Inhibition of Nitric Oxide (NO) Production in Lipopolysaccharide (LPS)-Activated Murine Macrophage RAW 264.7 Cells by the Norsesterterpene Peroxide, Epimuqubilin A
Abstract
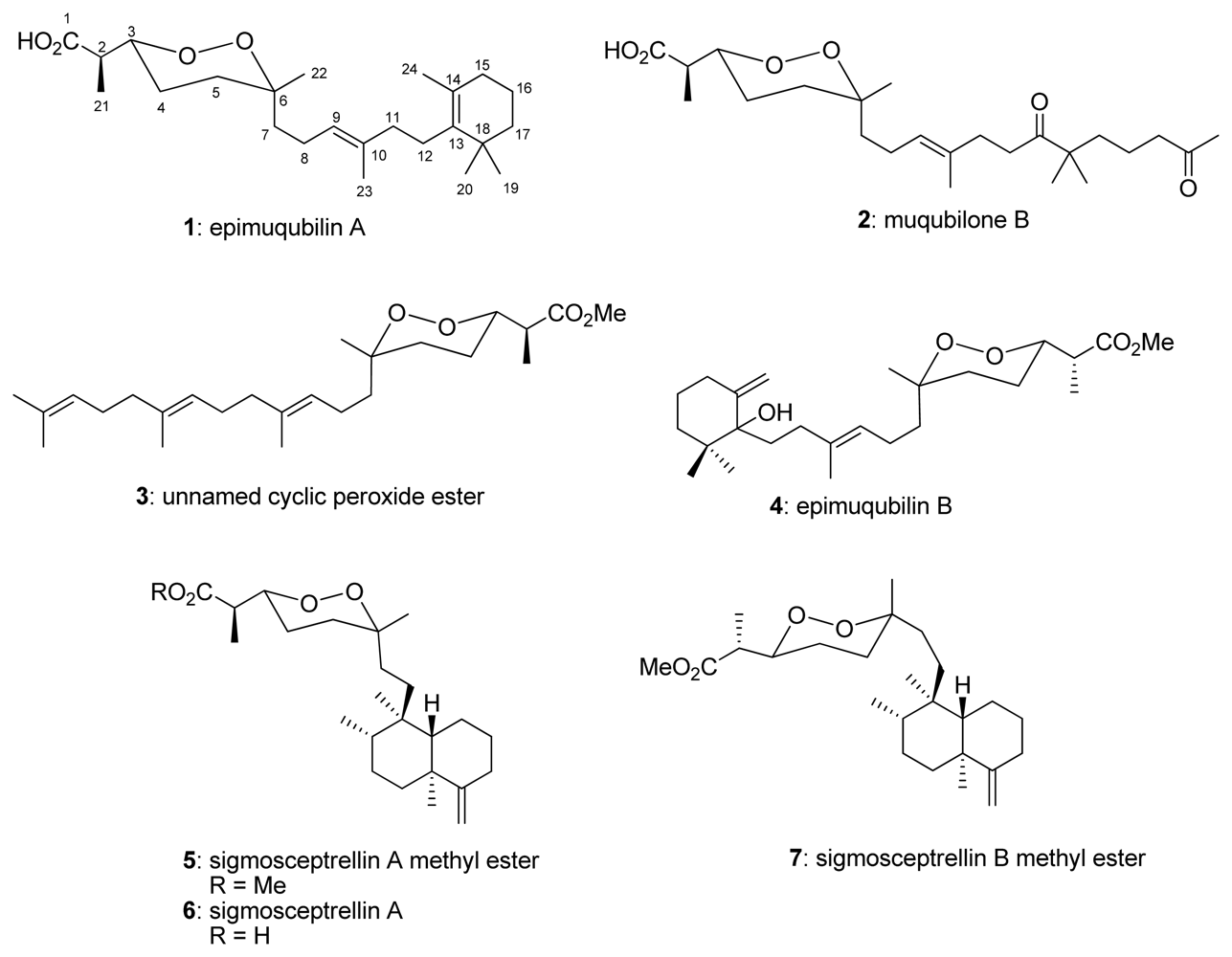
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Test Compounds
3.2. Assay for NO inhibitory effect using RAW264.7 cells
3.3. Glycogen synthase kinase-3β assay
Acknowledgements
- Samples Availability: Available from the authors.
