Domestic Asbestos Exposure: A Review of Epidemiologic and Exposure Data
Abstract
:1. Introduction
Early Reports of Domestic Exposure
2. Methods
2.1. Epidemiology Review and Analysis
2.2. Exposure Review
3. Results
3.1. Review of Domestic Epidemiologic Studies
3.1.1. Mesothelioma
| Author and Year | Population Studied | Occupation of Worker(s) | Results | Exposure Information |
|---|---|---|---|---|
| Wagner et al. 1960 [1] | 33 Pleural mesothelioma cases in Northwest Cape Province (South Africa) | Crocidolite miners | 25/33 cases had non-occupational exposure (76%). | Nearly exclusively neighborhood exposure |
| Lieben & Pistawka 1967 [14] | 42 Pleural and peritoneal mesothelioma cases in southeast Pennsylvania | Insulation plant workers | 3/42 (7%) cases had domestic exposure: 2 were daughters of insulation plant workers; 1 mother of two insulation plant workers | Amosite and chrysotile |
| Rusby 1968 [15] | Pleural mesothelioma in mother of factory workers | Asbestos factory workers | Mother of 3 daughters who worked in an asbestos factory | Laundered clothing for 1–2 years, 26 years prior; no other asbestos contact |
| Heller et al. 1970 [16] | 10 Pleural mesothelioma cases at Massachusetts General Hospital 1960–1967 | Pipefitter | 1 woman (10%) washed her pipefitter husband’s dusty work clothes; husband had asbestosis | Clothes washing |
| Bittersohl and Ose 1971 [17] (as cited in NIOSH 1995 [7]) | Wife of a chemical plant worker | Chemical plant worker | 1 woman with pleural mesothelioma whose husband was exposed to asbestos insulation at a chemical plant | Clothes washing |
| Champion 1971 [18] | Son of lagger | Lagger | Patient was never occupationally exposed to asbestos; father was a lagger who wore work overalls home; emphysematous changes seen in mother; sister had pleural plaques. | --- |
| Knappmann 1972 [19] (as cited in NIOSH 1995 [7]) | Brother of asbestos worker | Asbestos factory worker | Case report of mesothelioma in a man who lived for several years with his sister who was an asbestos worker | --- |
| Greenberg & Davies 1974 [20] | 246 Pleural and peritoneal mesothelioma cases in England, Wales, Scotland (1967–1968) | Asbestos factory workers | 2/246 (0.8%) with potential domestic exposures: 1 case had husband who worked in asbestos factory; 1 case lived near asbestos factory; parents worked at factory | Cases had 2 and 14 years of exposure, respectively |
| Lillington et al. 1974 [21] | Mesothelioma in husband and wife | Industrial exposure to asbestos | Husband had “industrial exposure”, wife washed his clothes; both were diagnosed with pleural mesothelioma | Clothes washing |
| Milne 1976 [22] | 32 Pleural mesothelioma cases in Victoria, Australia | Asbestos cement factory | 1/32 cases (3%) had domestic exposure; father worked in asbestos cement plant. | --- |
| Edge & Chaudhury 1978 [23] | 50 Mesothelioma cases from Barrow in Furness (British shipbuilding town; 1966–1976) | Shipyard plumber | 1/50 (2%) was married to a shipyard plumber. | Crocidolite |
| Li et al. 1978 [24] | Family in which father was pipe insulator in a shipyard. | Shipyard insulator | Father had asbestosis and lung cancer; wife washed his clothes and had mesothelioma; daughter had mesothelioma. | Clothes washing |
| Epler et al. 1980 [25] | 2 wives of asbestos workers | Asbestos factory workers | Mesothelioma in 2 wives of asbestos workers: 1 husband worked in an asbestos product factory for 23 years; 1 husband worked in an asbestos product factory and had asbestosis and mesothelioma. | --- |
| Vianna et al. 1981 [26] | 288 pleural and peritoneal mesothelioma cases in NY state (1973–1978) | Farmers, fireman | 7/288 (2.4%) cases with potential indirect exposure (1 male, 6 females); 5 females lived with a farmer; 1 lived with a fireman. | --- |
| Martensson et al. 1984 [27] | Two children of an asbestos worker | Foundry worker | Female with no occupational exposure; Father worked at foundry with insulation and hung his clothes where children played; Male, brother of female, grew up in same house and worked as a storekeeper for company supplying shipyard electrical equipment. | Exposure referred to as “slight household asbestos exposure during childhood”. |
| Krousel et al. 1986 [28] | Mother, daughter, and son with pleural mesothelioma | Factory workers | Mother worked as clothing sales person and candle-maker. First husband and second husband worked at lumber/shingle company. Family lived within a mile of lumber/shingle company that used asbestos wrap on pipes. Daughter worked as phone operator, husband was electrician. Son worked in submarine, shipyard, cement pipe maker, power company, and carpenter. | No microscopic evidence of asbestos fibers in son and daughter |
| Li et al. 1989 [29] | Family of asbestos worker | Insulator | Wife of insulator washed worker's laundry, used cloth sacks that were used to transport insulation as child's diapers. Child died of mesothelioma at age 32; mother died at age 49. Uncle who lived with family as teen and was briefly an insulator, developed mesothelioma at age 43. Father died of asbestosis at age 53. | Clothes washing and insulation cloth sacks as diapers. |
| Kane et al. 1990 [30] | 10 Cases of mesothelioma in patients 40 years old and under | Asbestos factory worker, shipyard insulator | Of 10 cases, 5 had household exposure (50%): Case 1: Father delivered asbestos products; Case 2: Father worked at glass factory that made asbestos products; Case 3: Father worked as shipyard pipe insulator; lived 6 km from shipyard; mother had mesothelioma; father had adenocarcinoma; Case 5: Brother-in-law worked in asbestos plant; lived 2 km from asbestos factory; Case 6: Exposed to father's dusty work clothing for one year; older sister developed lung cancer with same exposure. | 1–18 years of exposure |
| Konetzke et al. 1990 (German) [31] | 48 Cases of mesothelioma from the National Cancer Register in East Germany and 19 cases of pleural plaques were investigated for non-occupational exposure to asbestos | --- | 22/48 (46%) cases caused by cleaning by members of the family of working clothes contaminated with asbestos. | Clothes washing |
| Oern et al. 1991 [32] (Norwegian; as cited in NIOSH 1995 [7]) | Sister and husband of asbestos workers | Insulators | Family had 2 brothers, a sister and her husband. All males were insulators; 1 brother had asbestosis, other brother and sister had mesothelioma; woman who cleaned work clothes developed mesothelioma at age 79. | Clothes washing |
| Chellini et al. 1992 [33] | 100 Cases of pleural mesothelioma in Tuscany, Italy (1970–1988) | Construction, plumber in chemical manufacturing | 4/100 (4%) cases identified with “possible domestic” exposure—women whose husbands or members of the family were occupationally exposed (3 in construction and one as a plumber in chemical manufacturing) and who used to wash their spouses’ work clothes; same data also reported by Seniori-Constantini & Chellini 1997 [34]. | Clothes washing |
