporous silica nanoparticulate/b-TCP/BG composite drug delivery system for osteoarticular tuberculosis therapy. Biomaterials 2011, 32, 1986–1995. [Google Scholar] [CrossRef]
- OECD Synthetic Amorphous Silica and Silicates. Available online: http://www.chem.unep.ch/irptc/sids/OECDSIDS/Silicates.pdf (accessed on 17 December 2013).
- Regulation (EC) No. 1333/2008 of the European Parliament and of the Council of 16 December 2008 on Food Additives. Official Journal of the European Union 2008. Available online: http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32008R1333 (accessed on 19 May 2014).
- Sergent, J.A.; Paget, V.; Chevillard, S. Toxicity and genotoxicity of nano-SiO2 on human epithelial intestinal HT-29 cell line. Ann. Occup. Hyg. 2012, 56, 622–630. [Google Scholar]
- Napierska, D.; Thomassen, L.; Lison, D.; Martens, J.; Hoet, P. The nanosilica hazard: Another variable entity. Part Fibre Toxicol. 2010, 7. [Google Scholar] [CrossRef]
- So, S.J.; Jang, I.S.; Han, C.S. Effect of micro/nano silica particle feeding for mice. J. Nanosci. Nanotechnol. 2008, 8, 5367–5371. [Google Scholar] [CrossRef]
- Dekkers, S.; Bouwmeester, H.; Bos, P.M.; Peters, R.J.; Rietveld, A.G.; Oomen, A.G. Knowledge gaps in risk assessment of nanosilica in food: Evaluation of the dissolution and toxicity of different forms of silica. Nanotoxicology 2013, 7, 367–377. [Google Scholar]
- Nickols-Richardson, S.M. Nanotechnology: Implications for food and nutrition professionals. J. Amer. Diet. Assn. 2007, 107, 1494–1497. [Google Scholar] [CrossRef]
- Sekhon, B.S. Food nanotechnology–An overview. Nanotechnol. Sci. Appl. 2010, 3, 1–15. [Google Scholar]
- World Health Organization (WHO). Nanotechnology. 2008. Available online: http://www.who.int/entity/foodsafety/fs_management/No_01_nanotechnology_Feb08_en_rev1.pdf (accessed on 19 May 2014).
- Sheth, P.; Delos Santos, N.; Seth, A.; LaRusso, N.; Rao, R. Lipopolysaccharide disrupts tight junctions in cholangiocyte monolayers by c-Scc-, TLR4-, and LPB-dependent mechanism. Amer. J. Physiol. Gastrointest. Liver Physiol. 2007, 293, 308–318. [Google Scholar] [CrossRef]
- Gatti, A.M.; Rivasi, F. Biocompatibility of micro- and nanoparticles. Part I: In liver and kidney. Biomaterials 2002, 23, 2381–2387. [Google Scholar] [CrossRef]
- Gatti, A.M. Biocompatibility of Micro- and Nano-particles in the colon. Part II. Biomaterials 2004, 25, 385–392. [Google Scholar] [CrossRef]
- Powell, J.J.; Ainley, C.C.; Harvey, R.S.; Manson, I.M.; Kendall, M.D.; Sankey, E.A.; Dhillon, A.P.; Thompson, R.P. Characterization of inorganic microparticles in pigment cells of human gut associated lymphoid tissue. Gut 1996, 38, 390–395. [Google Scholar] [CrossRef]
- Powell, J.J.; Harvey, R.S.; Thompson, R.P. Microparticles in Crohn’s disease–Has the dust settled? Gut 1996, 392, 340–341. [Google Scholar]
- Roy, R.; Kumar, S.; Verma, A.K.; Sharma, A.; Chaudhari, B.P.; Tripathi, A.; Das, M.; Dwivedi, P.D. Zinc oxide nanoparticles provide an adjuvant effect to ovalbumin via a Th2 response in Balb/c mice. Int. Immunol. 2014, 26, 159–172. [Google Scholar] [CrossRef]
- Auttachoat, W.; McLoughlin, C.E.; White, K.L., Jr.; Smith, M.J. Route-dependent systemic and local immune effects following exposure to solutions prepared from titanium dioxide nanoparticles. J. Immunotoxicol. 2013. [Google Scholar] [CrossRef]
- Lu, X.; Liu, Y.; Kong, X.; Lobie, P.E.; Chen, C.; Zhu, T. Nanotoxicity: A growing need for study in the endocrine system. Small 2013, 9, 1654–1671. [Google Scholar] [CrossRef]
- Iavicoli, I.; Fontana, L.; Leso, V.; Bergamaschi, A. The effects of nanomaterials as endocrine disruptors. Int. J. Mol. Sci. 2013, 14, 16732–16801. [Google Scholar] [CrossRef]
- Walczyk, D.; Bombelli, F.B.; Monopoli, M.P.; Lynch, I.; Dawson, K.A. What the cell ‘‘sees’’ in bionanoscience. J. Am. Chem. Soc. 2010, 132, 5761–5768. [Google Scholar] [CrossRef]
- Gorbet, M.; Sefton, M. Endotoxin: The uninvited guest. Biomaterials 2005, 26, 6811–6817. [Google Scholar] [CrossRef]
- Magalhães, P.O.; Lopes, A.M.; Mazzola, P.G.; Rangel-Yagui, C.; Penna, T.C.; Pessoa, A., Jr. Methods of endotoxin removal from biological preparations: A review. J. Pharm. Pharm. Sci. 2007, 10, 388–404. [Google Scholar]
