Comparative Chemical Profiling and Antimicrobial/Anticancer Evaluation of Extracts from Farmed versus Wild Agelas oroides and Sarcotragus foetidus Sponges
Abstract
:1. Introduction
2. Results and Discussion
2.1. The Metabolomics Profile of Farmed and Wild Agelas oroides Sponges
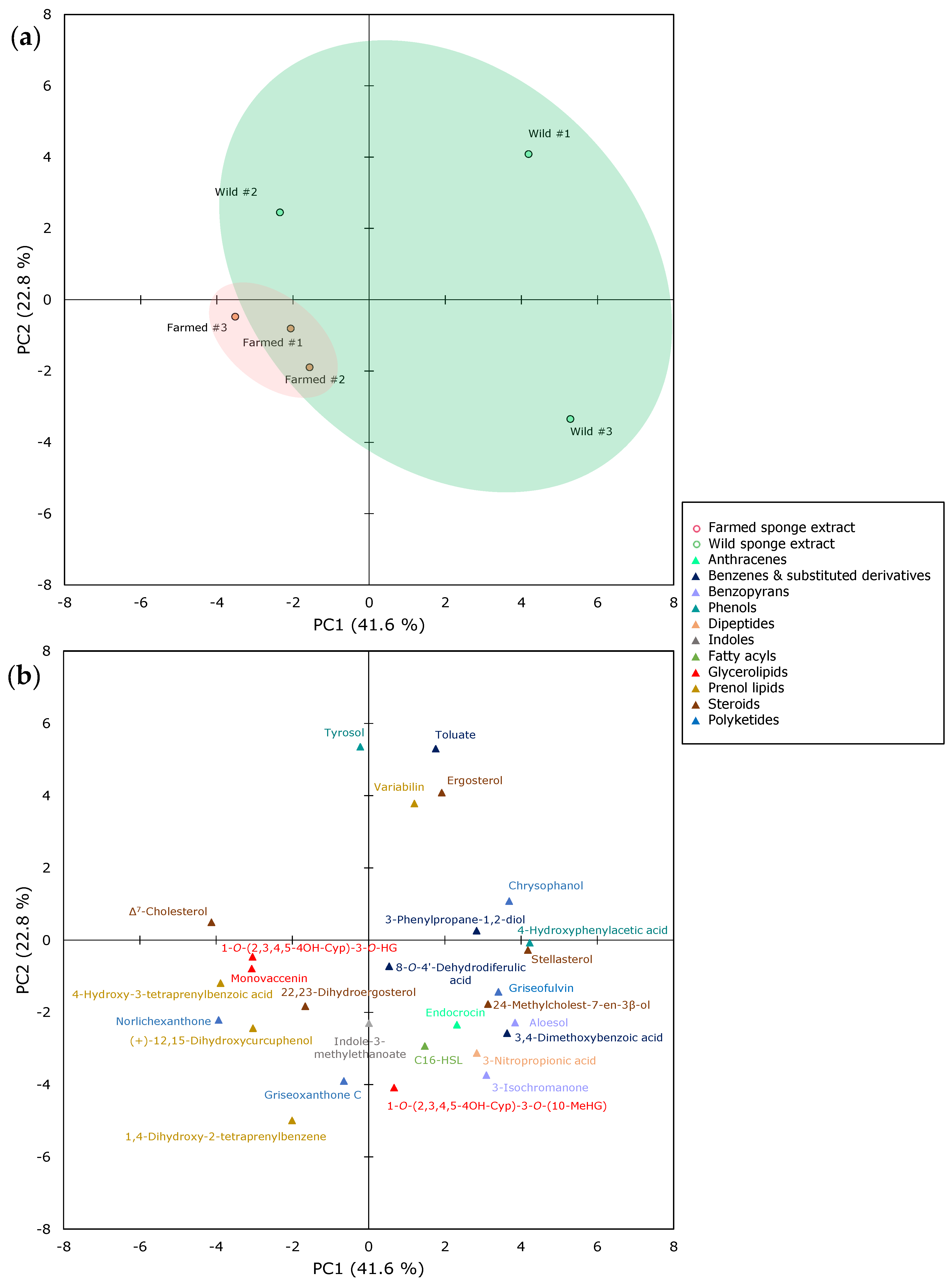
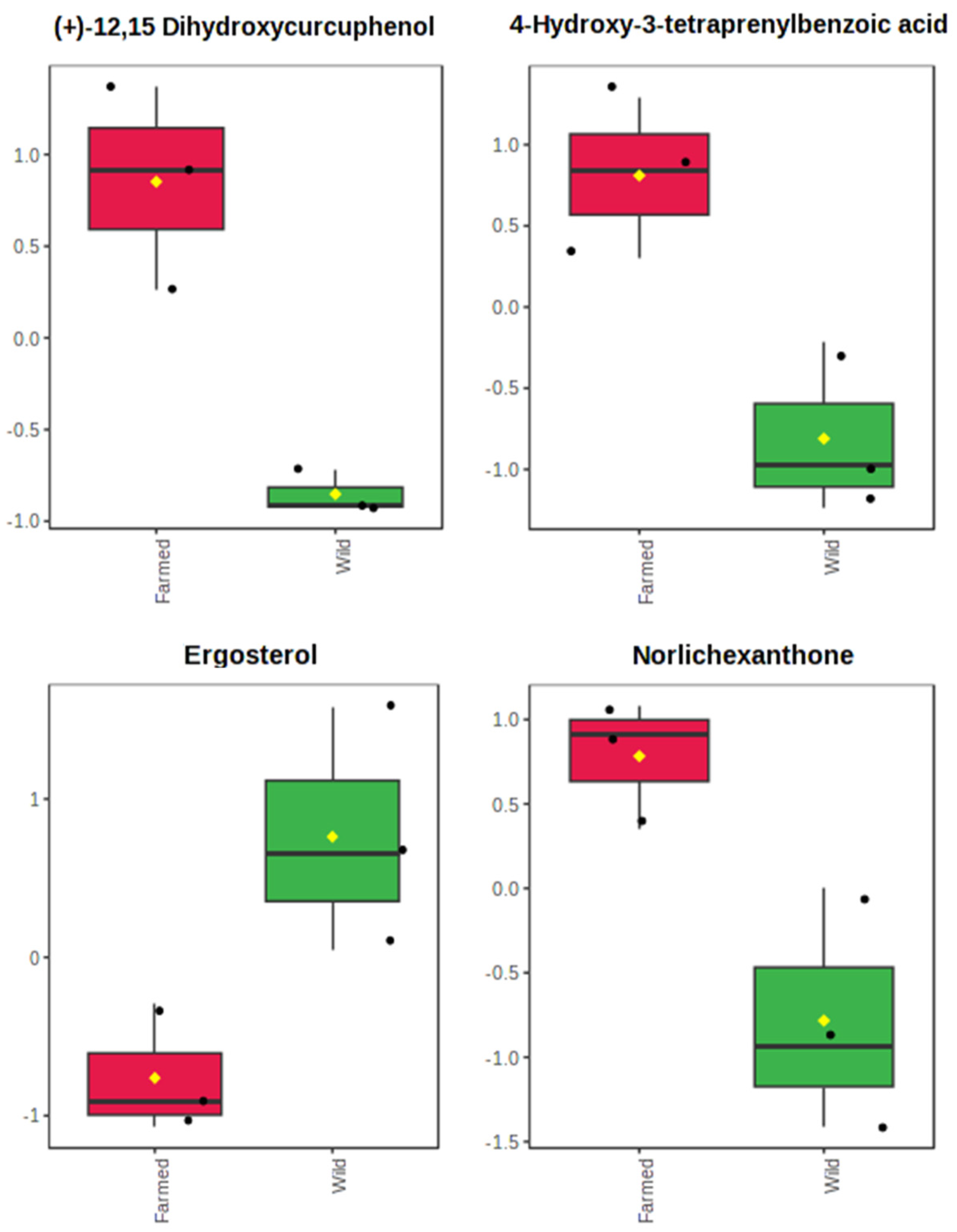
2.2. The Metabolomics Profile of Farmed and Wild Sarcotragus foetidus Sponges
2.3. Evaluation of the Sponges’ Biotechnological Potential
2.3.1. Antimicrobial Activity Evaluation
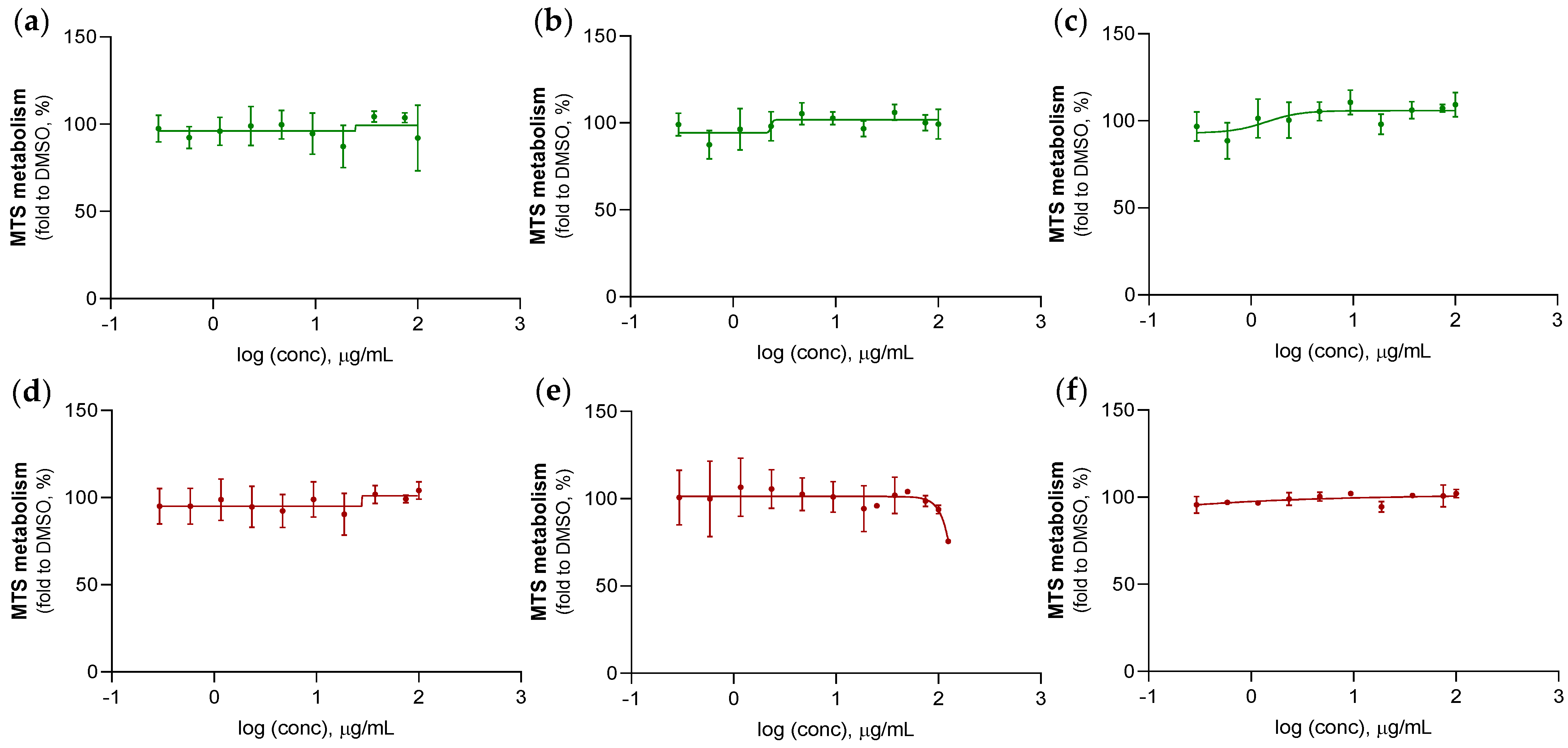
2.3.2. Anticancer Activity Evaluation
3. Materials and Methods
3.1. Sponge Material
3.2. Extraction
3.3. LC−MS/MS Analysis
3.3.1. Chemicals and Reagents
3.3.2. Instrumental LC–MS/MS Analysis
3.3.3. Sample Preparation for LC–MS/MS Analysis
3.3.4. Chromatographic and Mass Spectrometry Conditions
3.4. Antibacterial Assays
3.5. Anticancer Assays
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Garlock, T.; Asche, F.; Anderson, J.; Ceballos-Concha, A.; Love, D.C.; Osmundsen, T.C.; Pincinato, R.B.M. Aquaculture: The missing contributor in the food security agenda. Glob. Food Secur. 2022, 32, 100620. [Google Scholar] [CrossRef]
- Taranger, G.L.; Karlsen, O.; Bannister, R.J.; Glover, K.A.; Husa, V.; Karlsbakk, E.; Kvamme, B.O.; Boxaspen, K.K.; Bjorn, P.A.; Finstad, B.; et al. Risk assessment of the environmental impact of Norwegian Atlantic salmon farming. Ices J. Mar. Sci. 2015, 72, 997–1021. [Google Scholar] [CrossRef]
- Fu, W.T.; Sun, L.M.; Zhang, X.C.; Zhang, W. Potential of the marine sponge Hymeniacidon perleve as a bioremediator of pathogenic bacteria in integrated aquaculture ecosystems. Biotechnol. Bioeng. 2006, 93, 1112–1122. [Google Scholar] [CrossRef] [PubMed]
- Gokalp, M.; Mes, D.; Nederloff, M.; Zhao, H.W.; de Goeij, J.M.; Osinga, R. The potential roles of sponges in integrated mariculture. Rev. Aquacult 2021, 13, 1159–1171. [Google Scholar] [CrossRef]
- Longo, C.; Pierri, C.; Mercurio, M.; Trani, R.; Cardone, F.; Carbonara, P.; Alfonso, S.; Stabili, L. Bioremediation capabilities of Hymeniacidon perlevis (Porifera, Demospongiae) in a land-based experimental fish farm. J. Mar. Sci. Eng. 2022, 10, 874. [Google Scholar] [CrossRef]
- Milanese, M.; Chelossi, E.; Manconi, R.; Sara, A.; Sidri, M.; Pronzato, R. The marine sponge Chondrilla nucula Schmidt, 1862 as an elective candidate for bioremediation in integrated aquaculture. Biomol. Eng. 2003, 20, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Claus, G.; Madri, P.; Kunen, S. Removal of microbial pollutants from waste effluents by Redbeard sponge. Nature 1967, 216, 712–714. [Google Scholar] [CrossRef]
- Longo, C.; Corriero, G.; Licciano, M.; Stabili, L. Bacterial accumulation by the Demospongiae Hymeniacidon perlevis: A tool for the bioremediation of polluted seawater. Mar. Pollut. Bull. 2010, 60, 1182–1187. [Google Scholar] [CrossRef]
- Maldonado, M.; Zhang, X.C.; Cao, X.P.; Xue, L.Y.; Cao, H.; Zhang, W. Selective feeding by sponges on pathogenic microbes: A reassessment of potential for abatement of microbial pollution. Mar. Ecol. Prog. Ser. 2010, 403, 75–89. [Google Scholar] [CrossRef]
- Stabili, L.; Licciano, M.; Giangrande, A.; Longo, C.; Mercurio, M.; Marzano, C.N.; Corriero, G. Filtering activity of Spongia officinalis var. adriatica (Schmidt) (Porifera, Demospongiae) on bacterioplankton: Implications for bioremediation of polluted seawater. Water Res. 2006, 40, 3083–3090. [Google Scholar] [CrossRef]
- Wehrl, M.; Steinert, M.; Hentschel, U. Bacterial uptake by the marine sponge Aplysina aerophoba. Microb. Ecol. 2007, 53, 355–365. [Google Scholar] [CrossRef]
- Zhang, X.C.; Zhang, W.; Xue, L.Y.; Zhang, B.; Jin, M.F.; Fu, W.T. Bioremediation of bacteria pollution using the marine sponge Hymeniacidon perlevis in the intensive mariculture water system of turbot Scophthalmus maximus. Biotechnol. Bioeng. 2010, 105, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Frost, T.M. In situ measurements of clearance rates for freshwater sponge Spongilla lacustris. Limnol. Oceanogr. 1978, 23, 1034–1039. [Google Scholar] [CrossRef]
- Osinga, R.; Kleijn, R.; Groenendijk, E.; Niesink, P.; Tramper, J.; Wijffels, R.H. Development of in vivo sponge cultures: Particle feeding by the tropical sponge Pseudosuberites aff. andrewsi. Mar. Biotechnol. 2001, 3, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Riisgard, H.U.; Thomassen, S.; Jakobsen, H.; Weeks, J.M.; Larsen, P.S. Suspension-feeding in marine sponges Halichondria panicea and Haliclona urceolus: Effects of temperature on filtration rate and energy cost of pumping. Mar. Ecol. Prog. Ser. 1993, 96, 177–188. [Google Scholar] [CrossRef]
- Varamogianni-Mamatsi, D.; Anastasiou, T.I.; Vernadou, E.; Papandroulakis, N.; Kalogerakis, N.; Dailianis, T.; Mandalakis, M. A multi-species investigation of sponges’ filtering activity towards marine microalgae. Mar. Drugs 2022, 20, 24. [Google Scholar] [CrossRef]
- Camacho, F.G.; Chileh, T.; Garcia, M.C.C.; Miron, A.S.; Belarbi, E.H.; Gomez, A.C.; Grima, E.M. Sustained growth of explants from Mediterranean sponge Crambe crambe cultured in vitro with enriched RPMI 1640. Biotechnol. Prog. 2006, 22, 781–790. [Google Scholar]