| Dodoli et al. 1992 [35] | 262 Cases of pleural mesothelioma in Leghorn and La Spezia, Italy (1958–1988) | Shipyard workers, oil refinery worker | 10 (3.8%) women washed their relatives’ work clothes (9 shipyard workers, 1 oil refinery worker). | Clothes washing |
| Giarelli et al. 1992 [36] | 170 Cases of mesothelioma in Trieste, Italy (1968–1987) | Shipyard workers | 5/170 (2.9%) cases had domestic exposure and cleaned the clothes of their husbands who were shipyard workers. | 80% had no AB a; 20% had few AB; Clothes washing. |
| Schneider et al. 1996 [37] | 5 Pleural mesothelioma cases | Insulation mat manufacturing, turbine revision, roofer, asbestos cardboard manufacturing, and insulator. | “Causal relation established between the mesothelioma and inhalation of asbestos fibers while cleaning contaminated work-clothes and shoes”. | 7–23 years of exposure; cleaning clothes and shoes |
| Seniori-Constantini & Chellini 1997 [34] | 335 Pleural mesothelioma cases from registry in Tuscany, Italy (1970–1996) | NR b | 30%–35% of 59 female cases were housewives; Same data source as Chellini et al. 1992 [33]. | NR |
| Rees et al. 1999 [38] | 123 Cases in South Africa | Mining workers | 13/123 cases (11%) noted contaminated clothing as source of exposure, along with working with asbestos or living in mining district. “No subject exclusively exposed to contaminated work clothes brought home”. Three cases were reported to have only exposure to asbestos from contaminated clothing. | Mostly crocidolite and amosite; Contaminated clothing |
| Ascoli et al. 2000 (Italian) [39] | One female mesothelioma case | NR | Domestic exposure, duration of 20 years in an industrial town with a large chemical plant | NR |
| Barbieri et al. 2001 (Italian) [40] | 190 Cases of mesothelioma in Brescia, Italy diagnosed 1980–1999 | Asbestos hauler | 1/190 (0.5%) had domestic exposure; wife of asbestos hauler who washed his clothes | Clothes washing |
| Bianchi et al. 2001 [41] | 557 Malignant mesotheliomas of the pleura diagnosed 1968–2000 in the Trieste-Monfalcone area, Italy | Mainly shipbuilding town | 21/65 (32%) females and 0/492 males had histories of domestic exposure, cleaning clothes of an asbestos exposed worker; includes Giarelli et al. 1992 [36] cases. | 35% of domestic cases analyzed (n = 20) had AB; Clothes washing. |
| Mangone et al. 2002 (Italian) [42] | 323 Pleural and peritoneal mesothelioma cases in Emilia-Romagna, Italy (1996–2001) | NR | 13/325 (4%) were domestically exposed | NR |
| Miller 2005 [43] | 32 Pleural and peritoneal mesothelioma cases gathered from law firms (since 1990) | Shipyard workers, insulators, others. | 15 wives, 11 daughters, 3 sons, 1 sister-in-law, 1 niece, 1 boarder; Occupations of workers included: 13 shipyard workers, 7 insulators, 12 others | NR |
| Bianchi et al. 2007 [44] | 99 Cases in Trieste, Italy (2001–2006) | NR | 5 cases (5%) identified as “home exposure”, where patients had washed asbestos-exposed husbands’ work clothes | Clothes washing |
| Author and Year | Study Design | Population Studied (dates of death/incidence) | Controls/Unexposed | Disease | Fiber Type | Occupation of Worker(s) | Results a |
|---|---|---|---|---|---|---|---|
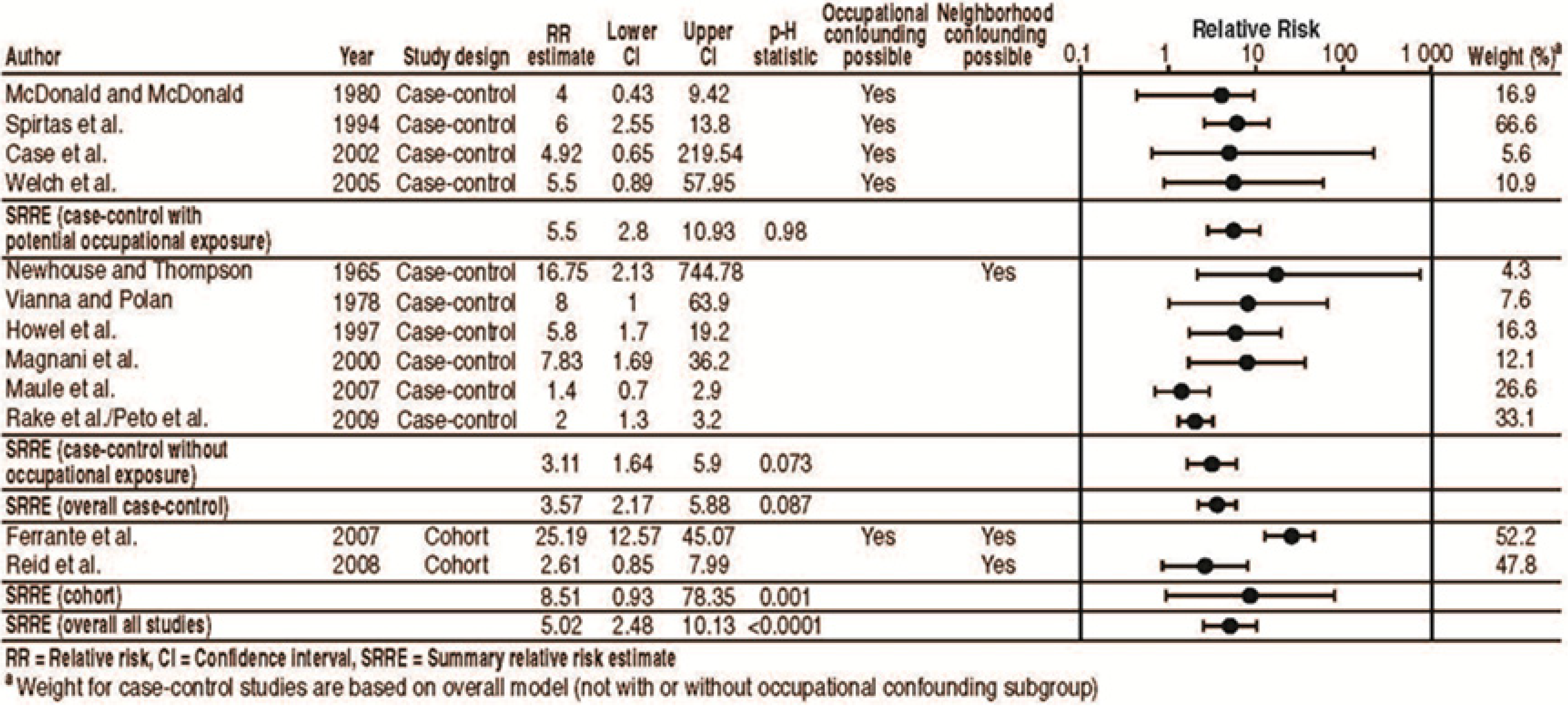
| Newhouse & Thompson 1965 a, b [2,3] | Case-control | 76 cases from London hospital (1956–1963) | 76 “in patient” series (patient in medical and surgical wards of the hospital during early summer 1964) matched by sex and date of birth | PL, PE | Crocidolite, chrysotile, amosite | Spinners, engine room worker, boiler coverer, asbestos factory foreman, docker, railway carriage builder, asbestos factory worker | 9 cases had relative who worked with asbestos (7 pleural, 2 peritoneal) vs. 36 cases with no occupational exposure. 1 of “in patient” series had relative who worked with asbestos vs. 67 with no occupational exposure. Crude OR = 16.75 (95% CI = 2.13–744.78) b,c,d |
| Ashcroft & Heppleston 1970 [45] | Case-control | 22 cases in Tyneside (British shipbuilding town) | 46 hospital controls matched for age and sex, free of malignant disease | PL, PE | NR | Asbestos worker | One case was the widow of an asbestos worker who, for a period of 3 years, had come home with asbestos dust on his hair and shoes. |
| McEwen et al. 1971 [46] | Case-control | 80 cases from Scotland (1950–1967) | 2 sets of hospital controls with coronary artery disease or lung or gastric cancer, matched for age and sex | PL, PE | For one case: “Blue and white” asbestos | For one case: dock worker | “...only a few [cases] had shared a household with relatives who were known to have worked with asbestos. There was no statistical difference with regard to either household or spare-time exposure to asbestos between the three groups [cases, cancer controls, cardiovascular controls]. One individual case, however, was interesting. The husband of one of the female cases had worked regularly with asbestos, both blue and white, as a dock labourer, and quite frequently had come home with asbestos on his overalls. His wife (the case) had washed them at home”. |
| Rubino et al. 1972 [47] | Case-control | 50 cases from Piedmont, Italy (1960–1970) | Patients with same sex, age, and at same institution | PL | NR | Asbestos industry | 3/50 cases had “family exposure” 0/50 controls had “family exposure” Thoracotomy cases: 1 (wife was employed in asbestos industry, no occupational exposure)/18 cases (3 with occupational exposure) 0 (no domestic or occupational exposure)/18 controls (no domestic or occupational exposure) No thoracotomy cases e: 2 (domestic exposure, unclear if could have occupational exposure)/32 cases (3 with occupational exposure) 0/32 controls (1 with occupational exposure) |
| Vianna & Polan 1978 [4] | Case-control | 52 female NY state residents 20+ year old (1967–1977) | 52 controls matched for age sex, race, marital status, county, year of death, and from non-cancer death | PL, PE | NR | Shoemaker, brake lining worker, pipefitter, heat insulation worker, heat electric wire worker, elevator insulation worker | 10 patients had husbands/fathers working in asbestos industry (9 pleural, 1 peritoneal), whereas their matched controls did not. 1 control had husband working in asbestos industry, whereas their matched case did not: RR = 10 (95% CI = 1.42–37.40) e. 8 patients after excluded own occupational exposure, whereas their matched controls did not. 1 control had husband working in asbestos industry, whereas their matched case did not (p < 0.02). OR = 8/1 = 8 (95% CI = 1–63.9) b |