- Cho, D.R.; Shanbhag, A.S.; Hong, C.Y.; Baran, G.R.; Goldring, S.R. The role of adsorbed endotoxin in particle-induced stimulation of cytokine release. J. Orthop. Res. 2002, 20, 704–713. [Google Scholar] [CrossRef]
- Regulation (EC) No 1935/2004 of the European Parliament and of the Council of 27 October 2004 on materials and articles intended to come into contact with food and repealing Directives 80/590/EEC and 89/109/EEC. Official Journal of the European Union. Available online: http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32004R1935 (accessed on 19 May 2014).
- EFSA. Guidance on the risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain. EFSA J. 2011, 9, 1–36. [Google Scholar]
- Benn, T.; Cavanagh, B.; Hristovski, K.; Posner, J.D.; Westerhoff, P. The release of nanosilver from consumer products used in the home. J. Environ. Qual. 2010, 39, 1875–1882. [Google Scholar] [CrossRef]
- EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids (CEF). Scientific Opinion on the Safety Evaluation of the Substance, Silver Zeolite A (Silver Zinc Sodium Ammonium Alumino Silicate), Silver Content 2%–5%, for Use in Food Contact Materials. EFSA J. 2011, 9, p. 1999. Available online: http://www.efsa.europa.eu/en/efsajournal/doc/1999.pdf (accessed on 19 May 2014).
- U.S. FDA (Food and Drug Administration). Food Additives Permitted for Direct Addition to Food for Human Consumption, Silver Nitrate and Hydrogen Peroxide. Feder. Regist. 2009, 74, 11476–11478. [Google Scholar]
- Directive 94/36 EC on colors for use in foodstuffs. Off. J. Eur. Commun. 1994, L237, 13–29.
- EFSA (European Food Safety Authority). The Potential Risks Arising from Nanoscience and Nanotechnologies on Food and Feed Safety. Available online: http://www.efsa.europa.eu/en/efsajournal/pub/958.htm (accessed on 17 December 2013).
- COT, COM, COC (Committees on Toxicity, Mutagenicity and Carcinogenicity of Chemicals in Food, Consumer Products and the Environment, Department of Health) Joint Statement on Nanomaterial Toxicology. December 2005. Available online: http://cot.food.gov.uk/cotstatements/cotstatementsyrs/cotstatements2007/cot200701 (accessed on 10 December 2013).
- Regulation (EC) No. 258/97 of the European Parliament and of the Council of 27 January 1997 concerning novel foods and novel food ingredients. Off. J. 1997, 43, pp. 1–7. Available online: http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:31997R0258 (accessed on 19 May 2014).
- Howlett, J.; Edwards, D.G.; Cockburn, P.; Hepburn, P.; Kleiner, J.; Knorr, D.; Kozianowski, G.; Muller, D.; Peijnenburg, A.; Perrin, I.; et al. The safety assessment of novel foods and concepts to determine their safety in use. Int. J. Food Sci. Nutr. 2003, 54, S1–S32. [Google Scholar]
- Constable, A.; Jonas, D.; Cockburn, A.; Davi, A.; Edwards, G.; Hepburn, P.; Herouet-Guicheney, C.; Knowles, M.; Moseley, B.; Oberdorfer, R.; et al. History of safe use as applied to the safety assessment of novel foods and foods derived from genetically modified organisms. Food Chem. Toxicol. 2007, 45, 2513–2525. [Google Scholar] [CrossRef]
- Hoet, P.; Bruske-Hohlfeld, I.; Salata, O. Nanoparticles-known and unkown health risks. J. Nanobiotechnol. 2004, 2, 12–27. [Google Scholar] [CrossRef]
- Wiebert, P.; Sanchez-Crespo, A.; Falk, R.; Philipson, K.; Lundin, A.; Larsson, S.; Mooler, M.; Kreyling, G.; Svartengren, M. No significant translocation of inhaled 35-nm carbon particles to the circulation in humans. Inhal. Toxicol. 2006, 18, 741–747. [Google Scholar] [CrossRef]
- Ballestri, M.; Baraldi, A.; Gatti, A.M.; Furci, L.; Bagni, A.; Loria, P.; Rapaa, M.; Carulli, N.; Albertazzi, A. Liver and kidney foreign bodies granulomatosis in a patient with malocclusion, bruxism, and worn dental prostheses. Gastroenterology 2001, 121, 1234–1238. [Google Scholar] [CrossRef]
- Gallegos Saliner, A.; Burello, E.; Worth, A. Review of Computational Approaches for Predicting the Physicochemical and Biological Properties of Nanoparticles. Available online: http://publications.jrc.ec.europa.eu/repository/handle/111111111/5155 (accessed on 20 December 2013).