- Aresta, A.; Marzano, C.N.; Lopane, C.; Corriero, G.; Longo, C.; Zambonin, C.; Stabili, L. Analytical investigations on the lindane bioremediation capability of the demosponge Hymeniacidon perlevis. Mar. Pollut. Bull. 2015, 90, 143–149. [Google Scholar] [CrossRef]
- Ribes, M.; Yahel, G.; Romera-Castillo, C.; Mallenco, R.; Morganti, T.M.; Coma, R. The removal of dissolved organic matter by marine sponges is a function of its composition and concentration: An in situ seasonal study of four Mediterranean species. Sci. Total Environ. 2023, 871, 161991. [Google Scholar] [CrossRef]
- Ferrante, M.; Vassallo, M.; Mazzola, A.; Brundo, M.V.; Pecoraro, R.; Grasso, A.; Copat, C. In vivo exposure of the marine sponge Chondrilla nucula Schmidt, 1862 to cadmium (Cd), copper (Cu) and lead (Pb) and its potential use for bioremediation purposes. Chemosphere 2018, 193, 1049–1057. [Google Scholar] [CrossRef]
- Gravina, M.F.; Longo, C.; Puthod, P.; Rosati, M.; Colozza, N.; Scarselli, M. Heavy metal accumulation capacity of Axinella damicornis (Esper, 1794) (Porifera, Demospongiae): A tool for bioremediation of polluted seawaters. Mediterr. Mar. Sci. 2022, 23, 125–133. [Google Scholar] [CrossRef]
- Fu, W.T.; Wu, Y.C.; Sun, L.M.; Zhang, W. Efficient bioremediation of total organic carbon (TOC) in integrated aquaculture system by marine sponge Hymeniacidon perleve. Biotechnol. Bioeng. 2007, 97, 1387–1397. [Google Scholar] [CrossRef] [PubMed]
- Pronzato, R. Sponge-fishing, disease and farming in the Mediterranean Sea. Aquat. Conserv. 1999, 9, 485–493. [Google Scholar] [CrossRef]
- Varamogianni-Mamatsi, D.; Anastasiou, T.I.; Vernadou, E.; Kouvarakis, N.; Kagiampaki, E.; Kalogerakis, N.; Dailianis, T.; Mandalakis, M. Uptake of aquaculture-related dissolved organic pollutants by marine sponges: Kinetics and mechanistic insights from a laboratory study. Sci. Total Environ. 2023, 899, 165601. [Google Scholar] [CrossRef] [PubMed]
- Han, B.-N.; Hong, L.-L.; Gu, B.-B.; Sun, Y.-T.; Wang, J.; Liu, J.-T.; Lin, H.-W. Natural products from sponges. In Symbiotic Microbiomes of Coral Reefs Sponges and Corals; Li, Z., Ed.; Springer: Dordrecht, The Netherlands, 2019; pp. 329–463. [Google Scholar]
- Sipkema, D.; Osinga, R.; Schatton, W.; Mendola, D.; Tramper, J.; Wijffels, R.H. Large-scale production of pharmaceuticals by marine sponges: Sea, cell, or synthesis? Biotechnol. Bioeng. 2005, 90, 201–222. [Google Scholar] [CrossRef]
- Carballo, J.L.; Yanez, B.; Zubia, E.; Ortega, M.J.; Vega, C. Culture of explants from the sponge Mycale cecilia to obtain bioactive mycalazal-type metabolites. Mar. Biotechnol. 2010, 12, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Munro, M.H.G.; Blunt, J.W.; Dumdei, E.J.; Hickford, S.J.H.; Lill, R.E.; Li, S.X.; Battershill, C.N.; Duckworth, A.R. The discovery and development of marine compounds with pharmaceutical potential. J. Biotechnol. 1999, 70, 15–25. [Google Scholar] [CrossRef]
- Bergmann, W.; Feeney, R.J. Contribution on the study of marine sponges, 32. The nucleosides of sponges. J. Org. Chem. 1951, 16, 981–987. [Google Scholar] [CrossRef]
- Bergmann, W.; Feeney, R.J. The isolation of a new thymine pentoside from sponges. J. Am. Chem. Soc. 1950, 72, 2809–2810. [Google Scholar] [CrossRef]
- Buchanan, R.A.; Hess, F. Vidarabine (Vira-a): Pharmacology and clinical experience. Pharmacol. Therapeut. 1980, 8, 143–171. [Google Scholar] [CrossRef]
- McConnell, O.J.; Longley, R.E.; Koehn, F.E. The discovery of marine natural products with therapeutic potential. In Discovery of Novel Natural Products with Therapeutic Potential; Gullo, V.P., Ed.; Elsevier: Kenilworth, NJ, USA, 1994; pp. 109–174. [Google Scholar]
- Paintsil, E.; Cheng, Y.-C. Antiviral agents. In Encyclopedia of Microbiology, 3rd ed.; Schaechter, M., Ed.; Academic Press: Cambridge, MA, USA, 2009; pp. 223–257. [Google Scholar]
- Bodey, G.P.; Freireich, E.J.; Monto, R.W.; Hewlett, J.S. Cytosine arabinoside (NSC-63878) therapy for acute leukemia in adults. Cancer Chemother. Rep. 1969, 53, 59–66. [Google Scholar]
- Belarbi, E.; Gomez, A.C.; Chisti, Y.; Camacho, F.G.; Grima, E.M. Producing drugs from marine sponges. Biotechnol. Adv. 2003, 21, 585–598. [Google Scholar] [CrossRef]
- Page, M.J.; Handley, S.J.; Northcote, P.T.; Cairney, D.; Willan, R.C. Successes and pitfalls of the aquaculture of the sponge Mycale hentscheli. Aquaculture 2011, 312, 52–61. [Google Scholar] [CrossRef]
- Schneider, X.T.; Stroil, B.K.; Tourapi, C.; Rebours, C.; Novoveska, L.; Vasquez, M.I.; Gaudêncio, S.P. Improving awareness, understanding, and enforcement of responsibilities and regulations in Blue Biotechnology. Trends Biotechnol. 2023, 41, 1327–1331. [Google Scholar] [CrossRef] [PubMed]
