High-Value Components and Bioactives from Sea Cucumbers for Functional Foods—A Review
Abstract
:1. Introduction
2. Classification, Distribution and Trade
2.1. Sea Cucumber Fishery
2.2. Sea Cucumber Trades
3. Food Value and Important Nutrients
4. High-Value Bioactives and Therapeutics
5. Biological Activities and Medicinal Health Functions
5.1. Anti-Angiogenic
5.2. Anticancer/Antiproliferative
5.3. Anti-Coagulant
5.4. Anti-Fatigue and Immune Functions
5.5. Anti-Hypertension and Angiotensin Converting Enzyme (ACE) Inhibition
5.6. Anti-Inflammatory
5.7. Antimicrobial
5.8. Antioxidant
5.9. Anti-Thrombotic
5.10. Antitumor
5.12. Osteoarthritis
5.13. Wound Healing
5.14. Other Properties
6. Future Prospectives
7. Conclusions
Acknowledgement
References
- Webb, GP. An Overview of Dietary Supplements and Functional Food. In Dietary Supplements and Functional Foods, 1st ed; Blackwell Publishing: Oxford, UK, 2006; pp. 1–35. [Google Scholar]
- Shahidi, F. Nutraceuticals and functional foods: Whole versus processed foods. Trends Food Sci. Technol 2009, 20, 376–387. [Google Scholar]
- Hu, S-Y. Food Plants of China; Chinese University Press: Hong Kong, China, 2005; pp. 3–11. [Google Scholar]
- Venugopal, V. Marine Habitat and Resources. In Marine Products for Healthcare: Functional and Bioactive Nutraceutical Compounds from the Ocean; Venugopal, V, Ed.; CRC Press Taylor &Francis Group: Boca Raton, FL, USA, 2009; pp. 23–50. [Google Scholar]
- Guerard, F; Decourcelle, N; Sabourin, C; Floch-laizet, C; Le Grel, L; Le Floch, P; Gourlay, F; Le Delezir, R; Jaouen, P; Bourseau, P. Recent developments of marine ingredients for food and nutraceutical applications: A review. J. Sci. Hal. Aquat 2011, 2, 21–27. [Google Scholar]
- Blunden, G. Biologically active compounds from marine organisms. Phytother. Res 2001, 15, 89–94. [Google Scholar]
- Taiyeb-Ali, TB; Zainuddin, SLA; Swaminathan, D; Yaacob, H. Efficacy of “Gamadent” toothpaste on the healing of gingival tissues: A preliminary report. J. Oral Sci 2003, 45, 153–159. [Google Scholar]
- Conand, C. The Fishery Resources of Pacific Island Countries Part 2: Holothurians; FAO Fisheries Technical Paper 2722; Food and Agriculture Organization of the United Nations: Rome, Italy, 1990; p. 143. [Google Scholar]
- Mehmet, A; Hüseyin, S; Bekir, T; Yilmaz, E; Sevim, K. Proximate composition and fatty acid profile of three different fresh and dried commercial sea cucumbers from Turkey. Int. J. Food Sci. Technol 2011, 46, 500–508. [Google Scholar]
- Conand, C. Overview of Sea Cucumbers Fisheries over the Last Decade—What Possibilities for a Durable Management? In Echinoderm 2000; Barker, M, Ed.; Swets & Zeitlinger: Lisse, The Netherlands, 2001; pp. 339–344. [Google Scholar]
- Conand, C; Byrne, M. A review of recent developments in the world sea cucumber fisheries. Mar. Fish. Rev 1993, 55, 1–13. [Google Scholar]
- Bruckner, AW; Johnson, KA; Field, JD. Conservation strategies for sea cucumbers: Can a CITES Appendix II listing promote sustainable international trade? SPC Beche-de-mer Inf. Bull 2003, 18, 24–33. [Google Scholar]
- Lawrence, AJ; Afifi, R; Ahmed, M; Khalifa, S; Paget, T. Bioactivity as an options value of sea cucumbers in the Egyptian Red Sea. Conserv. Biol 2009, 24, 217–225. [Google Scholar]
- Shiell, G. Field observations of juvenile sea cucumbers. SPC Beche-de-mer Inf. Bull 2004, 20, 6–11. [Google Scholar]
- Conand, C. Sea Cucumber Biology, Taxonomy, Distribution: Conversation Status. Proceeding of the Convention on International Trade in Endangered Species of Wild Fauna and Flora Tech Workshop on the Conversation of Sea Cucumbers in the Families Holothuridae and Stichopodidae, Kuala Lumpur, Malaysia, 1–3 March 2004.
- Advances in Sea Cucumber Aquaculture and Management. In FAO Fisheries Technical Paper No. 463; Lovatelli, A; Conand, C; Purcell, S; Uthicke, S; Hamel, J-F; Mercier, A (Eds.) Food and Agriculture Organization of the United Nations: Rome, Italy, 2004; p. 425.
- Huizeng, F. Sea cucumber: Ginseng of sea. Zhongguo Mar. Med 2001, 82, 37–44. [Google Scholar]
- Yaacob, HB; Kim, KH; Shahimi, M; Aziz, NS; Sahil, SM. Malaysian sea cucumber (Gamat): A prospect in health food and therapeutic. Proceeding of Asian Food Technology Seminar, Kuala Lumpur, Malaysia, 6–7 October 1997; p. 6.
- Jilin, L; Peck, G. Chinese Dietary Therapy; Churchill Livingstone: London, UK, 1995. [Google Scholar]
- Weici, T. Chinese medicinal materials from the sea. Abstr. Chin. Med 1987, 1, 571–600. [Google Scholar]
- Anderson, EN. The Food of China; Yale University Press: New Haven, CT, USA, 1988. [Google Scholar]
- Chen, J. Overview of sea cucumber farming and sea ranching practices in China. SPC Beche-de-mer Inf. Bull 2003, 18, 18–23. [Google Scholar]
- Wen, J; Hu, C; Fan, S. Chemical composition and nutritional quality of sea cucumbers. J. Sci. Food Agric 2010, 90, 2469–2474. [Google Scholar]
- Tian, F; Zhang, X; Tong, Y; Yi, Y; Zhang, S; Li, L; Sun, P; Lin, L; Ding, J. PE, a new sulfated saponin from sea cucumber, exhibits anti-angiogenic and anti-tumor activities in vitro and in vivo. Cancer Biol. Ther 2005, 4, 874–882. [Google Scholar]
- Roginsky, A; Singh, B; Ding, XZ; Collin, P; Woodward, C; Talamonti, MS; Bell, RH; Adrian, TE. Frondanol(R)-A5p from the sea cucumber, Cucumaria frondosa induces cell cycle arrest and apoptosis in pancreatic cancer cells. Pancreas 2004, 29, 335. [Google Scholar]
- Nagase, H; Enjyoji, K; Minamiguchi, K; Kitazato, KT; Kitazato, K; Saito, H; Kato, H. Depolymerized holothurian glycosaminoglycan with novel anticoagulant actions: Antithrombin III-and heparin cofactor II-independent inhibition of factor X activation by factor IXa-factor VIIIa complex and heparin cofactor II-dependent inhibition of thrombin. Blood 1995, 85, 1527–1534. [Google Scholar]
- Chen, S; Xue, C; Yin, L; Tang, Q; Yu, G; Chai, W. Comparison of structures and anticoagulant activities of fucosylated chondroitin sulfates from different sea cucumbers. Carbohydr. Polym 2011, 83, 688–696. [Google Scholar]
- Hamaguchi, P; Geirsdottir, M; Vrac, A; Kristinsson, HG; Sveinsdottir, H; Fridjonsson, OH; Hreggvidsson, GO. In vitro antioxidant and antihypertensive properties of Icelandic sea cucumber (Cucumaria frondosa). Presented at IFT 10 Annual Meeting & Food Expo, Chicago, IL, USA, 17–20 July 2010. presentation no. 282–04.