- Fubini, B.; Ghiazza, M.; Fenoglia, I. Physico-chemical features of engineered nanoparticles relevant to their toxicity. Nanotoxicology 2010, 4, 347–363. [Google Scholar] [CrossRef]
- Li, M.; Al-Jamal, K.T.; Kostarelos, K.; Reineke, J. Physiologically based pharmacokinetic modelling of nanoparticles. ACS Nano 2010, 4, 6303–6317. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, H.Y.; Ji, Z.X.; Rallo, R.; Xia, T.; Chang, C.H.; Nel, A.; Cohen, Y. Development of structure-activity relationship for metal oxide nanoparticles. Nanoscale 2013, 5, 5644–5653. [Google Scholar] [CrossRef]
- Tong, W.; Hong, H.; Xie, Q.; Shi, L.; Fang, H.; Perkins, R. Assessing QSAR limitations—A regulatory perspective. Curr. Comput.-Aid. Drug Des. 2005, 1, 195–205. [Google Scholar] [CrossRef]
- Dearden, J.C. In silico prediction of drug toxicity. J. Comput. Aid. Mol. Des. 2003, 17, 119–127. [Google Scholar] [CrossRef]
- Fourches, D.; Pu, D.; Tropsha, A. Exploring quantitative nanostructure-activity relationships (QNAR) modeling as a tool for predicting biological effects of manufactured nanoparticles. Comb. Chem. High Throughput Scr. 2011, 14, 217–225. [Google Scholar] [CrossRef]
- Puzyn, T.; Rasulev, B.; Gajewicz, A.; Hu, X.; Dasari, T.P.; Michalkova, A.; Hwang, H.M.; Toropov, A.; Leszczynska, D.; Leszczynski, J. Using nano-QSAR to predict the cytotoxicity of metal oxide nanoparticles. Nat. Nanotechnol. 2011, 6, 175–178. [Google Scholar] [CrossRef]
- Epa, V.C.; Burden, F.R.; Tassa, C.; Weissleder, R.; Shaw, S.; Winkler, D.A. Modeling biological activities of nanoparticles. Nano Lett. 2012, 12, 5808–5812. [Google Scholar] [CrossRef]
- Oberdörster, G. Safety assessment for nanotechnology and nanomedicine: Concepts of nanotoxicology. J. Intern. Med. 2010, 267, 89–105. [Google Scholar] [CrossRef]
- Peters, A. Particulate matter and heart disease: Evidence from epidemiological studies. Toxicol. Appl. Pharmacol. 2005, 207, S477–S482. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Martirosyan, A.; Schneider, Y.-J. Engineered Nanomaterials in Food: Implications for Food Safety and Consumer Health. Int. J. Environ. Res. Public Health 2014, 11, 5720-5750. https://doi.org/10.3390/ijerph110605720
Martirosyan A, Schneider Y-J. Engineered Nanomaterials in Food: Implications for Food Safety and Consumer Health. International Journal of Environmental Research and Public Health. 2014; 11(6):5720-5750. https://doi.org/10.3390/ijerph110605720
Chicago/Turabian StyleMartirosyan, Alina, and Yves-Jacques Schneider. 2014. "Engineered Nanomaterials in Food: Implications for Food Safety and Consumer Health" International Journal of Environmental Research and Public Health 11, no. 6: 5720-5750. https://doi.org/10.3390/ijerph110605720
APA StyleMartirosyan, A., & Schneider, Y.-J. (2014). Engineered Nanomaterials in Food: Implications for Food Safety and Consumer Health. International Journal of Environmental Research and Public Health, 11(6), 5720-5750. https://doi.org/10.3390/ijerph110605720