- Schneider, X.T.; Stroil, B.K.; Tourapi, C.; Rebours, C.; Gaudêncio, S.P.; Novoveska, L.; Vasquez, M.I. Responsible research and innovation framework, the Nagoya Protocol and other European Blue Biotechnology strategies and regulations: Gaps analysis and recommendations for increased knowledge in the marine biotechnology community. Mar. Drugs 2022, 20, 290. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Zahn, R.K.; Gasic, M.J.; Dogovic, N.; Maidhof, A.; Becker, C.; Diehiseifert, B.; Eich, E. Avarol, a cytostatically active compound from the marine sponge Dysidea avara. Comp. Biochem. Phys. C 1985, 80, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Schroder, H.C. Cell biological aspects of HIV-1 infection: Effect of the anti-HIV-1 agent avarol. Int. J. Sports Med. 1991, 12, S43–S49. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Schatton, W.F.H.; Gudrum, M. Verwendung von Avarol Oder Dessen Derivaten zur Bekämpfung von Entzündlichen Systemischen und Dermatologischen Erkrankungen. Patent Application DE 1991-4137093, 1991. [Google Scholar]
- Page, M.; West, L.; Northcote, P.; Battershill, C.; Kelly, M. Spatial and temporal variability of cytotoxic metabolites in populations of the New Zealand sponge Mycale hentscheli. J. Chem. Ecol. 2005, 31, 1161–1174. [Google Scholar] [CrossRef] [PubMed]
- Hadas, E.; Shpigel, M.; Ilan, M. Sea ranching of the marine sponge Negombata magnifica (Demospongiae, Latrunculiidae) as a first step for latrunculin B mass production. Aquaculture 2005, 244, 159–169. [Google Scholar] [CrossRef]
- De Rosa, S.; De Caro, S.; Iodice, C.; Tommonaro, G.; Stefanov, K.; Popov, S. Development in primary cell culture of demosponges. J. Biotechnol. 2003, 100, 119–125. [Google Scholar] [CrossRef]
- Müller, W.E.G.; Bohm, M.; Batel, R.; De Rosa, S.; Tommonaro, G.; Muller, I.M.; Schroder, H.C. Application of cell culture for the production of bioactive compounds from sponges: Synthesis of avarol by primmorphs from Dysidea avara. J. Nat. Prod. 2000, 63, 1077–1081. [Google Scholar] [CrossRef]
- Wulff, J.L. Resistance vs recovery: Morphological strategies of coral reef sponges. Funct. Ecol. 2006, 20, 699–708. [Google Scholar] [CrossRef]
- Duckworth, A.; Battershill, C. Sponge aquaculture for the production of biologically active metabolites: The influence of farming protocols and environment. Aquaculture 2003, 221, 311–329. [Google Scholar] [CrossRef]
- Dailianis, T.; Mandalakis, M. Enhancing the environmental and economical sustainability of aquaculture through integration of sponges: An assessment of current knowledge. In Proceedings of the 54th European Marine Biology Symposium, Dublin, Ireland, 25–29 August 2019. [Google Scholar]
- Chu, M.J.; Li, M.; Ma, H.; Li, P.L.; Li, G.Q. Secondary metabolites from marine sponges of the genus Agelas: A comprehensive update insight on structural diversity and bioactivity. RSC Adv. 2022, 12, 7789–7820. [Google Scholar] [CrossRef] [PubMed]
- Richelle-Maurer, E.; De Kluijver, M.J.; Feio, S.; Gaudêncio, S.; Gaspar, H.; Gomez, R.; Tavares, R.; Van de Vyver, G.; Van Soest, R.W.M. Localization and ecological significance of oroidin and sceptrin in the Caribbean sponge Agelas conifera. Biochem. Syst. Ecol. 2003, 31, 1073–1091. [Google Scholar] [CrossRef]
- Liu, Y.H.; Zhang, S.; Jung, J.H.; Xu, T.H. Variabilin, a chemotaxonomic marker for the family Irciniidae. Z. Naturforschung C 2007, 62, 473–476. [Google Scholar] [CrossRef]
- Wang, N.; Shin, J. Secondary metabolites of marine sponge Sarcotragus sp. J. Biotechnol. 2008, 136, S587. [Google Scholar] [CrossRef]
- Gul, W.; Hamann, M.T. Indole alkaloid marine natural products: An established source of cancer drug leads with considerable promise for the control of parasitic, neurological and other diseases. Life Sci. 2005, 78, 442–453. [Google Scholar] [CrossRef]
- Kobayashi, J.; Tsuda, M.; Murayama, T.; Nakamura, H.; Ohizumi, Y.; Ishibashi, M.; Iwamura, M.; Ohta, T.; Nozoe, S. Ageliferins, potent actomyosin atpase activators from the Okinawan marine sponge Agelas sp. Tetrahedron 1990, 46, 5579–5586. [Google Scholar] [CrossRef]
- Kobayashi, J.; Tsuda, M.; Ohizumi, Y. A potent actomyosin atpase activator from the Okinawan marine sponge Agelas cf nemoechinata. Experientia 1991, 47, 301–304. [Google Scholar] [CrossRef]
- Neumann, K.; Abdel-Lateff, A.; Wright, A.D.; Kehraus, S.; Krick, A.; Konig, G.M. Novel sorbicillin derivatives with an unprecedented carbon skeleton from the sponge-derived fungus Trichoderma species (pg 2268, 2007). Eur. J. Org. Chem. 2007, 2007, 4125. [Google Scholar] [CrossRef]
- Sauleau, P.; Moriou, C.; Al Mourabit, A. Metabolomics approach to chemical diversity of the Mediterranean marine sponge Agelas oroides. Nat. Prod. Res. 2017, 31, 1625–1632. [Google Scholar] [CrossRef]
- König, G.M.; Wright, A.D.; Linden, A. Antiplasmodial and cytotoxic metabolites from the maltese sponge Agelas oroides. Planta Med. 1998, 64, 443–447. [Google Scholar] [CrossRef]
- Kovalerchik, D.; Singh, R.P.; Schlesinger, P.; Mahajni, A.; Shefer, S.; Fridman, M.; Ilan, M.; Carmeli, S. Bromopyrrole alkaloids of the sponge Agelas oroides collected near the Israeli Mediterranean coastline. J. Nat. Prod. 2020, 83, 374–384. [Google Scholar] [CrossRef]
- Forenza, S.; Minale, L.; Riccio, R. New bromo-pyrrole derivatives from sponge Agelas oroides. J. Chem. Soc. D Chem. Commun. 1971, 18, 1129–1130. [Google Scholar] [CrossRef]