| McDonald & McDonald 1980 [5] | Case-control | 490 fatalities in Canada (1960–1972) and USA (1972) ascertained through 7,400 pathologists | Matched controls with pulmonary metastases from non-pulmonary malignancy by sex, age, year of death, and hospital | PL, PE | Chrysotile (at least 3 cases) and some amosite | Chrysotile production, insulation, or factory work | 2 males, 6 females exposed to dusty clothing of asbestos worker; none among matched controls; 2 controls were exposed, but the paired cases were not (p = 0.08). OR = 4.0 (95% CI = 0.43–9.42) b,e; 5/8 cases were exposed during childhood; 3/8 cases (1 control) were exposed by clothing of a Quebec chrysotile production worker; 5/8 cases (1 control) were exposed by clothing of a insulation/factory worker. |
| Muscat & Wynder 1991 [48] | Case-control | 124 cases entering NY City hospital between 1981–1990 | 267 controls with non-tobacco disease, matched for age, sex, hospital, race, month of interview | M | NR | Auto mechanic | 1/16 women without occupational exposure reported domestic contact with asbestos (one husband was auto mechanic); No information on controls; 1/105 males reported domestic exposure during childhood. |
| Spirtas et al. 1994 [49] | Case-control | 208 cases from Veterans Administration hospital files and Los Angeles county and New York state cancer registries (1975–1980) | 533 controls from death certificates or VA benefit files died of other causes excluding cancer, respiratory disease, suicide, or violence | PL, PE | NR | Asked if “cohabitant ever exposed to asbestos”. Separately asked if cohabitant performed any of 9 activities: (1) brake lining work/repair; (2) furnace/boiler installation/repair; (3) building demolition; (4) plumbing/heating; (5) insulation; (6) shipbuilding yard/repair; (7) elevator installation/repair; (8) production of textiles; (9) production of paper products. | OR for cohabitant ever exposed to asbestos: Men: 13.2 (95% CI = 3.4–54.7) (12 pleural) Women: 3.4 (95% CI = 0.3–61.3) Crude OR = 6 (95% CI = 2.55–13.8) b,e OR for cohabitant performed any of 9 activities: Men: 12.1 (95% CI = 4.6–33.3) (34 pleural, 4 peritoneal) Women: 1.4 (95% CI = 0.3–5.6) |
| Howel et al. 1997 [50] | Case-control | 185 cases of mesothelioma from mesothelioma and cancer registries and post-mortem records in Yorkshire, England (1979–1991) | 159 controls from necropsy records excluded if had mesothelioma, bronchial or ovarian cancer, or circumstances that made gathering information difficult, matched for age, sex, and year of death | PL, PE | Unknown although crocidolite and amosite identified at factory that provoked concern for the study | NR | ORs for para-occupational exposure: Excluding subjects with likely occupational exposure: Likely vs. possible and unlikely 5.6 (95% CI = 1.9–16.5); Likely and possible vs. unlikely 1.8 (95% CI = 0.87–3.6); Excluding those with likely or possible occupational exposure: Likely vs. possible and unlikely 61.7 (95% CI = 3.4–1104); Likely and possible vs. unlikely 5.8 (95% CI = 1.7–19.2). |
| Case et al. 2002 [51] | Case-control | 10 female residents aged ≥50 years of Quebec mining regions identified through hospital records (1970–1989) | 150 controls selected from previously interviewed sample matched on age and area | PL | Chrysotile with some tremolite contamination (Thetford mines) | Chrysotile miners | 10 cases identified: 9 (90%) had lived with one or more asbestos workers (vs. 65% of controls); Never lived with asbestos worker OR = 1 Lived with 1 or 2 workers OR = 3.4 (95% CI = 0.4–30.8); Lived with 3 or more workers OR = 9.0 (95% CI = 0.9–87.4) Crude OR = 4.92 (95% CI = 0.65–219.54) b,e |
| Magnani et al. 2000 [52] | Case-control | 53 mesothelioma cases in Italy, Spain, Switzerland without occupational exposure (1993–1997) | 232 controls from general population and hospitals without occupational exposure | PL | NR | Asbestos industry | OR for those with domestic exposure f and without environmental exposure: 4.92 (95% CI = 1.78–13.6); Probability domestic exposure f (adjusted for environmental exposure): Never exposed OR = 1; Low probability OR = 2.05 (95% CI = 0.83–5.09); Middle or high probability OR = 4.81 (95% CI = 1.77–13.1); Unknown OR = 0.74 (95% CI = 0.22–2.53); Intensity domestic exposure f (adjusted for environmental exposure): Never exposed OR = 1; Low intensity OR = 2.01 (95% CI = 0.84–5.06); Middle intensity OR = 5.68 (95% CI = 1.39–23.3); High intensity OR = 7.83 (95% CI = 1.69–36.2); Unknown OR = 0.75 (95% CI = 0.21–2.69) |
| Welch et al. 2005 [53] | Case-control | 24 male cases treated at Washington Cancer Institute, Washington, DC (1989–2001) | 24 patients with appendical cancer treated at Washington Cancer Institute 1990–2000, matched for age and sex | PE | NR | Same 9 activities specified in Spirtas et al. 1994 [49], except brake lining work is grouped with tire work. | 8/24 (33%) cases cohabitated with persons involved in 9 specified “high-risk-for-asbestos-exposure processes” 2/24 (8%) controls cohabitated with persons involved in 9 processes Crude OR = 5.5 (95% CI = 0.89–57.95) for co-habitating with one of the nine activities.b,e |
| Maule et al. 2007 [54] | Case-control | 103 cases from Casale Monferrato, Italy (1987–1993) | 272 controls matched by birth date, sex, vital status, date of death | PL | Crocidolite and chrysotile | Asbestos cement (AC) workers | OR for relative with AC occupation, adjusted for age, sex, and AC occupation: 2.4 (95% CI = 1.2–4.8); RR for relatives’ AC occupation accounting for age, sex, and domestic (home materials) exposure: Including distance to plant = 1.4 (95% CI = 0.7–2.9) Not including distance to plant = 2.1 (95% CI = 1.0–4.5) Update to Magnani et al. 2001 [55]. |
| Rake et al. 2009/Peto et al. 2009 [56,57] | Case-control | 622 patients in England, Wales and Scotland born since 1925 identified through physician records, cancer research network, and hospital records | 1,420 population controls matched for age and sex | M | Suggests that higher death rate in UK is due to amosite use. | NR | OR living with a potentially exposed worker before 30 years of age: 2.0 (95% CI = 1.3–3.2); Logistic regression results: OR living with a potentially exposed worker before 30 years of age (women): 2.3 (95% CI = 1.5–3.8) OR living with a potentially exposed worker before 30 years of age (men): 1.1 (95% CI = 0.9–1.4) OR living with a high-risk parentor sibling: 1.3 (95% CI = 1.0–1.6) OR living with a high-risk spouse: 2.1 (95% CI = 1.3–3.5) See also tables of Peto et al. 2009. |
| Anderson 1982 [58] | Cohort | 2,218 household contacts of Unarco amosite factory workers first employed between 1941–1945; 663 deaths | State of New Jersey, age and sex-specific | M | Amosite | Amosite insulation factory workers | After 30+ years from onset of exposure, mesothelioma deaths in 3/170 (1.8%) deceased household contacts (2 female and 1 male, all children of workers) c Observed/expected for respiratory cancer was 1.25 for females and 1.7 for males. Same cohort as Joubert et al. 1991 [59], Anderson 1979 [60]; Anderson 1976 [61], Selikoff 1981 [62]. |