- Collin, PD. Tissue fraction of sea cucumber for the treatment of inflammation. United State Patent 5,770,205, 23 June 1998. [Google Scholar]
- Collin, PD. Process for obtaining medically active fractions from sea cucumber. United State Patent 5,876,762, 2 March 1999. [Google Scholar]
- Collin, PD. Peptides having anti-cancer and anti-inflammatory activity. United State Patent 6,767,890, 27 July 2004. [Google Scholar]
- Beauregard, KA; Truong, NT; Zhang, H; Lin, W; Beck, G. The detection and isolation of a novel antimicrobial peptide from the echinoderm, Cucumaria frondosa. Adv. Exp. Med. Biol 2001, 484, 55–62. [Google Scholar]
- Hing, HL; Kaswandi, MA; Azraul-Mumtazah, R; Hamidah, SA; Sahalan, AZ; Normalawati, S; Samsudin, MW; Ridzwan, BH. Effect of methanol extracts from sea cucumbers Holothuria edulis and Stichopus chloronotus on Candida albicans. Microsc. Microanal 2007, 13, 270–271. [Google Scholar]
- Althunibat, OY; Ridzwan, BH; Taher, M; Jamaludin, MD; Ikeda, MA; Zali, BI. In vitro antioxidant and antiproliferative activities of three Malaysian sea cucumber species. Eur. J. Sci. Res 2009, 37, 376–387. [Google Scholar]
- Mourao, PAS; Guimaraes, B; Mulloy, B; Thomas, S; Gray, E. Antithrombotic activity of a fucosylated chondroitin sulphate from echinoderm: Sulphated fucose branches on the polysaccharide account for its antithrombotic action. Br. J. Haematol 1998, 101, 647–652. [Google Scholar]
- Pacheco, RG; Vicente, CP; Zancan, P; Mourão, PAS. Different antithrombotic mechanisms among glycosaminoglycans revealed with a new fucosylated chondroitin sulfate from an echinoderm. Blood Coagul. Fibrinolysis 2000, 11, 563–573. [Google Scholar]
- Zou, Z; Yi, Y; Wu, H; Wu, J; Liaw, C; Lee, K. Intercedensides A–C, three new cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Nat. Prod 2003, 66, 1055–1060. [Google Scholar]
- Tong, Y; Zhang, X; Tian, F; Yi, Y; Xu, Q; Li, L; Tong, L; Lin, L; Ding, J. Philinopside A, a novel marine-derived compound possessing dual anti-angiogenic and anti-tumor effects. Int. J. Cancer 2005, 114, 843–853. [Google Scholar]
- San Miguel-Ruiz, JE; García-Arrarás, JE. Common cellular events occur during wound healing and organ regeneration in the sea cucumber Holothuria glaberrima. BMC Dev. Biol 2007, 7, 1–19. [Google Scholar]
- Kerr, R; Chen, Z. In vivo and in vitro biosynthesis of saponins in sea cucumbers (Holothuroidea). J. Nat. Prod 1995, 58, 172–176. [Google Scholar]
- Miyamoto, T; Togawa, K; Higuchi, R; Komori, T; Sasaki, T. Constituents of Holothuroidea, II. Six newly identified biologically active triterpenoid glycoside sulfates from the sea cucumber Cucumaria echinata. Eur. J. Org. Chem 1990, 1990. 453–460. [Google Scholar]
- Aminin, DL; Chaykina, EL; Agafonova, IG; Avilov, SA; Kalinin, VI; Stonik, VA. Antitumor activity of the immunomodulatory lead Cumaside. Int. Immunopharmacol 2010, 10, 648–654. [Google Scholar]
- Vieira, RP; Mulloy, B; Mourão, PA. Structure of a fucose-branched chondroitin sulphate from sea cucumber. Evidence for the presence of 3-O-sulfo-β-D-glucuronosyl residues. J. Biol. Chem 1991, 266, 13530–13536. [Google Scholar]
- Mourao, PAS; Pereira, MS. Searching for alternatives to heparin: Sulfated fucans from marine invertebrates. Trends Cardiovasc. Med 1999, 9, 225–232. [Google Scholar]
- Goad, LJ; Garneau, FX; Simard, JL; ApSimon, JW; Girard, M. Isolation of Δ9(11)-sterols from the sea cucumber. Implications for holothurin biosynthesis. Tetrahedron Lett 1985, 26, 3513–3516. [Google Scholar]
- Mamelona, J; Pelletier, EM; Lalancette, KG; Legault, J; Karboune, S; Kermasha, S. Quantification of phenolic contents and antioxidant capacity of Atlantic sea cucumber, Cucumaria frondosa. Food Chem 2007, 104, 1040–1047. [Google Scholar]
- Rafiuddin, AM; Venkateshwarlu, U; Jayakumar, R. Multilayered peptide incorporated collagen tubules for peripheral nerve repair. Biomaterials 2004, 25, 85–94. [Google Scholar]
- Sugawara, T; Zaima, N; Yamamoto, A; Sakai, S; Noguchi, R; Hirata, T. Isolation of sphingoid bases of sea cucumber cerberosides and their cytotoxicity against human colon cancer cells. Biosci. Biotechnol. Biochem 2006, 70, 2906–2912. [Google Scholar]
- Mojica, ERE; Merca, FE. Lectin from the body walls of black sea cucumber (Holothuria atra Jäger). Philipp. J. Sci 2004, 133, 77–85. [Google Scholar]
- Mojica, ERE; Merca, FE. Biological properties of lectin from sea cucumber (Holothuria scabra Jäger). J. Biol. Sci 2005, 5, 472–477. [Google Scholar]
- Mojica, ERE; Merca, FE. Isolation and partial characterization of a lectin from the internal organs of the sea cucumber (Holothuria scabra Jäger). Int. J. Zool. Res 2005, 1, 59–65. [Google Scholar]
- Arnold, PW; Britles, RA. Soft Sediment Marine Invertebrates of Southeast Asia and Australia: A Guide to Identification; Australian Institute of Marine Science: Townsville, Austrialia, 1989. [Google Scholar]
- Ridzwan, BH. Sea Cucumbers, A Malaysian Heritage, 1st ed; Research Centre of International Islamic University Malaysia (IIUM): Kuala Lumpur Wilayah Persekutuan, Malaysia, 2007; pp. 1–15. [Google Scholar]
- Morgan, A; Archer, J. Overview: Aspects of sea cucucmber industry research and development in the south Pacific. SPC Beche-de-mer Inf. Bull 1999, 1, 15–17. [Google Scholar]
- Fell, HB. Phylum Echinodermata. In Text Book of Zoology Invertebrates, 7th ed; Marshall, AJ, Williams, WS, Eds.; American Elservier: New York, NY, USA, 1972. [Google Scholar]
- Higgins, M. Sea cucumbers in a deep pickle. Environmental News Network 2000. [Google Scholar]
- Zulfigar, Y; Kwang, SY; Shau-Hwai, AT; Shirayama, Y. Field Guide to the Echinoderms (Sea Cucumbers and Sea Stars) of Malaysia, 1st ed; Kyoto University Press: Kyoto, Japan, 2008; pp. 9–61. [Google Scholar]
- Clark, AM; Rowe, FW. Monograph of Shallow Water Indo-West Pacific Echinoderms; Publication No. 690; Trustees of the British Museum (Natural History): London, UK, 1971; pp. 171–209. [Google Scholar]
- Hamel, JF; Mercier, A. Early development, settlement, growth, and spatial distribution of the sea cucumber Cucumaria frondosa (Echinodermata: Holothuroidea). Can. J. Fish. Aquat. Sci 1996, 53, 253–271. [Google Scholar]
- Yamana, Y; Hamano, T; Goshima, S. Seasonal distribution pattern of adult sea cucumber Apostichopus japonicus (Stichopodidae) in Yoshimi Bay, western Yamaguchi Prefecture, Japan. Fish. Sci 2009, 75, 585–591. [Google Scholar]
- FAO Fisheries and Aquaculture Information and Statistics Service. Capture production 1950–2005. FISHSTAT Plus—Universal software for fishery statistical time series (online or CD-ROM); Food and Agriculture Organization of the United Nations: Rome, Italy, 2007. Available online: http://www.fao.org/fi/statist/FISOFT/FISHPLUS.asp accessed on 18 May 2011.
- Conand, C. Population Status, Fisheries and Trade of Sea Cucumbers in Africa and Indian Ocean. Sea Cucumbers A Global Review on Fishery and Trade; Toral-Granda, V, Lovatelli, A, Vasconcellos, M, Eds.; FAO Fisheries Technical Paper No 516. FAO: Rome, Italy, 2008. Available online: http://www.fao.org/docrep/011/i0375e/i0375e00.htm accessed on 15 April 2011.
- Toral-Granda, V. Population Status, Fisheries and Trade of Sea Cucumbers in Latin America and the Caribbean. Toral-Granda, V, Lovatelli, A, Vasconcellos, M, Eds.; FAO: Rome, Italy, 2008; pp. 213–229. [Google Scholar]
- Eeckhaut, I; Lavitra, T; Rasoforinina, R; Rabenevanana, MW; Gildas, P; Jangoux, M. Madagascar Holothurie SA: The first trade company based on sea cucumber aquaculture in Madagascar. SPC Beche-de-mer Inf. Bull 2008, 28, 22–23. [Google Scholar]
- Lavitra, T; Rachelle, D; Rasolofonirina, R; Jangoux, M; Eeckhaut, I. Processing and marketing of holothurians in the Toliara region, southwestern Madagascar. SPC Beche-de-mer Inf. Bull 2008, 28, 24–33. [Google Scholar]
- Robinson, G; Pascal, B. From hatchery to community—Madagascar’s first village-based holothurian mariculture programme. SPC Beche-de-mer Inf. Bull 2008, 29, 38–43. [Google Scholar]
- Tuwo, A. Status of Sea Cucumber Fisheries and farming in Indonesia. Lovatelli, A, Conand, C, Purcell, S, Uthicke, S, Hamel, J-F, Mercier, A, Eds.; FAO: Rome, Italy, 2004; pp. 49–55. [Google Scholar]
- Choo, PS. Population Status, Fisheries and Trade of Sea Cucumbers in Asia. Toral-Granda, V, Lovatelli, A, Vasconcellos, M, Eds.; FAO: Rome, Italy, 2008. [Google Scholar]
- Clarke, S. Trade in Asian Dried Seafood: Characterization, Estimation and Implications for Conservation; Wildlife Conservation Society Working Paper No. 22; Wildlife Conservation Society: Bronx, NY, USA, 2002. [Google Scholar]
- Chen, J. Present Status and Prospects of Sea Cucumber Industry in China. Lovatelli, A, Conand, C, Purcell, S, Uthicke, S, Hamel, J-F, Mercier, A, Eds.; FAO: Rome, Italy, 2004; pp. 25–38. [Google Scholar]
- Yuan, WP; Liu, CH; Wang, XJ; Meng, XM; Xia, XK; Zhang, MS; Hu, W. Sci. Technol. Food Ind. 2010. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-SPKJ201005091.htm accessed on 9 April 2010.