- Nakamura, H.; Ohizumi, Y.; Kobayashi, J.; Hirata, Y. Keramadine, a novel antagonist of serotonergic receptors isolated from the Okinawan Sea sponge Agelas sp. Tetrahedron Lett. 1984, 25, 2475–2478. [Google Scholar] [CrossRef]
- Cafieri, F.; Fattorusso, E.; Mangoni, A.; Taglialatela-Scafati, O. Dispacamides, anti-histamine alkaloids from Caribbean Agelas sponges. Tetrahedron Lett. 1996, 37, 3587–3590. [Google Scholar] [CrossRef]
- Chanas, B.; Pawlik, J.R.; Lindel, T.; Fenical, W. Chemical defense of the Caribbean sponge Agelas clathrodes (Schmidt). J. Exp. Mar. Biol. Ecol. 1997, 208, 185–196. [Google Scholar] [CrossRef]
- Kobayashi, J.; Ohizumi, Y.; Nakamura, H.; Hirata, Y. A novel antagonist of serotonergic receptors, hymenidin, isolated from the Okinawan marine sponge Hymeniacidon sp. Experientia 1986, 42, 1176–1177. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.; Nieto, R.M.; Hunter, L.M.; Diaz, M.C.; Crews, P.; Lobkovsky, E.; Clardy, J. Variation among known kalihinol and new kalihinene diterpenes from the sponge Acanthella cavernosa. Tetrahedron 1994, 50, 11079–11090. [Google Scholar] [CrossRef]
- Jimenez, C.; Crews, P. Mauritamide-A and accompanying oroidin alkaloids from the sponge Agelas mauritiana. Tetrahedron Lett. 1994, 35, 1375–1378. [Google Scholar] [CrossRef]
- Sharma, G.; Magdofffairchild, B. Natural products of marine sponges. 7. Constitution of weakly basic guanidine compounds, dibromophakellin and monobromophakellin. J. Org. Chem. 1977, 42, 4118–4124. [Google Scholar] [CrossRef]
- Umeyama, A.; Ito, S.; Yuasa, E.; Arihara, S.; Yamada, T. A new bromopyrrole alkaloid and the optical resolution of the racemate from the marine sponge Homaxinella sp. J. Nat. Prod. 1998, 61, 1433–1434. [Google Scholar] [CrossRef]
- Cafieri, F.; Fattorusso, E.; Taglialatela-Scafati, O. Novel bromopyrrole alkaloids from the sponge Agelas dispar. J. Nat. Prod. 1998, 61, 122–125. [Google Scholar] [CrossRef]
- Assmann, M.; Köck, M. Monobromoisophakellin, a new bromopyrrole alkaloid from the Caribbean sponge Agelas sp. Z. Naturforsch. C 2002, 57, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Freire, V.F.; Gubiani, J.R.; Spencer, T.M.; Hajdu, E.; Ferreira, A.G.; Ferreira, D.A.S.; Levatti, E.V.D.; Burdette, J.E.; Camargo, C.H.; Tempone, A.G.; et al. Feature-based molecular networking discovery of bromopyrrole alkaloids from the marine sponge Agelas dispar. J. Nat. Prod. 2022, 85, 1340–1350. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ma, Z.Q.; Wang, X.L.; De, S.; Ma, Y.Y.; Chen, C. Dimeric pyrrole-imidazole alkaloids: Synthetic approaches and biosynthetic hypotheses. Chem. Commun. 2014, 50, 8628–8639. [Google Scholar] [CrossRef] [PubMed]
- Eder, C.; Proksch, P.; Wray, V.; van Soest, R.W.M.; Ferdinandus, E.; Pattisina, L.A. Sudarsono, New bromopyrrole alkaloids from the indopacific sponge Agelas nakamurai. J. Nat. Prod. 1999, 62, 1295–1297. [Google Scholar] [CrossRef] [PubMed]
- Keifer, P.A.; Schwartz, R.E.; Koker, M.E.S.; Hughes, R.G.; Rittschof, D.; Rinehart, K.L. Bioactive bromopyrrole metabolites from the Caribbean sponge Agelas conifera. J. Org. Chem. 1991, 56, 2965–2975. [Google Scholar] [CrossRef]
- Walker, R.P.; Faulkner, D.J.; Vanengen, D.; Clardy, J. Sceptrin, an antimicrobial agent from the sponge Agelas sceptrum. J. Am. Chem. Soc. 1981, 103, 6772–6773. [Google Scholar] [CrossRef]
- Funayama, S.; Cordell, A.G. (Eds.) Alkaloids derived from terpenoids. In Alkaloids: A Treasury of Poisons and Medicines; Academic Press: New York, NY, USA, 2015; pp. 233–255. [Google Scholar]
- Cullen, E.; Devlin, J.P. Agelasine: A novel quaternary 9-methyladenine from sponge Agelas dispar. Can. J. Chem. 1975, 53, 1690–1691. [Google Scholar] [CrossRef]
- Nakamura, H.; Wu, H.M.; Ohizumi, Y.; Hirata, Y. Agelasine-A, -B, -C and -D, novel bicyclic diterpenoids with a 9-methyladeninium unit possessing inhibitory effects on Na,K-ATPase from the Okinawan sea sponge Agelas sp. Tetrahedron Lett. 1984, 25, 2989–2992. [Google Scholar] [CrossRef]
- Wu, H.; Nakamura, H.; Kobayashi, J.; Ohizumi, Y.; Hirata, Y. Agelasine-E and -F, novel monocyclic diterpenoids with a 9-methyladeninium unit possessing inhibitory effects on Na,K-ATPase isolated from the Okinawan sea sponge Agelas nakamurai Hoshino. Tetrahedron Lett. 1984, 25, 3719–3722. [Google Scholar] [CrossRef]
- Nakamura, H.; Wu, H.; Kobayashi, J.; Kobayashi, M.; Ohizumi, Y.; Hirata, Y. Agelasidines: Novel hypotaurocyamine derivatives from the Okinawan sea sponge Agelas nakamurai Hoshino. J. Org. Chem. 1985, 50, 2494–2497. [Google Scholar] [CrossRef]
- Yu, C.M.; Curtis, J.M.; Wright, J.L.C.; Ayer, S.W.; Fathi-Afshar, Z.R. An unusual fatty acid and its glyceride from the marine fungus Microsphaeropis olivacea. Can. J. Chem. 1996, 74, 730–735. [Google Scholar] [CrossRef]
- Tasdemir, D.; Topaloglu, B.; Perozzo, R.; Brun, R.; O’Neill, R.; Carballeira, N.M.; Zhang, X.J.; Tonge, P.J.; Linden, A.; Ruedi, P. Marine natural products from the Turkish sponge Agelas oroides that inhibit the enoyl reductases from Plasmodium falciparum, Mycobacterium tuberculosis and Escherichia coli. Bioorgan. Med. Chem. 2007, 15, 6834–6845. [Google Scholar] [CrossRef] [PubMed]
