Bacterial Diversity and Bioprospecting for Cold-Active Hydrolytic Enzymes from Culturable Bacteria Associated with Sediment from Nella Fjord, Eastern Antarctica
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of Cold-Adapted Bacteria from Sandy Marine Sediment from Nella Fjord
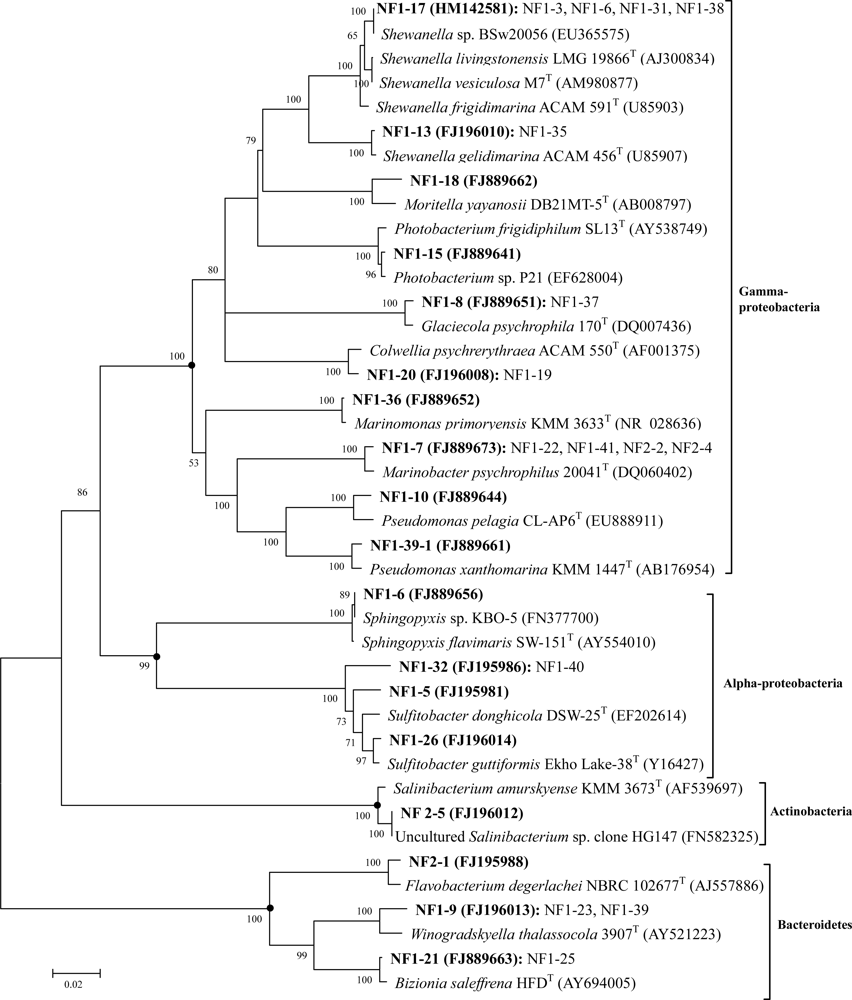
2.2. Phylogenetic Diversity
2.3. Hydrolytic Enzyme Activities of the Isolates
3. Experimental Section
3.1. Sediment Sample
3.2. Isolation of Bacterial Strains
3.3. 16S rDNA Amplification, Sequencing
3.4. Phylogenetic Analysis
3.5. Growth Temperature Range, Sodium Chloride Tolerance and Seawater Requirement
3.6. Hydrolase Activities
4. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References
- Melnikov, IA. Life siences. In RUSSIA National Report to SCAR for Year: 2008; Leitchenkov, GL, Troshichev, OA, Melnikov, IA, Eds.; Russian Committee on Antarctic Research: Moscow, Russia, 2008; p. 10. [Google Scholar]
- Tang, S; Kang, J; Zhou, S; Li, Z. Sea ice characteristics between the middle Weddell Sea and the Prydz Bay, Antarctica during the austral summer of 2003. Acta Oceanol. Sin 2005, 24, 9–15. [Google Scholar]
- Tang, S; Qin, D; Ren, J. Structure, salinity and isotopic composition of multi-year landfast sea ice in Nella Fjord, Antarctica. Cold Reg Sci Technol 2007, 49, 170–177. [Google Scholar]
- He, J; Chen, B. Seasonal change of ice algal and phytoplankton assemblages in the Nella Fjord near Zhongshan Station, East Antarctica. Chin J Polar Sci 2000, 12, 113–122. [Google Scholar]
- Thomson, PG; McMinn, A; Kiessling, I; Watson, M; Goldworthy, PM. Composition and succession of dinoflagellates and chrysophytes in the upper fast ice of Davis Station, East Antarctica. Polar Biol 2006, 29, 337–345. [Google Scholar]
- Sirenko, BI; Gagayev, SYu; Dzhurinsky, VP. Hydrobiological Research Activities in the Nella Fjord, Prydz Bay; IPY-2007/08 NEWS, N 9–10; November/December 2007; Russian Federation National Research Center of Arctic and Antarctic Research Institute (AARI): Petersburg, Russia, 2007; pp. 28–29.