| Ferrante et al. 2007 [63] | Cohort | Cohort of 1,780 wives of asbestos cement workers in Casale Monferrato, Italy (deaths from Registrar’s office, incidence from mesothelioma registry) Deaths: 1965–2003Incidence: 1990–2001 | Used age and sex specific rates in Piedmont, Italy for reference | PL, PE | Crocidolite and chrysotile | Asbestos cement workers | Peritoneal cancer SMR = 2.51 (95% CI = 0.52–7.35) Pleural cancer SMR = 18.00 (95% CI = 11.14–27.52) Pleural malignant mesothelioma SIR = 25.19 (95% CI = 12.57–45.07) d,e Update to Magnani et al. 1993 [64]. |
| Reid et al. 2008 [65] | Cohort | Followed 2,552 women and girls who lived in Wittenoom (crocidolite mining town) from 1943 to 1992 and were not involved in mining/milling (1950–2004) | Western Australia female population from 1970–2004 | PL (0 PE) | Crocidolite | Crocidolite miners | The risk of death from mesothelioma was increased, but not significantly, in residents known to have lived with (HR = 2.67, 95% CI = 0.77–9.21) e or washed the clothes of an Australian Blue Asbestos Company asbestos worker (HR = 2.61, 95% CI = 0.85–7.99) d; Update to Hansen et al. 1993 [66]. |
| Bourdès et al. 2000 [10] | Meta-analysis | Five studies: Yazicioglu et al. 1980 [67]; Newhouse & Thompson 1965 [3]; McDonald & McDonald 1980 [5]; Magnani et al. 1993 [64]; Howel et al. 1997 [50] | -- | PL | Chrysotile (McDonald & McDonald 1980 [5]); the rest are amosite or mixed fibers | -- | Meta RR: 8.1 (95% CI = 5.3–12) e |
| Author and Year | Study Design | Population Studied | Comparison Group | Disease | Fiber Type | Occupation of Worker | Results |
|---|---|---|---|---|---|---|---|
| Navratil & Trippe 1972 [68] | Cohort | 114 Blood relatives of asbestos workmen in Czechoslovakia | “General population” of district N with no known exposure | Pleural calcifications | Chrysotile | Asbestos product plant | 4/114 (3.5%) of blood relatives had pleural calcifications. Observed/expected = 4/0.39 |
| Anderson 1982 [58] | Cohort | 679 Household contacts (no occupational asbestos exposure) of amosite factory workers in Paterson, NJ employed between 1941–1954 | 325 urban NJ residence controls matched by sex, age, and residential community without asbestos exposure | Small opacities (≥1/0) and pleural abnormalities (pleural thickening, pleural calcification, pleural plaques) (1971 ILO a) | Amosite | Amosite asbestos factory workers | Household resident during index worker employment period. Cases (N = 679): Small opacities = 114 (17%), Pleural abnormalities = 178 (26%), Both = 239 (35%) (p < 0.001 compared to Controls). Controls (N = 325): Small opacities = 10 (3%), Pleural abnormalities = 6 (2%), Both = 15 (5%) Found statistically significant relationship between duration of exposure and year of first exposure and pleural thickening, pleural calcification, and both together, but not small opacities alone. |
| Ferrante et al. 2007 [63] | Cohort | Cohort of 1,780 wives of asbestos cement workers in Casale Monferrato, Italy (deaths from Registrar’s office: 1965–2003) | Used age and sex specific rates in Piedmont, Italy for reference | Nonmalignant respiratory disease | Crocidolite and chrysotile | Asbestos cement workers | SMR b = 0.86 (0.47–1.45) |
| Kilburn et al. 1986 [69] | Cross-sectional | 274 Wives, 79 sons, and 140 daughters of shipyard workers from Long Beach, CA who started work before 1961. Subjects volunteered and had no occupational exposure. | 1,347 Members of Long Beach Census tract in 1975 and random sample of adult population of Michigan during 1978–1979 without occupational asbestos exposure | Asbestosis and pleural abnormalities (refer to all as asbestosis) (ILO 1980, ≥1/0, and/or presence of pleural thickening or plaques) | NR | Shipyard workers | Asbestosis prevalence: Wives: 11% Sons: 8% Daughters: 2% Comparison populations: Long Beach men: 3.7% Long Beach women: 0.6% Michigan men: 0.5% Michigan women: 0.0% Wives with abnormalities: Pleural only: 39% Parenchymal only: 52% Parenchymal and pleural: 10% 75% of wives with asbestosis had husbands with asbestosis. |
| Sider et al. 1987 [70] | Cross-sectional | 93 wives > 40 years of age of current and retired insulators screened from January to March 1986 at Northwestern Memorial Hospital in Chicago with no occupational exposure | Wives without radiographic abnormalities | Parenchymal opacities and pleural changes according to ILO 1980 | NR | Pipe covers and asbestos removers (insulation workers) | 18/93 (19.4%) demonstrated pleural changes consistent with asbestos exposure: pleural plaques (N = 16, 88.9%), diaphragm plaques (N = 6, 27.8%), pleural calcification (N = 3, 16.6%), and diffuse pleural thickening (N = 1, 5.5%). No parenchymal opacities. Radiographs of the husbands were compared for 17 of the 18 wives with pleural abnormalities. 14 of the husbands (82%) demonstrated more severe pleural involvement than their wives and 6 had parenchymal abnormalities. The remaining 3 wives with more severe changes had at least one household contact in addition to her husband. Only year of initial exposure was statistically different from the comparison group. |
| Peipins et al. 2003 [71] | Cross-sectional | 6,668 Participants ≥ 18 years of age who had lived, worked, or played in Libby, MT for at least 6 months before December 31, 1990 | None | Pleural abnormality (any unilateral or bilateral pleuralcalcification on the diaphragm, chest wall, or other site or any unilateral or bilateralpleural thickening or plaque on the chest wall, diaphragm, or costophrenic angle site, consistent with asbestos-related pleural disease, using the PA view, the oblique views, or a combination of those views) and interstitial abnormality (ILO 1980, ≥1/0). | Libby amphibole (tremolite, actinolite, winchite, richterite) | Vermiculite miners | Lived with W.R. Grace workers (n = 1,376): Pleural abnormality N = 358 (26.0%); Interstitial abnormality N = 17 (1.2%); Does not exclude occupational or non-occupational exposure; Using logistic regression found having been ahousehold contact of a vermiculite miner associated with pleural abnormalities. |
3.1.2. Lung Cancer
3.1.3. Pleural and Interstitial Abnormalities
3.2. Review of Domestic Exposure Studies
3.2.1. Exposures in the Home Environment
3.2.2. Exposures from Clothing
| Author and Year | Population or Task Studied | Asbestos Fiber Type | Reported Exposure Information | |
|---|---|---|---|---|
| Studies reporting measurements of airborne or settled dust in homes of asbestos workers | ||||