- Costantino, V.; Dell’Aversano, C.; Fattorusso, E.; Mangoni, A. Ecdysteroids from the caribbean sponge Iotrochota birotulata. Steroids 2000, 65, 138–142. [Google Scholar] [CrossRef]
- Zhao, P.C.; Li, J.J.; Wang, Y.; Jiang, H.B. Broad-spectrum antimicrobial activity of the reactive compounds generated in vitro by Manduca sexta phenoloxidase. Insect Biochem. Mol. Biol. 2007, 37, 952–959. [Google Scholar] [CrossRef]
- Page, M.J.; Northcote, P.T.; Webb, V.L.; Mackey, S.; Handley, S.J. Aquaculture trials for the production of biologically active metabolites in the New Zealand sponge Mycale hentscheli (Demospongiae: Poecilosclerida). Aquaculture 2005, 250, 256–269. [Google Scholar] [CrossRef]
- De Voogd, N.J. The mariculture potential of the Indonesian reef-dwelling sponge Callyspongia (Euplacella) biru: Growth, survival and bioactive compounds. Aquaculture 2007, 262, 54–64. [Google Scholar] [CrossRef]
- Turon, X.; Becerro, M.A.; Uriz, M.J. Seasonal patterns of toxicity in benthic invertebrates: The encrusting sponge Crambe crambe (Poecilosclerida). Oikos 1996, 75, 33–40. [Google Scholar] [CrossRef]
- Rohde, S.; Gochfeld, D.J.; Ankisetty, S.; Avula, B.; Schupp, P.J.; Slattery, M. Spatial variability in secondary metabolites of the Indo-Pacific sponge Stylissa massa. J. Chem. Ecol. 2012, 38, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Turon, X.; Marti, R.; Uriz, M.J. Chemical bioactivity of sponges along an environmental gradient in a Mediterranean cave. Sci. Mar. 2009, 73, 387–397. [Google Scholar] [CrossRef]
- Becerro, M.A.; Turon, X.; Uriz, M.J. Natural variation of toxicity in encrusting sponge Crambe crambe (Schmidt) in relation to size and environment. J. Chem. Ecol. 1995, 21, 1931–1946. [Google Scholar] [CrossRef]
- Rohde, S.; Molis, M.; Wahl, M. Regulation of anti-herbivore defence by Fucus vesiculosus in response to various cues. J. Ecol. 2004, 92, 1011–1018. [Google Scholar] [CrossRef]
- Agell, G.; Uriz, M.J.; Cebrian, E.; Marti, R. Does stress protein induction by copper modify natural toxicity in sponges? Environ. Toxicol. Chem. 2001, 20, 2588–2593. [Google Scholar] [CrossRef] [PubMed]
- Becerro, M.A.; Paul, V.J. Effects of depth and light on secondary metabolites and cyanobacterial symbionts of the sponge Dysidea granulosa. Mar. Ecol. Prog. Ser. 2004, 280, 115–128. [Google Scholar] [CrossRef]
- Uriz, M.J.; Turon, X.; Becerro, M.A.; Galera, J.; Lozano, J. Patterns of resource allocation to somatic, defensive, and reproductive functions in the Mediterranean encrusting sponge Crambe crambe (Demospongiae, Poecilosclerida). Mar. Ecol. Prog. Ser. 1995, 124, 159–170. [Google Scholar] [CrossRef]
- Chanas, B.; Pawlik, J.R. Defenses of Caribbean sponges against predatory reef fish. II. Spicules, tissue toughness, and nutritional quality. Mar. Ecol. Prog. Ser. 1995, 127, 195–211. [Google Scholar] [CrossRef]
- Assmann, M.; Lichte, E.; Pawlik, J.R.; Kock, M. Chemical defenses of the Caribbean sponges Agelas wiedenmayeri and Agelas conifera. Mar. Ecol. Prog. Ser. 2000, 207, 255–262. [Google Scholar] [CrossRef]
- Kloppel, A.; Pfannkuchen, M.; Putz, A.; Proksch, P.; Brummer, F. Ex situ cultivation of Aplysina aerophoba close to in situ conditions: Ecological, biochemical and histological aspects. Mar. Ecol. 2008, 29, 259–272. [Google Scholar] [CrossRef]
- Duckworth, A.R.; Samples, G.A.; Wright, A.E.; Pomponi, S.A. In vitro culture of the tropical sponge Axinella corrugata (Demospongiae): Effect of food cell concentration on growth, clearance rate, and biosynthesis of stevensine. Mar. Biotechnol. 2003, 5, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Ternon, E.; Perino, E.; Manconi, R.; Pronzato, R.; Thomas, O.P. How environmental factors affect the production of guanidine alkaloids by the Mediterranean sponge Crambe crambe. Mar. Drugs 2017, 15, 181. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, A.L.; Taylor, T.A.; Beatty, J.T.; Greenberg, E.P. Long-chain acyl-homoserine lactone quorum-sensing regulation of Rhodobacter capsulatus gene transfer agent production. J. Bacteriol. 2002, 184, 6515–6521. [Google Scholar] [CrossRef]
- Bojko, B.; Onat, B.; Boyaci, E.; Psillakis, E.; Dailianis, T.; Pawliszyn, J. Application of in situ solid-phase microextraction on Mediterranean sponges for untargeted exometabolome screening and environmental monitoring. Front. Mar. Sci. 2019, 6, 632. [Google Scholar] [CrossRef]
- Saurav, K.; Burgsdorf, I.; Teta, R.; Esposito, G.; Bar-Shalom, R.; Costantino, V.; Steindler, L. Isolation of marine Paracoccus sp. Ss63 from the sponge Sarcotragus sp. and characterization of its quorum-sensing chemical-signaling molecules by LC-MS/MS analysis. Isr. J. Chem. 2016, 56, 330–340. [Google Scholar] [CrossRef]
- Cimino, G.; De Stefan, S.; Minale, L. Prenylated quinones in marine sponges: Ircinia sp. Experientia 1972, 28, 1401–1402. [Google Scholar] [CrossRef]
- Liu, Y.H.; Jung, J.H.; Ji, H.; Zhang, S. Glycerolipids from a Sarcotragus species sponge. Molecules 2006, 11, 714–719. [Google Scholar] [CrossRef]
- Gil, J.H.; Seo, J.; Kim, K.J.; Jung, J.H.; Jung, O.S.; Kim, M.S.; Hong, J. Structural determination of cyclitol derivatives by fast-atom bombardment tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2006, 20, 1253–1256. [Google Scholar] [CrossRef]