- Melnikov, IA. Life siences. In RUSSIA National Report to SCAR for Year: 2009; Klepikov, A, Leitchenkov, G, Melnikov, IA, Eds.; Russian Committee on Antarctic Research: Moscow, Russia, 2010; pp. 9–10. [Google Scholar]
- Zhang, XY; Zhang, YJ; Yu, Y; Li, HJ; Gao, ZM; Chen, XL; Chen, B; Zhang, YZ. Neptunomonas antarctica sp. nov., isolated from marine sediment. Int J Syst Evol Microbiol 2010, 60, 1958–1961. [Google Scholar]
- Bowman, JP; McCammon, SA; Gibson, JAE; Robertson, L; Nichols, PD. Prokaryotic Metabolic Activity and Community Structure in Antarctic Continental Shelf Sediments. Appl Environ Microbiol 2003, 69, 2448–2462. [Google Scholar]
- Helmke, E; Weyland, H. Psychrophilic versus psychrotolerant bacteria-occurrence and significance in polar and temperate marine habitats. Cell Mol Biol 2004, 50, 553–556. [Google Scholar]
- Huston, AL. Biotechnological Aspects of Cold-Adapted Enzymes. In Psychrophiles: From Biodiversity to Biotechnology; Margesin, R, Schinner, F, Max, J-C, Gerday, C, Eds.; Springer-Verlag: Berlin, Germany, 2008; pp. 347–363. [Google Scholar]
- Morita, RY. Psychrophilic bacteria. Bacteriol Rev 1975, 39, 44–167. [Google Scholar]
- Wayne, LG; Brenner, DJ; Colwell, RR; Grimont, PAD; Kandler, O; Krichevsky, MI; Moore, LH; Moore, WEC; Murray, RGE; Stackebrandt, E; Starr, MP; Trüper, HG. Report of the ad hoc committee on reconciliation of approaches to bacterial systematics. Int J Syst Bacteriol 1987, 37, 463–464. [Google Scholar]
- Stackebrandt, E; Goebel, BM. Taxonomic note: a place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. Int J Syst Bacteriol 1994, 44, 846–849. [Google Scholar]
- Balcazar, JL; Lee, NM; Pintado, J; Planas, M. Phylogenetic characterization and in situ detection of bacterial communities associated with seahorses (Hippocampus guttulatus) in captivity. Syst Appl Microbiol 2010, 33, 71–77. [Google Scholar]
- Srinivas, TNR; Nageswara Rao, SSS; Vishnu Vardhan Reddy, P; Pratibha, MS; Sailaja, B; Kavya, B; Hara Kishore, K; Begum, Z; Singh, SM; Shivaji, S. Bacterial Diversity and Bioprospecting for Cold-Active Lipases, Amylases and Proteases, from Culturable Bacteria of Kongsfjorden and Ny-Ålesund, Svalbard, Arctic. Curr Microbiol 2009, 59, 537–547. [Google Scholar]
- Staley, JT; Gosink, JJ. Poles apart: biodiversity and biogeography of sea ice bacteria. Ann Rev Microbiol 1999, 53, 189–215. [Google Scholar]
- Zeng, Y; Zheng, T; Yu, Y; Chen, B; He, J. Relationships between Arctic and Antarctic Shewanella strains evaluated by a polyphasic taxonomic approach. Polar Biol 2010, 33, 531–541. [Google Scholar]
- Fox, GE; Wisotzkey, JD; Jurtshuk, P, Jr. How close is close: 16S rRNA sequence identity may not be sufficient to guarantee species identity. Int J Syst Bacteriol 1992, 42, 166–170. [Google Scholar]
- Chrost, RJ. Environmental control of the synthesis and activity of aquatic microbial ectoenzymes. In Microbial Enzymes in Aquatic Environments; Chrost, RJ, Ed.; Springer-Verlag: New York, NY, USA, 1991; pp. 84–95. [Google Scholar]