| Nicholson et al. 1980 [81] | Homes of chrysotile miners in Copperopolis, California and Baie Verte, Newfoundland | Chrysotile | Homes of miners: 100 to < 5,000 ng/m3 (approx. 0.003–0.15 f/cc a,b) (nc = 13) Homes of non-miners (Baie Verte): 32, 45, 65 ng/m3 | |
| Selikoff and Lee 1978 [82] | Settled dust in asbestos workers’ homes | Amosite | “...small amounts of amosite were found 20–25 years later in the settled dust of asbestos workers' houses from factory operations over the period 1941–1954, and up to 400 yards downwind in the neighboring houses of nonasbestos workers”. | |
| WHO d 1986 [83] | Asbestos miners’ homes | NR | Residences of asbestos miners in South Africa: Mean = 0.006 f/cc (range, 0.002–0.011 f/cc); Para-occupational range: 0.01–1.0 f/cc | |
| Study of clothing and laundering | ||||
| Sawyer et al. 1977 [85] | Asbestos abatement workers | Chrysotile | Mean of personal samples (n = 12): 0.4 f/cc (max = 1.2 f/cc) Mean of area samples: During picking up clothing (n = 4): 0.4 f/cc Loading washer (n = 5): 0.4 f/cc Loading dryer (n = 6): 0.0 f/cc | |
| Exposure simulation studies | ||||
| Jiang et al. 2008 [88] | Unpacking and repacking clutches | Chrysotile | 30 min PCM e -adjusted mean following clothing handling = 0.002 ± 0.002 f/cc (n = 4) Estimated 8 h TWA f= 0.0001 f/cc. | |
| Madl et al. 2008 [89] | Unpacking and repacking brakes | Chrysotile | 30 min PCM-adjusted mean (range) following clothing handling (n = 5): 0.011 f/cc (0.002–0.015 f/cc ) | |
| Madl et al. 2009 [90] | Mechanics performing brake repair on heavy equipment | Chrysotile | 30 min PCM-adjusted mean (range) following clothing handling: For primary worker (n = 2): 0.036 f/cc (0.032–0.039 f/cc) For bystander (n = 2): 0.010 f/cc (0.003–0.018 f/cc) | |
| Mowat et al. 2007 [91] | Roofers removing dried material from laundered clothing | Chrysotile | 30 min PCM-E g mean (n = 12): 0.0017 f/cc (range = 0–0.011 f/cc) Calculated TWAs = 0.003–0.002 f/cc | |
| Weir et al. 2001 [92] | Brake mechanics | Chrysotile | Agitation of operator’s coveralls (30 min) = 0.72 f/cc Background concentration in laboratory ≤ 0.065 f/cc | |
| Lung burden studies | ||||
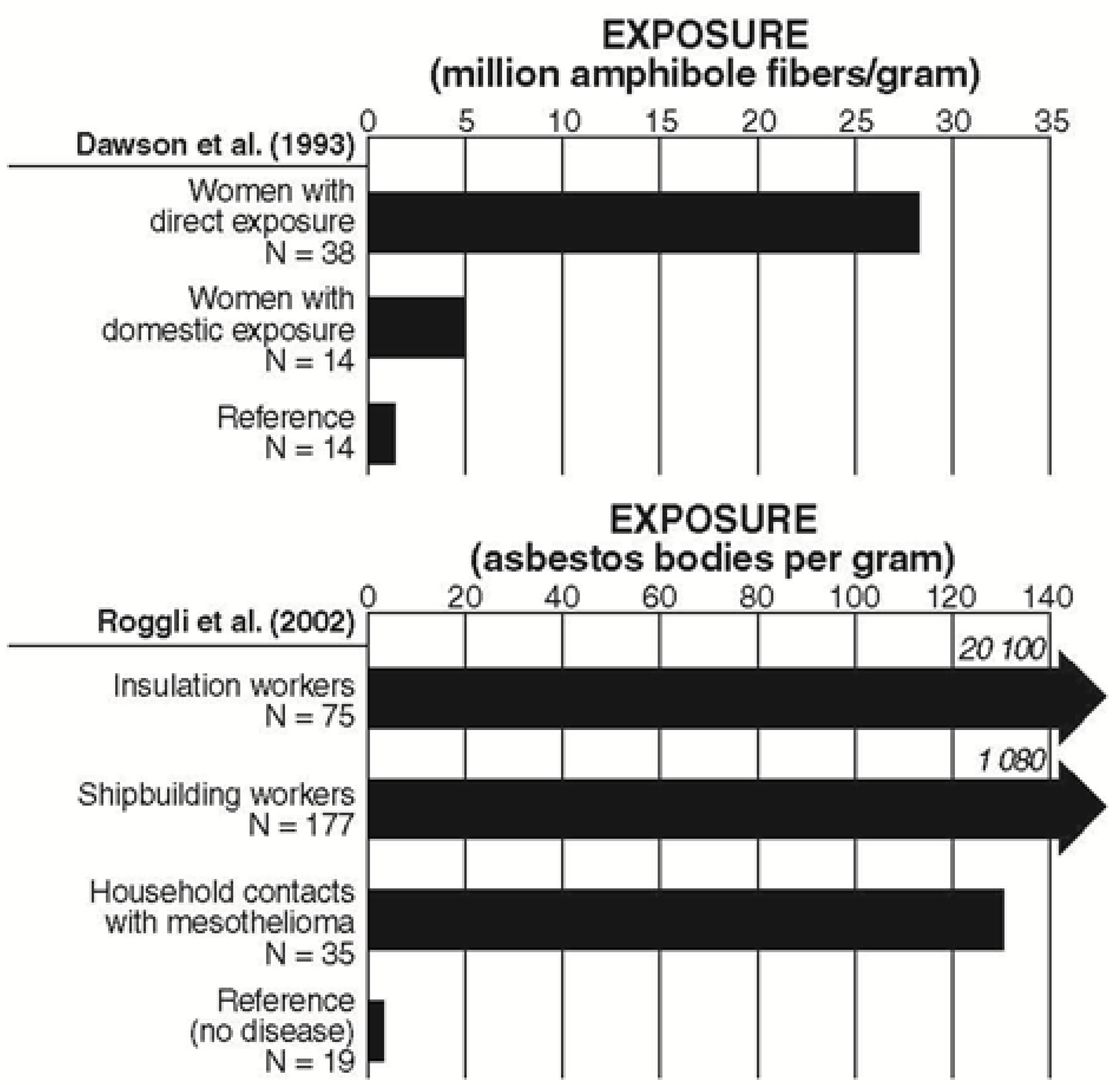
| Dawson et al. 1993 [93] | Women with mesothelioma (n = 170) | Mixed | Women with domestic exposure (n = 14): Total amphiboles = 4.9 × 106 f/g h (range = 0–251) Chrysotile = 12.7 × 106 f/g (range = 0–2506) Control group (n = 31): Total amphiboles = 0.04 × 106 f/g (range = 0–1.0); Unknown = 4.4 × 106 f/g (range = 0–20.1) | |
| Dodson et al. 2003 [94] | Women with mesothelioma (n = 15) | Mixed | 4 women had potential domestic exposure through their father’s/husband’s work; 2/4 had ferruginous bodies (wife of crocidolite worker and wife of laborer/ship scaler/cement worker/etc.); 1 had uncoated amosite and tremolite (daughter of shipyard worker); 1 had uncoated tremolite and no commercial amphiboles (daughter of maintenance worker and wife of shipyard worker/painter/etc. | |
| Giarelli et al. 1992 [36] | Family members of shipyard workers with mesothelioma in Trieste, Italy | --- | 5/170 (2.9%) cases had domestic exposure, cleaned clothes of spouse:80% had no AB i 20% had few AB (1–5 AB/section) | |
| Gibbs et al. 1989 [95] | Mesothelioma cases with para-occupational exposure (n = 13) | Mixed | Mean (range) fiber counts of Group II para-occupational, e.g., wives of males working with asbestos (n = 13): Total: 277.8 (5.6–2507) Amosite: 1.5 (0–6.1) Crocidolite: 31.8 (0–251.1) Chrysotile: 218.9 (1.9–2507) Mean (range) fiber counts of Group V, unexposed (n = 21): Total: 42.5 (0–188.3) Amosite: 0.7 (0–4.6) Crocidolite: 5.5 (0–101.7) Chrysotile: 19.6 (0–76.5) Units in fiber × 106/g dry lung | |
| Gibbs et al. 1989 [95] | Mesothelioma cases without occupational exposure (n = 84). | Mixed | Para-occupational group averages (range) in dry lung: Amosite: 1.5 × 106 f/g (0–6.1) Crocidolite: 31.8 × 106 f/g (0–251) Chrysotile: 218.9 × 106 f/g (1.9–2507) Unexposed group averages (range) in dry lung: Amosite: 0.7 × 106 f/g (0–4.6) Crocidolite: 5.5 × 106 f/g (0–102) Chrysotile: 19.6 × 106 f/g (0–77) | |
| Gibbs et al. 1990 [96] | Mesothelioma cases with para-occupational exposure (n = 10) | Mixed | 9 exposed to their husbands’ work clothes and 1 was the daughter of a man who had died of asbestosis. | |
| Huncharek 1989 [97] | Wife of shipyard machinist | Mixed | Chrysotile: 2.5 × 106 f/g ACj: 0.8 × 106 f/g TAAk: 3.2 × 106 f/g (in dry lung) | |
| Roggli & Longo 1991 [98] | Women whose only known exposure was household contact with an asbestos worker with asbestos-related disease (n = 6) | NR | Household contacts: median = 1,700 AB/g (range, 2–8,200) Uncoated fibers (UF l): median = 24,300 UF/g (range, 17,000–120,000) Normal range: median = 3,100 UF/g (range, 0–20) | |
| Roggli 1992 [99] | Household contacts with mesothelioma (n = 3) | NR | Wife of shipyard insulator: 8,200 AB/g (29 yr exposure) Daughter of insulator: 2,330 AB/g, 17,000 UF/g (25 yr exposure) Wife of shipyard worker: 2 AB/g, 24,300 UF/g (1–2 yr exposure) Normal lungs: 0–22 AB/g, 1,600–5,600 UF/g | |
| Roggli et al. 2002 [100] | Household contacts with mesothelioma (asbestosis confirmed in 8.3%) | Mixed | Mean (range) lung burden in wet lung of household: 130 AB/g (2–14,100) AC: 3,400 f/g (450–116,000) TAA: 5,200 f/g (980–22,400) chrysotile: 1,800 f/g Mean (range) lung burden in wet lung of reference cases: AB: 3 f/g (2–22) AC: <600 f/g (<100–<2,540) TAA: 158,000 f/g (1700–455,000) Chrysotile: <600 f/g (<100–<2,540) | |
3.2.3. Exposure Modeling and Simulation
3.2.4. Lung-Burden Studies
4. Discussion and Conclusions
Acknowledgements
Conflicts of Interest
References
- Wagner, J.C.; Sleggs, C.A.; Marchand, P. Diffuse pleural mesothelioma and asbestos exposure in the north western Cape Province. Brit. J. Ind. Med. 1960, 17, 260–271. [Google Scholar]
- Newhouse, M.L.; Thompson, H. Epidemiology of mesothelial tumors in the London area. Ann. N. Y. Acad. Sci. 1965, 132, 579–588. [Google Scholar]
- Newhouse, M.L.; Thompson, H. Mesothelioma of pleura and peritoneum following exposure to asbestos in the London area. Brit. J. Ind. Med. 1965, 22, 261–269. [Google Scholar]
- Vianna, N.J.; Polan, A.K. Non-occupational exposure to asbestos and malignant mesothelioma in females. Lancet 1978, 1, 1061–1063. [Google Scholar] [CrossRef]
- McDonald, A.D.; McDonald, J.C. Malignant mesothelioma in North America. Cancer 1980, 46, 1650–1656. [Google Scholar]
- Grandjean, P.; Bach, E. Indirect exposures: The significance of bystanders at work and at home. Amer. Ind. Hyg. Assn. J. 1986, 47, 819–824. [Google Scholar]
- National Institute for Occupational Safety and Health (NIOSH). Report to Congress on Workers’ Home Contamination Study Conducted under the Workers’ Family Protection Act; United States; Department of Health and Human Services, Centers for Disease Control and Prevention: Cincinnati, OH, USA, 1995.