- Elissawy, A.M.; Ebada, S.S.; Ashour, M.L.; Ozkaya, F.C.; Ebrahim, W.; Singab, A.B.; Proksch, P. Spiroarthrinols a and B, two novel meroterpenoids isolated from the spongederived fungus Arthrinium sp. Phytochem. Lett. 2017, 20, 246–251. [Google Scholar] [CrossRef]
- Casellas, M.; Grifoll, M.; Bayona, J.M.; Solanas, A.M. New metabolites in the degradation of fluorene by Arthrobacter sp. strain F101. Appl. Environ. Microb. 1997, 63, 819–826. [Google Scholar] [CrossRef] [PubMed]
- Kashiwada, Y.; Nonaka, G.; Nishioka, I. Studies on rhubarb (Rhei rhizoma). V. Isolation and characterization of stilbenes. Chem. Pharm. Bull. 1984, 32, 3501–3517. [Google Scholar]
- Liu, Y.H.; Jung, J.H.; Zhang, S. Indole alkaloids from a sponge Sarcotragus species. Biochem. Syst. Ecol. 2006, 34, 453–456. [Google Scholar] [CrossRef]
- Kingston, D.G.I.; Chen, P.N.; Vercellotti, J.R. Metabolites of Aspergillus versicolor: 6,8-Di-O-methylnidurufin, griseofulvin, dechlorogriseofluvin, and 3,8-dihydroxy-6-methoxy-1-methylaxanthone. Phytochemistry 1976, 15, 1037–1039. [Google Scholar] [CrossRef]
- Danielsen, K.; Aksnes, D.W. NMR study of some anthraquinones from rhubarb. Magn. Reson. Chem. 1992, 30, 359–360. [Google Scholar] [CrossRef]
- Grove, J.F.; Macmillan, J.; Mulholland, T.P.C.; Rogers, M.A.T. 759. Griseofulvin. Part I. J. Chem. Soc. 1952, 3949–3958. [Google Scholar] [CrossRef]
- Wells, J.M.; Cole, R.J.; Kirksey, J.W. Emodin, a toxic metabolite of Aspergillus wentii isolated from weevil-damaged chestnuts. Appl. Microbiol. 1975, 30, 26–28. [Google Scholar] [CrossRef]
- Kurobane, I.; Vining, L.C.; Mcinnes, A.G. Biosynthetic relationships among the secalonic acids. Isolation of emodin, endocrocin and secalonic acids from Pyrenochaeta terrestris and Aspergillus aculeatus. J. Antibiot. 1979, 32, 1256–1266. [Google Scholar] [CrossRef]
- Williams, M.C. 3-Nitropropionic acid and 3-nitro-1-propanol in species of Astragalus. Can. J. Bot. 1982, 60, 1956–1963. [Google Scholar] [CrossRef]
- Wu, T.S.; Chan, Y.Y.; Leu, Y.L. The constituents of the root and stem of Aristolochia cucurbitifolia hayata and their biological activity. Chem. Pharm. Bull. 2000, 48, 1006–1009. [Google Scholar] [CrossRef]
- Mao, S.; Lee, S.J.; Hwangbo, H.; Kim, Y.W.; Park, K.H.; Cha, G.S.; Park, R.D.; Kim, K.Y. Isolation and characterization of antifungal substances from Burkholderia sp. culture broth. Curr. Microbiol. 2006, 53, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Cross, B.E.; Galt, R.H.B.; Hanson, J.R. 706. New metabolites of Gibberella fujikuroi. Part IV. Structures of 7,18-dihydroxy- and 7,16,18-trihydroxy-kaurenolide. J. Chem. Soc. 1963, 3783–3790. [Google Scholar] [CrossRef]
- Barrow, C.J.; Blunt, J.W.; Munro, M.H.G.; Perry, N.B. Variabilin and related compounds from a sponge of the genus Sarcotragus. J. Nat. Prod. 1988, 51, 275–281. [Google Scholar] [CrossRef]
- Bassaganya-Riera, J.; Skoneczka, J.; Kingston, D.G.J.; Krishnan, A.; Misyak, S.A.; Guri, A.J.; Pereira, A.; Carter, A.B.; Minorsky, P.; Tumarkin, R.; et al. Mechanisms of action and medicinal applications of abscisic acid. Curr. Med. Chem. 2010, 17, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Dini, A.; Sica, D.; Boniforti, L. 2 New delta-5,7-sterols from 2 Spongiidae sponges. Comp. Biochem. Phys. B 1984, 78, 741–744. [Google Scholar] [CrossRef]
- Yang, J.X.; Qiu, S.X.; She, Z.G.; Lin, Y.C. A new xanthone derivative from the marine fungus Phomopsis sp. (No. SK7RN3G1). Chem. Nat. Compd. 2013, 49, 246–248. [Google Scholar] [CrossRef]
- Correa, E.; Sterner, O.; Echeverri, F. Prenylhydroxybenzoic acid compounds with pungent activity from Piper arieianum (CDC) leaves. Phytochemistry 2011, 72, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Ahluwalia, V.; Kumar, J.; Rana, V.S.; Sati, O.P.; Walia, S. Comparative evaluation of two Trichoderma harzianum strains for major secondary metabolite production and antifungal activity. Nat. Prod. Res. 2015, 29, 914–920. [Google Scholar] [CrossRef]
- Liu, S.Y.; Lo, C.T.; Shibu, M.A.; Leu, Y.L.; Jen, B.Y.; Peng, K.C. Study on the anthraquinones separated from the cultivation of Trichoderma harzianum strain Th-R16 and their biological activity. J. Agric. Food Chem. 2009, 57, 7288–7292. [Google Scholar] [CrossRef]
- Dias, A.C.D.; Couzinet-Mossion, A.; Ruiz, N.; Lakhdar, F.; Etahiri, S.; Bertrand, S.; Ory, L.; Roussakis, C.; Pouchus, Y.F.; Nazih, E.; et al. Steroids from marine-derived fungi: Evaluation of antiproliferative and antimicrobial activities of eburicol. Mar. Drugs 2019, 17, 372. [Google Scholar] [CrossRef]
- Kusama, T.; Tanaka, N.; Takahashi-Nakaguchi, A.; Gonoi, T.; Fromont, J.; Kobayashi, J. Bromopyrrole alkaloids from a marine sponge Agelas sp. Chem. Pharm. Bull. 2014, 62, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, M.A.; Lourenco, A.; Tavares, M.R.; Curto, M.J.M.; Feio, S.S.; Roseiro, J.C. (−)-Agelasidine A from Agelas clathrodes. Z. Naturforsch. C 2006, 61, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.Y.; Meng, F.P.; Peng, X.L.; Lin, Y.F.; Wang, G.S.; Wu, J.Y. Kinetic analysis and degradation mechanism for natural attenuation of xylenes under simulated marine conditions. Ecotoxicol. Environ. Safe 2019, 168, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Loots, D.T.; van der Westhuizen, F.H.; Botes, L. Aloe ferox leaf gel phytochemical content, antioxidant capacity, and possible health benefits. J. Agric. Food Chem. 2007, 55, 6891–6896. [Google Scholar] [CrossRef] [PubMed]