- Hoppe, HG. Microbial extracellular enzyme activity: a new key parameter in aquatic ecology. In Microbial Enzymes in Aquatic Environments; Chrost, RJ, Ed.; Springer-Verlag: New York, NY, USA, 1991; pp. 60–83. [Google Scholar]
- Suzuki, MT; Rappé, MS; Haimberger, ZW; Winfield, H; Adair, N; Strobel, J; Giovannoni, SJ. Bacterial diversity among small-subunit rRNA Gene clones and cellular isolates from the same seawater sample. Appl Environ Microbiol 1997, 63, 983–989. [Google Scholar]
- Weisburg, WG; Burns, SM; Pelletier, DA; Lane, DJ. 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 1991, 173, 697–703. [Google Scholar]
- Chun, J; Lee, JH; Jung, Y; Kim, M; Kim, S; Kim, BK; Lim, YW. EzTaxon: a web-based tool for the identification of prokaryotes based on 16S ribosomal RNA gene sequences. Int J Syst Evol Microbiol 2007, 57, 2259–2261. [Google Scholar]
- National Center for Biotechnology Information. Available online: http://www.ncbi.nlm.nih.gov/ (accessed on 16 December 2010).
- Thompson, JD; Gibson, TJ; Plewniak, F; Jeanmougin, F; Higgins, DG. The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 1997, 25, 4876–4882. [Google Scholar]
- Hall, TA. BioEdit: a user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acids Symp Ser 1999, 41, 95–98. [Google Scholar]
- Saitou, N; Nei, M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 1987, 4, 406–425. [Google Scholar]
- Tamura, K; Dudley, J; Nei, M; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 2007, 24, 1596–1599. [Google Scholar]
- Smibert, RM; Krieg, NR. Phenotypic characterization. In Methods for General and Molecular Bacteriology; Gerhardt, P, Murray, RGE, Wood, WA, Krieg, NR, Eds.; American Society for Microbiology: Washington, DC, USA, 1994; pp. 607–654. [Google Scholar]
- Kouker, G; Jaeger, KE. Specific and sensitive plate assay for bacterial lipases. Appl Environ Microbiol 1987, 53, 211–213. [Google Scholar]
- West, PA; Colwell, RR. Identification and classification of the Vibrionaceae—an overview. In Vibrios in the Environment; Colwell, RR, Ed.; John Wiley: Chichester, UK, 1984; pp. 285–363. [Google Scholar]
- Edberg, SC; Trepeta, RW; Kontnick, CM; Torres, AR. Measurement of active constitutive beta-D-glucosidase (esculinase) in the presence of sodium desoxycholate. J Clin Microbiol 1985, 21, 363–365. [Google Scholar]
- Teather, RM; Wood, PJ. Use of Congo red-polysaccharide interactions in enumeration and characterization of cellulolytic bacteria from bovine rumen. Appl Environ Microbiol 1982, 43, 777–780. [Google Scholar]
| Genus | Strain | Temperature range (°C) | NaCl tolerance (%) | Seawater requirement † | Hydrolase activities * | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Proteae | Esterase | Lipase | Chitinase | Amylase | β-Glucosidase | β-Galactosidase | |||||
| Alphaproteobacteria | |||||||||||
| Sphingopyxis | NF1-6 | 0–20 | 1–6 | + | − | + | − | − | − | − | − |
| Sulfitobacter | NF1-5 | 0–25 | 1–5 | + | − | + | + | − | − | − | − |
| NF1-26 | 0–25 | 1–5 | + | + | + | + | − | − | − | − | |
| NF1-32 | 0–25 | 1–5 | + | − | − | − | − | − | − | − | |