- National Institute for Occupational Safety and Health (NIOSH). Protecting Workers’ Families: A Research Agenda; United States Department of Health and Human Services, Centers for Disease Control and Prevention: Cincinnati, OH, USA, 2002.
- Institut National de la Santé de la Recherche Médicale (INSERM). Effets sur la santé des principaux types d’exposition à l’amiante. INSERM: Paris, France, 1997. [Google Scholar]
- Bourdès, V.; Boffetta, P.; Pisani, P. Environmental exposure to asbestos and risk of pleural mesothelioma: Review and meta-analysis. Eur. J. Epidemiol. 2000, 16, 411–417. [Google Scholar]
- Donovan, E.P.; Donovan, B.L.; McKinley, M.A.; Cowan, D.M.; Paustenbach, D.P. Evaluation of take home (para-occupational) exposure to asbestos and disease: A review of the literature. Crit. Rev. Toxicol. 2012, 42, 703–731. [Google Scholar]
- Sutton, A.J.; Jones, D.R.; Abrams, K.R.; Sheldon, T.A.; Song, F. Systematic reviews and meta-analysis: A structured review of the methodological literature. J. Health. Serv. Res. Policy 1999, 4, 49–55. [Google Scholar]
- Andersson, T.; Ahlbom, A. Episheet software: Spreadsheets for the analysis of epidemiologic data. 2004. Available online: http://www.google.com/url?sa=t&rct=j&q=&esrc=s&frm=1&source=web&cd=1&ved=0CC0QFjAA&url=http%3A%2F%2Fkrothman.hostbyet2.com%2FEpisheet.xls&ei=SeTyUZrgPOSqiALi2IDwDQ&usg=AFQjCNHeAwcg2RhXfkGM_b8RdX498JSs9A&sig2=U6MjH7LGvrV4FIowPto19g&bvm=bv.49784469,d.cGE (accessed on 23 October 2013).
- Lieben, J.; Pistawka, H. Mesothelioma and asbestos exposure. Arch. Environ. Health 1967, 14, 559–563. [Google Scholar]
- Rusby, N.L. Pleural manifestations following the inhalation of asbestos in relation to malignant change. J. Roy. Nav. Med. Serv. 1968, 54, 142–148. [Google Scholar]
- Heller, R.M.; Janower, M.L.; Weber, A.L. The radiological manifestations of malignant pleural mesothelioma. Amer. J. Roentgenol. 1970, 108, 53–59. [Google Scholar] [CrossRef]
- Bittersohl, G.; Ose, H. The epidemiology of pleural mesotheliomas. (in German). Z. Gesante Hyg. 1971, 17, 861–864. [Google Scholar]
- Champion, P. Two cases of malignant mesothelioma after exposure to asbestos. Amer. Rev. Resp. Dis. 1971, 103, 821–826. [Google Scholar]
- Knappmann, J. Observations made at post mortem examination of 251 cases of mesothelioma in Hamburg (1958–1968). (in German). Pneumonology 1972, 148, 60–65. [Google Scholar] [CrossRef]
- Greenberg, M.; Davies, T.A.L. Mesothelioma register 1967−1968. Br. J. Ind. Med. 1974, 34, 91–104. [Google Scholar]
- Lillington, G.A.; Jamplis, R.W.; Differding, J.R. Conjugal malignant mesothelioma. N. Engl. J. Med. 1974, 291, 583–584. [Google Scholar]
- Milne, J.E.H. Thirty-two cases of mesothelioma in Victoria, Australia: A retrospective survey related to occupational asbestos exposure. Br. J. Ind. Med. 1976, 33, 115–122. [Google Scholar]
- Edge, J.R.; Choudhury, S.L. Malignant mesothelioma of the pleura in Barrow-in-Furness. Thorax 1978, 33, 26–30. [Google Scholar]
- Li, F.P.; Lokich, J.; Lapey, J.; Neptune, W.B.; Wilkins, E.W. Familial mesothelioma after intense asbestos exposure at home. J. Amer. Med. Assn. 1978, 240. [Google Scholar] [CrossRef]
- Epler, G.R.; Gerald, M.X.; Gaensler, E.A.; Carrington, C.B. Asbestos-related disease from household exposure. Respiration 1980, 39, 229–240. [Google Scholar]
- Vianna, N.J.; Maslowsky, J.; Roberts, S.; Spellman, G.; Patton, R.B. Malignant mesothelioma: Epidemiologic patterns in New York State. N. Y. State J. Med. 1981, 81, 735–738. [Google Scholar]
- Martensson, G.; Larsson, S.; Zettergren, L. Malignant mesothelioma in two pairs of siblings: Is there a hereditary predisposing factor? Eur. J. Resp. Dis. 1984, 65, 179–184. [Google Scholar]
- Krousel, T.; Garcas, N.; Rothschild, H. Familial clustering of mesothelioma: A report on three affected persons in one family. Amer. J. Prev. Med. 1986, 2, 186–188. [Google Scholar]
- Li, F.P.; Drefus, M.G.; Antman, K.H. Asbestos-contaminated nappies and familial mesothelioma. Lancet 1989, 1, 909–910. [Google Scholar]
- Kane, M.J.; Chahinian, A.P.; Holland, J.F. Malignant mesothelioma in young adults. Cancer 1990, 65, 1449–1455. [Google Scholar]
- Konetzke, G.W.; Beck, B.; Mehnert, W.H. Uber berufliche und auberberufliche asbestwirkungen—Remarks on occupational and non-occupational effects of asbestos. Pneumologie 1990, 44, 858–861. [Google Scholar]
- Oern, S.; Odden, S.; Osnes, M. Familial clustering of asbestos-related disease. (in Norwegian). Tidssker Nor Laegeforen 1991, 111, 1099–1101. [Google Scholar]
- Chellini, E.; Fornaciai, G.; Merler, E.; Paci, E.; Costantini, A.S.; Silvestri, S.; Zappa, M.; Buiatti, E. Pleural malignant mesothelioma in Tuscany, Italy (1970−1988): II. Identification of occupational exposure to asbestos. Amer. J. Ind. Med. 1997, 21, 577–585. [Google Scholar]
- Seniori-Costantini, A.; Chellini, E. The experience of the mesothelioma registry of Tuscany in assessing health hazard associated with asbestos exposure. Med. Lav. 1997, 88, 310–315. [Google Scholar]
- Dodoli, D.; del Nevo, M.; Fiumalbi, C.; Iaia, T.E.; Cristaudo, A.; Comba, P.; Viti, C.; Battista, G. Environmental household exposures to asbestos and occurrence of pleural mesothelioma. Amer. J. Ind. Med. 1992, 21, 681–687. [Google Scholar]
- Giarelli, L.; Bianchi, C.; Grandi, G. Malignant mesothelioma of the pleura in Trieste, Italy. Amer. J. Prev. Med. 1992, 22, 521–530. [Google Scholar]
- Schneider, J.; Straif, K.; Woitowitz, H.J. Pleural mesothelioma and household asbestos exposure. Rev. Environ. Health 1966, 11, 65–70. [Google Scholar]
- Rees, D.; Goodman, K.; Fourie, E.; Chapman, R.; Blignaut, C.; Bachmann, M.O.; Myers, J. Asbestos exposure and mesothelioma in South Africa. S. Afr. Med. J. 1990, 89, 627–634. [Google Scholar]
- Ascoli, V.; Fantini, F.; Carnovale, S.C.; Blasetti, F.; Bruno, C.; di Domenicantonio, R.; lo Presti, E.; Pasetto, R.; Nardi, F.; Comba, P. Malignant mesothelioma in the industrial area of Colleferro. Med. Lav. 2000, 91, 547–564. [Google Scholar]
- Barbieri, P.G.; Lombardi, S.; Candela, A.; Pezzotti, C.; Binda, I. Incidence of malignant mesothelioma (1980–1999) and asbestos exposure in 190 cases diagnosed in the population of the Province of Brescia. Med. Lav. 2001, 92, 249–262. [Google Scholar]
- Bianchi, C.; Brollo, A.; Ramani, L.; Bianchi, T.; Giarelli, L. Asbestos exposure in malignant mesothelioma of the pleura: A survey of 557 cases. Ind. Health 2001, 39, 161–167. [Google Scholar]
- Mangone, L.; Romanelli, A.; Campari, C.; Candela, S. Malignant mesothelioma in Emilia-Romagna: Incidence and asbestos exposure. Epidemiol. Prev. 2002, 26, 124–129. [Google Scholar]
- Miller, A. Mesothelioma in household members of asbestos-exposed workers: 32 United States cases since 1990. Amer. J. Ind. Med. 2005, 47, 458–462. [Google Scholar] [CrossRef]
- Bianchi, C.; Bianchi, T.; Tommasi, M. Mesothelioma of the pleura in the province of Trieste. Med. Lav. 2007, 98, 374–380. [Google Scholar]
- Ashcroft, T.; Heppelston, A.G. Mesothelioma and asbestos on tyneside. In Pneumoconiosis: Proceedings of the International Conference, Johannesburg 1969; Shapiro, H.A., Ed.; Oxford University Press: Cape Town, South Africa, 1970; pp. 177–179. [Google Scholar]
- McEwen, J.; Finlayson, A.; Mair, A.; Gibson, A.A. Asbestos and mesothelioma in Scotland: An epidemiological study. Int. Arch. Arbeitsmed. 1971, 28, 301–311. [Google Scholar]
- Rubino, G.F.; Scansetti, G.; Donna, A.; Palestro, G. Epidmilogy of pleural mesotheioma in north-western Italy (Piedmont). Br. J. Ind. Med. 1972, 29, 436–442. [Google Scholar]