- Taboada, MC; Gonzalez, M; Rodriguez, E. Value and effects on digestive enzymes and serum lipids of the marine invertebrate Holothuria forskali. Nutr. Res 2003, 23, 1661–1670. [Google Scholar]
- Qin, Z; Jing-feng, W; Yong, X; Yi, W; Sen, G; Min, L; Chang-hu, X. Comparative study on the bioactive components and immune function of three species of sea cucumber. J. Fish. Sci. China. 2008. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-ZSCK200801022.htm accessed on 3 April 2011.
- Saito, M; Kunisaki, N; Urano, N. Collagen as the major edible component of sea cucumber. J. Food Sci 2002, 67, 1319–1322. [Google Scholar]
- Zhao, Y; Li, B; Liu, Z; Dong, S; Zhao, X; Zeng, M. Antihypertensive effect and purification of an ACE inhibitory peptide from sea cucumber gelatin hydrolysate. Process Biochem 2007, 42, 1586–1591. [Google Scholar]
- Jian, J; Bao-ling, Y. Studies on resources and bioactive substances of sea cucumber. Lett. Biotechnol. 2004. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-SWTX200405031.htm accessed on 10 May 2011.
- Fredalina, BD; Ridzwan, BH; Zainal Abidin, AA; Kaswandi, MA; Zaiton, H; Zali, I; Kittakoop, P; Jais, AM. Fatty acid compositions in local sea cucumber, Stichopus chloronotus for wound healing. Gen. Pharmacol 1999, 33, 337–340. [Google Scholar]
- Svetashev, VI; Levin, VS; Lam, CN; Nga, DT. Lipid and fatty acid composition of holothurians from tropical and temperate waters. Comp. Biochem. Physiol 1991, 4, 489–494. [Google Scholar]
- Drazen, JC; Phleger, CF; Guest, MA; Nichols, PD. Lipid, sterols and fatty acid composition of abyssal holothurians and ophiuroids from the North-East Pacific Ocean: Food web implications. Comp. Biochem. Physiol 2008, 151, 79–87. [Google Scholar]
- Gil, A. Polyunsaturated fatty acids and inflammatory disease. Biomed. Pharmacother 2002, 56, 388–396. [Google Scholar]
- Mat, JAM; McCulloch, R; Croft, K. Fatty acid and amino acid composition in haruan as a potential role in wound healing. Gen. Pharmacol 1994, 25, 947–950. [Google Scholar]
- Harper, CR; Jacobson, TA. Usefulness of omega-3 fatty acids and the prevention of coronary heart disease. Am. J. Anat 2005, 96, 1521–1529. [Google Scholar]
- Roynette, CE; Calder, PC; Dupertuis, YM; Pichard, C. n-3 Polyunsaturated fatty acids and colon cancer prevention. Clin. Nutr 2004, 23, 139–151. [Google Scholar]
- Imanari, T; Washio, Y; Huang, Y; Toyoda, H; Suzuki, A; Toida, T. Oral absorption and clearance of partially depolymerized fucosyl chondroitin sulfate from sea cucumber. Thromb. Res 1999, 93, 129–135. [Google Scholar]
- Kariya, Y; Mulloy, B; Imai, K; Tominaga, A; Kaneko, T; Asari, A; Suzuki, K; Masuda, H; Kyogashima, M; Ishii, T. Isolation and partial characterization of fucan sulfates from the body wall of sea cucumber Stichopus japonicus and their ability to inhibit osteoclastogenesis. Carbohydr. Res 2004, 339, 1339–1346. [Google Scholar]
- Yamada, S; Sugahara, K. Potential therapeutic application of chondroitin sulfate/dermatan sulfate. Curr. Drug Discov. Technol 2008, 5, 289–301. [Google Scholar]
- Chen, SG; Li, GY; Yin, LA; Huang, WC; Dong, P; Xu, J; Chang, YG; Xue, CH. Identification of eight species of sea cucumber chondroitin sulfates by high temperature ~1H NMR. J. Instrum. Anal 2010, 29, 8. [Google Scholar]
- McClure, MO; Moore, JP; Blanc, DF; Scotting, P; Cook, GMW; Keynes, RJ; Weber, JN; Davies, D; Weiss, RA. Investigations into the mechanism by which sulfated polysaccharides inhibit HIV infection in vitro. AIDS Res. Hum. Retroviruses 1992, 8, 19–26. [Google Scholar]
- Beutler, JA; McKee, TC; Fuller, RW; Tischler, M; Cardellina, JH; Snader, KM; McCloud, TG; Boyd, MR. Frequent occurrence of HIV-inhibitory sulphated polysaccharides in marine invertebrates. Antivir. Chem. Chemother 1993, 4, 167–172. [Google Scholar]
- Kaswandi, MA; Hing, HL; Sahalan, AZ; Farah, F; Ridzwan, BH; Samsudin, MW; Yasin, MSM; Ali, AM. Saponin from sea cucumber Stichopus badionotus sluiter as potential cytotoxic agent on CEM-SS T-lymphoblastic cell. J. Microsc. Soc. Thailand 2004, 18, 79–84. [Google Scholar]
- Tian, F; Zhu, C; Zhang, X; Xie, X; Xin, X; Yi, Y; Lin, L; Geng, M; Ding, J. Philinopside E, a new sulfated saponin from sea cucumber, blocks the interaction between kinase insert domain-containing receptor (KDR) and αvβ3 integrin via binding to the extracellular domain of KDR. Mol. Pharmacol 2007, 72, 545–552. [Google Scholar]
- Van Dyck, S; Gerbaux, P; Flammang, P. Qualitative and quantitative saponin contents in five sea cucumbers from the Indian Ocean. Mar. Drugs 2010, 8, 173–189. [Google Scholar]
- Caulier, G; van Dyck, S; Gerbaux, P; Eeckhaut, I; Flammang, P. Review of saponin diversity in sea cucumbers belonging to the family Holothuridae. SPC Beche-de-mer Inf. Bull 2011, 31, 48–54. [Google Scholar]
- Hu, X; Wang, Y; Wang, J; Xue, Y; Li, Z; Nagao, K; Yanagita, T; Xue, C. Dietary saponins of sea cucumber alleviate orotic acid-induced fatty liver in rats via PPARα and SREBP-1c signalling. Lipids Health Dis 2010, 9, 25. [Google Scholar]
- Silchenko, AS; Avilov, SA; Kalinin, VI; Kalinovsky, AI; Dmitrenok, PS; Fedorov, SN; Stepanov, VG; Dong, Z; Stonik, VA. Constituents of the sea cucumber Cucumaria okhotensis. Structures of okhotosides B1–B3 and cytotoxic activities of some glycosides from this species. J. Nat. Prod 2007, 71, 351–356. [Google Scholar]
- Hegde, VR; Chan, TM; Pu, H; Gullo, VP; Patel, MG; Das, P; Wagner, N; Parameswaran, PS; Naik, CG. Two selective novel triterpene glycosides from sea cucumber, Telenata Ananas: Inhibitors of chemokine receptor-5. Bioorg. Med. Chem. Lett 2002, 12, 3203–3205. [Google Scholar]
- Zhang, SL; Li, L; Yi, YH; Zou, ZR; Sun, P. Philinopgenin A, B, and C, three new triterpenoid aglycones from the sea cucumber Pentacta quadrangulasis. Mar. Drugs 2004, 2, 185–191. [Google Scholar]
- Kalinin, VI; Silchenko, AS; Avilov, SA; Stonik, VA; Smirnov, AV. Sea cucumbers triterpene glycosides, the recent progress in structural elucidation and chemotaxonomy. Phytochem. Rev 2005, 4, 221–236. [Google Scholar]
- Zou, Z; Yi, Y; Wu, H; Yao, X; Du, L; Jiuhong, W; Liaw, CC; Lee, KH. Intercedensides D–I, cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Nat. Prod 2005, 68, 540–546. [Google Scholar]
- Kalinin, VI; Aminin, DL; Avilov, SA; Silchenko, AS; Stonik, VA. Triterpene glycosides from sea cucucmbers (holothurioidea, echinodermata). Biological activities and functions. Stud. Nat. Prod. Chem 2008, 35, 135–196. [Google Scholar]
- Sun, P; Yi, YH; Li, L; Tang, HF. Studies on chemical constituents from the sea cucumber Holothuria impatiens. Chin. J. Mar. Drugs 2008, 27, 1–7. [Google Scholar]
- Antonov, AS; Avilov, SA; Kalinovsky, AI; Anastyuk, SD; Dmitrenok, PS; Kalinin, VI; Taboada, S; Bosh, A; Avila, C; Stonik, VA. Triterpene glycosides from Antarctic sea cucumbers. 2. Structure of Achlioniceosides A1, A2, and A3 from the sea cucumber Achlionice violaecuspidata (=Rhipidothuria racowitzai). J. Nat. Prod 2009, 72, 33–38. [Google Scholar]
- Han, H; Yi, Y; Xu, Q; La, M; Zhang, H. Two new cytotoxic triterpene glycosides from the sea cucumber Holothuria scabra. Planta Med 2009, 75, 1608–1612. [Google Scholar]
- Han, H; Yi, Y; Li, L; Liu, B; Pan, M; Yan, B; Wang, X. Triterpene glycosides from Sea cucumber Holothuria leucospilota. Chin. J. Nat. Med 2009, 7, 346–350. [Google Scholar]
- Zhang, S; Yi, Y; Tang, H. Bioactive triterpene glycosides from the Sea Cucumber Holothuria fuscocinerea. J. Nat. Prod 2006, 69, 1492–1495. [Google Scholar]
- Kitagawa, I. Structure of holothurin A a biologically active triterpene-oligoglycoside from the sea cucumber Holothuria leucospilota Brandt. Tetrahedron Lett 1979, 20, 1419–1422. [Google Scholar]
- Himeshima, T; Hatakeyama, T; Yamasaki, N. Amino acid sequence of a lectin from the sea cucumber, Stichopus japonicus, and its structural relationship to the C-type animal lectin family. J. Biochem 1994, 115, 689–692. [Google Scholar]
- Gowda, NM; Goswami, U; Islam Khan, M. Purification and characterization of a T-antigen specific lectin from the coelomic fluid of a marine invertebrate, sea cucumber (Holothuria scabra). Fish Shellfish Immunol 2009, 24, 450–458. [Google Scholar]
- Ikeda, Y; Inagaki, M; Yamada, K; Miyamoto, T; Higuchi, R; Shibata, O. Langmur monolayers of cerebroside with different head groups originated from sea cucumber: Binary systems with dipalmitoylphosphatidylcholine (DPPC). Colloids Surf B Biointerfaces 2009, 72, 272–283. [Google Scholar]
- Lu, Y; Kang, C; Xue, C. Separation and purification of cerebrosides from sea cucumbers. Food Sci. 2009. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-SPKX200911006.htm accessed on 18 April 2011.