References and Notes
- Albericci, M; Collart-Lempereur, M; Braekman, JC; Daloze, D; Tursch, B. Chemical studies of marine invertebrates. XLI. Sigmosceptrellin-A methyl ester a nor-sesterterpenoid peroxide from the sponge Sigmosceptrella laevis. Tetrahedron Lett 1979, 20, 2687–2690. [Google Scholar]
- Albericci, M; Braekman, JC; Daloze, D; Tursch, B. Chemical studies of marine invertebrates— XLV: The chemistry of three norsesterterpene peroxides from the sponge Sigmosceptrella laevis. Tetrahedron 1982, 38, 1881–1890. [Google Scholar]
- Ovenden, SPB; Capon, RJ. Trunculins G–I: New norsesterterpene cyclic peroxides from a Southern Australian marine sponge, Latrunculia sp. Aust J Chem 1998, 51, 573–579. [Google Scholar]
- Capon, RJ; MacLeod, JK; Willis, AC. Trunculins A and B, norsesterterpene cyclic peroxides from a marine sponge Latrunculia brevis. J Org Chem 1987, 52, 339–342. [Google Scholar]
- Ibrahim, SRM; Ebel, R; Wray, V; Muller, WEG; Edrada-Ebel, R; Proksch, P. Diacarperoxides, norterpene cyclic peroxides from the sponge Diacarnus megaspinorhabdosa. J Nat Prod 2008, 71, 1358–1364. [Google Scholar]
- Dai, J; Liu, Y; Zhou, Y.-D; Nagle, DG. Hypoxia-selective antitumor agents: norsesterterpene peroxides from the marine sponge Diacarnus levii preferentially suppress the growth of tumor cells under hypoxic conditions. J Nat Prod 2007, 70, 130–133. [Google Scholar]
- Groweiss, A; Shmueli, U; Kashman, Y. Marine toxins of Latrunculia magnifica. J Org Chem 1983, 48, 3512–3516. [Google Scholar]
- Perry, NB; Blunt, JW; McCombs, JD; Munro, MHG. Discorhabdin C, a highly cytotoxic pigment from sponge of the genus Latrunculia. J Org Chem 1986, 51, 5476–5478. [Google Scholar]
- Ford, J; Capon, RJ. Discorhabdin R: A new antibacterial pyrroloiminoquinone from two Latrunculiid marine sponges, Latrunculia sp. and Negombata sp. J Nat Prod 2000, 63, 1527–1528. [Google Scholar]
- Reyes, F; Martin, R; Rueda, A; Fernandez, R; Montalvo, D; Gomez, C; Sanchez-Puelles, JM. Discorhabdins I and L, cytotoxic alkaloids from the sponge Latrunculia brevis. J Nat Prod 2004, 67, 463–465. [Google Scholar]
- Sepe, V; D’Orsi, R; Borbone, N; D’Auria, MV; Bifulco, G; Monti, MC; Catania, A; Zampella, A. Callipeltins F-I: New antifungal peptides from the marine sponge Latrunculia sp. Tetrahedron 2006, 62, 833–840. [Google Scholar]
- D’Auria, MV; Sepe, V; D’Orsi, R; Bellotta, F; Debitus, C; Zampella, A. Isolation and structural elucidation of callipeltins J-M: antifungal peptides from the marine sponge Latrunculia sp. Tetrahedron 2007, 63, 131–140. [Google Scholar]
- Butler, MS; Capon, RJ. Trunculin-F and contrunculin-A and –B: novel oxygenated norterpenes from a Southern Australian marine sponge Latrunculia conulosa. Aust J Chem 1993, 46, 1363–1374. [Google Scholar]
- Sperry, S; Valeriote, FA; Corbett, TH; Crews, P. Isolation and cytotoxic evaluation of marine sponge-derived norterpene peroxides. J Nat Prod 1998, 61, 241–247. [Google Scholar]
- Manes, LV; Bakus, GJ; Crews, P. Bioactive marine sponge norditerpene and norsesterterpene peroxides. Tetrahedron Lett 1984, 25, 931–934. [Google Scholar]
- El Sayed, KA; Hamann, MT; Hashish, NE; Shier, WT; Kelly, M; Khan, AA. Antimalarial, antiviral, and antitoxoplasmosis norsesterterpene peroxide acids from the Red Sea sponge Diacarnus erythraeanus. J Nat Prod 2001, 64, 522–524. [Google Scholar]
- Schottenfeld, D; Beebe-Dimmer, J. Alleviating the burden of cancer: A perspective on advances, challenges, and future directions. Cancer Epidemiol Biomarkers Prev 2006, 15, 2049–2055. [Google Scholar]
- Coussens, LM; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar]
- Kopydlowski, KM; Salkowski, CA; Cody, MJ; van Rooijen, N; Major, J; Hamilton, TA; Vogel, SN. Regulation of macrophage chemokine expression by lipopolysaccharide in vitro and in vivo. J Immunol 1999, 163, 1537–1544. [Google Scholar]
- Bingle, L; Brown, NJ; Lewis, CE. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol 2002, 196, 254–265. [Google Scholar]