| NF1-40 | 0–25 | 1–5 | + | − | − | − | − | − | − | − | |
| Gammaproteobacteria | |||||||||||
| Colwellia | NF1-19 | 0–25 | 1–5 | + | − | − | + | − | + | − | − |
| NF1-20 | 0–25 | 1–5 | + | + | − | − | − | − | + | − | |
| Glaciecola | NF1-8 | 0–20 | 1–6 | − | − | + | − | − | − | − | − |
| NF1-37 | 0–20 | 1–6 | − | − | − | + | − | − | + | − | |
| Marinomona | NF1-36 | 0–30 | 0–6 | − | − | − | − | − | − | − | + |
| Marinobacter | NF1-7 | 0–25 | 2–8 | − | − | + | − | − | − | + | − |
| NF1-22 | 0–25 | 2–8 | − | − | + | − | − | − | − | − | |
| NF1-41 | 0–25 | 2–8 | − | − | + | − | − | − | + | − | |
| NF2-2 | 0–25 | 2–8 | − | − | + | − | − | − | + | − | |
| NF2-4 | 0–25 | 2–8 | − | − | + | − | − | − | + | − | |
| Moritella | NF1-18 | 0–20 | 1–6 | + | + | − | − | − | − | − | − |
| Photobacterium | NF1-15 | 0–20 | 1–4 | + | + | − | + | − | + | + | − |
| Pseudomonas | NF1-10 | 0–30 | 0.5–8 | − | − | + | + | − | + | − | − |
| NF1-39-1 | 0–37 | 0–8 | − | + | + | + | − | + | + | − | |
| Shewanella | NF1-3 | 0–25 | 0–9 | − | + | − | − | − | − | + | + |
| NF1-16 | 0–25 | 0–9 | − | + | + | − | − | − | + | − | |
| NF1-17 | 0–30 | 0–5 | − | + | + | − | + | − | − | − | |
| NF1-13 | 0–20 | 1–6 | + | − | − | − | + | − | + | − | |
| NF1-31 | 0–30 | 0–6 | − | − | − | − | − | − | + | − | |
| NF1-35 | 0–20 | 1–6 | + | − | − | − | + | − | − | − | |
| NF1-38 | 0–30 | 0–5 | − | + | + | − | + | − | + | − | |
| Bacteroidetes | |||||||||||
| Bizionia | NF1-21 | 0–25 | 1–6 | + | + | − | − | − | − | − | − |
| NF1-25 | 0–25 | 1–6 | + | + | − | − | − | − | − | − | |
| NF2-1 | 0–30 | 0–5 | − | − | − | − | − | + | + | − | |
| Flavobacterim | NF1-9 | 0–37 | 1–8 | − | + | + | − | − | − | + | − |
| NF1-23 | 0–37 | 1–8 | − | + | + | − | − | − | + | + | |
| NF1-39 | 0–37 | 1–8 | − | + | + | − | − | − | + | − | |
| Actinobacteria | |||||||||||
| Salinibacterium | NF2-5 | 0–37 | 0–10 | − | + | − | − | − | − | − | − |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yu, Y.; Li, H.-R.; Zeng, Y.-X.; Chen, B. Bacterial Diversity and Bioprospecting for Cold-Active Hydrolytic Enzymes from Culturable Bacteria Associated with Sediment from Nella Fjord, Eastern Antarctica. Mar. Drugs 2011, 9, 184-195. https://doi.org/10.3390/md9020184
Yu Y, Li H-R, Zeng Y-X, Chen B. Bacterial Diversity and Bioprospecting for Cold-Active Hydrolytic Enzymes from Culturable Bacteria Associated with Sediment from Nella Fjord, Eastern Antarctica. Marine Drugs. 2011; 9(2):184-195. https://doi.org/10.3390/md9020184
Chicago/Turabian StyleYu, Yong, Hui-Rong Li, Yin-Xin Zeng, and Bo Chen. 2011. "Bacterial Diversity and Bioprospecting for Cold-Active Hydrolytic Enzymes from Culturable Bacteria Associated with Sediment from Nella Fjord, Eastern Antarctica" Marine Drugs 9, no. 2: 184-195. https://doi.org/10.3390/md9020184
APA StyleYu, Y., Li, H.-R., Zeng, Y.-X., & Chen, B. (2011). Bacterial Diversity and Bioprospecting for Cold-Active Hydrolytic Enzymes from Culturable Bacteria Associated with Sediment from Nella Fjord, Eastern Antarctica. Marine Drugs, 9(2), 184-195. https://doi.org/10.3390/md9020184