- Kariya, Y; Watabe, S; Kyogashima, M; Ishihara, M; Ishii, T. Structure of fucose branches in the glycosaminoglycan from the body wall of the sea cucumber Stichopus japonicus. Carbohydr. Res 1997, 297, 273–279. [Google Scholar]
- Liu, HH; Ko, WC; Hu, ML. Hypolipidemic effect of glycosaminoglycans from the sea cucumber Metriatyla scabra in rats fed a cholesterol-supplemented diet. J. Agric. Food Chem 2002, 50, 3602–3606. [Google Scholar]
- Wu, M; Xu, S; Zhao, J; Kang, H; Ding, H. Free-radical depolymerization of glycosaminoglycan from sea cucumber Thelenata ananas by hydrogen peroxide and copper ions. Carbohydr. Polym 2010, 8, 1116–1124. [Google Scholar]
- Findlay, JA; Daljeet, A; Moharir, YE. Some constituents of the sea cucumber Cucumaria frondosa. Mar. Chem 1983, 12, 228. [Google Scholar]
- Findlay, JA; Daljeet, A. A New Aglycone from the Sea Cucumber Cucumaria Frondosa. J. Nat. Prod 1984, 47, 320–324. [Google Scholar]
- Yamada, K; Matsubara, R; Kaneko, M; Miyamoto, T; Higuchi, R. Constituents of holothuroidea. 10. Isolation and structure of a biologically active ganglioside molecular species from the sea cucumber Holothuria leucospilota. Chem. Pharm. Bull 2001, 49, 447–452. [Google Scholar]
- Maier, MS; Roccatagliata, AJ; Kuriss, A; Chludil, H; Seldes, AM; Pujol, CA; Damonte, EB. Two new cytotoxic and virucidal trisulfated triterpene glycosides from the Antarctic sea cucumber Staurocucumis liouvillei. J. Nat. Prod 2001, 64, 732–736. [Google Scholar]
- Murray, AP; Muniaín, C; Seldes, AM; Maier, MS. Patagonicoside A: A novel antifungal disulfated triterpene glycoside from the sea cucumber Psolus patagonicus. Tetrahedron 2001, 57, 9563–9568. [Google Scholar]
- Zhang, Y; Song, S; Song, D; Liang, H; Wang, W; Ji, A. Proliferative effects on neural stem/progenitor cells of a sulfated polysaccharide purified from the sea cucumber Stichopus japonicas. J. Biosci. Bioeng 2010, 109, 67–72. [Google Scholar]
- Wu, P; Chen, Y; Fang, J; Su, W. Studies on the chemical constituents from sea cucumber Mensamaria intercedens IV isolation, properties and antitumor activity of the glycoprotein from Mensamaria intercedens. Chin. J. Mar. Drugs. 2000. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-HYYW200005001.htm accessed on 18 September 2011.
- Jawahar, AT; Nagarajan, J; Shanmugam, SA. Antimicrobial substances of potential biomedical importance from holothurian species. Indian J. Mar. Sci 2002, 31, 161–164. [Google Scholar]
- Lu, Y; Wang, BL. The research progress of antitumorous effectiveness of Stichopus japonicus acid mucopolysaccharide in north of China. Am. J. Med. Sci 2009, 337, 195–198. [Google Scholar]
- Han, H; Xu, Q; Tang, H; Yi, Y; Gong, W. Cytotoxic Holostane-Type Triterpene Glycosides from the Sea Cucumber Pentacta quadrangularis. Planta Med 2010, 76, 1900–1904. [Google Scholar]
- Janakiram, NB; Mohammed, A; Zhang, Y; Choi, CI; Woodward, C; Collin, P; Steele, VE; Rao, CV. Chemopreventive effects of frondanol A5, a Cucumaria frondosa extract, against rat colon carcinogenesis and inhibition of human colon cancer cell growth. Cancer Prev. Res 2010, 3, 82–91. [Google Scholar]
- Chanley, JD; Ledeen, R; Wax, J; Nigrelli, RF; Sobotka, H. Holothurin I. Isolation, properties, and sugar components of holothurin A. J. Am. Chem. Soc 1959, 81, 5180–5183. [Google Scholar]
- Borsing, L; Wang, L; Cavalcante, MCM; Cardilo-Reis, L; Ferreira, PL; Mourão, PAS; Esko, JD; Pavão, MSG. Selectin blocking activity of a fucosylated chondroitin sulfate glycosaminoglycan from sea cucumber, effect on tumor metastasis and neutrophil recruitment. J. Biol. Chem 2007, 282, 14984–14991. [Google Scholar]
- Mourao, PAS; Pereira, MS; Pavão, MS; Mulloy, B; Tollefsen, DM; Mowinckel, MC; Abildgaard, U. Structure and anticoagulant activity of a fucosylated chondroitin sulphate from echinoderm. Sulphated fucose branches on the polysaccharide account for its high anticoagulant action. J. Biol. Chem 1996, 271, 23973–23984. [Google Scholar]
- Poh-Sze, C. Fisheries, Trade and Utilization of Sea Cucumbers in Malaysia. In Advances in Sea Cucumber Aquaculture and Management; FAO Fisheries Technical Paper No 463; FAO: Rome, Italy, 2004. [Google Scholar]
- Ming, S. Investigation on Component and Pharmacology of Sea Cucumber. Chin. Tradit. Pat. Med. 2001. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-ZCYA200110021.htm accessed on 11 February 2011.
- Collin, PD. Inhibition of angiogenesis by sea cucumber fractions. United States Patent 5,985,330, 16 November 1999. [Google Scholar]
- Ogushi, M; Yoshie-stark, M; Suzuki, T. Cytostatic activity of hot water extracts from the sea cucumber in Caco-2. Food Sci. Technol. Res 2005, 11, 202–206. [Google Scholar]
- Li, X; Roginsky, A; Ding, X; Woodward, C; Collin, P; Newman, RA; Bell, RH; Adrian, TE. Review of the Apoptosis Pathways in Pancreatic Cancer and the Anti-apoptotic Effects of the Novel Sea Cucumber Compound, Frondoside A. Ann. N. Y. Acad. Sci 2008, 1138, 181–198. [Google Scholar]
- Li, X; Roginsky, A; Ding, X; Woodward, C; Collin, P; Talamonti, MS; Bell, RH; Adrian, TE. The triterpenoid frondoside A from the sea cucumber Cucumaria frondosa inhibits proliferation of human pancreatic cancer cells in vivo and in vitro. J. Nutr 2008, 35, 135–196. [Google Scholar]
- Zhao, Q; Xue, Y; Liu, Z; Li, H; Wang, J; Li, Z; Wang, Y; Dong, P; Xue, C. Differential effects of sulfated triterpene glycosides, holothurin A1, and 24-Dehydroechinoside A, on antimetastasic activity via regulation of the MMP-9 signal pathway. J. Food Sci 2010, 75, 280–288. [Google Scholar]
- Mulloy, B; Mourão, PAS; Gray, E. Structure/function studies of anticoagulant sulphated polysaccharides using NMR. J. Biotechnol 2000, 77, 123–135. [Google Scholar]
- Wu, M; Xu, S; Zhao, J; Kang, H; Ding, H. Physicochemical characteristics and anticoagulant activities of low molecular weight fractions by free-radical depolymerization of a fucosylated chondroitin sulphate from sea cucumber Thelenata ananas. Food Chem 2010, 122, 716–723. [Google Scholar]
- Fu, X; Cui, Z. Anti-fatigue effects of lower polypeptide from sea cucumber on mice. Food Sci. Technol. 2007. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-SSPJ200704086.htm accessed on 18 May 2011.