- Muscat, J.E.; Wynder, E.L. Cigarette smoking, asbestos exposure, and malignant mesothelioma. Cancer Res. 1991, 51, 2263–2267. [Google Scholar]
- Spirtas, R.; Heineman, E.F.; Bernstein, L.; Beebe, G.W.; Keehn, R.J.; Stark, A.; Harlow, B.L.; Benichou, J. Malignant mesothelioma: Attributable risk of asbestos exposure. Occup. Environ. Med. 1994, 51, 804–811. [Google Scholar]
- Howel, D.; Arblaster, L.; Sinburne, L.; Schweiger, M.; Renvoize, E.; Hatton, P. Routes of asbestos exposure and the development of mesothelioma in an English region. Occup. Environ. Med. 1997, 54, 403–409. [Google Scholar]
- Case, B.W.; Camus, M.; Richardson, L.; Parent, M.E.; Desy, M.; Siemiatycki, J. Preliminary findings for pleural mesothelioma among women in the Quebec chrysotile mining regions. Ann. Occup. Hyg. 2002, 46, S128–S131. [Google Scholar]
- Magnani, C.; Agudo, A.; Gonzalez, C.A.; Andrion, A.; Calleja, A.; Chellini, E.; Dalmasso, P.; Escolar, A.; Hernandez, S.; Lvaldi, C.; et al. Multicentric study on malignant pleural mesothelioma and non-occupational exposure to asbestos. Br. J. Cancer 2000, 83, 104–111. [Google Scholar]
- Welch, L.S.; Acherman, Y.I.Z.; Haile, E.; Sokas, R.K.; Sugarbaker, P.H. Asbestos and peritoneal mesothelioma among college-educated men. Int. J. Occup. Environ. Health 2005, 11, 254–258. [Google Scholar]
- Maule, M.M.; Magnani, C.; Dalmasso, P.; Mirabelli, D.; Merletti, F.; Biggeri, A. Modeling mesothelioma risk associated with environmental asbestos exposure. Environ. Health Perspect. 2007, 115, 1066–1071. [Google Scholar]
- Magnani, C.; Dalmasso, P.; Biggeri, A.; Ivaldi, C.; Mirabelli, D.; Terracini, B. Increased risk of malignant mesothelioma of the pleura after residential or domestic exposure to asbestos: A case-control study in Casale Monferrato, Italy. Environ. Health Perspect. 2001, 109, 915–919. [Google Scholar]
- Rake, C.; Gilham, C.; Hatch, J.; Darnson, A.; Hodgson, J.; Peto, J. Occupational, domestic and environmental mesothelioma risks in the British population: A case-control study. Br. J. Cancer 2009, 100, 1175–1183. [Google Scholar]
- Peto, J.; Rake, C.; Gilham, C.; Hatch, J. Occupational, Domestic and Environmental Mesothelioma Risks in Britain: A Case-Control Study; Health and Safety Executive: Norwich, UK, 2009.
- Anderson, H.A. Family contact exposure. In Proceedings of the World Symposium on Asbestos; Canadian Asbestos Information Centre: Montreal, Canada, 1982; pp. 349–362. [Google Scholar]
- Joubert, L.; Seidman, H.; Selikoff, I.J. Mortality experience of family contacts of asbestos factory workers. Ann. N. Y. Acad. Sci. 1991, 643, 416–418. [Google Scholar]
- Anderson, H.A.; Lilis, R.; Daum, S.M.; Selikoff, I.J. Asbestosis among household contacts of asbestos factory workers. Ann. N. Y. Acad. Sci. 1979, 330, 387–399. [Google Scholar]
- Anderson, H.A.; Lilis, R.; Daum, S.M.; Fischbein, A.S.; Selikoff, I.J. Household-contact asbestos neoplastic risk. Ann. N. Y. Acad. Sci. 1976, 271, 311–323. [Google Scholar]
- Selikoff, I.J. Household risks with inorganic fibers. Bull. N. Y. Acad. Med. 1981, 57, 947–961. [Google Scholar]
- Ferrante, D.; Bertolotti, M.; Todesco, A.; Mirabelli, D.; Terracini, B.; Magnani, C. Cancer mortality and incidence of mesothelioma in a cohort of wives of asbestos workers in Casale Monferrato, Italy. Environ. Health Perspect. 2007, 115, 1401–1405. [Google Scholar]
- Magnani, C.; Terracini, B.; Ivaldi, C.; Botta, M.; Budel, P.; Mancini, A.; Zanetti, R. A cohort study on mortality among wives of workers in the asbestos cement industry in Casale Monferrato, Italy. Br. J. Ind. Med. 1993, 50, 779–784. [Google Scholar]
- Reid, A.; Heyworth, J.; de Klerk, N.H.; Musk, B. Cancer incidence among women and girls environmentally and occupationally exposed to blue asbestos at Wittenoom, Western Australia. Int. J. Cancer 2008, 122, 2337–2344. [Google Scholar]
- Hansen, J.; de Klerk, N.H.; Eccles, J.L.; Musk, A.W.; Hobbs, M.S. Malignant mesothelioma after environmental exposure to blue asbestos. Int. J. Cancer 1993, 54, 578–581. [Google Scholar]
- Yazicioglu, S.; Ilcayto, R.; Balci, K.; Sayli, B.S.; Yorulmaz, B. Pleural calcification, pleural mesotheliomas, and bronchial cancers caused by tremolite dust. Thorax 1980, 35, 564–569. [Google Scholar]
- Navratil, M.; Trippe, F. Prevalence of pleural calcification in persons exposed to asbestos dust, and in the general population in the same district. Environ. Res. 1972, 5, 210–216. [Google Scholar]
- Kilburn, K.H.; Warshaw, R.; Thornton, J.C. Asbestos diseases and pulmonary symptoms and signs in shipyard workers and their families in Los Angeles. Arch. Int. Med. 1986, 146, 2213–2220. [Google Scholar]
- Sider, L.; Holland, E.A.; Davis, T.M.; Cugell, D.W. Changes on radiographs of wives of workers exposed to asbestos. Radiology 1987, 164, 723–726. [Google Scholar]
- Peipins, L.A.; Lewin, M.; Campolucci, S.; Lybarger, J.A.; Miller, A.; Middleton, D.; Weis, C.; Spence, M.; Black, B.; Kapil, V. Radiographic abnormalities and exposure to asbestos-contaminated vermiculite in the community of Libby, Montana, USA. Environ. Health Perspect. 2003, 111, 1753–1759. [Google Scholar]
- Hodgson, J.T.; Darnton, A. The quantitative risks of mesothelioma and lung cancer in relation to asbestos exposure. Ann. Occup. Hyg. 2000, 44, 565–601. [Google Scholar]
- Yarborough, C.M. Chrysotile as a cause of mesothelioma: An assessment based on epidemiology. Crit. Rev. Toxicol. 2006, 36, 165–187. [Google Scholar]
- Berman, W.D.; Crump, K.S. A meta-analysis of asbestos-related cancer risk that addresses fiber size and mineral type. Crit. Rev. Toxicol. 2008, 38, S49–S73. [Google Scholar]
- Berman, W.D.; Crump, K.S. Update of potency factors for asbestos-related lung cancer and mesothelioma. Crit. Rev. Toxicol. 2008, 38, S1–S47. [Google Scholar]
- Kiviluoto, T. Pleural plaques and asbestos: Further observations on endemic and other nonoccupational asbestosis. Ann. N. Y. Acad. Sci. 1965, 132, 235–239. [Google Scholar]
- Dalquen, P.; Hinz, I.; Dabbert, A.F. Pleural plaques, asbestosis and exposure to asbestos: An epidemiological study from the Hamburg Area. Pneumonologie 1970, 143, 23–42. [Google Scholar]
- Ebihara, I. Asbestos related pulmonary disorders in Japan: Occupational exposure, contact in the home and air pollutional exposure to asbestos in Japanese urban districts. Rodo Kagaku (Journal of Science of Labour—Japanese Edition) 1981, 57, 363–396. [Google Scholar]
- Lander, F.; Viskum, B. The occurrence of benign pulmonary changes in the spouses of previously employed asbestos workers. Ugeskr. Laeg. 1985, 147, 1805–1806. [Google Scholar]
- Bianchi, C.; Brollo, A.; Ramani, L.; Berte, R. Exposure to asbestos in Monfalcone, Italy. A necropsy-based study. In Autopsy in Epidemiology and Medical Research; Ribloi, E., Delendi, M., Eds.; International Agency for Research on Cancer (IARC): Lyon, France, 1991; pp. 127–140. [Google Scholar]
- Nicholson, W.J.; Rohl, A.N.; Weisman, I.; Selikoff, I.J. Environmental asbestos concentrations in the United States. In Biological Effects of Mineral Fibres; Wagner, J.C., Ed.; International Agency for Research on Cancer (IARC): Lyon, France, 1980; Volume 2, pp. 823–827. [Google Scholar]
- Selikoff, I.J.; Lee, D.H.K. Asbestos and Disease; Academic Press: New York, NY, USA, 1978. [Google Scholar]
- World Health Organization (WHO). Asbestos and Other Natural Mineral Fibres; World Health Organisation: Geneva, Swizerland, 1986.