- Mussinan, C.J.; Walradt, J.P. Organic acids from fresh California strawberries. J. Agric. Food Chem. 1975, 23, 482–484. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Romero, M.C.; Murphy, T.; Morris, M.; Cummins, E.; Kerry, J.P. Antimicrobial activity of chitosan, organic acids and nano-sized solubilisates for potential use in smart antimicrobially-active packaging for potential food applications. Food Control 2013, 34, 393–397. [Google Scholar] [CrossRef]
- Sullivan, D.J.; Azlin-Hasim, S.; Cruz-Romer, M.; Cummins, E.; Kerry, J.P.; Morris, M.A. Antimicrobial effect of benzoic and sorbic acid salts and nano-solubilisates against Staphylococcus aureus, Pseudomonas fluorescens and chicken microbiota biofilms. Food Control 2020, 107, 106786. [Google Scholar] [CrossRef]
- Andrade, J.C.; Morais-Braga, M.F.B.; Guedes, G.M.M.; Tintino, S.R.; Freitas, M.A.; Menezes, I.R.A.; Coutinho, H.D.M. Enhancement of the antibiotic activity of aminoglycosides by alpha-toocopherol and other cholesterol derivates. Biomed. Pharmacother. 2014, 68, 1065–1069. [Google Scholar] [CrossRef]
- Tintino, S.R.; Oliveira-Tintino, C.D.M.; Campina, F.F.; Costa, M.S.; Cruz, R.P.; Pereira, R.L.S.; Andrade, J.C.; Sousa, E.O.; Siqueira-Junior, J.P.; Coutinho, H.D.M.; et al. Cholesterol and ergosterol affect the activity of Staphylococcus aureus antibiotic efflux pumps. Microb. Pathog. 2017, 104, 133–136. [Google Scholar] [CrossRef]
- Govinden-Soulange, J.; Marie, D.; Kauroo, S.; Beesoo, R.; Ramanjooloo, A. Antibacterial properties of marine sponges from Mauritius waters. Trop. J. Pharm. Res. 2014, 13, 249–254. [Google Scholar] [CrossRef]
- Dyson, L.; Wright, A.D.; Young, K.A.; Sakoff, J.A.; McCluskey, A. Synthesis and anticancer activity of focused compound libraries from the natural product lead, oroidin. Bioorgan. Med. Chem. 2014, 22, 1690–1699. [Google Scholar] [CrossRef] [PubMed]
- Kwon, O.S.; Kim, D.; Kim, H.; Lee, Y.J.; Lee, H.S.; Sim, C.J.; Oh, D.C.; Lee, S.K.; Oh, K.B.; Shin, J. Bromopyrrole alkaloids from the sponge Agelas kosrae. Mar. Drugs 2018, 16, 513. [Google Scholar] [CrossRef] [PubMed]
- Ang, A.M.G.; Tabugo, S.R.M.; Uy, M.M. Antiproliferative, proapoptotic, and antimigration activities of marine sponges against human colon cancer cell line (HCT116). J. Appl. Pharm. Sci. 2023, 13, 186–192. [Google Scholar] [CrossRef]
- Steenkamp, V.; Gouws, M.C. Cytotoxicity of six South African medicinal plant extracts used in the treatment of cancer. S. Afr. J. Bot. 2006, 72, 630–633. [Google Scholar] [CrossRef]
- Faulkner, D.J. Marine natural products. Nat. Prod. Rep. 2002, 19, 1–48. [Google Scholar] [PubMed]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2005, 22, 15–61. [Google Scholar] [CrossRef] [PubMed]
- Voultsiadou, E. Demosponge distribution in the eastern Mediterranean: A NW-SE gradient. Helgol. Mar. Res. 2005, 59, 237–251. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2014, 31, 160–258. [Google Scholar] [CrossRef]
- Ferretti, C.; Vacca, S.; De Ciucis, C.; Marengo, B.; Duckworth, A.R.; Manconi, R.; Pronzato, R.; Domenicotti, C. Growth dynamics and bioactivity variation of the Mediterranean demosponges Agelas oroides (Agelasida, Agelasidae) and Petrosia ficiformis (Haplosclerida, Petrosiidae). Mar. Ecol. 2009, 30, 327–336. [Google Scholar] [CrossRef]
- Idan, T.; Goren, L.; Shefer, S.; Ilan, M. Sponges in a changing climate: Survival of Agelas oroides in a warming Mediterranean Sea. Front. Mar. Sci. 2020, 7, 603593. [Google Scholar] [CrossRef]
- Manconi, R.; Cadeddu, B.; Ledda, F.; Pronzato, R. An overview of the Mediterranean cave-dwelling horny sponges (Porifera, Demospongiae). Zookeys 2013, 281, 1–68. [Google Scholar] [CrossRef] [PubMed]
- Konstantinou, D.; Gerovasileiou, V.; Voultsiadou, E.; Gkelis, S. Sponges-cyanobacteria associations: Global diversity overview and new data from the Eastern Mediterranean. PLoS ONE 2018, 13, e0195001. [Google Scholar] [CrossRef] [PubMed]
- Altunok, M.; Ozkaya, F.C.; Engin, S.; Tanrikul, T.T.; Aydinlik, S.; Ulukaya, E. In vitro antibacterial activity of sponge-associated fungi against bacterial aquaculture pathogens. Fresenius Environ. Bull. 2015, 24, 2158–2166. [Google Scholar]
- Pinto-Almeida, A.; Bauermeister, A.; Luppino, L.; Grilo, I.R.; Oliveira, J.; Sousa, J.R.; Petras, D.; Rodrigues, C.F.; Prieto-Davó, A.; Tasdemir, D.; et al. The diversity, metabolomics profiling, and the pharmacological potential of actinomycetes isolated from the Estremadura Spur pockmarks (Portugal). Mar. Drugs 2022, 20, 21. [Google Scholar] [CrossRef]
- Andrews, J.M. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother. 2001, 48 (Suppl. 1), 5–16. [Google Scholar] [CrossRef]