- Wang, H; Yin, H; Jin, H; Ha, J. The study of anti-fatigue effects of sea cucumber polypeptide on mice. Food Mach. 2007. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-SPJX200703028.htm accessed on 7 March 2011.
- Liu, C; Wang, X; Yuan, W; Meng, X; Xia, X; Zhang, M; Tang, J; Hu, W; Sun, Y; Liu, J. Anti-fatigue and immune functions of sea cucumber oral liquid. Mod. Food Sci. Technol 2009, 25, 1115–1119. [Google Scholar]
- Bing, L; Jing-feng, W; Jia, F; Xiao-lin, L; Hui, L; Qin, Z; Chang-hu, X. Antifatigue effect of sea cucumber Stichopus japonicus in mice. Food Sci 2010, 31, 244–247. [Google Scholar]
- Zhao, Y; Li, B; Zeng, M; Dong, S; Liu, Z. Study on antihypertensive activity of a lower-value sea cucumber protein hydrolysate. Fish. Mod. 2009. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-HDXY200901013.htm accessed on 25 July 2010.
- Zhao, YB; Dong, S; Liu, Z; Zhao, X; Wang, J; Zeng, M. A novel ACE inhibitory peptide isolated from Acaudina molpadioidea hydrolysate. Peptides 2009, 30, 1028–1033. [Google Scholar]
- Smith, AC. A proposed phylogenetic relationship between sea cucumber Polian vesicles and the vertebrate lymphoreticular system. J. Invertebr. Pathol 1978, 31, 353–357. [Google Scholar]
- Whitehouse, MW; Fairlie, DP. Anti-inflammatory activity of a holothurian (sea cucumber) food supplement in rats. Inflammopharmacology 1994, 9, 23–28. [Google Scholar]
- Herencia, F; Ubeda, A; Ferrándiz, ML; Terencio, MC; Alcaraz, MJ; García-Carrascosa, M; Capaccioni, R; Payá, M. Anti-inflammatory activity in mice of extracts from mediterranean marine invertebrates. Life Sci 1998, 62, 115–120. [Google Scholar]
- Ridzwan, BH; Kaswandi, MA; Azman, Y; Fuad, M. Screening for antibacterial agents in three species of sea cucumbers from coastal areas of Sabah. Gen. Pharmacol 1995, 26, 1539–1543. [Google Scholar]
- Haug, T; Kjuul, AK; Styrvold, OB; Sandsdalen, E; Olsen, ØM; Stensvåg, K. Antibacterial activity in Strongylocentrotus droebachiensis (Echinoidea), Cucumaria frondosa (Holothuroidea), and Asterias rubens (Asteroidea). J. Invertebr. Pathol 2002, 8, 94–102. [Google Scholar]
- Kumar, R; Chaturvedi, AK; Shuklab, PK; Lakshmia, V. Antifungal activity in triterpene glycosides from the sea cucumber Actinopyga lecanora. Bioorg. Med. Chem. Lett 2007, 17, 4387–4391. [Google Scholar]
- Farouk, AEA; Ghouse, FAH; Ridzwan, BH. New bacterial species isolated from Malaysian sea cucumbers with optimized secreted antibacterial activity. Am. J. Biochem. Biotechnol 2007, 3, 60–65. [Google Scholar]
- Ismail, H; Lemriss, S; Ben Aoun, Z; Mhadhebi, L; Dellai, A; Kacem, Y; Boiron, P; Bouraoui, A. Antifungal activity of aqueous and methanolic extracts from the Mediterranean sea cucumber, Holothuria polii. J. Mycol. Med 2008, 18, 23–26. [Google Scholar]
- Chludil, HD; Muniain, CC; Seldes, AM; Maier, MS. Cytotoxic and antifungal triterpene glycosides from the Patagonian sea cucumber Hemoiedema spectabilis. J. Nat. Prod 2002, 65, 860–865. [Google Scholar]
- Muniai, C; Centurion, R; Careaga, VP; Maier, MS. Chemical ecology and bioactivity of triterpene glycosides from the sea cucumber Psolus patagonicus (Dendrochirotida: Psolidae). J. Mar. Biol. Assoc. UK 2008, 88, 817–823. [Google Scholar]
- Yuan, W; Yi, Y; Tang, H; Liu, B; Wang, Z; Sun, G; Zhang, W; Li, L; Sun, P. Antifungal triterpene glycosides from the sea cucumber Bohadschia marmorata. Planta Med 2009, 75, 168–173. [Google Scholar]
- Zhong, Y; Ahmad Khan, M; Shahidi, F. Compositional characteristics and antioxidant properties of fresh and processed sea cucumber (Cucumaria frondosa). J. Agric. Food Chem 2007, 55, 1188–1192. [Google Scholar]
- Zeng, M; Xiao, F; Li, B; Zhao, Y; Liu, Z; Dong, S. Study on free radical scavenging activity of sea cucumber (Paracaudina chinens var.) gelatin hydrolysate. J. Ocean Univ. China 2007, 6, 255–258. [Google Scholar]
- Chenghui, L; Beiwei, Z; Xiuping, D; Liguo, C. Study on the separation and antioxidant activity of enzymatic hydrolysates from sea cucumber. Food Ferment. Ind 2007, 33, 50–53. [Google Scholar]
- Mamelona, J; Saint-Louis, R; Pelletier, E. Nutritional composition and antioxidant properties of protein hydrolysates prepared from echinoderm byproducts. Int. J. Food Sci. Technol 2010, 45, 147–154. [Google Scholar]
- Su, Y; Liu, S; Wu, C. Optimization of the preparation procedure and the antioxidant activity of polypeptide from sea cucumber. J. Fujian Fish. 2009. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-FJSC200902001.htm accessed on 28 June 2011.
- Wang, J; Wang, Y; Tang, Q; Wang, Y; Chang, Y; Zhao, Q; Xue, C. Antioxidation activities of low-molecular-weight gelatin hydrolysate isolated from the sea cucumber Stichopus japonicas. J. Ocean Univ. China 2010, 9, 94–98. [Google Scholar]
- Huihui, C; Ping, Y; Jianrong, L. The preparation of collagen polypeptide with free radical scavenging ability purified from Acaudina molpadioides Semper. J. Chin. Inst. Food Sci. Technol. 2010. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-ZGSP201001002.htm accessed on 12 February 2011.
- Hawa, I; Zulaikah, M; Jamaludin, M; Zainal Abidin, AA; Kaswandi, MA; Ridzwan, BH. The potential of the coelomic fluid of sea cucumber as an antioxidant. Mal. J. Nutr 1999, 5, 55–59. [Google Scholar]
- Dolmatova, LS; Eliseikina, MG; Romashina, VV. Antioxidant enzymatic activity of coelomocytes of the Far East sea cucumber Eupentacta fraudatrix. J. Evol. Biochem. Phys 2004, 40, 126–135. [Google Scholar]
- Zancan, P; Mourão, PAS. Venous and arterial thrombosis in rat models: Dissociation of the antithrombotic effects of glycosaminoglycans. Blood Coagul. Fibrinolysis 2004, 15, 45–54. [Google Scholar]
- Suzuki, N; Kitazato, K; Takamatsu, J; Saito, H. Antithrombotic and anticoagulant activity of depolymerized fragment of the glycosaminoglycan extracted from Stichopus japonicus Selenka. Thromb. Haemost 1991, 65, 369–373. [Google Scholar]
- Li, Z; Wang, H; Li, J; Zhang, G; Gao, C. Basic and clinical study on the antithrombotic mechanism of glycosaminoglycan extracted from sea cucumber. Chin. Med. J. (Engl. ) 2000, 113, 706–711. [Google Scholar]
- Wei-wei, Z; Yin, L. Study advance of Holothuria in antitumor activities. China J. Tradit. Chin. Med. Pharm. 2010. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-BXYY201001035.htm accessed on 8 June 2011.