- Committee on Nonoccupational Health Risks of Asbestiform Fibers; Board on Toxicology and Environmental Health Hazards; Commission on Life Sciences; National Research Council (NRC). Asbestiform. Fibers: Nonoccupational Health Risks; National Academy Press: Washington, DC, USA, 1984.
- Sawyer, R.N. Asbestos exposure in a Yale building. Environ. Res. 1977, 13, 146–169. [Google Scholar]
- Seixas, N.; Ordin, D. Health Hazard Evaluation Report; National Institute for Occupational Safety and Health (NIOSH): Cincinnati, OH, USA, 1986.
- Driscoll, R.J.; Elliott, L.J. Health Hazard Evaluation Report; National Institute for Occupational Safety and Health (NIOSH): Trenton, MI, USA, 1990.
- Jiang, G.C.T.; Madl, A.K.; Ingmundson, K.J.; Murbach, D.M.; Fehling, K.A.; Paustenbach, D.J.; Finley, B.L. A study of airborne chrysotile concentrations associated with handling, unpacking, and repacking boxes of automobile clutch discs. Regul. Toxicol. Pharmacol. 2008, 51, 87–97. [Google Scholar]
- Madl, A.K.; Scott, L.L.; Murbach, D.M.; Fehling, K.A.; Finley, B.L.; Paustenbach, D.J. Exposure to chrysotile asbestos associated with unpacking and repacking boxes of automobile brake pads and shoes. Ann. Occup. Hyg. 2008, 52, 463–479. [Google Scholar]
- Madl, A.K.; Gaffney, S.H.; Balzer, J.L.; Paustenbach, D.J. Airborne asbestos concentrations associated with heavy equipment brake removal. Ann. Occup. Hyg. 2009, 53, 839–857. [Google Scholar]
- Mowat, F.; Weidling, R.; Sheehan, P. Simulation tests to assess occupational exposure to airborne asbestos from asphalt-based roofing products. Ann. Occup. Hyg. 2007, 51, 451–462. [Google Scholar]
- Weir, F.W.; Tolar, G.; Meraz, L.B. Characterization of vehicular brake service personnel exposure to airborne asbestos and particulate. Appl. Occup. Environ. Hyg. 2001, 16, 1139–1146. [Google Scholar]
- Dawson, A.; Gibbs, A.R.; Pooley, F.D.; Griffiths, D.M.; Hoy, J. Malignant mesothelioma in women. Thorax 1993, 48, 269–274. [Google Scholar]
- Dodson, R.F.; O'Sullivan, M.; Brooks, D.R.; Hammar, S.P. Quantitative analysis of asbestos burden in women with mesothelioma. Amer. J. Ind. Med. 2003, 43, 188–195. [Google Scholar]
- Gibbs, A.R.; Jones, J.S.P.; Pooley, F.D.; Griffiths, D.M.; Wagner, J.C. Non-occupational malignant mesotheliomas. In Non-Occupational Exposure to Mineral Fibres; Bignon, J., Peto, J., Saracci, R., Eds.; International Agency for Research on Cancer: Lyon, France, 1989; pp. 219–228. [Google Scholar]
- Gibbs, A.R.; Griffiths, D.M.; Pooley, F.D.; Jones, J.S.P. Comparisons of fibre types and size distributions in lung tissues of paraoccupational and occupational cases of malignant mesothelioma. Br. J. Ind. Med. 1990, 47, 621–626. [Google Scholar]
- Huncharek, M.; Capotorto, J.V.; Muscat, J. Domestic asbestos exposure, lung fibre burden, and pleural mesothelioma in a housewife. Br. J. Ind. Med. 1989, 46, 354–355. [Google Scholar]
- Roggli, V.L.; Longo, W.E. Mineral fiber content of lung tissue in patients with environmental exposures: Household contacts vs. building occupants. Ann. N. Y. Acad. Sci. 1991, 643, 511–518. [Google Scholar]
- Roggli, V.L. Mineral fiber content of lung tissue in patients with malignant mesothelioma. In Malignant Mesothelioma; Henderson, D.W., Shilkin, K.B., Langlois, S.L., Witaker, D, Eds.; Hemisphere Publishing Corp.: New York, NY, USA, 1992; pp. 201–222. [Google Scholar]
- Roggli, V.L.; Sharma, A.; Butnor, K.J.; Sporn, T.; Vollmer, R.T. Malignant mesothelioma and occupational exposure to asbestos: A clinicopathological correlation of 1445 cases. Ultrastruct. Pathol. 2002, 26, 55–65. [Google Scholar]
- Sheehy, J.W.; Cooper, T.C.; O’Brien, D.M.; McGlothlin, J.D.; Froehlich, P.A. Control. of Asbestos Exposure During Brake Drum Service; U.S. National Institute for Occupational Safety and Health: Cincinnati, OH, USA, 1989.
- Roggli, V.L. Asbestos bodies and nonasbestos ferruginous bodies. In Pathology of Asbestos-associated Diseases, 2nd ed.; Oury, T.D., Sporn, T.A., Eds.; Springer-Verlag: New York, NY, USA, 2004; pp. 34–70. [Google Scholar]
- Camus, M.; Siemiatycki, J.; Meek, B. Nonoccupational exposure to chrysotile asbestos and the risk of lung cancer. N. Engl. J. Med. 1998, 338, 1565–1571. [Google Scholar] [CrossRef]
- American Thoracic Society (ATS). Diagnosis and initial management of nonmalignant diseases related to asbestos. Amer. J. Respir. Crit. Care Med. 2004, 170, 691–715. [Google Scholar]
- Paustenbach, D.J.; Richter, R.O.; Finley, B.L.; Sheehan, P.J. An evaluation of the historical exposures of mechanics to asbestos in brake dust. Appl. Occup. Environ. Hyg. 2003, 18, 786–804. [Google Scholar]
- Finley, B.L.; Richter, R.O.; Mowat, F.S.; Mlynarek, S.; Paustenbach, D.J.; Warmerdam, J.M.; Sheehan, P.J. Cumulative asbestos exposure for US automobile mechanics involved in brake repair (circa 1950s−2000). J. Expo. Sci. Enviro. Epidemiol. 2007, 17, 644–655. [Google Scholar]
- Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Asbestos; Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2001.
- Hessel, P.A.; Teta, M.J.; Goodman, M.; Lau, E. Mesothelioma among brake mechanics: An expanded analysis of a case-control study. Risk Anal. 2004, 24, 547–552. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goswami, E.; Craven, V.; Dahlstrom, D.L.; Alexander, D.; Mowat, F. Domestic Asbestos Exposure: A Review of Epidemiologic and Exposure Data. Int. J. Environ. Res. Public Health 2013, 10, 5629-5670. https://doi.org/10.3390/ijerph10115629
Goswami E, Craven V, Dahlstrom DL, Alexander D, Mowat F. Domestic Asbestos Exposure: A Review of Epidemiologic and Exposure Data. International Journal of Environmental Research and Public Health. 2013; 10(11):5629-5670. https://doi.org/10.3390/ijerph10115629
Chicago/Turabian StyleGoswami, Emily, Valerie Craven, David L. Dahlstrom, Dominik Alexander, and Fionna Mowat. 2013. "Domestic Asbestos Exposure: A Review of Epidemiologic and Exposure Data" International Journal of Environmental Research and Public Health 10, no. 11: 5629-5670. https://doi.org/10.3390/ijerph10115629
APA StyleGoswami, E., Craven, V., Dahlstrom, D. L., Alexander, D., & Mowat, F. (2013). Domestic Asbestos Exposure: A Review of Epidemiologic and Exposure Data. International Journal of Environmental Research and Public Health, 10(11), 5629-5670. https://doi.org/10.3390/ijerph10115629