- Florindo, P.R.; Pereira, D.M.; Borralho, P.M.; Costa, P.J.; Piedade, M.F.M.; Rodrigues, C.M.P.; Fernandes, A.C. New [(η(5)-C5H5)Ru(N-N)(PPh3)][PF6] compounds: Colon anticancer activity and GLUT-mediated cellular uptake of carbohydrate-appended complexes. Dalton Trans. 2016, 45, 11926–11930. [Google Scholar] [CrossRef]
- Prieto-Davo, A.; Dias, T.; Gomes, S.E.; Rodrigues, S.; Parera-Valadez, Y.; Borralho, P.M.; Pereira, F.; Rodrigues, C.M.; Santos-Sanches, I.; Gaudêncio, S.P. The Madeira Archipelago as a significant source of marine-derived actinomycete diversity with anticancer and antimicrobial potential. Front. Microbiol. 2016, 7, 1594. [Google Scholar] [CrossRef]
- Pontes, M.d.C.N.D. Estudo Químico e Biológico das Esponjas Agelas e Agelas dispar do Nordeste do Brasil. Ph.D. Thesis, Universidade Federal do Rio Grande do Norte, Natal, Brazil, 2019. [Google Scholar]
- Hao, E.; Fromont, J.; Jardine, D.; Karuso, P. Natural products from sponges of the genus Agelas—On the trail of a [2+2]-photoaddition enzyme. Molecules 2001, 6, 130–141. [Google Scholar] [CrossRef]
- Riyanti; Marner, M.; Hartwig, C.; Patras, M.A.; Wodi, S.I.M.; Rieuwpassa, F.J.; Ijong, F.G.; Balansa, W.; Schaberle, T.E. Sustainable low-volume analysis of environmental samples by Semi-Automated Prioritization of Extracts for Natural Product Research (SeaPEPR). Mar. Drugs 2020, 18, 649. [Google Scholar] [CrossRef]
- Kang, D.; Kim, J.; Choi, J.N.; Liu, K.H.; Lee, C.H. Chemotaxonomy of Trichoderma spp. using mass spectrometry-based metabolite profiling. J. Microbiol. Biotechnol. 2011, 21, 5–13. [Google Scholar] [CrossRef]
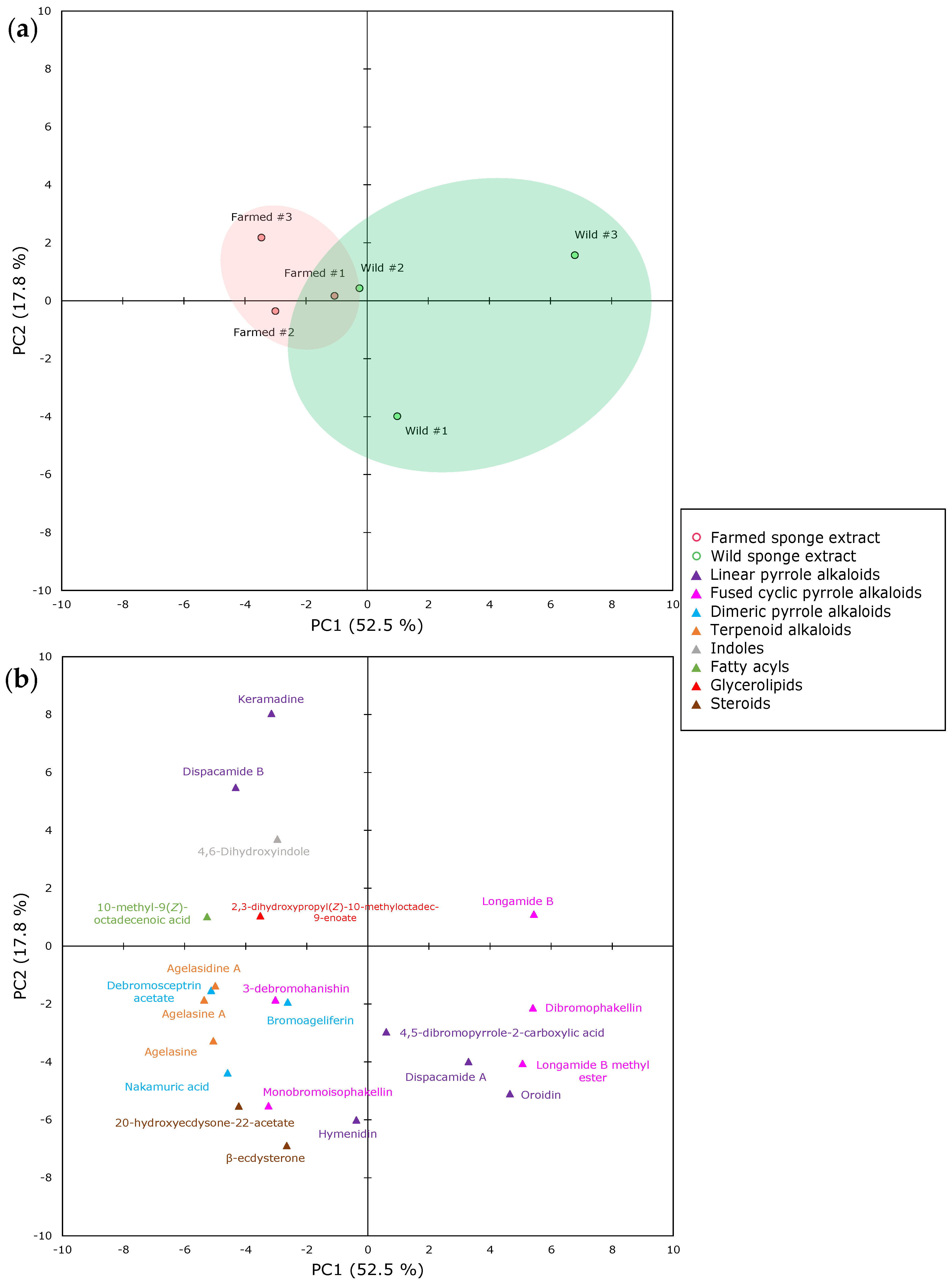




| Superclass | Class | Subclass | Wild (%) | Farmed (%) |
|---|---|---|---|---|
| Alkaloids | Pyrrole alkaloids | Linear pyrrole alkaloids | 97.2 (1.4) | 95.1 (0.9) |
| Fused cyclic pyrrole alkaloids | 0.9 (0.1) | 0.7 (0.0) | ||
| Dimeric pyrrole alkaloids | 0.2 (0.2) | 0.4 (0.1) | ||
| Terpenoid alkaloids | ND | 0.6 (0.5) | 1.3 (0.1) | |
| Indoles | ND | ND | <0.03 | <0.03 |
| Lipids | Fatty acyls | ND | 1.0 (0.9) | 2.4 (0.8) |
| Glycerolipids | ND | <0.002 | <0.003 | |
| Steroids | ND | <0.002 | <0.001 |
| Superclass | Class | Wild (%) | Farmed (%) |
|---|---|---|---|
| Benzenoids | Anthracenes | <0.01 | <0.01 |
| Benzene and substituted derivatives | 4.2 (0.3) | 3.2 (0.4) | |
| Benzopyrans | 0.5 (0.2) | 0.4 (0.0) | |
| Phenols | <0.01 | <0.01 | |
| Dipeptides | ND | <0.02 | <0.02 |
| Indoles | ND | 0.1 (0.0) | 0.1 (0.1) |
| Lipids | Fatty acyls | 55.5 (1.2) | 56.2 (0.5) |
| Glycerolipids | 7.2 (0.4) | 7.4 (0.3) | |
| Prenol lipids | 1.5 (0.4) | 3.1 (0.5) | |
| Steroids | 30.1 (0.7) | 28.7 (0.4) | |
| Polyketides | ND | 0.8 (0.1) | 0.8 (0.0) |
| Species | Type of Sponge Population | Sponge Replicate | MIC Values (µg/mL) for S. aureus MRSA COL | MIC Values (µg/mL) for E. coli ATCC 25922 |
|---|---|---|---|---|
| Agelas oroides | Wild | #1 | NA | NA |
| #2 | NA | NA | ||
| #3 | 250 | NA | ||
| Farmed | #1 | NA | NA | |
| #2 | 250 | NA | ||
| #3 | 250 | 250 | ||
| Sarcotragus foetidus | Wild | #1 | 125 | NA |
| #2 | 125 | NA | ||
| #3 | 250 | NA | ||
| Farmed | #1 | NA | NA | |
| #2 | 250 | NA | ||
| #3 | NA | NA | ||
| Positive control | 1.9 | 3.9 | ||
| Species | Type of Population | Replicate | IC50 (µg/mL) | 95% CI |
|---|---|---|---|---|
| Sarcotragus foetidus | Wild | #1 | 70.8 | 70.3–80.4 |
| #2 | 70.4 | 65.7–74.7 | ||
| #3 | 71.5 | 68.4–73.8 | ||
| Farmed | #1 | 80.5 | 74.1–93.3 | |
| #2 | 41.2 | 30.1–50.0 | ||
| #3 | 74.2 | 70.8–79.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varamogianni-Mamatsi, D.; Nunes, M.J.; Marques, V.; Anastasiou, T.I.; Kagiampaki, E.; Vernadou, E.; Dailianis, T.; Kalogerakis, N.; Branco, L.C.; Rodrigues, C.M.P.; et al. Comparative Chemical Profiling and Antimicrobial/Anticancer Evaluation of Extracts from Farmed versus Wild Agelas oroides and Sarcotragus foetidus Sponges. Mar. Drugs 2023, 21, 612. https://doi.org/10.3390/md21120612
Varamogianni-Mamatsi D, Nunes MJ, Marques V, Anastasiou TI, Kagiampaki E, Vernadou E, Dailianis T, Kalogerakis N, Branco LC, Rodrigues CMP, et al. Comparative Chemical Profiling and Antimicrobial/Anticancer Evaluation of Extracts from Farmed versus Wild Agelas oroides and Sarcotragus foetidus Sponges. Marine Drugs. 2023; 21(12):612. https://doi.org/10.3390/md21120612
Chicago/Turabian StyleVaramogianni-Mamatsi, Despoina, Maria João Nunes, Vanda Marques, Thekla I. Anastasiou, Eirini Kagiampaki, Emmanouela Vernadou, Thanos Dailianis, Nicolas Kalogerakis, Luís C. Branco, Cecília M. P. Rodrigues, and et al. 2023. "Comparative Chemical Profiling and Antimicrobial/Anticancer Evaluation of Extracts from Farmed versus Wild Agelas oroides and Sarcotragus foetidus Sponges" Marine Drugs 21, no. 12: 612. https://doi.org/10.3390/md21120612
APA StyleVaramogianni-Mamatsi, D., Nunes, M. J., Marques, V., Anastasiou, T. I., Kagiampaki, E., Vernadou, E., Dailianis, T., Kalogerakis, N., Branco, L. C., Rodrigues, C. M. P., Sobral, R. G., Gaudêncio, S. P., & Mandalakis, M. (2023). Comparative Chemical Profiling and Antimicrobial/Anticancer Evaluation of Extracts from Farmed versus Wild Agelas oroides and Sarcotragus foetidus Sponges. Marine Drugs, 21(12), 612. https://doi.org/10.3390/md21120612