- Rodriguez, J; Castro, R; Riguera, R. Holothurinosides: New antitumor non sulphated triterpenoid glycosides from the sea cucumber Holothuria forskali. Tetrahedron 1991, 47, 4753–4762. [Google Scholar]
- Ogushi, M; Yoshie-stark, M; Suzuki, T. Apoptosis-inducing activity of hot water extracts from the sea cucumber in human colon tumor cells. Food Sci. Technol. Res 2006, 12, 290–294. [Google Scholar]
- Wu, J; Yi, YH; Tang, HF; Wu, HM; Zou, ZR; Lin, HW. Nobilisides A–C, three new triterpene glycosides from the sea cucumber Holothuria nobilis. Planta Med 2006, 72, 932–935. [Google Scholar]
- Wu, J; Yi, YH; Tang, HF; Wu, HM; Zhou, ZR. Hillasides A and B, two new cytotoxic triterpene glycosides from the sea cucumber Holothuria hilla Lesson. J. Asian Nat. Prod. Res 2007, 9, 609–615. [Google Scholar]
- Sun, P; Liu, BS; Yi, YH; Li, L; Gui, M; Tang, HF; Zhang, DZ; Zhang, SL. A new cytotoxic lanostane-type triterpene glycoside from the sea cucumber Holothuria impatiens. Chem. Biodivers 2007, 4, 450–457. [Google Scholar]
- Yaacob, HB; Kim, KH; Shahimi, MM; Jamalulail, SMS. Water extract of Stichopus sp. 1 improves wound healing. J. Perubatan Univ. Kebangsaan Malays 1994, 16, 19–29. [Google Scholar]
- Menton, DN; Eisen, AZ. Cutaneous wound healing in the sea cucumber, Thyone briareus. J. Morphol 1973, 141, 185–203. [Google Scholar]
- Ridzwan, BH; Dayang, KM; Fredalina, B; Haniza, H; Suhaina, S. Crude Extracts of Three Local Species of Sea-Cucumbers: Their Inhibitory Effects on Histamine Induced Anaphylactic Shock in Guinea Pigs; University Kebangsaan: Kuala Lumpur, Malaysia, 2000. [Google Scholar]
- Kim, KH; Chin, KY; Chin, KC; Yaacob, HB. Anti-anaphylactic shock properties of sea cucumber water extract (“Air Gamat”). Proceedings of the Malaysian Science and Technology Congress, Melaka, Malaysia, 8–10 October 2001; p. 89.
- Ridzwan, BH; Zarina, MZ; Kaswandi, MA; Nadirah, M; Shamsuddin, AF. The antinocieptive effects of extracts from Stichopus chloronotus Brandt. Pak. J. Biol. Sci 2001, 4, 244–246. [Google Scholar]
- Ridzwan, BH; Leong, TC; Idid, SZ. The antinociceptive effects ofwater extracts from sea cucumbers Holothuria leocospilota Brandt, Bohadschia marmoratavitiensis Jaeger and coelomic fluid from Stichopus hermanii. Pak. J. Biol. Sci 2003, 6, 2068–2072. [Google Scholar]
- Singh, N; Kumar, R; Gupta, S; Dube, A; Lakshmi, V. Antileishmanial activity in vitro and in vivo of constituents of sea cucumber Actinopyga lecanora. J. Parasitol. Res 2008, 103, 351–354. [Google Scholar]
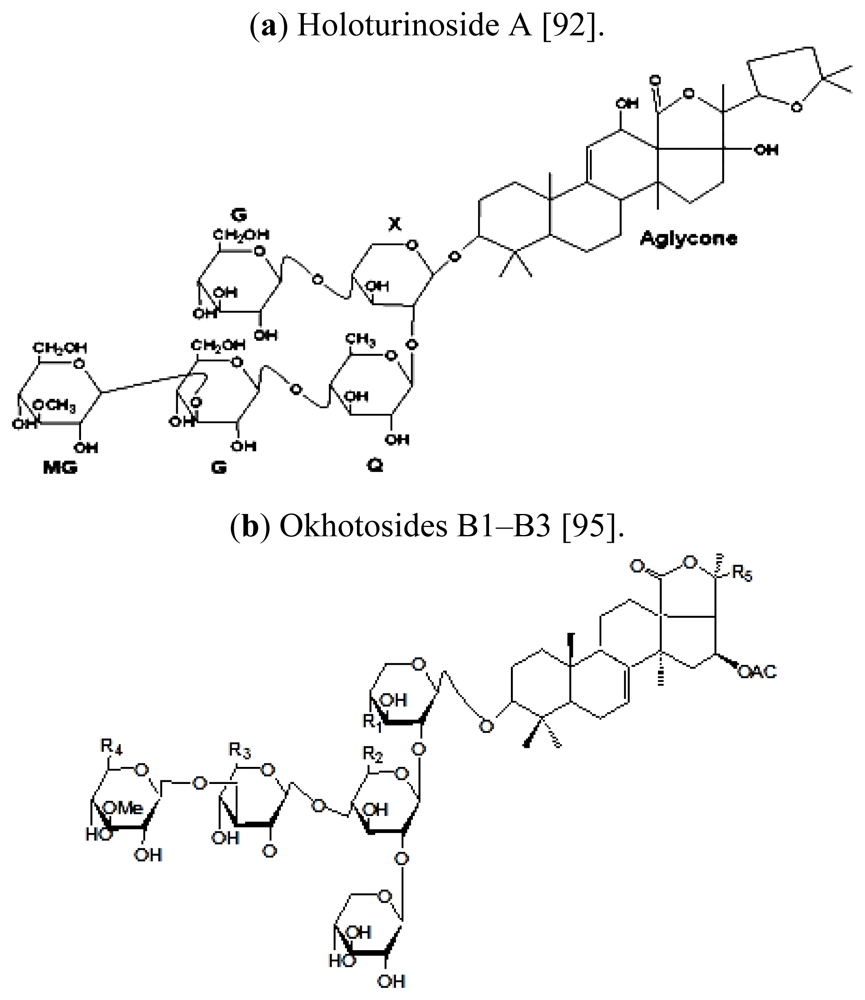
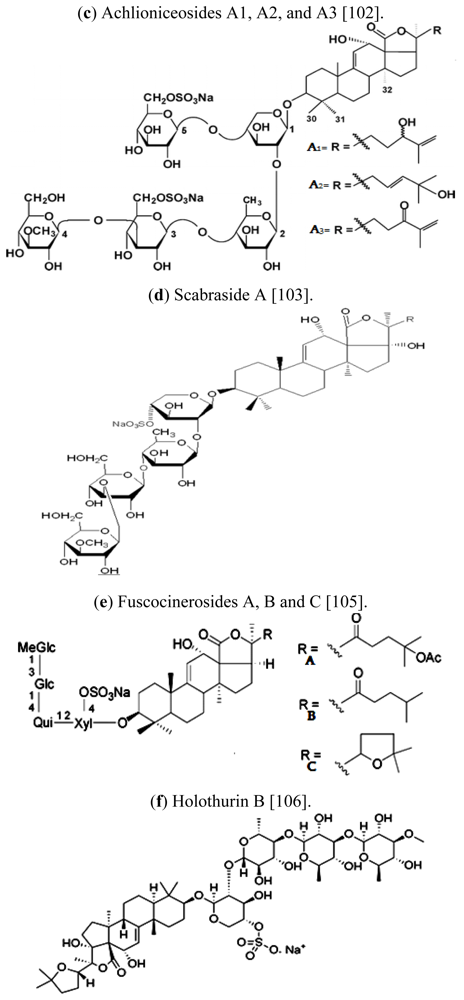
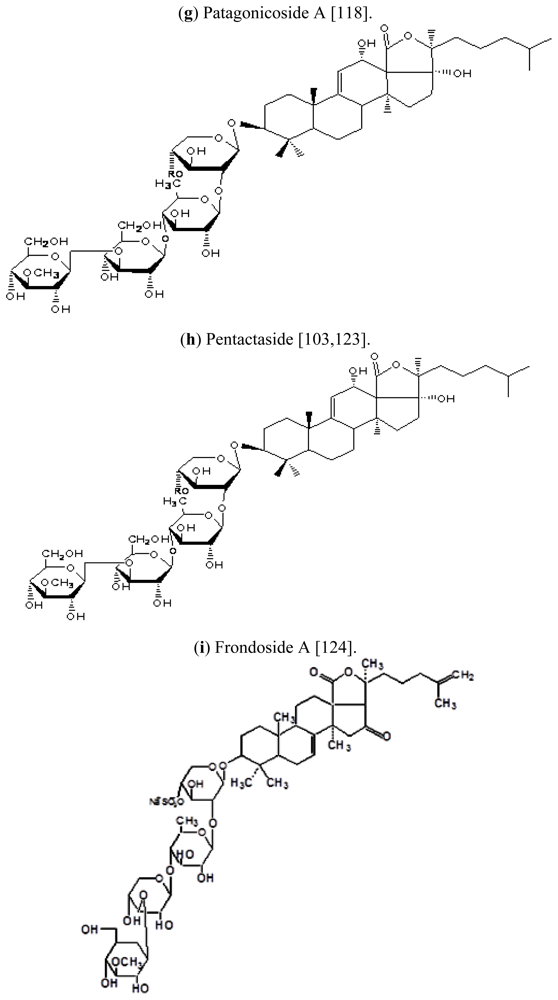
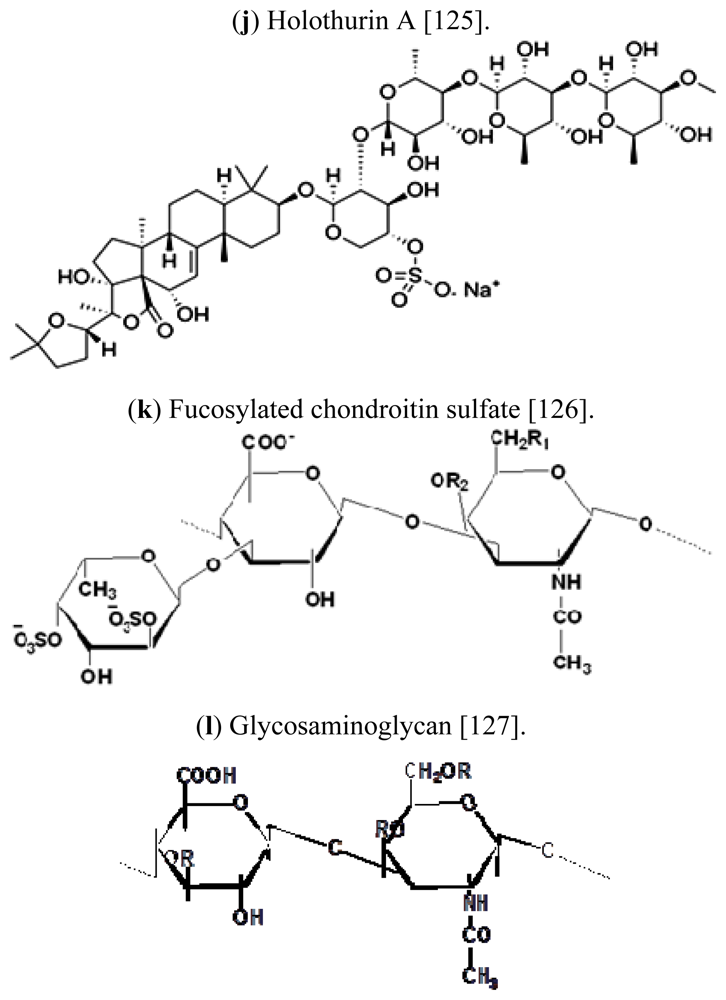
| Scientific (Binomial) name | Common name | Distribution | References |
|---|---|---|---|
| Actinopyga echinites (Jaeger, 1833) | Brown fish (deep water red fish) | South-Pacific | [12] |
| Actinopyga lecanora (Jaeger, 1833) | Stone fish | Indo-West Pacific and South China Sea | [12,57] |
| Actinopyga mauritiana (Quoy & Gaimard, 1834) | Surf red fish, White-spotted or Speckled sea cucumber | Indo-Pacific, South China Sea, Africa and Hawaii | [12,58] |
| Actinopyga miliaris (Quoy & Gaimard, 1833) | Black fish | South-Pacific | [12] |
| Actinopyga obese (Selenka, 1867) | Plump sea cucumber | Western and Central Pacific and Hawaii | [58] |
| Bohadshia argus (Jaeger, 1833) | Spotted or Argus fish, Leopard sea cucumber | Indo-Pacific South-Pacific, South East and South China Sea | [12,57,58] |
| Bohadshia graeffei (Semper, 1868) | Orange fish | South-Pacific, South East Asia | [12] |
| Bohadshia marmorata (Jaeger, 1833) | Chalky fish/chalky sea cucumber | Indo-Pacific, South-Pacific, Red Sea and South China Sea | [12,57] |
| Bohadschia paradoxa (Selenka, 1867) | Paradoxical sea cucumber | Indo-Pacific and Hawaii | [58] |
| Bohadshia vitiensis (Semper, 1868) | Brown sandfish | South-Pacific, Indian Ocean | [12] |
| Cucumaria frondosa (Gunnerus, 1767) | Phenix sea cucumber, pumpkins; orange footed sea cucumber | Indo-West Pacific, and North East coast of Scotland, Shetland and Orkney, West Atlantic | [12,57,59] |
| Holothuria arenicola (Semper, 1868) | Sand sea cucumber | Indo-Pacific and Tropical West Atlantic | [58] |
| Holothuria atra (Jaeger, 1833) | Lollyfish or Black sea cucumber | Indo-Pacific, South-Pacific, South China Sea, Persian Gulf, Africa, Red Sea to Hawaii | [12,58] |
| Holothuria cinerascens (Brandt, 1835) | Ashy sea cucumber | Indo-Pacific, South China Sea, Red sea to Hawaii | [58] |
| Holothuria dificillis (Semper, 1868) | Difficult sea cucumber | Indo-Pacific, South China Sea, Red Sea to Hawaii | [58] |
| Holothuria edulis (Lesson, 1830) | Burnt hotdog or Pink fish | Indo-Pacific, South-Pacific, South China Sea, Red Sea to Hawaii | [12,58] |
| Holothuria fuscogilva (Cherbonnier, 1980) | White teatfish | South-Pacific, Indian Ocean, South East Asia | [12] |
| Holothuria fuscopunctata (Jaeger, 1833) | Elephant trunkfish | South-Pacific, South East Asia | [12] |