- Patel, RP; McAndrew, J; Sellak, H; White, CR; Jo, H; Freeman, BA; Darley-Usmar, VM. Biological aspects of reactive nitrogen species. Biochim Biophys Acta 1999, 1411, 385–400. [Google Scholar]
- Doble, BW; Woodgett, JR. GSK-3β: Tricks of the trade for a multi-tasking kinase. J Cell Sci 2003, 116, 1175–1186. [Google Scholar]
- Cohen, P; Goedert, M. GSK3 inhibitors: Development and therapeutic potential. Nat Rev Drug Disc 2004, 3, 479–487. [Google Scholar]
- Rubio, BK; Tenney, K; Ang, K.-H; Abdulla, M; Arkin, M; McKerrow, JH; Crews, P. The marine sponge Diacarnus bismarckensis as a source of peroxiterpene inhibitors of Trypanosoma brucei, the causative agent of sleeping sickness. J Nat Prod 2009, 72, 218–222. [Google Scholar]
- Capon, RJ; MacLeod, JK. Structural and stereochemical studies on marine norterpene cyclic peroxides. Tetrahedron 1985, 41, 3391–3404. [Google Scholar]
- D’Ambrosio, M; Guerriero, A; Debitus, C; Waikedre, J; Francesco, P. Relative contributions to antitumoral activity of lipophilic vs. polar reactive moieties in marine terpenoids. Tetrahedron Lett 1997, 38, 6285–6288. [Google Scholar]
- Kossuga, MH; Nascimento, AM; Reimao, JQ; Tempone, AG; Taniwaki, NN; Veloso, K; Ferreira, AG; Cavalcanti, BC; Pessoa, C; Moraes, MO; Mayer, AMS; Hajdu, E; Berlinck, RGS. Antiparasitic, antineuroinflammatory, and cytotoxic polyketides from the marine sponge Plakortis angulospiculatus collected in Brazil. J Nat Prod 2008, 71, 334–339. [Google Scholar]
- Min, HY; Kim, MS; Jang, DS; Park, EJ; Seo, EK; Lee, SK. Suppression of lipopolysaccharide-stimulated inducible nitric oxide synthase (iNOS) expression by a novel humulene derivative in macrophage cells. Int Immunopharmacol 2009, 9, 844–849. [Google Scholar]
- Andoh, T; Hirata, Y; Kikuchi, A. Yeast glycogen synthase kinase 3 is involved in protein degradation in cooperation with Bul1, Bul2, and Rsp5. Mol Cell Biol 2000, 20, 6712–6720. [Google Scholar]
| Compounds | NO IC50, μM) | Cytotoxic activity | Yeast GSK-3βa (mm) at 37 °C(μg/disk) | ||||
|---|---|---|---|---|---|---|---|
| % Survivalb | IC50, μM | 40 | 20 | 10 | 5 | ||
| Epimuqubilin A (1) | 7.4 | 36.1 | 37.1 | 10 | 9 | nac | na |
| Muqubilone B (2) | 23.8 | 99.0 | - | na | - | - | - |
| Unnamed cyclic peroxide ester (3) | 46.0 | >100 | - | ntd | - | - | - |
| Epimuqubilin B (4) | 25.6 | >100 | - | 10 | 8 | na | na |
| Sigmosceptrellin A methyl ester (5) | >100 | 90.6 | - | na | - | - | - |
| Sigmosceptrellin A (6) | 9.9 | 43.0 | 42.7 | 13 | 11 | 10 | 8 |
| Sigmosceptrellin B methyl ester (7) | 65.7 | >100 | - | na | - | - | - |
| L-NMMAe | 25.5 | >100 | - | - | - | - | - |
| TDZD-8f at 37°C | 15 | 11 | 10 | 9 | |||
| at 25°C | 12 | 10 | 9 | 8 | |||
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cheenpracha, S.; Park, E.-J.; Rostama, B.; Pezzuto, J.M.; Chang, L.C. Inhibition of Nitric Oxide (NO) Production in Lipopolysaccharide (LPS)-Activated Murine Macrophage RAW 264.7 Cells by the Norsesterterpene Peroxide, Epimuqubilin A. Mar. Drugs 2010, 8, 429-437. https://doi.org/10.3390/md8030429
Cheenpracha S, Park E-J, Rostama B, Pezzuto JM, Chang LC. Inhibition of Nitric Oxide (NO) Production in Lipopolysaccharide (LPS)-Activated Murine Macrophage RAW 264.7 Cells by the Norsesterterpene Peroxide, Epimuqubilin A. Marine Drugs. 2010; 8(3):429-437. https://doi.org/10.3390/md8030429
Chicago/Turabian StyleCheenpracha, Sarot, Eun-Jung Park, Bahman Rostama, John M. Pezzuto, and Leng Chee Chang. 2010. "Inhibition of Nitric Oxide (NO) Production in Lipopolysaccharide (LPS)-Activated Murine Macrophage RAW 264.7 Cells by the Norsesterterpene Peroxide, Epimuqubilin A" Marine Drugs 8, no. 3: 429-437. https://doi.org/10.3390/md8030429
APA StyleCheenpracha, S., Park, E.-J., Rostama, B., Pezzuto, J. M., & Chang, L. C. (2010). Inhibition of Nitric Oxide (NO) Production in Lipopolysaccharide (LPS)-Activated Murine Macrophage RAW 264.7 Cells by the Norsesterterpene Peroxide, Epimuqubilin A. Marine Drugs, 8(3), 429-437. https://doi.org/10.3390/md8030429