| Holothuria hilla (Lesson, 1830) | Light-spotted sea cucumber | Red Sea to Hawaii, Indo-Pacific, South-Pacific, South China Sea, Persian Gulf | [58] |
| Holothuria impatiens (Forskaal, 1775) | Slender sea cucumber or Impatient sea cucumber | Indo-Pacific, Persian Gulf, South China Sea, Southern California, Hawaii, Caribbean (Mexico) and other Tropical Waters | [12,58] |
| Holothuria Mexicana (Ludwig, 1875) | Donkey dung | Caribbean (Venezuela) | [12] |
| Holothuria nobilis (Selenka, 1867) | Black teatfish | Indo-Pacific, South Pacific, South China Sea, SE Asia, Red Sea to Hawaii, Africa | [58] |
| Holothuria pardalis (Selenka, 1867) | Leopard sea cucumber | Indo-Pacific and Eastern Pacific, Red Sea to Hawaii | [58] |
| Holothuria pervicax (Selenka, 1867) | Stubborn sea cucumber | Indo-Pacific, Africa and Hawaii | [58] |
| Holothuria scarab (Jaeger, 1833) | Sandfish | Africa, Red Sea, South China Sea, South-Pacific, South East Asia, Indian Ocean | [12,58] |
| Holothuria scabra versicolor (Conand, 1986) | Golden sandfish | South-Pacific, South East Asia | [12] |
| Isostichopus badionotus (Selenka, 1867) | Three-rowed sea cucumber | Caribbean (Venezuela) | [12] |
| Stichopus californicus (Stimpson, 1857) | Giant red sea cucumber | East Pacific (US/Canada) | [12] |
| Stichopus chloronotus (Brandt, 1835) | Black knobby or green fish | Indo-West Pacific, Eastern Africa to Hawaii (rarely), Indian Ocean and the South-Pacific | [12,58] |
| Stichopus hermanni (Semper, 1868) | Curry fish or Hermann’s sea cucumber | Indo-West Pacific, South East Asia and South-Pacific | [12,58] |
| Stichopus japonicus (Selenka, 1867) | Japanese sea cucumber | North West pacific and Japan Coastal Areas | [12,60] |
| Stichopus horrens (Selenka, 1867) | Golden sea cucumber | Indo-Pacific, South-Pacific and Hawaii | [58] |
| Stichopus mollis (Hutton, 1872) | New Zealand sea cucumber | New Zealand, Australia, Tasmania | [12] |
| Thelenota ananas (Jaeger, 1833) | Prickly redfish | South-Pacific | [12] |
| Thelenota anax (Clark, 1921) | Amber fish | South-Pacific | [12] |
| Year | Nicaragua | Ecuador | Mexico | Chile | Total in the Region | World harvest total | Percentage from region |
|---|---|---|---|---|---|---|---|
| 1988 | - | 3 | - | - | 3 | 19,905 | 0.02 |
| 1989 | - | 10 | - | - | 10 | 17,467 | 0.05 |
| 1990 | - | 12 | - | - | 12 | 19,976 | 0.06 |
| 1991 | - | 29 | - | - | 29 | 21,790 | 0.15 |
| 1992 | - | 152 | - | 237 | 389 | 20,892 | 1.95 |
| 1993 | - | 12 | - | 13 | 25 | 19,348 | 0.13 |
| 1994 | - | 12 | - | 4 | 16 | 24,505 | 0.08 |
| 1995 | - | 12 | - | 106 | 118 | 24,050 | 0.59 |
| 1996 | - | 12 | - | 115 | 127 | 26,795 | 0.64 |
| 1997 | - | 15 | - | 1 | 16 | 24,672 | 0.08 |
| 1998 | - | 15 | 271 | 30 | 316 | 22,004 | 1.59 |
| 1999 | - | 15 | 234 | 108 | 357 | 20,462 | 1.79 |
| 2000 | - | 15 | 426 | 1510 | 1951 | 24,509 | 9.80 |
| 2001 | - | 15 | 481 | 107 | 603 | 20,431 | 3.03 |
| 2002 | - | 15 | 290 | 106 | 411 | 23,445 | 2.06 |
| 2003 | - | 15 | 285 | 307 | 607 | 28,085 | 3.05 |
| 2004 | - | 15 | 265 | 234 | 514 | 27,540 | 2.58 |
| 2005 | 51 | 15 | 312 | 153 | 531 | 26,002 | 2.67 |
| Total | 51 | 389 | 2564 | 3031 | 6035 | 411,878 | 1.46 |
| Bioactive compounds | Sea cucumber species | References |
|---|---|---|
| Triterpene glycoside (Saponin) | Pentaca quadrangularis, Holothuria atra, Actinopyga echinites, Bohadschia subrubra, Pearsonothuria graeffei (Holothuria forskali), Psolus patagonicus, Mensamria intercedens, Thelenota ananas, Holothuria fuscocinerea, Holothuria nobilis, Holothuria hilla, Holothuria impatiens, Cucumaria frondosa, Holothuria leucospilota | [24,25,37,38,40,41,90–94,96–106] |
| Sulfated triterpene glycosides | Hemoiedema spectabilis, Cucumaria japonica, Staurocucumis liouvillei | [117,118] |
| Cerberoside | Bohadschia argus | [48,109,110] |
| (Fucosylated) Chondroitin sulfates | Ludwigothurea grisea, Thelenota ananas, Pearsonothuria graeffei, Stichopus tremulus, Holothuria vagabunda, Isostichopus badionotus | [27,35,36,43,84–87] |
| Glycosaminoglycan | Stichopus japonicas, Holothuria (Metriatyla) scabra, Thelenota ananas | [26,36,111–113] |
| Lectin | Stichopus japonicus, Holothuria atra, Holothuria scabra | [49–51,107,108] |
| Sulfated polysaccharide | Ludwigothurea grisea, Stichopus japonicus | [44,88,89,119] |
| Sterol (glycosides, sulfates) | Cucumaria frondosa | [45,114,115] |
| Bioactive peptides {protein (gelatin & collagen) hydrolysates} | Cucumaria frondosa, Acaudina molpadioides, Paracaudina chilensis, Acaudina molpadioidea, Stichopus japonicas | [31,32,47,73–75] |
| Phenols and flavonoids | Holothuria scabra, Holothuria leucospilota, Stichopus chloronotus, Cucumaria frondosa | [34,46] |
| Triterpene oligoglycosides | Cucumaria okhotensis | [95] |
| Glycoprotein | Mensamaria intercedens | [120] |
| Steroidal sapogenins | Actinopyga echinites, Actinopyga miliaris, Holothuria atra, Holothuria scabra | [121] |
| Mucopolysaccharide (SJAMP) | Stichopus japonicas | [122] |
| Polyunsaturated fatty acids (PUFA): arachidonic acid (AA C20:4 n-6), eicosapentaenoic acid (EPA C20:5 n-3), docosahexaenoic acid (DHA C22:6 n-3) | Stichopus herrmanni, Thelenota ananas, Thelenota anax, Holothuria fuscogilva, Holothuria fuscopunctata, Actinopyga mauritiana, Actinopyga caerulea, Bohadschia argus, Stichopus chloronotus, Holothuria tubulosa, Holothuria polii, Holothuria mammata | [9,23,77–83] |
| Sea cucumber species | Bioactive compounds | Pharmacologica/medicinal activity | References |
|---|---|---|---|
| Pentacta quadrangularius, Cucumaria frondosa | Sulfated saponin [Philinospide A], Philinospide E (PE), Sea cucumber fractions: B1000 and Fucosylated chondroitin sulfate | Antiangiogenic | [24,38,130] |
| Holothuria scabra, Holothurialeucos pilota, Stichopus chloronotus, Cucumaria frondosa, Cucumaria okhotensis, Mensamaria intercedens, Pearsonothuria graeffei, Stichopus japonicus, Stichopus variegates | Triterpenoid [Frondoside A], Triterpene oligoglycosides [Okhotosides B1, B2, and B3], Triterpene glycosides [Intercedensides A, B, and C], Glycolipid [Frondanol A], Triterpene oligoglycosides [Holothurin A and 24-dehydroechinoside], Frondanol(R)-A5p, sphingoid base composition of cerebrosides | Anticancer | [34,37,48,124,131–134] |
| Ludwigothurea grisea, Thelenota ananas | Fucosylated chondroitin sulfate, Fucosylated chondroitin sulfate | Anticoagulant | [127,135,136] |
| Stichopus japonicas | Low molecular weight polypeptides, Polypeptides Acidic mucopolysaccharides, collagen and bioactive amino acids (all together) | Anti-fatigue | [137–140] |
| Actinopyga echinites, Actinopyga miliari, Holothuria atra, Holothuria scabra, Bohadshia argus, Cucumaria frondosa, Holothuria poli, Hemoiedema spectabilis, Psolus patagonicus, Actinopyga lecanora, Holothuria atra, Psolus patagonicus, Bohadschia marmorata, Cucumaria frondosa | Steroidal sapogenins, (Phosphate-buffered saline [PBS]), Sulfated triterpene glycosides [Hemoiedemosides A and B], Triterpene glycoside [patagonicoside A], Triterpene glycoside [holothurin B (saponin)], Triterpene glycoside [patagonicoside A], Holostan-type triterpene glycosides [marmoratoside A, 17α-hydroxy impatienside A, impatienside A and bivittoside D], Bioactive peptides | Antimicrobial Antibacterial and antifungal | [32,118,121,146–153] |
| Cucumariafrondosa, Stichopus japonicus, Paracaudina chilensis, Cucumaria frondosa, Cucumaria frondosa, Holothuria scabra, Holothuria leucospilota, Stichopus chloronotus, Acaudina molpadioides | Gelatin hydrolysate, Gelatin hydrolysate, Protein hydrolysate [bioactive peptides], Bioactive peptides, Phenols and flavonoids, Phenols, Gelatin hydrolysate [Bioactive peptides], Collagen polypeptides | Antioxidation | [34,46,154–160] |
| Stichopus japonicas | Glycosaminoglycan, Holothurian glycosaminoglycan | Antithrombotic | [126,163–165] |
| Mensamaria intercedens, Mensamaria intercedens, Holothuria hilla, Pentacta quadrangularis, Holothuria forskali, Stichopus japonicus, Holothuria nobilis, Holothuria fuscocinerea, Stichopus japonicus, Holothuria impatiens, Ludwigothurea grisea, Cucumaria japonica | Triterpene glycosides, [intercedensides D–I], Glycoprotein (GPMI I), Triterpene glycosides [hillasides A and B], Sulfated saponins [Philinopside A], Triterpene glycosides [holothurinosides A, B, C and D; and desholothurin A], Mucopolysaccharide (SJAMP), Triterpene glycosides [nobilisides A, B and C], Triterpene glycosides [fuscocinerosides A, B, and C], Monosulfated triterpene glycosides, Lanostane-type triterpene glycoside [impatienside A], Sulfated polysaccharide, Monosulfated triterpene glycosides [cumaside] | Antitumour | [37,38,42,120,122,166–171] |
| Staurocucumis liouvillei | Trisulfated triterpene glycosides [liouvillosides A and B] | Antiviral | [117] |
| Stichopus japonicas | Fucan sulfate, Glucosamin, Chondroitin | Osteoarthritis | [85] |
| Thyone briareus, Stichopus chloronotus, Stichopus herrmanni, Thelenota ananas, Thelenota anax, Holothuria fuscogilva, Holothuria fuscopunctata, Actinopyga mauritiana, Actinopyga caerulea, Bohadschia argus, Stichopus chloronotus, Holothuria tubulosa, Holothuria polii, Holothuria mammata | Polyunsaturated fatty acids, (arachidonic acid, eicosapentaenoic acid, docosahexaenoic acid ) | Wound healing | [9,23,77–79,172] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bordbar, S.; Anwar, F.; Saari, N. High-Value Components and Bioactives from Sea Cucumbers for Functional Foods—A Review. Mar. Drugs 2011, 9, 1761-1805. https://doi.org/10.3390/md9101761
Bordbar S, Anwar F, Saari N. High-Value Components and Bioactives from Sea Cucumbers for Functional Foods—A Review. Marine Drugs. 2011; 9(10):1761-1805. https://doi.org/10.3390/md9101761
Chicago/Turabian StyleBordbar, Sara, Farooq Anwar, and Nazamid Saari. 2011. "High-Value Components and Bioactives from Sea Cucumbers for Functional Foods—A Review" Marine Drugs 9, no. 10: 1761-1805. https://doi.org/10.3390/md9101761
APA StyleBordbar, S., Anwar, F., & Saari, N. (2011). High-Value Components and Bioactives from Sea Cucumbers for Functional Foods—A Review. Marine Drugs, 9(10), 1761-1805. https://doi.org/10.3390/md9101761




